Abstract
Purpose:
The purpose of this study is to develop an accessible step-wise management algorithm for the management of pediatric spinal osteoid osteomas (OOs) based on a systematic review of the published literature regarding the diagnostic evaluation, treatment, and outcomes following surgical resection.
Methods:
A systematic review of the literature was conducted on PubMed to locate English language studies reporting on the management of pediatric spinal OOs. Data extraction of clinical presentation, management strategies and imaging, and treatment outcomes were performed.
Results:
Ten studies reporting on 85 patients under the age of 18 years presenting with OOs were identified. Back pain was the most common presenting symptom, and scoliosis was described in 8 out of 10 studies, and radicular pain in 7 out of 10 studies. Diagnostic, intraoperative, and postoperative assessment included radiographs, computed tomography (CT) scans, magnetic resonance imaging (MRI), bone scans, and frozen section. Treatment options varied, including conservative management, open surgical resection with or without intraoperative imaging, and percutaneous image-guided treatment. All included studies described partial or complete resolution of pain in the immediate postoperative period.
Conclusions:
The proposed algorithm provides a suggested framework for management of pediatric spinal OOs based on the available evidence (levels of evidence: 3, 4). This review of the literature indicated that a step-wise approach should be utilized in the management of pediatric spinal OOs.
Introduction
Osteoid osteomas (OOs) are benign bone tumors that usually affect long bones and account for 2–3% of all primary bone tumors.1 –4 However, 6–20% of OOs can also occur in the spine,2,5 –7 where they may cause significant deformity, pain, and disability. When presenting in childhood, untreated spinal OOs, or those treated in a delayed fashion, may cause persistent disability to patients.8 –10 Spinal OOs are relatively rare, and they can be especially challenging to manage due to their location and proximity to vital structures.
Various management options for spinal OOs have been proposed, including observation, nonsteroidal anti-inflammatory drugs (NSAIDs), open surgical resection (OSR), minimally invasive surgery (MIS), and percutaneous imaging-guided treatment. The optimal treatment for a given patient depends on a range of factors, such as tumor location and size, patient age and overall health, and severity of symptoms, among other considerations.
Despite the availability of these treatment options, there is currently no established management algorithm for pediatric spinal OO. This lack of consensus can create uncertainty and variability in the management of spinal OO, potentially leading to suboptimal outcomes. To address this gap in knowledge, a systematic review may be used to aggregate and synthesize the available evidence on the treatment of pediatric spinal OOs and develop a management algorithm that can help guide clinical decision-making.
The primary objective of this systematic review is to identify and evaluate the presentation, diagnostic evaluation, treatment, and outcomes following spinal OOs in the pediatric population. Through an overview of the current literature, we hope to introduce a management algorithm to improve the care of pediatric patients with spinal OO, while additionally identifying gaps in the literature and suggesting avenues for future research.
Methods
Search strategy
A systematic review was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) research methods and reporting guidelines. A digital search of the online medical literature database, MEDLINE (PubMed) was done between August 1, 2022, and August 9, 2022. The search strategy included the following keyword terms: “pediatric” or “adolescent” or “child,” and “osteoid osteoma,” and “spine.” All potential studies were stored in Papers (www.papersapp.com), an open-source software program used for bibliographic citation management.
Study selection
We assessed all studies that were retrieved by the following search process. Studies were initially screened by title and abstract. A full-text review was conducted for the remaining studies, selecting published literature that described therapeutic measures for pediatric OOs of the spine. Studies were excluded for the following reasons: (a) studies involving adult-only cases; (b) studies that included a mixture of pediatric and adult cases but that did not distinguish the data extracted from pediatric and adult patients; (c) case series with fewer than five pediatric patients; (d) case series without appropriate postoperative follow-up, defined as at least 6 months on average; (e) studies involving cadaveric specimens; and (f) studies published in languages other than English were excluded.
Data extraction
The following information was extracted from each eligible study: first author name; year of publication; years of included cases; number of patients per study; mean age; procedure type; presenting symptoms; lesion location; duration of symptoms prior to treatment; preoperative imaging; intraoperative imaging; postoperative imaging; imaging characteristics; postoperative status; follow-up period; follow-up imaging; rate of recurrence; nonrecurrence complications; and rates of additional procedures. For studies that did not clearly delineate the described categories above, we left the associated data unrecorded.
Results
Search results
Our search strategy found 251 studies for screening. After title and abstract screening, 84 studies underwent full-text review. Ten studies qualified for this study after exclusion per the criteria described above (Figure 1).
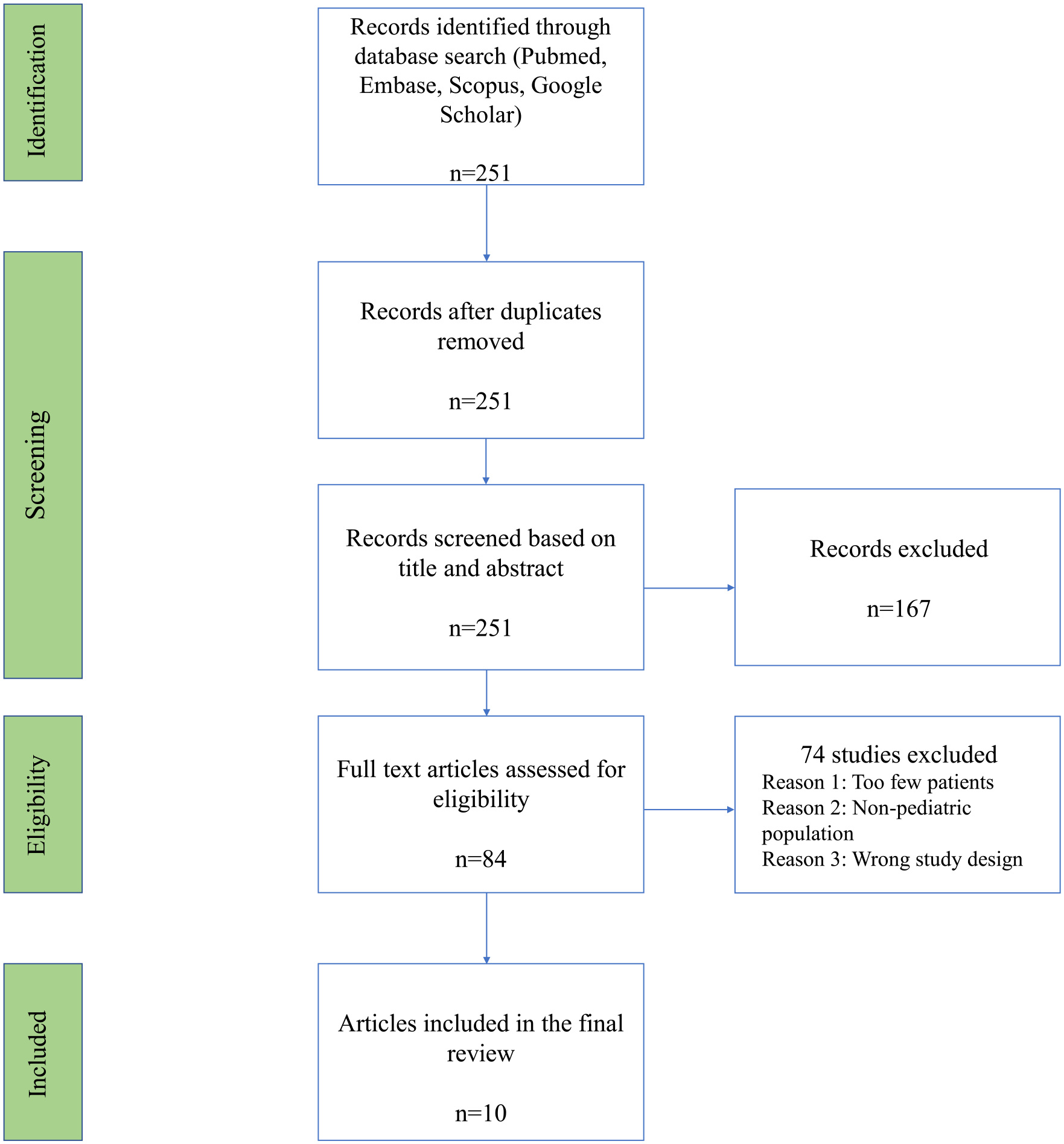
PRISMA flow diagram detailing the results of the database search and exclusion process.
Study characteristics
Among the ten studies that qualified for this review, seven were case series (level of evidence 11 : 4),4,8,12 –16 and three were retrospective cohort studies (level of evidence: 3).17 –19 In aggregate, these studies included 85 pediatric patients under the age of 18 with spinal OO. The results are presented in chronological order to describe the evolution in the evaluation of OO.
Diagnosis and workup
Clinical presentation
A complete set of data describing clinical presentation, including number of patients per study, mean age, a complete list of presenting symptoms, and duration of symptoms prior to treatment are listed in Table 1. The mean age among included studies ranged from 10.5 to 13.0 years. Among these studies, all described back pain as a presenting symptom.4,8,12 –19 Eight out of ten studies described scoliosis.4,8,12 –15,17,18 Seven out of ten studies described radicular pain.4,12 –14,16,18,19 Other symptoms included back pain that worsens at night, back pain that is relieved by NSAIDs, neck pain, neck stiffness, decreased range of motion (ROM) of the neck and spine, pathologic spinal fracture, and torticollis.
Summary of presenting characteristics, including average patient age, presenting symptoms, and duration of symptoms prior to management as described in included studies.
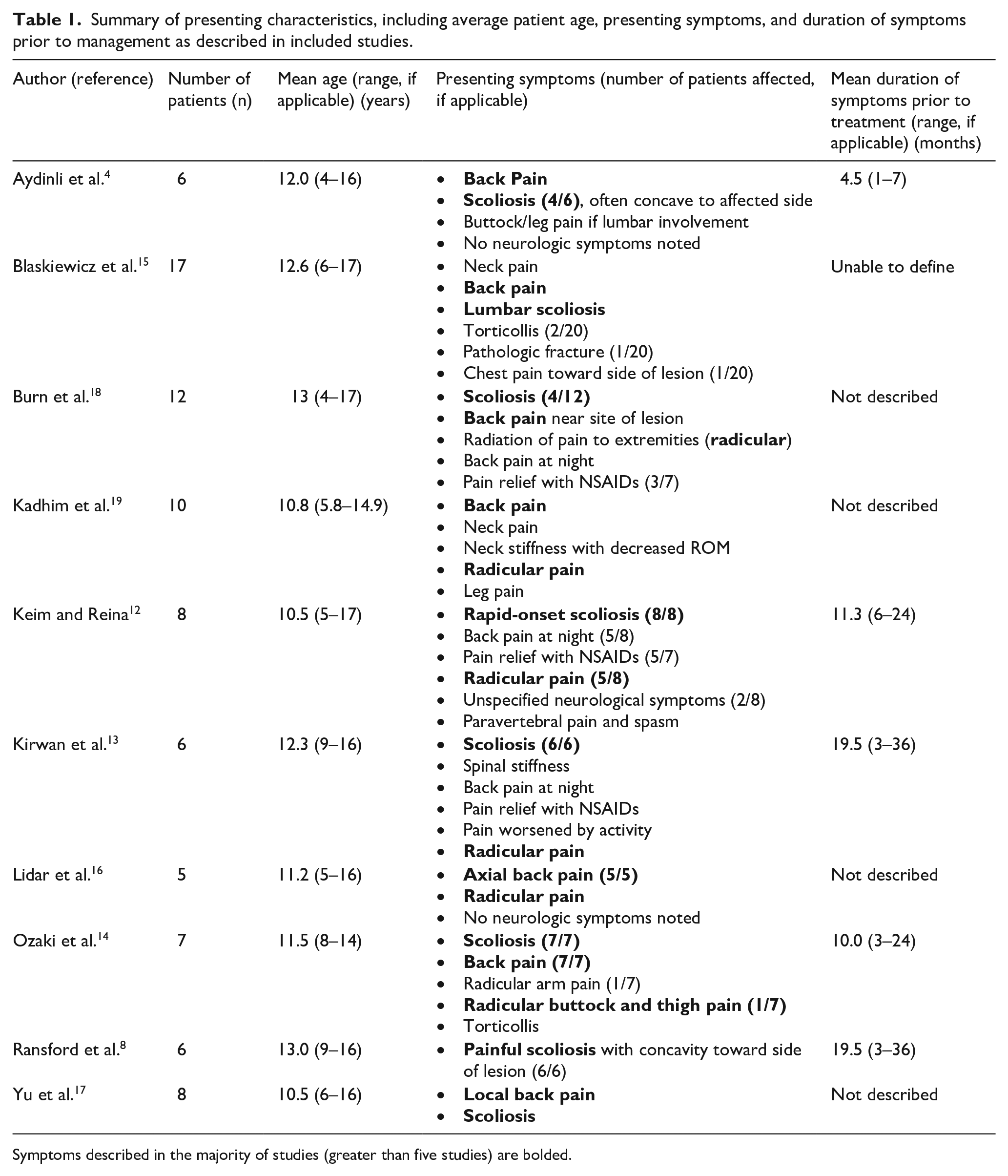
Symptoms described in the majority of studies (greater than five studies) are bolded.
Evaluation
A complete set of data describing evaluation of patients with spinal OO is listed in Table 2. The studies included in this review evaluated patients using a combination of radiographs (XR), computed tomography (CT) scans, magnetic resonance imaging (MRI), bone scans, and/or frozen section. Imaging was utilized preoperatively for diagnosis, intraoperatively, and/or postoperatively, depending on the study. XR findings were commonly described as osteosclerotic or radiodense lesions with or without a nidus.4,12,14 Scoliosis confirmed on XR was often associated with a lesion on the concave side of the curve.4,8,12,13 Keim et al. noted that XR obtained for 3/8 patients failed to identify the tumor, while all CT scans on these patients identified the lesion. 12 Interestingly, all lesions were able to be identified on XR retrospectively. CT findings typically included hypodense lesions, often with an identifiable nidus, with surrounding sclerotic bone.12,13,15 –17 MRI findings included high intensity signals in surrounding bone and muscle via T2 sequence;14,18 nidus identification was not often described in comparison to CT. Bone scan was described as showing an increased area of radionuclide activity. 15
Summary of patient evaluation, including preoperative, intraoperative, and postoperative imaging utilized in included studies.
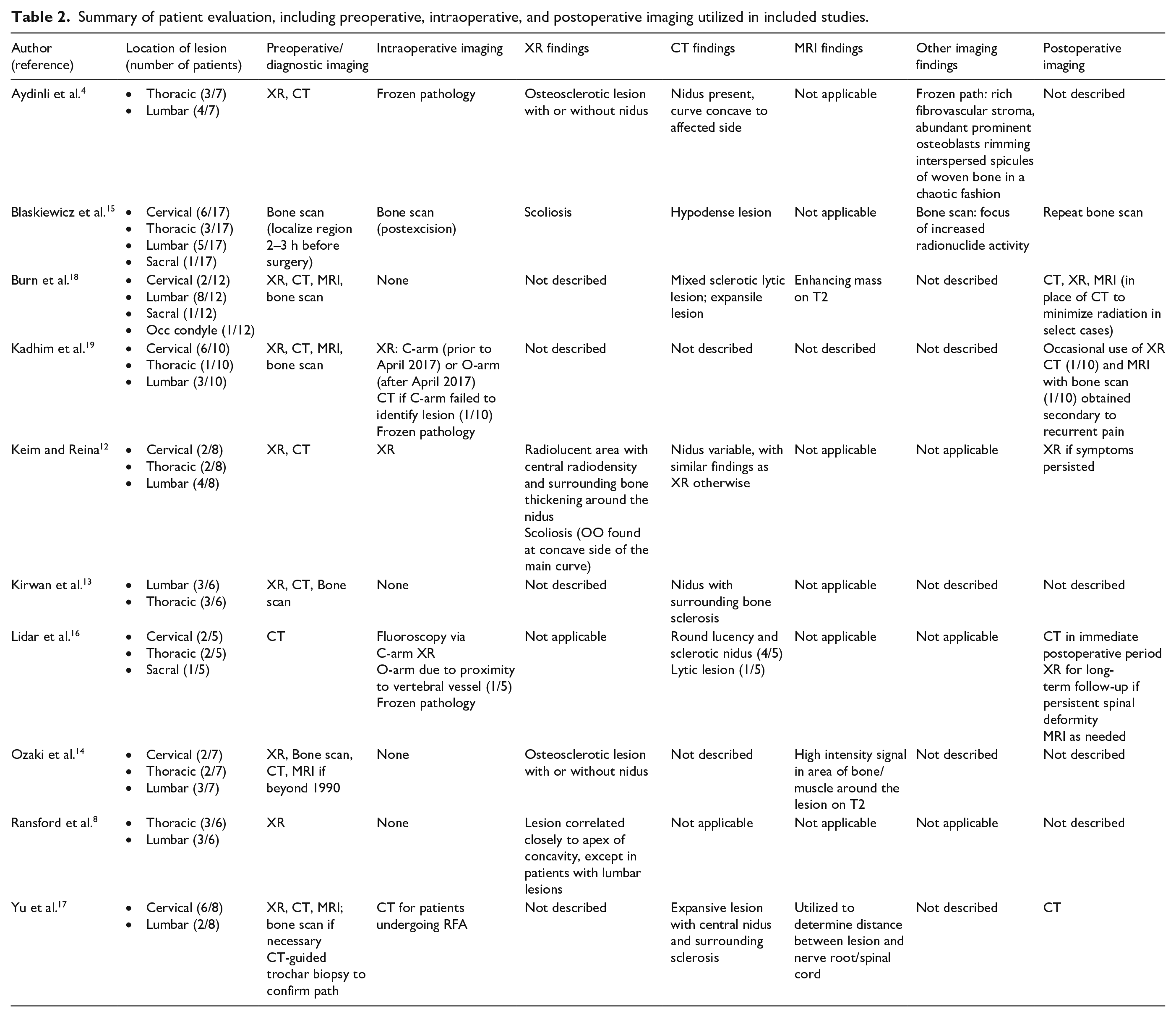
Intraoperative assessment included bone scan, 15 XR,12,16,19 CT,17,19 and frozen pathology.4,16,19 When utilizing XR intraoperatively, Kadhim et al. suggested that O-arm was beneficial in localization of the lesion when compared to C-arm, despite increased radiation. 19 Lidar et al. 16 similarly utilized O-arm in the case of one patient found to have a tumor near the vertebral vessels. Bone scans were noted to be particularly effective in aiding intraoperative detection of total resection. Blaskiewicz et al. reported that, on average, 2.8 intraoperative bone scan images were obtained per patient for lesion localization and to confirm resection. In 60% of their patients, more than two intraoperative scans were required due to insufficiency of the initial resection attempt. 15 Notably, five patients in their study had initially presented with a collective history of 12 prior surgeries for OO, all performed without intraoperative bone scan. Following resection with intraoperative bone scans, these patients have been free of recurrence with a mean follow-up time of 37 months (range 8–120). Postoperative imaging consisted of bone scan,15,19 XR,12,16,18,19 CT,16 –19 and/or MRI16,18,19 depending on surgeon preference and various patient-related indications that were often not described unless noted in Table 2.
Treatment options
Nonoperative
Five of the 12 patients included in the study by Burn et al. 18 were treated nonoperatively. These were the only patients included in this review to be managed without surgery. Indications for operative treatment included symptoms that were poorly controlled with conservative treatment, specifically the use of NSAIDs. Increased local concentration of prostaglandins have been found to be produced within the nidus of OOs, explaining the efficacy of NSAIDs and their ability to reduce prostaglandin, prostacyclin, and cyclooxygenase-2 levels. 18
Operative
The eight patients included in the study by Keim and Reina 12 were all treated by OSR with intraoperative XR.
The nineteen total patients included in the studies by Kirwan et al., 13 Ransford et al., 8 and Ozaki et al. 14 were treated by OSR without intraoperative imaging.
The six patients included in the study by Aydinli et al. 4 were treated by OSR with intraoperative biopsy.
The seventeen patients included in the study by Blaskiewicz et al. 15 were treated with intraoperative bone scan-assisted resection.
The remaining seven patients included in the study by Burn et al. 18 were managed with OSR. One patient required a transoral approach. Intraoperative imaging was not used.
The ten patients included in the study by Kadhim et al. 19 were treated by OSR with intraoperative XR (C-arm or O-arm, as described above). The C-arm failed to identify the tumor in one patient, who required intraoperative transport to CT for accurate localization.
The eight patients included in the study by Yu et al. 17 were treated with either CT-guided biopsy or radiofrequency ablation (3/8) under local anesthesia, or OSR (5/8) without intraoperative imaging.
The six patients included in the study by Lidar et al. were treated by OSR with C-arm fluoroscopy and intraoperative biopsy. O-arm was used in one case given the proximity of the tumor to the vertebral vessels.
Outcomes
A complete set of data describing outcomes, mean follow-up period, recurrence rate, nonrecurrence complications, and descriptions of additional procedures is included in Table 3. All included studies described partial or complete resolution of pain in the immediate postoperative period. In the percutaneously-managed group, postoperative pain was reported to be more effectively controlled than patients who underwent OSR. 17 There was variation in recurrence of pain in the long-term follow-up period. Six studies described partial or complete resolution of scoliosis that was found on presentation in the immediate postoperative period.4,8,12 –15 Other studies described improved back stiffness and ROM13,17 and neurologic symptom resolution, particularly radicular pain that was noted on presentation.14,15 Nevertheless, some patients demonstrated persistent or permanent structural deformity of the spine despite operative management.8,13
Summary of postmanagement outcomes, recurrence rate, nonrecurrence complications, and secondary procedures among included studies.
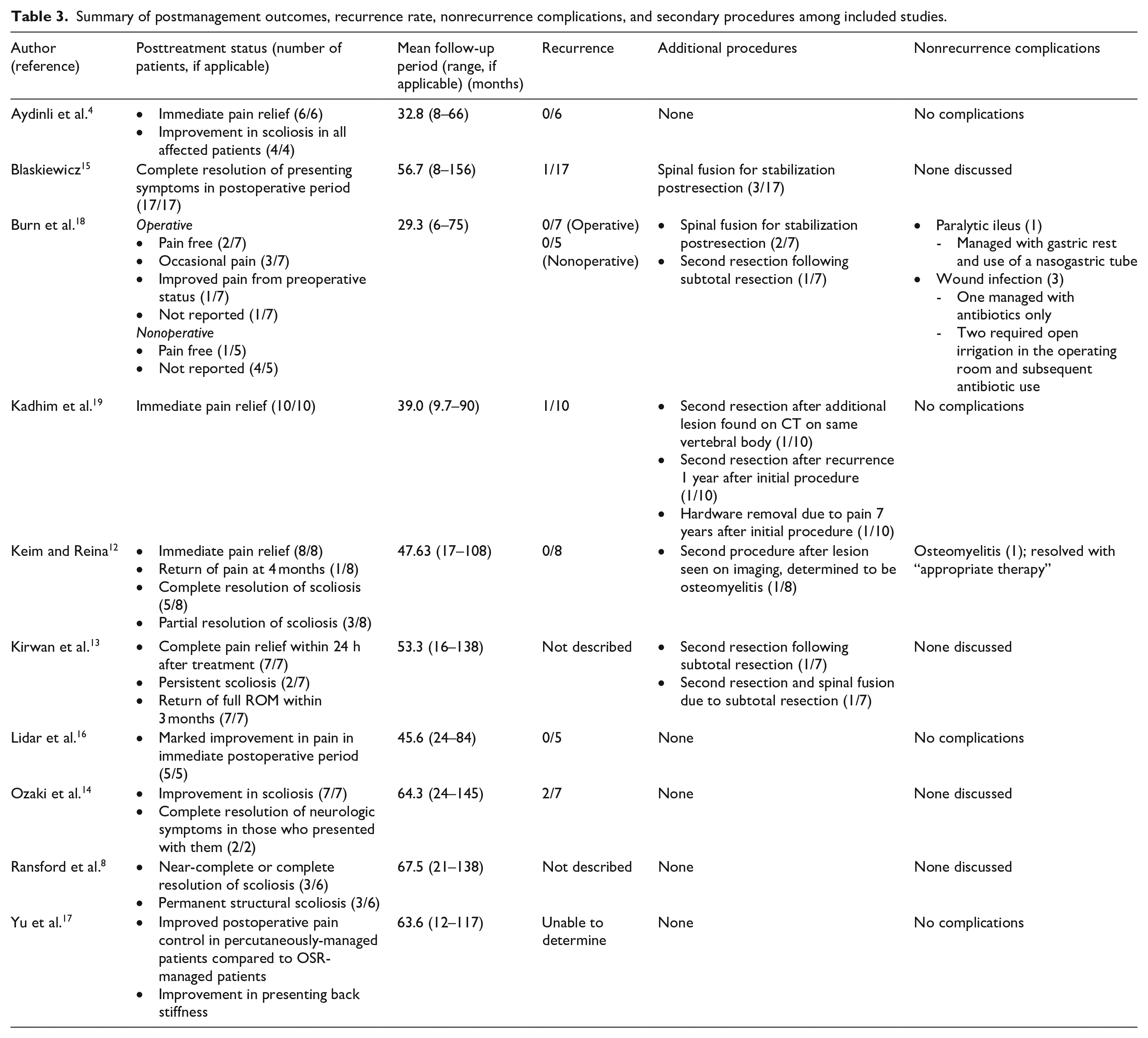
Several studies that utilized OSR commented on necessity of spinal fusion to improve stability after resection.13,15,18 Recurrence rate was relatively low across all studies regardless of treatment modality, ranging from 0.0% to 28.6% (2/7), 14 with an overall rate of 6.7% (4/60) among operatively managed patients with reported data. Among patients who were found to have a recurrent lesion, they often presented initially with recurrent symptoms resembling those on initial presentation, particularly back pain, back stiffness with decreased ROM, or recurrent scoliosis.15,17,19 Recurrent lesions were confirmed with imaging and a second resection. Mean follow-up period ranged from 29.3 to 67.5 months.
Nonrecurrence complications were seen in two studies12,18 and were limited to paralytic ileus, wound infection, and osteomyelitis (Table 3).
Management algorithm
Based on review of the existing literature on management of spinal OOs in the pediatric population, we propose a step-wise management algorithm (Figure 2). When a pediatric patient is found by history and physical exam to have common symptoms, such as rapid-onset scoliosis and stiffness, nocturnal or constant back pain, or pain improved with NSAIDs, clinical suspicion for spinal OO should be raised. Radiographs of the affected area of the spine are traditionally first line of imaging. However, with high clinical suspicion, CT with or without bone scan may instead be preferred, as these studies are significantly more sensitive for spinal OO detection. Negative radiographs in the setting of high clinical suspicion warrant further imaging with CT with or without bone scan. MRI may be used per surgeon preference if there is concern for soft tissue involvement, or if necessary for RFA planning. Following diagnosis, a trial of conservative treatment, utilizing NSAIDs and closing monitoring, may be attempted. If the patient demonstrates persistent pain or progressing spinal deformity during this period, operative treatment modalities (OSR, MIS, or RFA) should be considered. When deciding between the described treatment choices, special consideration should be paid to the location, size, and vascularity of the lesion. Other relative indications and contraindications are listed in Figure 2. If available, intraoperative imaging modalities, preferably bone scan, should be utilized. Following operative management, the patient should be closely monitored for any persistent or recurrent symptoms. Postoperative imaging may be used per surgeon preference. If the patient experiences recurrence of presenting symptoms or has evidence of partially resected OO, immediate re-evaluation is indicated, and the management algorithm is recycled. If the patient remains asymptomatic within the six-week follow-up period, further monitoring and appropriate follow-up should occur with imaging at the surgeon’s discretion. It is important to note that, while this algorithm may help provide a general reference for management of pediatric spinal OOs, referral to specialists with expertise in treating tumors of the spine should be considered in cases of unusual or unique patient presentations and characteristics.
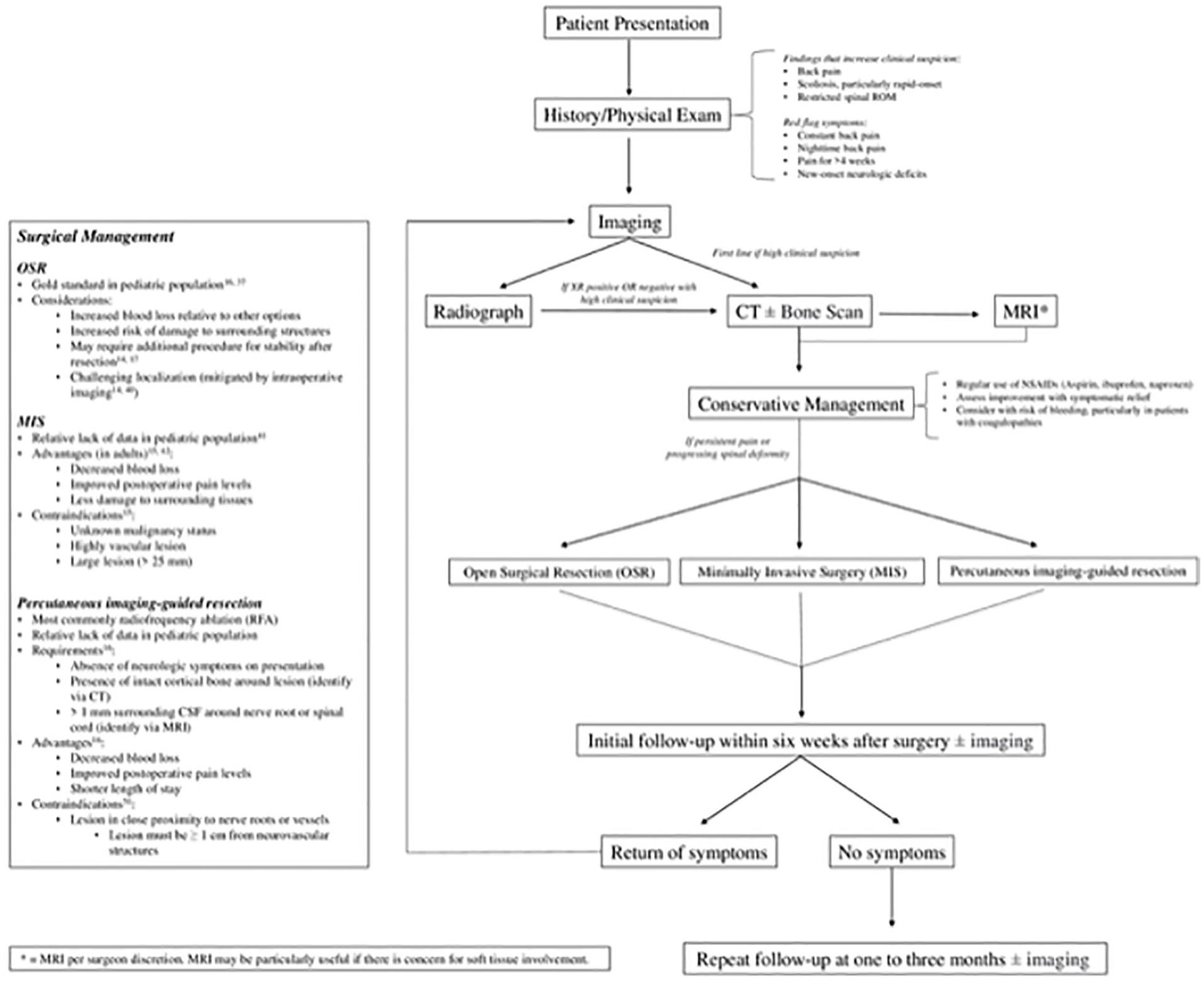
Management algorithm for pediatric spinal osteoid osteoma.
Discussion
In this review, we aimed to evaluate the current literature on spinal OOs in pediatric patients in order to establish a management algorithm. With such a wide variety of treatment modalities available, there is no consensus for management of patients with spinal OO. This may be due in part to limited literature; as a relatively rare,1 –7 finding that often goes underdiagnosed, even with appropriate initial imaging, spinal OO has yet to be thoroughly studied in the pediatric population. By evaluating the literature that currently exists, we have compiled the most common presenting symptoms, the development of diagnostic strategies, and the evolution of treatment options, while also considering outcomes and complications.
Diagnosis and workup
The presenting symptoms are often nonspecific. The most common presenting symptoms included back pain, scoliosis (33.3% to 100.0% among reported values) that was often rapid-onset, and back stiffness with restricted ROM. Other common symptoms included nighttime pain improved with aspirin, radicular pain, neurologic deficits, and torticollis; however, these were less consistent, and thus, less reliable.
Once OO is suspected, further evaluation is warranted. The studies included in this review utilized a wide range of imaging, including XR, CT, MRI, or bone scans in the diagnosis stage. XR is typically the indicated initial imaging modality for back pain in children, particularly with red flag symptoms such as constant pain, night pain, pain for greater than four weeks, or neurologic signs. 20 Only one of the ten studies included in this review did not comment on use of radiograph as the first-line imaging modality. Despite its ubiquity, XR may not be reliable enough to detect OOs consistently; radiographs were reported to miss lesions that were later detected by other imaging modalities.13,21 CT was often selected as the second-line imaging modality for patients with negative radiographs but strong clinical evidence for OO. There were no reports of missed lesions via CT, consistent with existing literature on the high sensitivity of CT for detection of spinal OO.22 –26 Diagnosis of OO is characterized by identification of the nidus, the highly mineralized center of the tumor that is often missed on XR due to surrounding sclerosis, but is more consistently detected by CT.23,24 Several studies additionally used bone scintigraphy to confirm diagnosis13 –15,17 –19 with a sensitivity of nearly 100% due to the high-turnover nidus that is distinct to OO.27,28 Bone scintigraphy was also found to be effective for intraoperative monitoring of resection.14,15 MRI was commonly utilized in our included studies.4,17 –19 However, the use of MRI in this setting is controversial. Several studies, conducted in pediatric 29 and nonpediatric populations,22,30,31 indicate that the accuracy of CT surpasses that of MRI. Davies et al. 31 suggested that a negative MRI with concerning surrounding soft tissue features would require follow-up with CT or bone scintigraphy, negating its advantage as a nonradiating imaging modality while also further delaying diagnosis. It has been proposed that, when contrast-enhanced, MRI may be relatively more successful in spinal OO detection. 32 The benefits of limiting radiation exposure to pediatric patients via MRI should be considered on a case-by-case basis, balanced against the risk of misdiagnosis. Preoperative or intraoperative biopsy is an additional highly useful tool for surgeons to utilize to ensure accurate diagnosis. Three of the ten studies in this review utilized biopsy as part of their diagnostic approach, in addition to other imaging modalities. While OO is typically a clinical and radiographic diagnosis, confirmation by histology may be useful for cases with unusual characteristics.33 –36
Treatment options
OOs may gradually resolve over years,33,37 with shortened resolution intervals when treated with aspirin or NSAIDs. 2 However, studies have suggested that insufficiently treated OOs can pose a significant risk for long-term disability, in addition to short-term debilitating pain and deformity.8,16 Long-term NSAID use also comes with the risk of bleeding that can be considered unnecessary given the efficacy of operative/percutaneous intervention. 38 All patients, except for five in the study by Burn et al., 18 underwent operative or percutaneous imaging-guided treatment with good success. Outcomes for these five nonoperative patients were not well-described, but notably, none demonstrated OO recurrence.
Surgical options described in this review included OSR versus an MIS approach. OSR is historically the standard treatment option for spinal OO in pediatric patients17,39 and involves removal of the nidus with additional removal of surrounding sclerotic bone as allowed. Noted challenges include induction of a bone defect, which may require additional procedures to address instability,15,18 as well as challenging localization that may lead to a repeat resection.14,18 The latter may be addressed with intraoperative bone scan.15,40 Intraoperative bone scan may be preferred over intraoperative CT because of OO location; given that OOs are often located posteriorly near nerve roots and vessels, continuous detection by bone scan signal, rather than periodic evaluation via intraoperative CT, may prove safer. 14 MIS for removal of spine tumors in children has not been well-studied. Lidar et al. presented a case series of six patients with spinal OO successfully treated by this technique. MIS approaches are still a relatively recent development in the management of spinal tumors and are better studied in adults at this time. 41 While the results demonstrated by Lidar et al. 16 suggest it may be a promising surgical option—particularly with advantages of less damage to surrounding tissues, decreased pain, and decreased blood loss 42 —further studies are needed to establish its role in the pediatric population. In addition, this approach should not be used if malignancy status is unknown, if the lesion is highly vascular, or if it is very large (defined as greater than 25 mm by Lidar et al. 16 ).
Percutaneous imaging-guided treatment has gained popularity when used in the appropriate clinical setting. Options include radiofrequency ablation (RFA), cryoablation, laser thermocoagulation, and trephine excision, all most often guided by CT;43 –46 these studies were not exclusive to lesions of the spine, nor focused on a pediatric population. For this reason, while RFA has become increasingly considered to be a potential replacement for OSR,47 –51 further research on its viability, specifically in treating spinal OO in the pediatric population remains necessary. Only one study included in this review utilized percutaneous RFA treatment, 17 and no complications relating to RFA use were reported. Yu et al. 17 describe prerequisites for RFA include the absence of neurologic symptoms on presentation, the presence of intact cortical bone around CT-identified lesions, and a greater than 1 mm cushion of CSF between the lesion and the nerve root or spinal cord as detected by MRI. In this study, RFA demonstrated strong results when compared to OSR. RFA patients had decreased blood loss, improved pain control postoperatively, and a shorter hospital stay. One of twelve patients total, including both adult and pediatric patients, who were managed with RFA were reported to have recurrence, consistent with previous studies that have indicated a recurrence rate of up to 21% following management with RFA; however, most studies report lower values.17,52 –57 This recurrence rate has been suggested to be due to possible incomplete eradication of the tumor, 58 an issue also identified following other treatment modalities. Given that RFA works by administering extreme levels of heat to the local area, its main limitation is the location of the OO to nearby neurovascular structures. The existing recommendation is that the lesion must be at least one centimeter from any neurovascular structure, 51 however, ongoing efforts are being made to evaluate the validity of this protocol. 57 While RFA has been shown to be efficacious and safe,53,59 additional research is needed in the pediatric population with spinal lesions to make a definitive claim. At this time, the significant potential risk for devastating injury may favor alternative management options for lesions in close proximity to the cord in pediatric patients.
Outcomes and follow-up
Risk of complications is essential to consider when electing operative management. The primary complication evaluated was the recurrence rate. Recurrence overall was relatively uncommon, at 6.6% overall after operative management. Nonrecurrence complications, including wound infection (three patients total), osteomyelitis (one patient total), and paralytic ileus (one patient total), were exceedingly rare across all included studies and adequately resolved with standard treatment. Thus, while complications must be discussed, outcomes are particularly favorable with operative management. Follow-up after treatment should consist of both clinical status and imaging. All studies noted a resolution of pain and back stiffness, with a marked improvement in spinal deformity present preoperatively. Return of these symptoms should cause concern for possible recurrence and merits further evaluation. The use of follow-up imaging in this review was relatively inconsistent. Seven of ten studies described use of postoperative imaging, which includes XR, CT, MRI, or bone scintigraphy. Based on our evaluation of the current literature, we cannot recommend a standard follow-up imaging plan. However, importantly, all patients with recurrence for which a presentation was reported demonstrated new-onset or worsening of previous symptoms.15,17,19 While it may be possible that recurrence would occur without symptoms, this was not found in our review. Regarding timing, we recommend follow-up for symptom evaluation within six weeks after surgery, followed by another visit within one to three months, then further spaced appointments subsequently. Recurrence was noted within the first one to six months after surgery by one study; 17 however, this timeline varies depending on the study and likely requires additional research to be definitively established. Nonetheless, if symptoms recur, imaging is warranted. If obtaining imaging under high suspicion for recurrence, we recommend CT or bone scan, rather than radiograph; we noted a trend away from radiographs, favoring CT and bone scan in this setting as more recent studies were conducted,15 –19 supporting our previous assertion that radiographs may be unreliable for OO detection. MRI may be useful for the detection of other postoperative complications, including osteomyelitis and soft tissue injury, that may help determine prognosis while causing symptoms that resemble recurrence.50,60 However, the same limitations for MRI, when compared to CT, exist in the postoperative period as in the preoperative period, as previously described.
Limitations
There are limitations to this study that warrant discussion. First, during our search process, we did not find any relevant randomized trials. Thus, the studies involved in this review are comprised of observational cohort studies and case series only. Second, several of the studies involved were relatively small. While we hoped to mitigate the decreased power that comes with smaller study sizes by excluding those with fewer than five patients, many qualified studies remained under ten patients. Third, because there has been no standardized approach for the management of spinal OO in pediatric patients, we discovered heterogeneity in the diagnostic, treatment, and follow-up approaches used by each study, with varying descriptions and information provided for each. This heterogeneity led to variable data collection from some papers. There was additional heterogeneity across included studies regarding the various management options, including OSR, MIS, percutaneous imaging-guided resection, and conservative treatment of spinal OOs. Among our ten included manuscripts, only two compared MIS 16 and percutaneous radioablation, 17 respectively, to OSR. OSR has been considered the gold standard prior to development of less invasive techniques, 61 although this mindset may be shifting already toward RFA. 62 Therefore, it remains unclear which modality is superior in the pediatric population from this systematic review. Further research with larger sample sizes and, thus, higher power is likely necessary to definitively determine the most appropriate management method. However, given the current state of the literature and the potential for OOs to cause significant, long-term damage if mismanaged, we believe this review presents a robust and reliable synthesis of the relevant literature that exists today.
Conclusion
In this study, we sought to conduct a review of the current literature on the management of spinal OO in the pediatric population, resulting in a proposed management algorithm, as one has yet to be published on this topic. It is our hope that this algorithm may be utilized to help physicians appropriately approach this relatively rare, but debilitating, condition.
Supplemental Material
sj-pdf-1-cho-10.1177_18632521231192477 – Supplemental material for Spinal osteoid osteoma in the pediatric population: A management algorithm and systematic review
Supplemental material, sj-pdf-1-cho-10.1177_18632521231192477 for Spinal osteoid osteoma in the pediatric population: A management algorithm and systematic review by Alexander R Farid, David S Liu, Mary M Morcos and Grant D Hogue in Journal of Children’s Orthopaedics
Footnotes
Author contributions
AF: Writing—original draft; Writing—review & editing; Methodology; Analysis; Visualization.
DL: Conceptualization; Writing—original draft; Writing—review & editing; Methodology.
MM: Writing—original draft; Writing—review & editing; Methodology; Analysis; Visualization.
GH: Conceptualization; Methodology; Writing—reviewing & editing; Supervision.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical statement
Not applicable for this type of study.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
