Abstract
Purpose:
Literature regarding total hip arthroplasty for pediatric hip diseases is scarce. This review aims to portray the various orthopedic conditions of childhood that can lead to significant impairment of the hip joint and, ultimately, to total hip arthroplasty in adolescence and adulthood.
Methods:
In total, 61 out of 3666 articles were selected according to (1) the diagnosis of one of the 12 pediatric hip pathologies (Legg–Perthes–Calvé disease, developmental dysplasia of the hip, slipped capital femoral epiphysis, neuromuscular hip dysplasia, post-traumatic avascular necrosis of the proximal femur, juvenile rheumatoid arthritis, achondroplasia, spondyloepiphyseal dysplasia, mucopolysaccharidosis, mucolipidosis, hip infections, and tumors) that required total hip arthroplasty; (2) minimum follow-up of 16 months; (3) assessed outcome with a clinical or radiologic score; (4) Methodological Items for Non-Randomized Studies quality score of 9 or higher. The following information for each pathology was retrieved: mean age at total hip arthroplasty, reason for total hip arthroplasty, type of total hip arthroplasty, surgical technique, mean follow-up, and outcomes.
Results:
Overall, the mean age at total hip arthroplasty for pediatric hip disease is in the sixth and seventh decade, except for tumors and skeletal dysplasias. The reason for performing total hip arthroplasty is often osteoarthrosis and abnormal anatomy. Prosthesis types change based on patient’s conditions and technological advances; custom-made implants are used for tumors, juvenile rheumatoid arthritis, and skeletal dysplasias; for other diseases, the most frequent are modular cementless implants. Outcomes are generally good, and all studies portray functional and pain improvements.
Conclusion:
Total hip arthroplasty is performed more frequently than in the past in patients with pediatric hip pathologies; it enhances patients’ quality of life by reducing pain and improving function. However, revision rate in these patients is not negligible.
Introduction
The body of literature on total hip arthroplasty (THA) resulting from pediatric hip pathology proves to be scarce and considerably mixed. Establishing a causal relationship between pediatric hip disease and THA poses a significant challenge, often necessitating follow-up over many decades.
The causes of pediatric hip disorders that may lead to THA in adulthood range from classic conditions, such as Legg–Calvè–Perthes disease (LCPD), developmental dysplasia of the hip (DDH), and slipped capital femoral epiphysis (SCFE), to rarer ones such as neuromuscular conditions, immunity disorders, post-traumatic sequelae, or skeletal dysplasias.
Pediatric hip pathology can potentially result in THA with wide variability depending on the type of disease and its severity. The occurrence of THA in patients with LCPD or SCFE is below 10% compared to over 35% in individuals with DDH treated after the walking age. The incidence of rare pathologies is unclear due to the limited number of patients and the paucity of literature reports.
This review examines the various orthopedic conditions of childhood that can lead to significant impairment of the hip joint and, ultimately, to THA in adolescence and adulthood. In addition, this review examines the current status of THA for patients with pediatric hip disorders, the appropriate timing and indications for THA, and outcomes and revision rates.
Material and methods
The co-authors of the current study met virtually twice for the selection of the most relevant pediatric hip pathologies for the study. After two rounds of discussion 2 weeks apart, the following pathologies (n = 12) were selected: LCPD, DDH, SCFE, neuromuscular hip dysplasia, post-traumatic avascular necrosis (AVN) of the proximal femur, juvenile rheumatoid arthritis (JRA), skeletal dysplasias, such as achondroplasia, spondyloepiphyseal dysplasia (SED), mucopolysaccharidosis (MPS), and mucolipidosis (ML), hip infections, and tumors.
Literature search
A literature search was conducted independently for each pathology using MEDLINE, PubMed, Web of Science, Embase, OVID, and Cochrane Library databases from the earliest date possible through June 2023. Retrospective, prospective, and longitudinal cohort studies were included in the review. Narrative and systemic reviews, editorials, and letters were also part of the review. The relevant studies were screened for eligibility for inclusion (Figure 1).
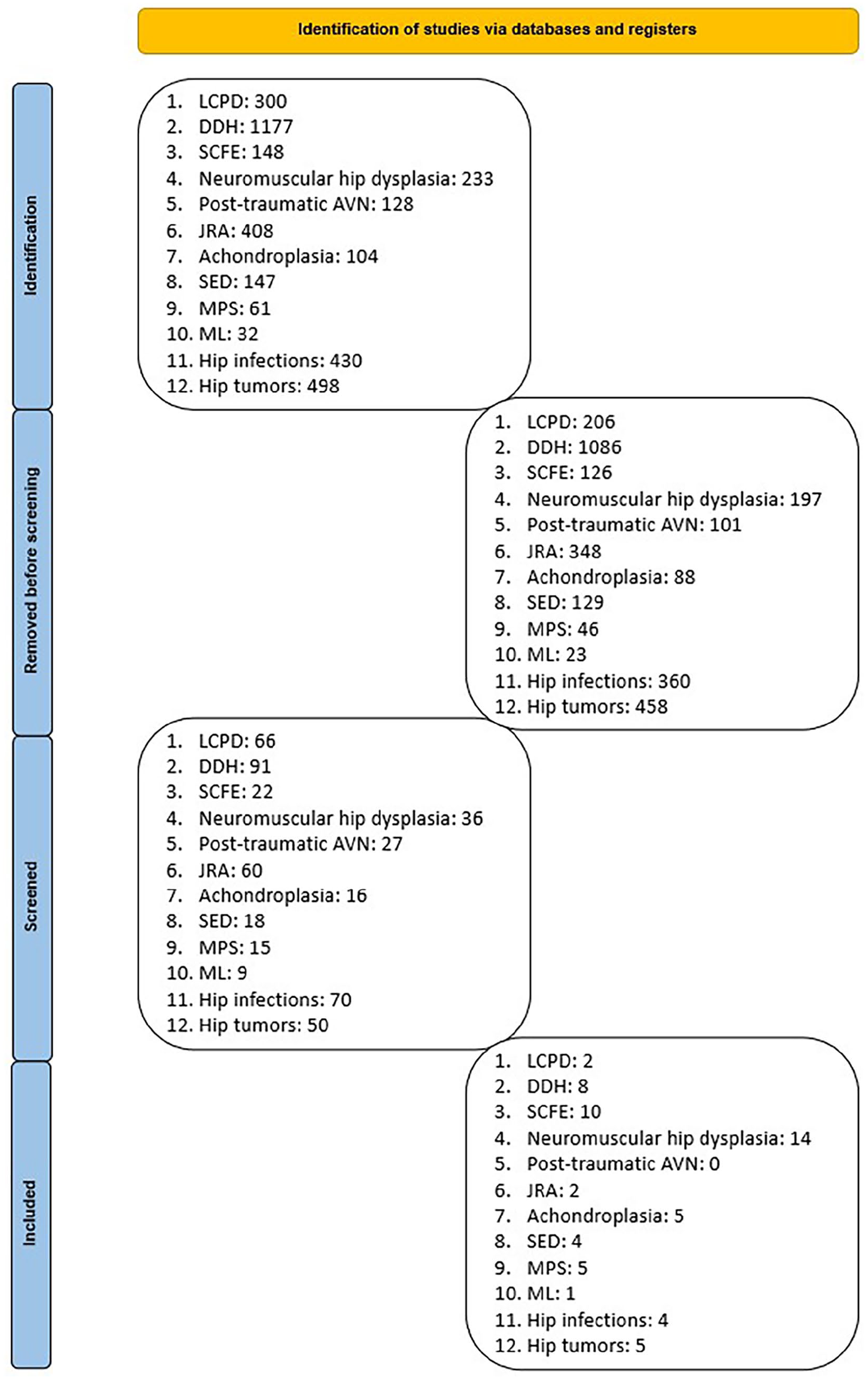
Search strategy.
In the study design, the authors had planned to include the chapter “Post-traumatic AVN of the proximal femoral epiphysis.” However, after searching the aforementioned databases for [post-traumatic OR traumatic] AND [AVN OR avascular necrosis OR osteonecrosis] AND [pediatric OR children] AND [THA OR THR OR total hip arthroplasty], no relevant articles were found. Therefore, the total number of pediatric hip disorders analyzed was reduced to 11.
Table 1 shows the bibliographic database search strategy (Table 1).
Hip disorders, keywords, and time frame of the literature review.
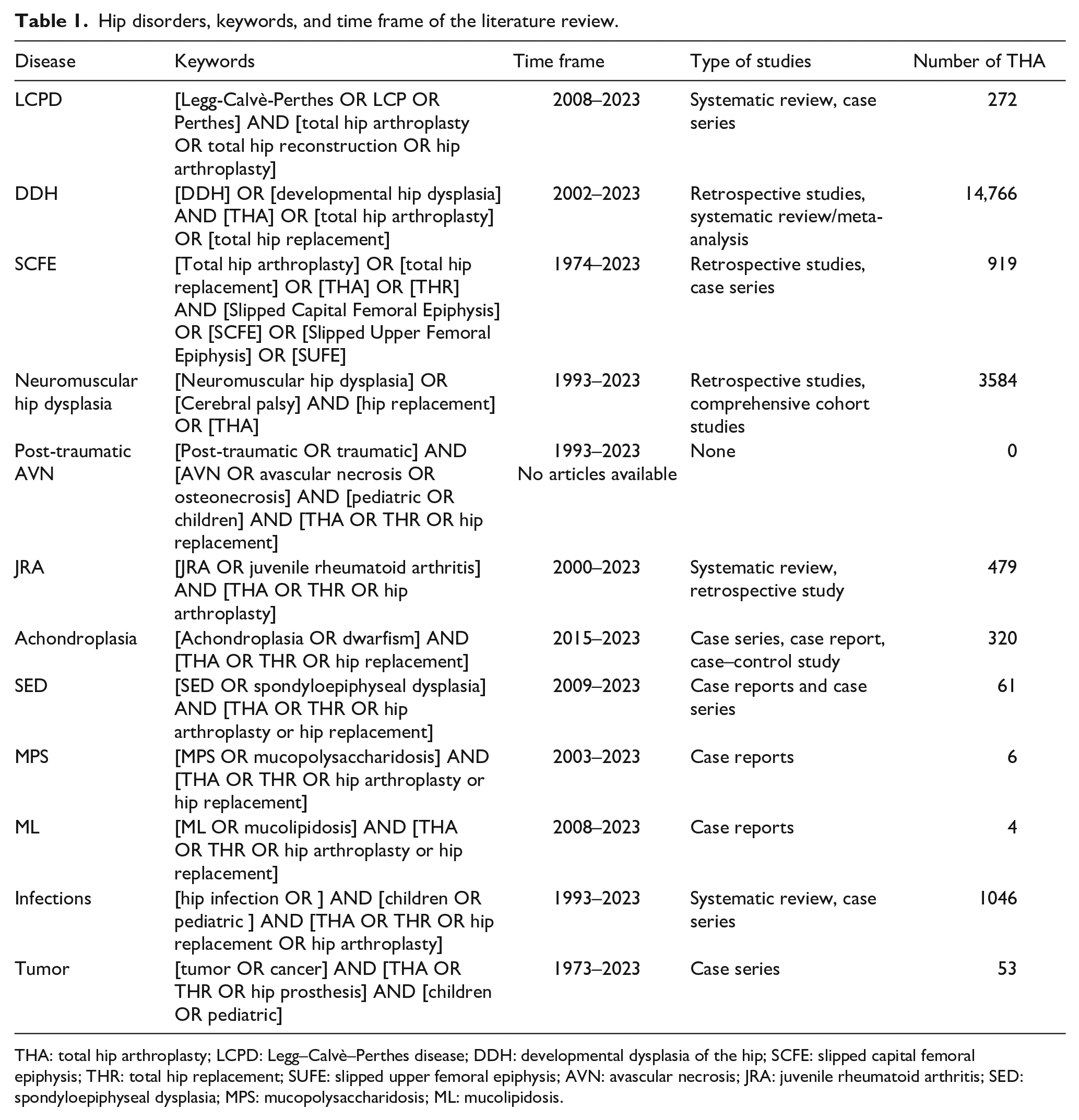
THA: total hip arthroplasty; LCPD: Legg–Calvè–Perthes disease; DDH: developmental dysplasia of the hip; SCFE: slipped capital femoral epiphysis; THR: total hip replacement; SUFE: slipped upper femoral epiphysis; AVN: avascular necrosis; JRA: juvenile rheumatoid arthritis; SED: spondyloepiphyseal dysplasia; MPS: mucopolysaccharidosis; ML: mucolipidosis.
Selection criteria
Studies met the inclusion criteria if they (1) included children with a confirmed diagnosis of 1 of the 12 pediatric hip pathologies included in this review that required THA; (2) presented a minimum follow-up of 16 months; (3) assessed outcome with a clinical or radiologic score; (4) had a Methodological Items for Non-Randomized Studies (MINORS) quality score of 9 or higher. 1
Exclusion criteria were as follows: (1) studies that did not meet the inclusion criteria; (2) studies with MINORS quality score less than 8 points.
Quality evaluation
The MINORS 12-item quality assessment checklist was used to assess the literature’s quality. The global ideal score is 16 points for noncomparative studies and 24 points for comparative studies. 1
Literature screening and data extraction
Screening (titles and abstracts) and data extraction were performed independently by four junior researchers (S.D.S., N.M., M.S., and R.S.). The full text of all the articles that were considered to be relevant after the initial title and abstract screening was retrieved and assessed for eligibility. The references of all selected articles were also reviewed to identify potential missed articles. Remaining ineligible articles were excluded and duplicate articles were removed. Disagreements were resolved by junior and senior authors’ (F.C., A.A., L.L., and A.D.) consensus. Analyses followed Preferred Reporting Items for Systematic Review and Meta-analysis (PRISMA) guidelines for a systematic review.2,3
For each pediatric orthopedic pathology, the extracted data included (1) the characteristics of selected literature, including first author, year of publication, nationality, number of patients, case collection time, and study type; (2) patient clinical characteristics, including age at diagnosis, gender, indication for surgery, age at THA, type of implant, surgical technique, outcomes, complications, and revision rate.
Results
Table 1 summarizes the keywords, time frame of the literature review, type of studies included, and number of THA according to the pediatric hip pathology (Table 1). The mean MINORS criteria of papers included in this review varied according to the pathology; the mean score was 14 ± 0.5 in LCPD, 12 ± 1 in DDH, 13 ± 1 in SCFE, 11 ± 1.5 in neuromuscular hip dysplasia, 12 ± 0.5 in JRA, 11 ± 0.5 in achondroplasia, 10 ± 0.5 in SED, 10 ± 0.5 in MPS, 12 ± 0.5 in osteoarticular infection of the hip, and 11 ± 0.5 in tumors; the MINOR score was not calculated for ML because only case reports were available for review.
LCPD
The authors found a systematic review published in 2017 covering this review’s topic and a meta-analysis published in 2023 focusing on THA incidence in LCPD patients.4,5 Successively, additional works were evaluated to cover other aspects of the THA treatment for a complete overview. 6 The search time frame was 15 years (2008–2023).
A total of 272 hips with LCPD underwent THA. The average age at the time of THA was 46.9 years. According to a recent study, the incidence of THA in LCPD patients is 6.8% after conservative treatment and 5.14% after surgical one. 6 Age, containment, and follow-up are the main features paving the way to a THA treatment.4,5
Cementless prostheses were used for 248 hips, hybrid for 22, and cemented for 2. The postoperative mean Harris hip score (HHS) was 90, with a mean difference in HHS before and after THA of 39 points.
Complications (n = 51; 18.8%) included 27 intraoperative fractures, 13 cases of aseptic loosening, 7 sciatic nerve palsies, and 4 heterotopic ossifications.
Revision surgery was required in 16 out of 272 hips (5.9%), with an average of 7.5 years from the original THA. The acetabular component was revised in nine hips, the femoral stem in six hips, and both components in one hip. The primary cause for revision was aseptic loosening in 13 cases, infection in 2 cases, and excessive lengthening resulting in nerve palsy in 1 case.
In general, THA in patients with LCPD has comparable functional results as patients with primary osteoarthritis (OA), although some studies have shown that they have a higher revision rate at midterm follow-up. 4 Moreover, these patients have an increased risk of sciatic nerve palsy, mainly related to having had antecedent surgical hip procedures4-6 (Figure 2).
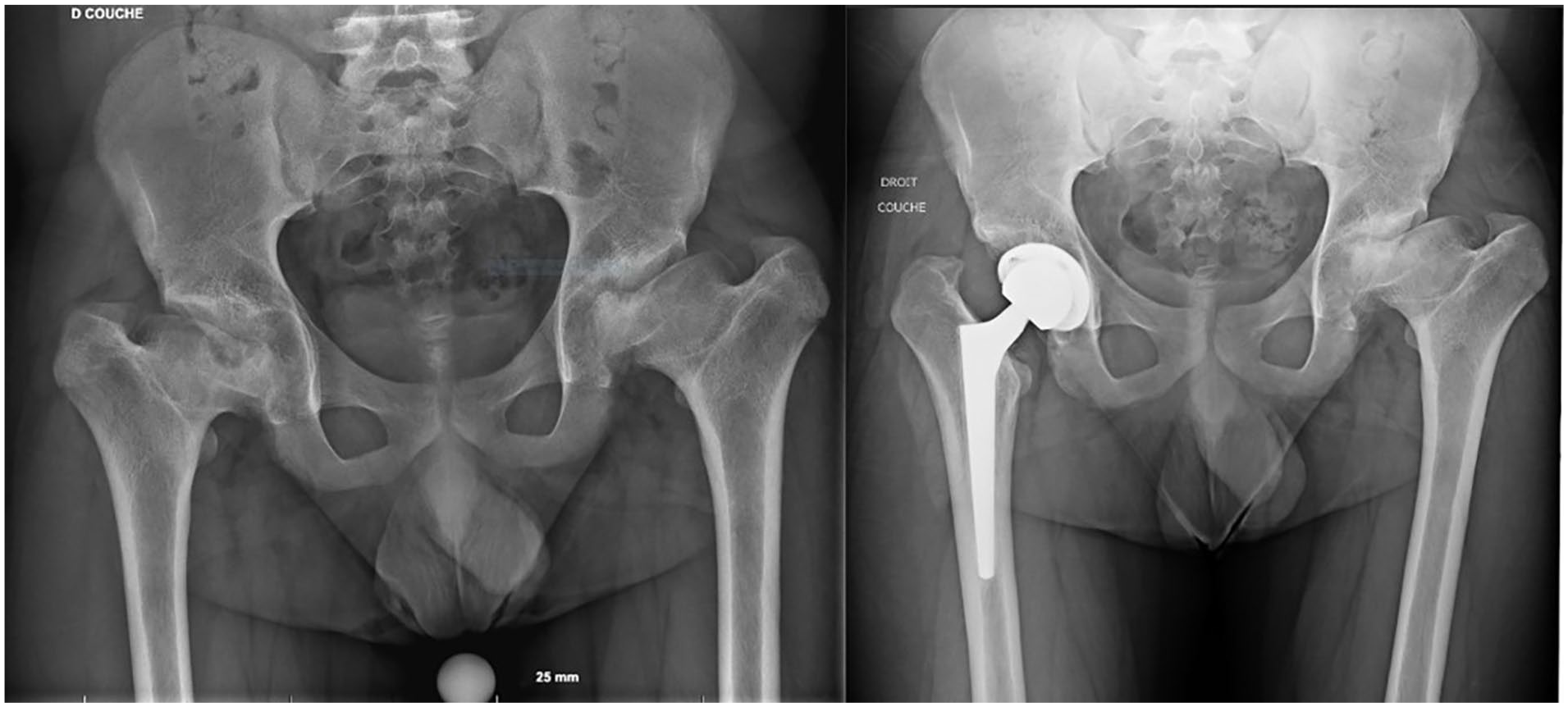
Male, 17 years old, with LCPD. THA performed at 19 years of age.
DDH
Four registry studies were reviewed, involving 14,766 hips that underwent THA due to a confirmed diagnosis of DDH7-10 (Table 2). The search time frame was 21 years (2002–2023).
DDH.
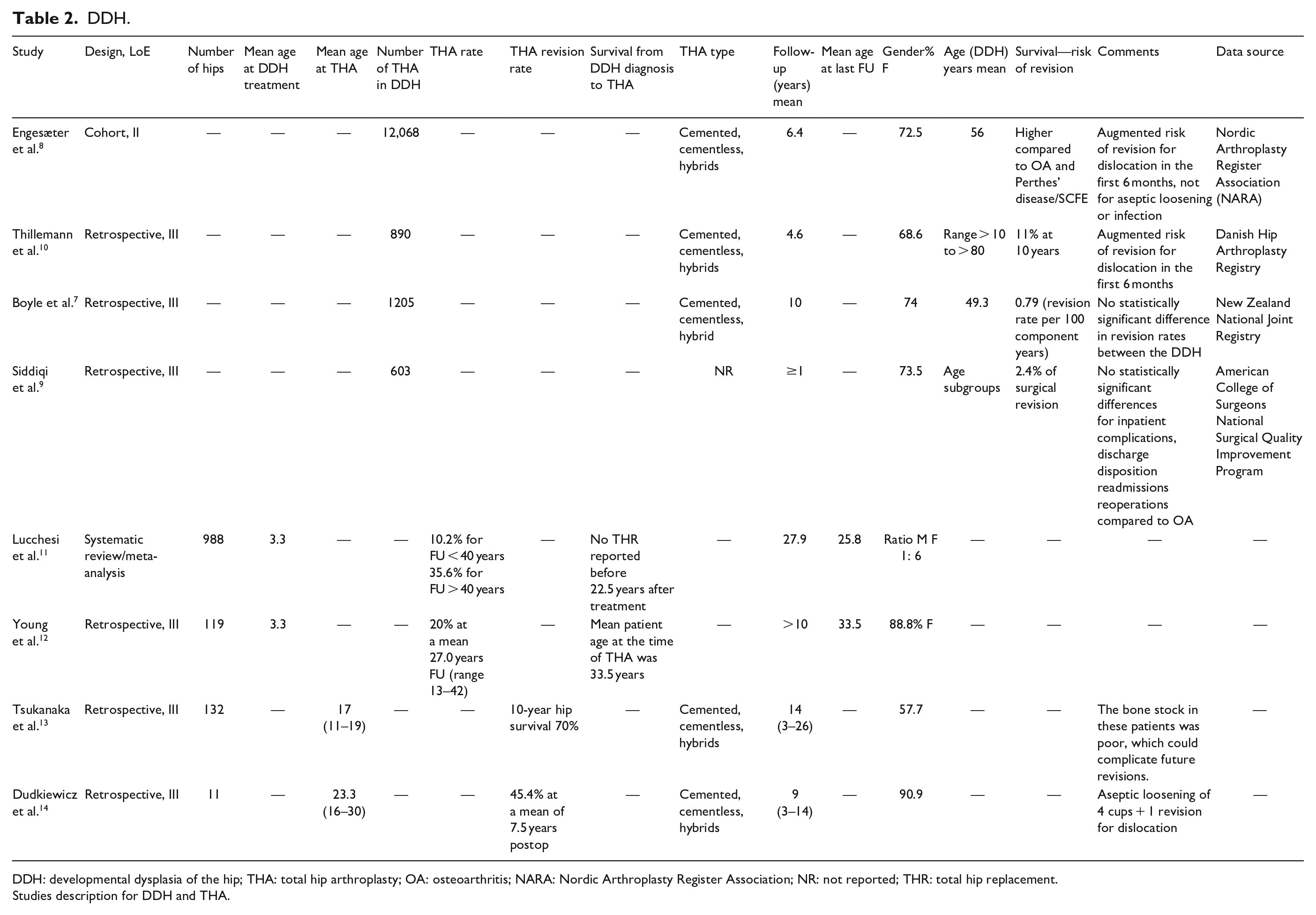
DDH: developmental dysplasia of the hip; THA: total hip arthroplasty; OA: osteoarthritis; NARA: Nordic Arthroplasty Register Association; NR: not reported; THR: total hip replacement.
Studies description for DDH and THA.
Lucchesi et al. 11 concluded, in a metanalysis of 988 hips diagnosed with DDH after the walking age, that signs of OA manifest on average three decades after the initial procedure. After surgery, there is a further deterioration during 30–40 years, and then the hip continues to deteriorate for 40–50 years after the initial treatment. Restoration of normal hip anatomy appears to be the most critical factor in preventing joint degradation. Another study conducted at the Mayo Clinic, which followed patients for at least 10 years, concluded that closed reduction treatment for DDH hips did not necessitate THA. 12 However, patients who received treatment after the age of 4 years exhibited lower hip survivorship rates at the 35-year mark, with a 50% survival rate compared to 85% for those treated before 4 years of age. These findings suggest that early referral and treatment may be advantageous for the longevity of hip function. Two studies revealed an increased risk of revision due to dislocation within the first 6 months after THA,8,10 while the other two studies found no statistically significant differences in inpatient complications, discharge disposition, readmissions, or reoperations compared to THA for OA.7,9
Femoral osteotomy was found to be correlated with subsequent THA11,12 (Table 2). Table 2 also presents two studies that investigated THA outcomes for DDH patients under the ages of 20 and 30 years, respectively13,14 (Table 2). The mean postoperative HHS for both groups was acceptable, with values of 83 and 90.6. However, a high revision rate of up to 45.4% (5 out of 11 patients) was reported with a mean follow-up of 7.5 years, while the THA survival rate was only 70% at 10 years. This underscores high revision rates and poor overall implant survival, predominantly associated with poor bone stocks resulting from previous DDH corrective surgeries and aseptic loosening (Figure 3).
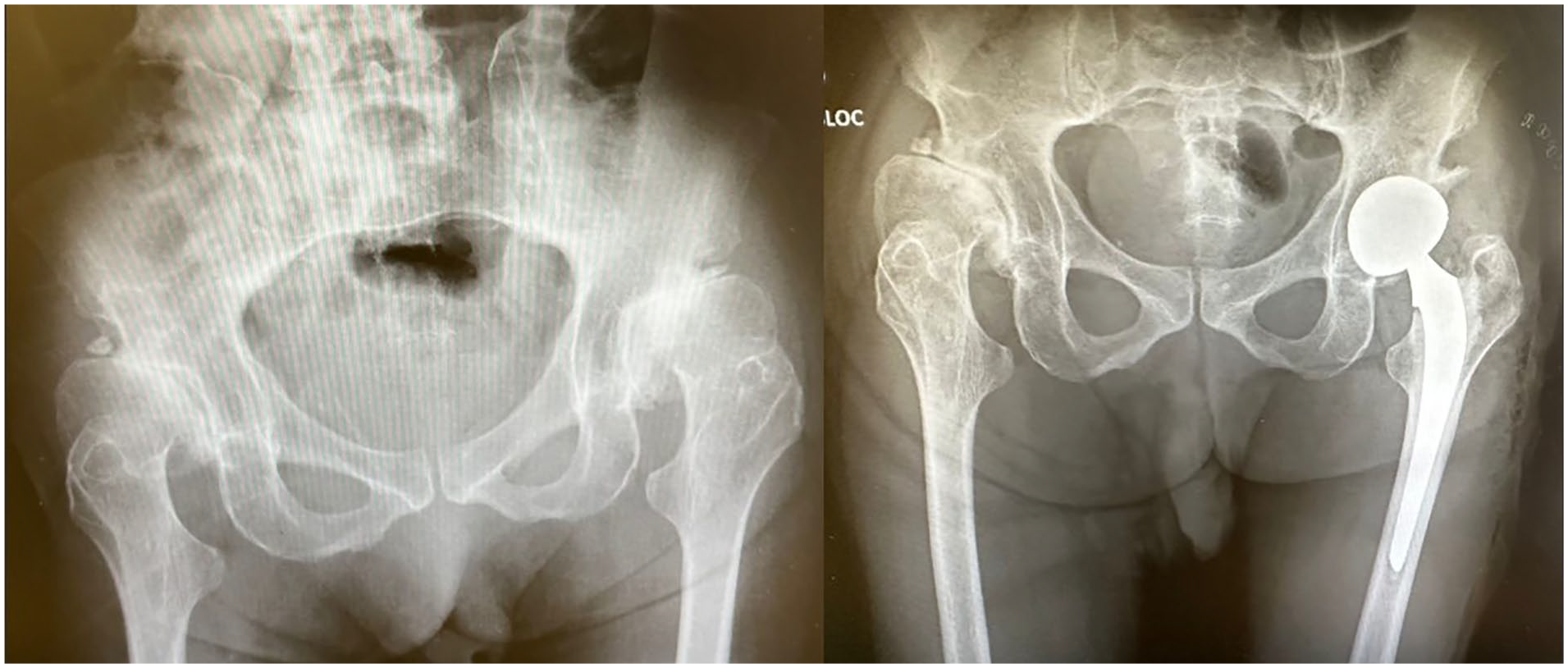
Male, diagnosed with DDH, THA performed at 39 years of age.
SCFE
Although the search time frame was 49 years (1974–2023), included studies were published between 2008 and 2018. They included six retrospective patient cohorts and four cohort studies from national registries.7,8,10,15-21 Overall, the studies examined 919 patients with a mean age of 41 years who received THA. Seven studies indicated a mean implant survival rate of 89% (range, 81%–100%) during a mean follow-up period of 9.4 (range, 4.4–15.2) years.
The most common reason for revision surgery was aseptic loosening, and uncemented implants were used most frequently (Table 3). Five studies reported an average increase in 41.5 points (range, 31.6–59) in the HHS among 120 patients. Lehmann et al. 17 discovered that SCFE had a significantly higher EQ-5D score than LCPD and DDH. Engesaeter et al. 8 found that the risk of revision for THA following childhood hip diseases was 1.4 times higher than for OA. However, Boyle et al. 22 found no disparities between the SFCE group and the OA group in terms of functional outcomes (Table 3; Figure 4).
Overview of the selected studies for SCFE and THA.
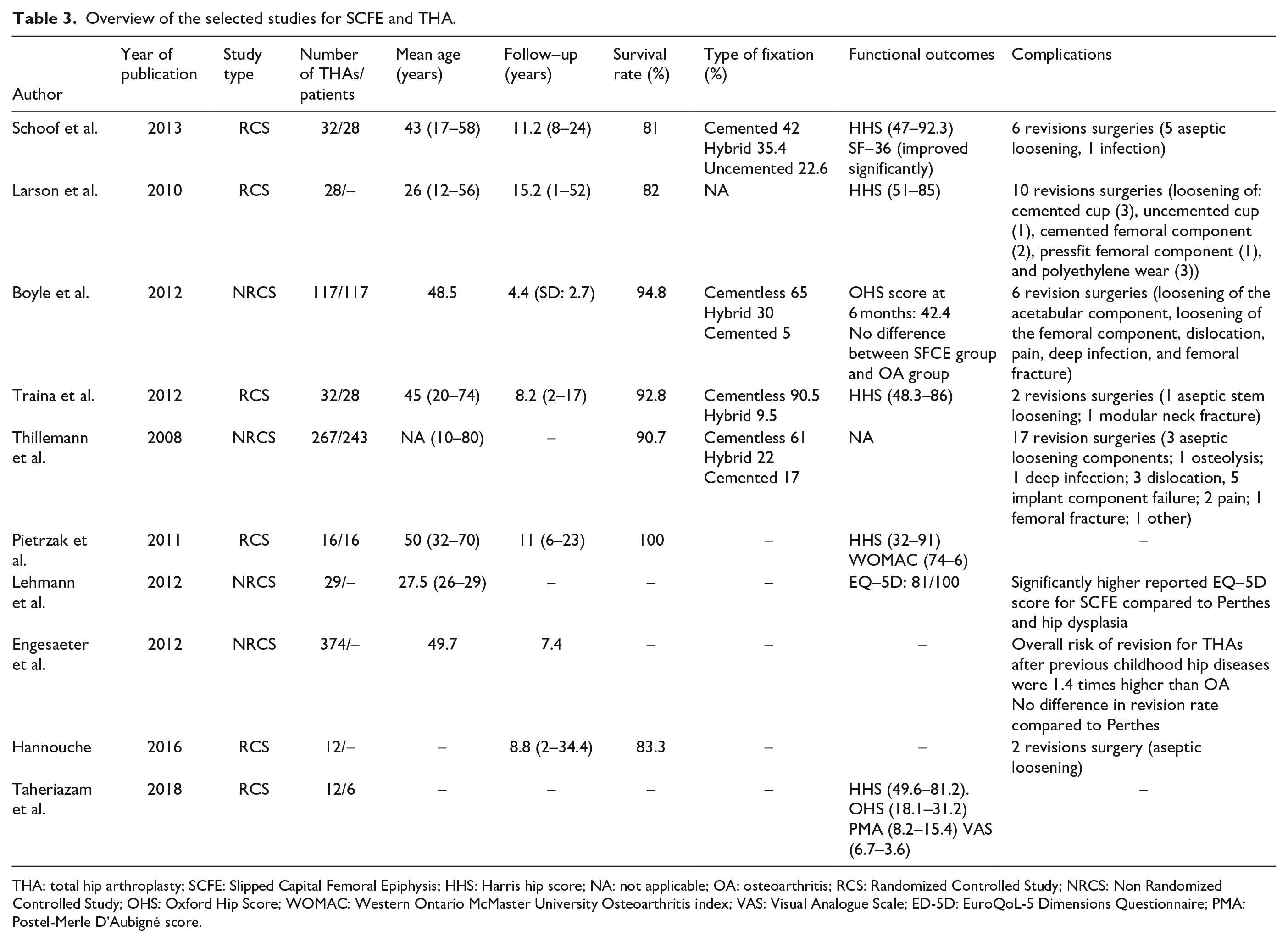
THA: total hip arthroplasty; SCFE: Slipped Capital Femoral Epiphysis; HHS: Harris hip score; NA: not applicable; OA: osteoarthritis; RCS: Randomized Controlled Study; NRCS: Non Randomized Controlled Study; OHS: Oxford Hip Score; WOMAC: Western Ontario McMaster University Osteoarthritis index; VAS: Visual Analogue Scale; ED-5D: EuroQoL-5 Dimensions Questionnaire; PMA: Postel-Merle D’Aubigné score.
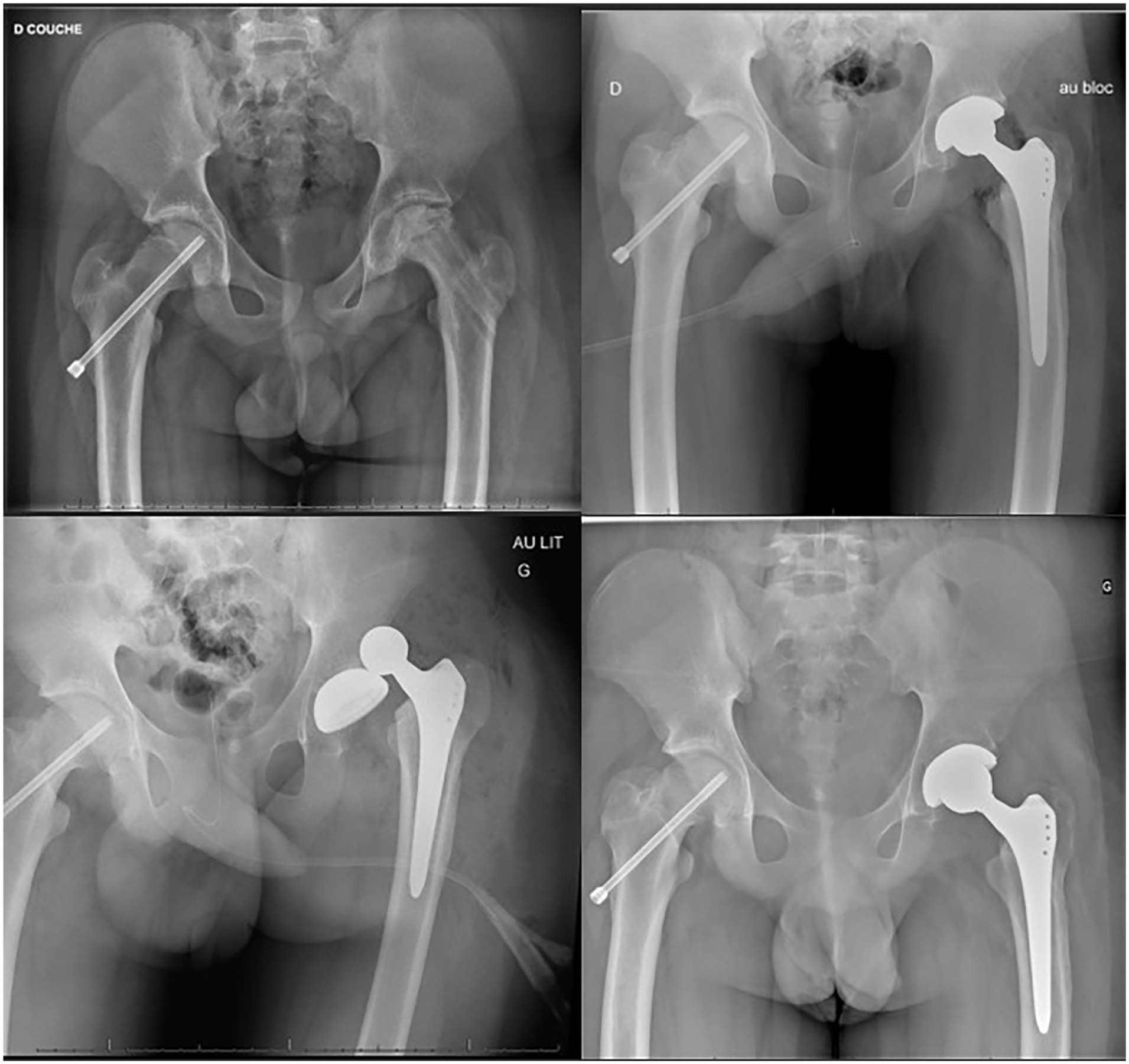
Male, 15 years old, affected by SCFE with AVN. Dislocation of the first THA implant at Day 1.
Neuromuscular hip dysplasia
The authors found 14 studies regarding neuromuscular hip dysplasia and THA.23–36 Going from 1993 to 2021, this selection mainly includes retrospective cohort studies.
The total number of THA is 3584. Mean age at THA was 51.6 years. Mean follow-up ranged from 2 to 12 years. At 10 years, the survival rate collected by three studies was of 83.8%.29,31,35 Implant preferences are specified in 11 studies,23,25,26,29–36 and 243 cemented, 231 uncemented, 85 hybrid, and 14 reverse hybrid. Two studies describe the use of hip resurfacing techniques,25,36 and one study describes the use of humeral stem in 12 hips 30 (Table 4).
Description of the studies used for neuromuscular hip dysplasia and THA.
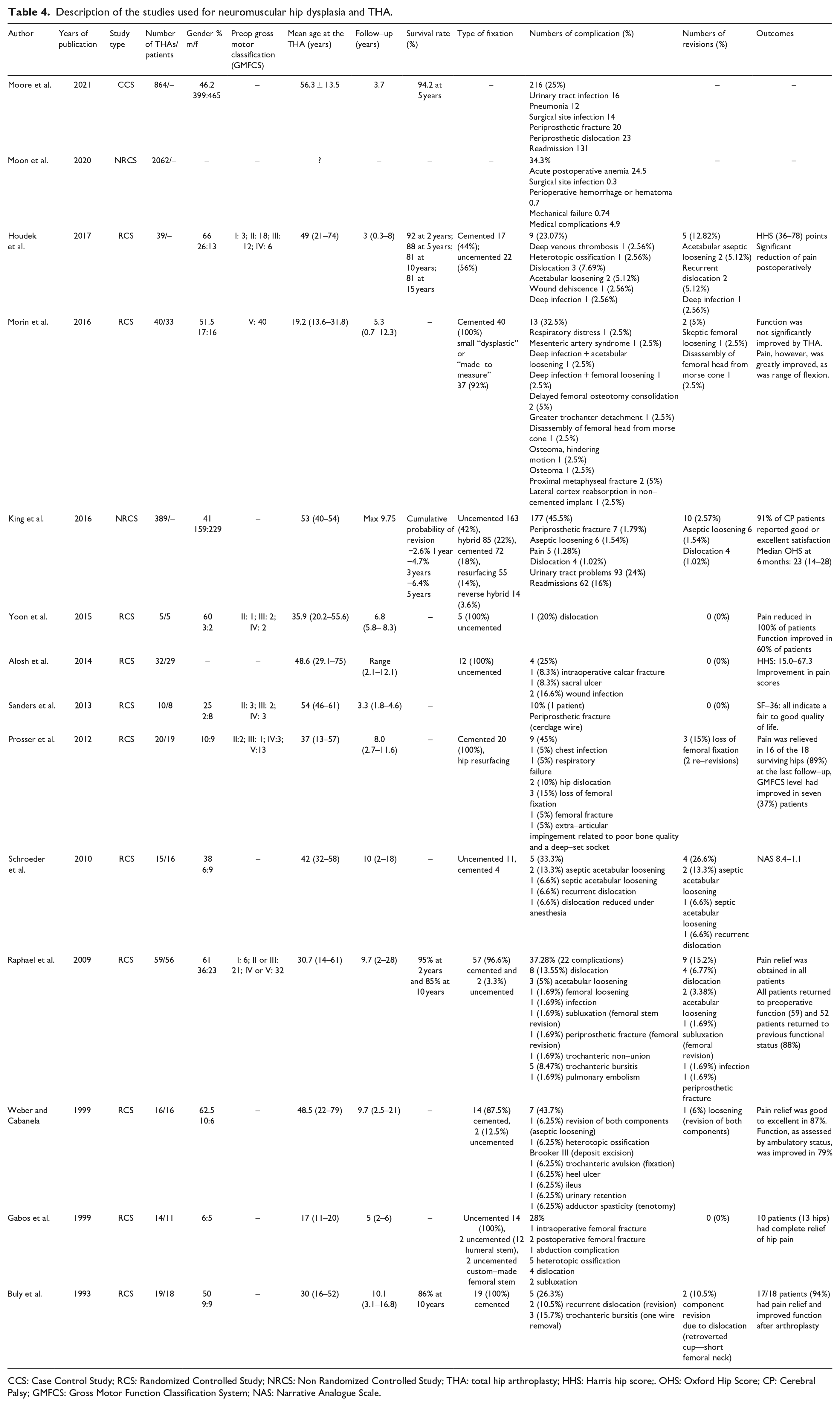
CCS: Case Control Study; RCS: Randomized Controlled Study; NRCS: Non Randomized Controlled Study; THA: total hip arthroplasty; HHS: Harris hip score;. OHS: Oxford Hip Score; CP: Cerebral Palsy; GMFCS: Gross Motor Function Classification System; NAS: Narrative Analogue Scale.
Complications rate goes from 10% to 45.5% with a mean of 30.6%. Frequent complications are urinary tract infections, surgical site infection, periprosthetic fractures, dislocations, and aseptic loosening. In all studies investigating the outcomes, those were satisfactory: pain relief was present in all cases, quality of life improved, and the majority of cases also function. One study reports no significant improvements in function 26 (Figure 5).
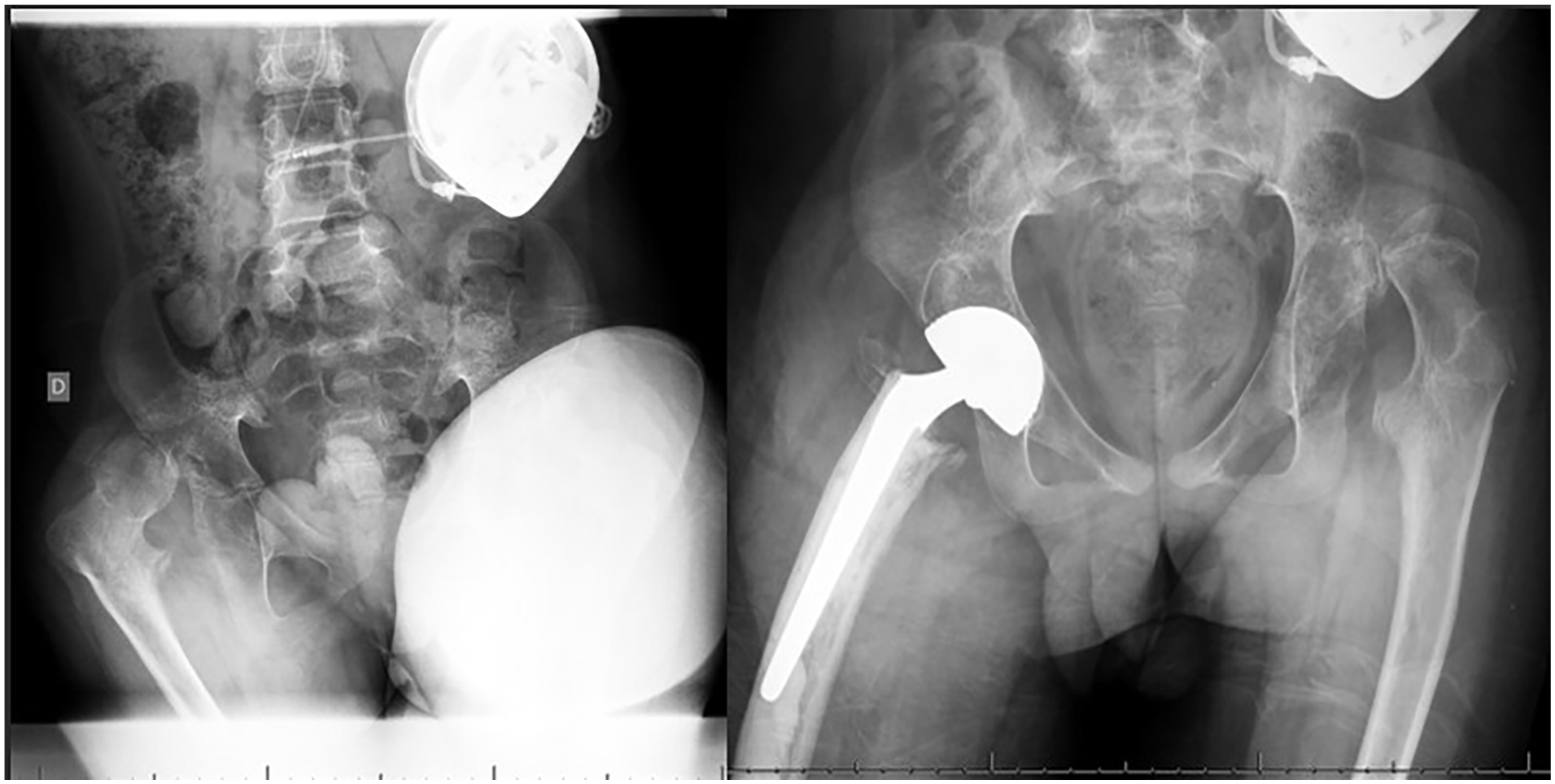
Male, 14 years old, affected by neurologic hip dislocation, previously treated with varus derotation shortening osteotomy.
JRA
Two studies examined 307 patients with a total of 479 hips over a period of 23 years (2000–2023).37,38 The mean age at THA ranged from 14 to 30 (range, 10–60) years. The mean follow-up period ranged from 16 months to 36 years. The direct lateral approach was the most frequently used (168 hips), followed by the posterior approach (116 hips). The most commonly implanted prosthesis type was uncemented (398 hips).
Nerve palsies and surgical site infections were the most frequently occurring complications, with five incidences each. The majority of revisions, making up 22% (92 hips), were necessary for the acetabular component, followed by the femoral part with 20 hips. Aseptic loosening was the most common cause of revision for the acetabular component. HHS was not conducted for all patients, but those who did undergo it demonstrated an improvement in quality of life, pain, and function37,38 (Figure 6).
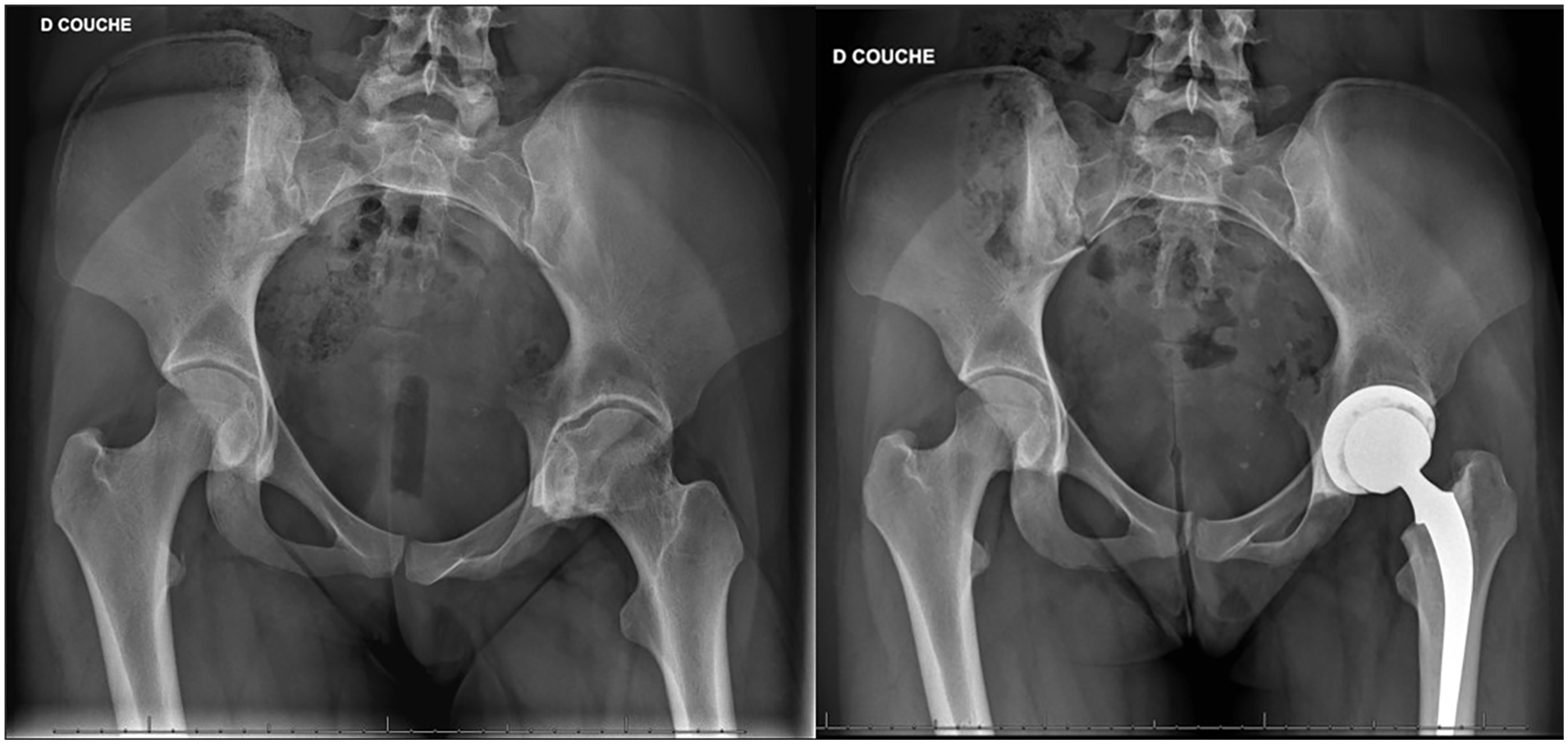
Female with JRA (laminar coxitis) and THA at 15 years of age.
Achondroplasia
One case series, one case report, and one case–control retrospective study were found;39–41 the search time frame was 8 years (2015–2023). A total of 320 THAs were performed in patients with achondroplasia, comprising 234 females and 67 males with a median age of 59.8 years at the time of THA. Of the 320 patients, 285 underwent THA due to a diagnosis of OA, 13 due to hip dysplasia, 8 due to dislocation, and 1 due to ankylosis. The average follow-up period was 6.2 years.
A retrospective case–control study conducted by Modi et al. 39 found no significant differences in operating time, 2-, 5-, 10-year survivorship, and revision rate (14/102, 13.7%) between patients with THA affected by achondroplasia and those without. In the group with achondroplasia, postoperative prosthetic fractures (5/102, 4.9%), mechanical wear and osteolysis of non-cross-linked polyethylene lines (4/102, 3.9%), periprosthetic joint infections (3/102), loosening of the femoral and acetabular components (1/102), and instability (1/102) were the reasons for revision. However, these results were not statistically significant compared to the control group. Statistically significant results were found in the analysis of radiographic values for head size, preoperative, and postoperative offset, all of which were higher in the control group. Moore et al. 40 examined postoperative complications within 90 days in patients with achondroplasia. They found no significant difference in the overall complication rate of 90 days after the surgery for individual complications, but did find a statistically significant difference in the rate of postoperative surgical site infections and readmission within the first 90 days after the surgery. Periprosthetic fractures and dislocations did not show any significant differences.
The study by Oba et al. 41 had the longest follow-up with a mean of 12.3 years, and all 68 hips underwent a cementless standard THA via a posterolateral approach. Patients were assessed using the Japanese Orthopedic Association score and exhibited a statistically significant mean difference of 38 points from pre-operation to post-operation. Complications included five dislocations, two of which required revision surgery, and one ceramic implant failure. The implant-associated survival rate was 94.1% at 10 years in this study.
SED
Studies on SED and THA are limited; the search time frame was 14 years (2009–2023).42,43 In SED’s studies, 35 patients (16 males, 19 females), with a mean age of 40 years at the time of THA, were included, of which 26 had bilateral THA, resulting in a total of 61 hips. The follow-up period was 9.5 years on average.
In Wyles et al.’s 42 study, prior to THA, 12 hips out of 50 had undergone at least one other surgical procedure (24%), such as femoral or pelvic osteotomy, or interposition arthroplasty. The posterolateral approach was used for 23 patients, transtrochanteric for 16, and anterolateral for 11. THA was performed with uncemented components in 32 cases, hybrid components in 11 cases, cemented components in 6 cases, and reverse hybrid components in 1 case. With regard to the bearing surface, metal on polyethylene was used for 40 hips, while ceramic-on-polyethylene was used for 6 and ceramic-on-ceramic was used for 4. Out of a total of 46 polyethylene prosthesis, 24 were high-cross-linked polyethylene and 22 were conventional polyethylene. The mean survival rates at 5, 10, and 20 years were 96%, 85%, and 55%, respectively.
There were 13 revisions in this cohort (26%): in 12 cases, conventional polyethylene was used as the bearing surface. The indication for revision surgery was polyethylene wear in six cases (two uncemented and four hybrid), aseptic loosening in five cases, periprosthetic fracture in three cases, and infection in one case. Specifically, there were three revisions for both the acetabular and femoral implant, three for the acetabular implant, five for the femoral implant, and seven for the head and liner. The mean preoperative HHS was 47, and postoperative was 87; 64% of the population used gait aid before the surgery, which decreased to 34% after the surgery.
MPS
THA and MPS works found are mainly case reports; the search time frame was 20 years (2003–2023). We have analyzed only articles on MPS type I and IV, for the scarcity of literature on the other types of MPS, mainly due to the reduced average lifespan of these patients.
Four case reports on MPS and THA were found through literature analysis,44–47 describing a total of six THA (four patients; two males, two females; and two bilateral THA). Of these individuals, one had MPS type I, one had type IV-A, and two had type IV-B. The median age at THA was 20.5 years. The indication for THA was the early onset of OA, pain, and decreased mobility. The surgical approach was posterior in three patients (one unspecified). There were three prostheses without cement, one computer navigation-aided case, and one cemented. The average follow-up was 4 years, with a range of 7 months to 8 years. All studies reported improved pain and range of motion post-surgery.
ML
The literature regarding ML and THA is poor and focuses on ML-III, since the lifespan of patients affected by other ML’s types is generally not long enough for a THA. The search time frame was 15 years (2008–2023).
A case report was found through literature analysis, describing a total of four THA (two bilateral). 46 The patients in both cases were affected by ML-III, with a mean age of 18 years at implantation, and received cemented implants. The mean follow-up period was 4 years. Both patients reported experiencing less pain compared to before surgery and improved mobility, despite one having a periprosthetic fracture and still exhibiting a Trendelenburg gait.
Osteoarticular infections
A 2021 systematic review examined the outcomes of THA in patients treated for osteoarticular infection.48–51 The authors conducted a literature search (1993–2023) that included recent studies and analyzed various aspects of the topic.
A total of 1046 patients with hip infections during childhood requiring a THA in adulthood were identified in the literature. The mean age at THA was 45 years. According to a study, 50 the average age at infection was 9.6 years, while another study reported 7.1 years. 48 However, the age at infection varied between 1 and 11 years.
Post-THA follow-up ranged from 2 to 38 years, depending on the study.48,49 The most common pathogens that caused the infections were
In cases of high dislocation after childhood infection, it has been observed that THA with subtrochanteric shortening osteotomy can reduce the lower limb discrepancy, ameliorate overall HHS, decrease pain, and improve the range of movement. Moreover, this technique has shown better results if paired with the fixation of autogenous cortical bone struts, which may enhance the union osteotomy site.
The HHS was significantly higher post THA in all studies, with a mean improvement of 40.7 points. Complications observed included sciatic nerve palsy, perioperative prosthetic fractures, infection, femoral nerve injury, and dislocation, among others. Sciatic nerve palsy is the most common complication, with 30 reported cases (30/1046; 2.9%); however, in the majority of cases (24), it was transient, with an 80% resolution rate. Post-hip arthroplasty infection rates are generally not higher in these patients, with a rate of 1%. When comparing studies conducted in the past 10 years, the rate decreases to 0.7%, except when THA is performed within 10 years of the initial hip infection. 49
The revision rate varies widely across different studies, ranging from 6.6% (with a mean follow-up of 6.1 years) to 54% (with a mean follow-up of 31.5 years). It was observed that longer follow-up times correlated with higher rates of revision.
Tumors
Five studies were identified through literature search and could be included in the present review; the search time frame was 50 years (1973–2023).52–54 One of them focused on corticosteroid-induced osteonecrosis for hematologic malignancies while the others focused on solid tumors around the hip. 55
The collected data for solid tumors around the hip include 53 patients with a mean age of 12.5 years at prosthesis. The most common tumor types were Ewing sarcoma (28), Osteosarcoma (21), malignant peripheral nerve sheath tumor (2), and Garthan disease (1). Depending on the extent of the lesion, the type of prosthesis and replacement structure were adjusted accordingly.
VanKampen et al. 54 reported that 36 patients received proximal femoral replacement, with 24 fixed and 12 extendable implants, and 4 patients underwent total femoral replacement, with 2 extendable implants. There was an observed change in the hip joint reconstruction components: from cemented high-density polyethylene acetabular cups to unipolar hemiarthroplasty components, to uncemented acetabular components. Out of the 27 patients who received a cemented acetabular component, 15 were alive during the follow-up period (6–15 years old). Of the 10 patients who underwent unipolar hemiarthroplasty, 6 survived. All three patients treated with an uncemented acetabular component died from metastases. However, 14 patients received extendable prostheses, with 5 of them dying. The remaining patients had between 1 and 24 lengthenings, up to a maximum of 150 mm, and no acute dislocations were observed after lengthening. Moreover, 10 patients underwent surgery with an acetabular cemented component before the age of 11 years. Acetabular growth and development caused verticalization of cups, resulting in loosening of the acetabular component. Among the seven surviving patients, four required acetabular revisions between 4 and 9 years after the implantation. Only one patient older than 11 years of age needed a revision for mechanical loosening 5 years after implantation. Two dislocations were treated without requiring revision surgery, while three cases of deep infection occurred, with one resulting in amputation. At the latest follow-up, which had a mean duration of 11.2 years, 13 of the 40 patients required revision surgery, with 3 undergoing amputation (1 of which resulted from a revision surgery), while 16 patients passed away.
In their study, Schwameis et al. 53 used hip replacement in 12 patients with malignant pelvis bone tumors. However, 10 patients underwent endoprosthesis surgery with pelvic reconstruction, using a cementless stem and proximal or total femur replacement with Brunswick acetabular cups. An additional two patients received an allograft and prosthesis with a cementless femoral component. Wound healing was the most frequent complication, occurring in 12 patients, followed by infection in 4 patients, transient nerve palsy in 4 patients, hematoma in 2 patients, and 1 case each of loosening, implant wear, periarticular ossification, and stem subsidence. Another cause of THA related to tumors could be osteonecrosis induced by corticosteroids for hematologic malignancies. In their study, Bernhard et al. 56 gathered data from 27 patients who had undergone 41 THAs for this reason, with a minimum follow-up of 5 years. Of these hips, 33 had undergone prior core decompression. THA was offered only to skeletally mature patients in the study, and all surgeries were performed via a posterior approach with cementless components. The authors of this study emphasize a temporal shift observed in implant choices, moving from modular stems to smaller options and from ceramic-on-ceramic and metal-on-metal to polyethylene on ceramics.
In patients with hematologic malignancy, described by Bernhard et al., 56 the median age at diagnosis was 14.9 years, and most cases were diagnosed with acute lymphoblastic leukemia. All hips underwent conservative treatment (including intra-articular injections, core decompression, and femoral head resurfacing) at a median age of 19.77 years prior to THA. All the implants used were pressfit. Complications included one nondisplaced calcar fracture during surgery, one dislocation 6 days after surgery, and one revision for a cracked ceramic liner 21 months post-operation. Significant improvements were observed in range of motion, pain, and daily activities. 56
Discussion
Based on the review’s findings, it is important to note that the majority of pediatric hip disorders typically require THA between the ages of 50 and 70 years, with the exception of skeletal dysplasias and tumors that often require THA before the age of 40 years and within the first two decades of life, respectively (Table 5). Furthermore, tumors are often found in younger individuals undergoing THA, while those diagnosed with DDH after walking age experience a higher rate of THA revisions and shorter prosthetic longevity. Among the conditions necessitating custom prostheses, tumors are the most common, followed by skeletal dysplasias and JRA. However, it should be noted that the substantial variability among the studies included and the prevalence of multiple case reports present a challenge in reasonably comparing THA rates and outcomes for distinct pediatric hip pathologies.
Summary of the main findings on THA for each disease.
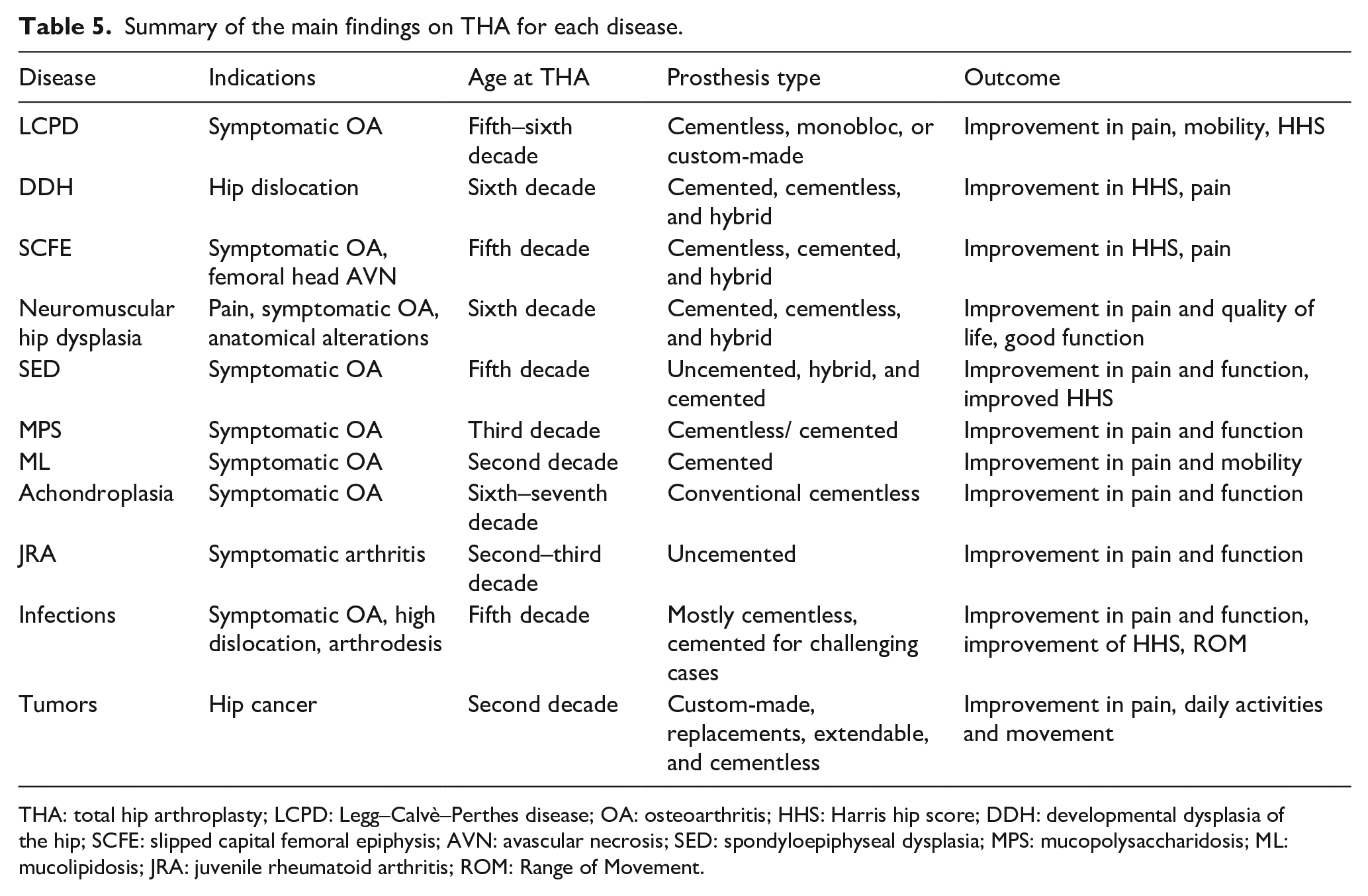
THA: total hip arthroplasty; LCPD: Legg–Calvè–Perthes disease; OA: osteoarthritis; HHS: Harris hip score; DDH: developmental dysplasia of the hip; SCFE: slipped capital femoral epiphysis; AVN: avascular necrosis; SED: spondyloepiphyseal dysplasia; MPS: mucopolysaccharidosis; ML: mucolipidosis; JRA: juvenile rheumatoid arthritis; ROM: Range of Movement.
Overall, most THAs are performed between the ages of 50 and 70 years. However, age at THA varies significantly among different pathologies due to the varying development of OA. OA generally develops earlier in patients with DDH and later in patients with achondroplasia. Patients with DDH and LCPD may experience initial OA symptoms in their second or third decade of life, with a THA needed in their fifth or sixth decade. Hip disruptions in patients with skeletal dysplasia are commonly attributed to irregular skeletal growth or abnormal load distribution in the hip joint. Surgical intervention is typically necessary once OA becomes symptomatic during early adulthood. Consequently, skeletal dysplasias (excluding achondroplasia) typically require THA at an earlier age (patients below 40 years of age) than the general population (> 70 years) or other pediatric hip pathologies (50–70 years). In particular, the average age for THA in patients with SED is 40 years. Similarly, children who undergo treatment for hip osteoarticular infection may develop severe hip OA, hip dislocation, and ankylosis, resulting in the need for THA at a younger age. Typically, THA is performed during the fourth or fifth decade of life in these patients. However, there have been rare instances of DDH observed in patients with skeletal dysplasias, particularly achondroplasia, where no THA procedures were performed during childhood.
It is important to note that childhood treatment can impact the timing of THA. For example, conservative treatment for LCPD in children older than seven increases the likelihood of THA. 6 In addition, the risk of THA increases over time, with a higher incidence observed after 40 years of follow-up in patients diagnosed with DDH, LCPD, SCFE, and achondroplasia. Nonetheless, the majority of pediatric patients with hip pathology typically undergo osteotomies during childhood and adolescence for femoral head alignment and coverage of the acetabular roof. This procedure aims to reduce potential hip joint degeneration and limit the risk of future THA. These procedures delay THA’s timing and alter the joint’s anatomy, potentially rendering THA surgery more complex. Custom-made implants may be necessary for significant anatomical changes. In particular, there is promising research for monobloc cementless femoral stems in patients with LCPD.57,58 Severe femoral dysplasia may require a custom-made femoral design or conical modular implants, as they appear to be associated with a lower incidence of intraoperative fractures. 59 Hybrid implants have been used in cases of severe anatomical deformities resulting from hip joint infections.
The choice of implant depends on the pediatric orthopedic condition. Specifically, patients with tumors, skeletal dysplasias, and JRA have higher rates of custom-made implants. In addition, the osteopenic nature of bone in MPS patients favors the use of cementless implants over cemented ones. 60 However, the ongoing debate regarding the type of THA necessitates consideration. These populations exhibit a high prevalence of osteopenia. Consequently, despite longevity concerns, a cemented prosthesis may be a viable option for certain patients. THA in patients with pediatric hip pathology is linked to a greater risk of complications compared to the general population. These complications include periprosthetic intraoperative fracture, femoral and sciatic nerve palsy, infection, heterotopic ossification, and dislocations, irrespective of the initial pathology. It is crucial to differentiate between patients receiving THA for tumors and those without, as the prognosis is heavily influenced by factors, such as histology, grading, and disease staging. Regardless of the quality of the implant, however, these patients have a higher rate of implant infection and a higher overall readmission rate in the first 90 days postoperatively. 61 Patients with SED, MPS, ML, and a history of osteoarticular infections have a higher likelihood of perioperative complications, increased risk of postoperative infections, and elevated rate of perioperative bleeding. While orthopedic pathologies inherently pose difficulties, surgical time does not significantly vary across different categories, except for cases involving tumors, which require a longer duration of surgery.
Despite the heterogeneous nature of orthopedic conditions and the high risk of complications, THA improves hip function, reduces hip pain, and enhances the quality of life for all patients, irrespective of their initial diagnosis. However, the revision rate is significant across groups, primarily due to aseptic loosening.
Conclusion
Overall, THA is primarily recommended for pediatric orthopedic patients suffering from hip joint pathology that induces OA and abnormal anatomy, leading to early hip joint degeneration. THA effectively improves the quality of life by reducing pain and improving function. The incidence of revision and complications linked to total hip replacement surgery is higher than that of the general population and correlates with the patient’s age at the surgery’s implantation. Irrespective of the initial diagnosis, patients undergoing THA at a younger age are at a greater risk of developing complications.
Supplemental Material
sj-pdf-1-cho-10.1177_18632521241229608 – Supplemental material for Total hip arthroplasty in patients with common pediatric hip orthopedic pathology
Supplemental material, sj-pdf-1-cho-10.1177_18632521241229608 for Total hip arthroplasty in patients with common pediatric hip orthopedic pathology by Sara De Salvo, Ricardo Sacco, Nicolas Mainard, Ludovico Lucenti, Marco Sapienza, Alain Dimeglio, Antonio Andreacchio and Federico Canavese in Journal of Children’s Orthopaedics
Footnotes
Author contributions
F.C., A.A., and A.D. contributed to study design. All authors performed data collection. F.C., S.D.S., N.M., and R.S. conducted data analysis. S.D.S., R.S., N.M., M.S., and F.C. participated in article preparation. A.A., A.D., L.L., and F.C. provided critical review of the article. F.C. involved in supervision. All authors provided approval of the final version to be published.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical statement
Approval by the Institutional Review Board (IRB) is not required as it is a literature review with no patient contact.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
