Abstract
Purpose:
To explore scoliosis risk factors and outcomes in children with dysplastic spondylolisthesis undergoing surgical reduction and internal fixation.
Methods:
We retrospectively analyzed 56 children with dysplastic spondylolisthesis who underwent surgical reduction and internal fixation. Patients were grouped according to presence of scoliosis before surgery. Radiographic parameters measured before surgery included pelvic incidence, pelvic tilt, sacral slope, coronal Cobb angle, slip percentage, Dubousset’s lumbosacral angle, lumbar lordosis, sagittal vertical axis, and Spinal Deformity Study Group index. Groups were compared using logistic regression. Receiver operating characteristic analysis was performed to determine the optimal Spinal Deformity Study Group index cut-off value. All patients were followed up for at least 2 years.
Results:
The scoliosis group comprises 36 patients (mean age: 9.6 ± 2.7 years), while the no scoliosis group comprises 20 (mean age: 9.1 ± 2.4 years). Slip percentage and Spinal Deformity Study Group index were significantly higher in the scoliosis group (p < 0.01). Sacral slope and pelvic incidence were lower in the scoliosis group (p < 0.05). Univariate logistic regression analysis showed that slip percentage, Spinal Deformity Study Group index, pelvic incidence, and sacral slope were significantly associated with scoliosis. In the multivariate logistic regression analysis, only Spinal Deformity Study Group index was an independent risk factor for scoliosis. The optimal cut-off value for Spinal Deformity Study Group index was 0.288. Mean Cobb angle decreased from 20.3° ± 8.8° before surgery to 8.5° ± 8.9° at last follow-up; the mean scoliosis correction rate was 59.3%.
Conclusion:
Severe S1 dysplasia and high slip percentage may be risk factors for developing scoliosis in patients with dysplastic spondylolisthesis. Scoliosis resolved spontaneously after spondylolisthesis reduction and fixation in most patients.
Level of evidence
3.
Introduction
Spondylolisthesis is common in children. Five main types of spondylolisthesis have been defined: dysplastic, isthmic, degenerative, traumatic, and pathological.1,2 Dysplastic spondylolisthesis is the most frequent type in children, who typically present with local L5-S1 kyphosis, L5 wedging, and doming of S1. Degree of spondylolisthesis can rapidly progress in children because of growth and development. Symptoms of slip progression include back pain, crouch gait, and neurological deficit.
Patients with spondylolisthesis may also develop scoliosis.3 –5 Reported incidence rates of scoliosis in patients with lumbar spondylolisthesis range from 23% to 48%, which are much higher than the rate in the general population.5 –7 However, few studies have focused on scoliosis in children with dysplastic spondylolisthesis or have reported changes in scoliosis after surgery for spondylolisthesis. Therefore, this study aimed to investigate scoliosis risk factors and analyze scoliosis outcomes after spondylolisthesis surgery in children with dysplastic spondylolisthesis.
Materials and methods
Subjects
We retrospectively reviewed 56 children (age <18 years) with dysplastic L5-S1 spondylolisthesis who underwent spondylolisthesis surgery in our hospital between July 2013 and November 2020. Data were obtained from the medical record and review of radiographic studies. Dysplastic spondylolisthesis was diagnosed according to the Wiltse classification. 2 Spondylolisthesis with dysplastic changes at L5/S1, such as L5 wedging and S1 doming, is diagnosed as dysplastic spondylolisthesis, regardless of the presence of elongation or separation of the pars interarticularis. Patients with a history of previous spinal trauma or surgery and incomplete radiographic data were excluded. We also excluded those with less than 2 years of follow-up.
A total of 78 patients diagnosed with dysplastic spondylolisthesis were initially enrolled. After excluding 16 patients with incomplete radiographic data, 5 with insufficient follow-up time, and 1 with a history of spine surgery, the final cohort consisted of 56 patients. Patients were grouped according to presence of scoliosis before surgery (scoliosis and no scoliosis groups). Scoliosis was defined as coronal Cobb angle >10°. Based on the apex location, patients with scoliosis can be classified into three types: thoracic (T2–T11), thoracolumbar (T12–L1), and lumbar (L2–L4). Decompression, spondylolisthesis reduction, kyphosis correction, and internal fixation were performed in all patients.
Radiographic evaluation
Radiographic images were measured twice by one experienced orthopedic surgeon using Surgimap software version: 2.3.2.1 (Nemaris, New York, NY). Standing posteroanterior and lateral full spine radiographs were obtained before and after surgery and throughout follow-up. The S1 upper endplate was identified using Labelle’s method. 8 The best fit line along the anterior (A) and posterior (B) border of the sacrum was drawn. Two lines lose contact with the S1 in the point a and b, line (ab) is considered to be the S1 upper endplate (Figure 1(a)). The following imaging parameters were measured on preoperative and the last follow-up radiography: (1) pelvic incidence (PI), the angle between a line perpendicular to the S1 upper endplate at its midpoint and a line joining the center of the S1 upper endplate to the axis of the femoral heads; (2) pelvic tilt (PT), the angle between the line connecting the center of the S1 upper endplate to the axis of the femoral heads and vertical reference line; (3) sacral slope (SS), the angle between the horizontal line and the S1 upper endplate; (4) coronal Cobb angle of the main curve; (5) slip percentage (SP), L5 vertebral slip distance as a percentage of the S1 upper endplate length; (6) Dubousset’s lumbosacral angle (Dub-LSA), the angle between the upper endplate of L5 and the posterior border line of S1 (Figure 1(b)); (7) the Spinal Deformity Study Group (SDSG) index (a measure of S1 dysplasia severity), the percentage ratio of height of the sacral dome to the length of the S1 upper endplate; 9 (8) lumbar lordosis (LL), the angle between the superior endplate of L1 and the inferior endplate of L5; and (9) sagittal vertical axis (SVA), the horizontal distance from the C7 plumb line to the posterosuperior corner of S1. To measure the height of the sacral dome, Line (E) is drawn parallel to line (ab) and touch the most rostral part of the sacral dome (point c). The line (cd) represents the linear distance from point c to line (ab), which is considered to be the height of the sacral dome (Figure 1(c)). Spondylolisthesis was graded using Meyerding’s 10 classification. The scoliosis correction rate was calculated as (preoperative Cobb angle−final follow-up Cobb angle)/preoperative Cobb angle × 100%.
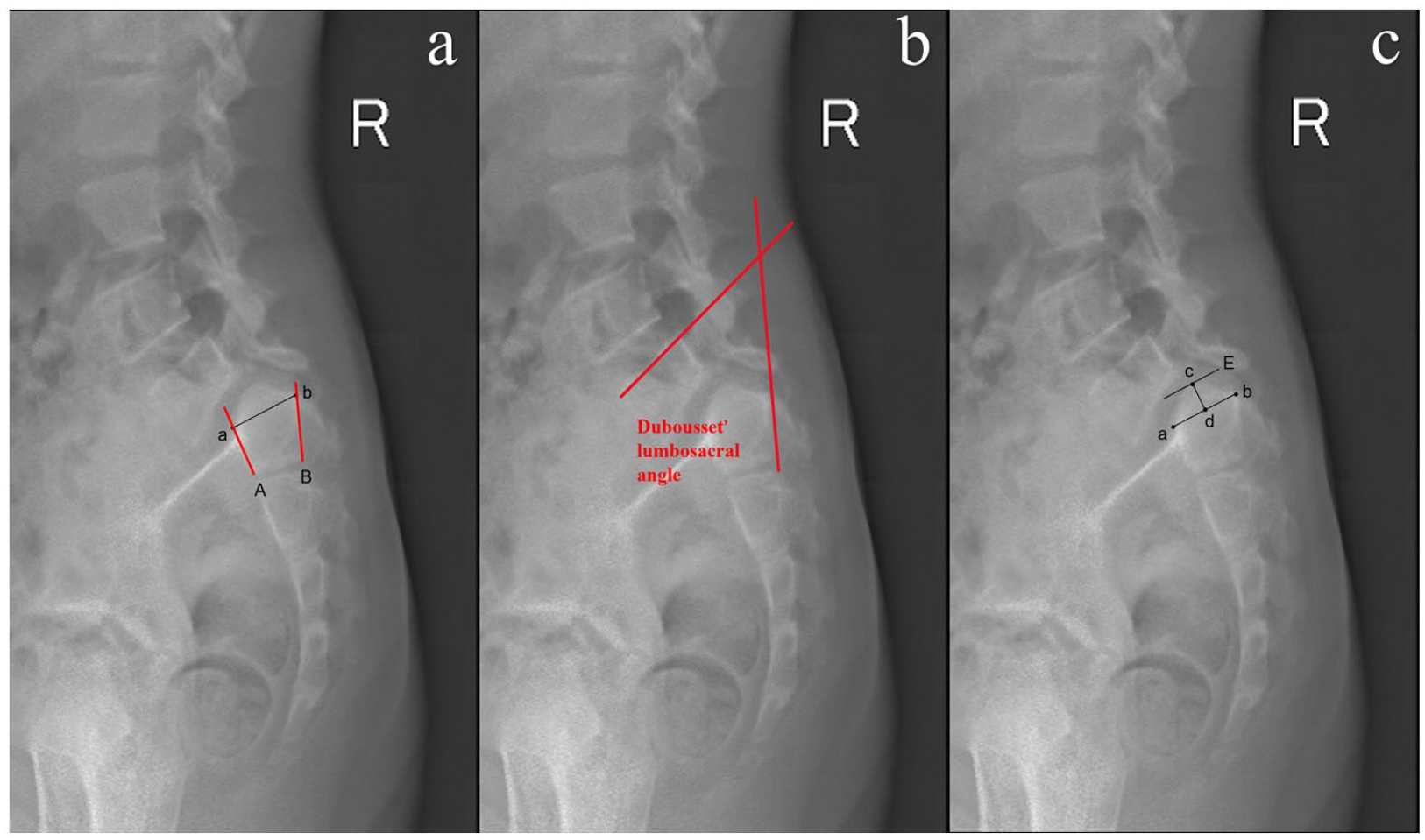
Methods for measuring sagittal parameters. (a) Method to identify the S1 upper endplate. (b) Measurement of Dubousset’s lumbosacral angle. (c) Measurement of the height of the sacral dome.
Statistical analysis
Statistical analyses were performed using SPSS software version 26.0 (IBM Corp., Armonk, NY). Quantitative data were tested for normality using the Shapiro–Wilk test. Normally distributed quantitative data were compared using the independent samples t test. The Mann–Whitney rank sum test was used to compare non-normally distributed data. Categorical data were compared using the chi-square test. P < 0.05 was considered significant. Univariate logistic regression was performed with the presence of scoliosis as the dependent variable and risk factors as independent variables. Factors found significant in the univariate analyses were included in the multivariate logistic regression analysis. Receiver operating characteristic (ROC) analysis was performed to determine the optimal SDSG index cut-off value.
Results
Mean age of the 56 study patients (5 males and 51 females) was 9.4 years (range: 4–15). Mean SP was 55.6%. Spondylolisthesis grade was I in 3 patients, II in 21, III in 21, IV in 8, and V in 3. On admission, 42 patients were with back and leg pain, 22 with gait abnormality, 4 with limited lower extremity movement, and 1 with urinary incontinence.
The scoliosis group comprises 36 patients (4 males and 32 females; mean age, 9.6 ± 2.7 years) while the no scoliosis group comprises 20 (1 male and 19 females; mean age, 9.1 ± 2.4 years). Patient characteristics according to group are presented in Table 1. Age, gender, Dub-LSA, LL, SVA, and PT did not significantly differ between the two groups. SP (61.1% vs 45.6%; p = 0.009) and SDSG index (0.31 ± 0.10 vs 0.22 ± 0.10; p = 0.003) were significantly higher in the scoliosis group. SS (29.8 ± 12.1 vs 39.6 ± 12.3; p = 0.006) and PI (68.3 ± 10.8 vs 76.8 ± 11.2; p = 0.035) were lower in the scoliosis group.
Comparison of parameters between patients with and without scoliosis.
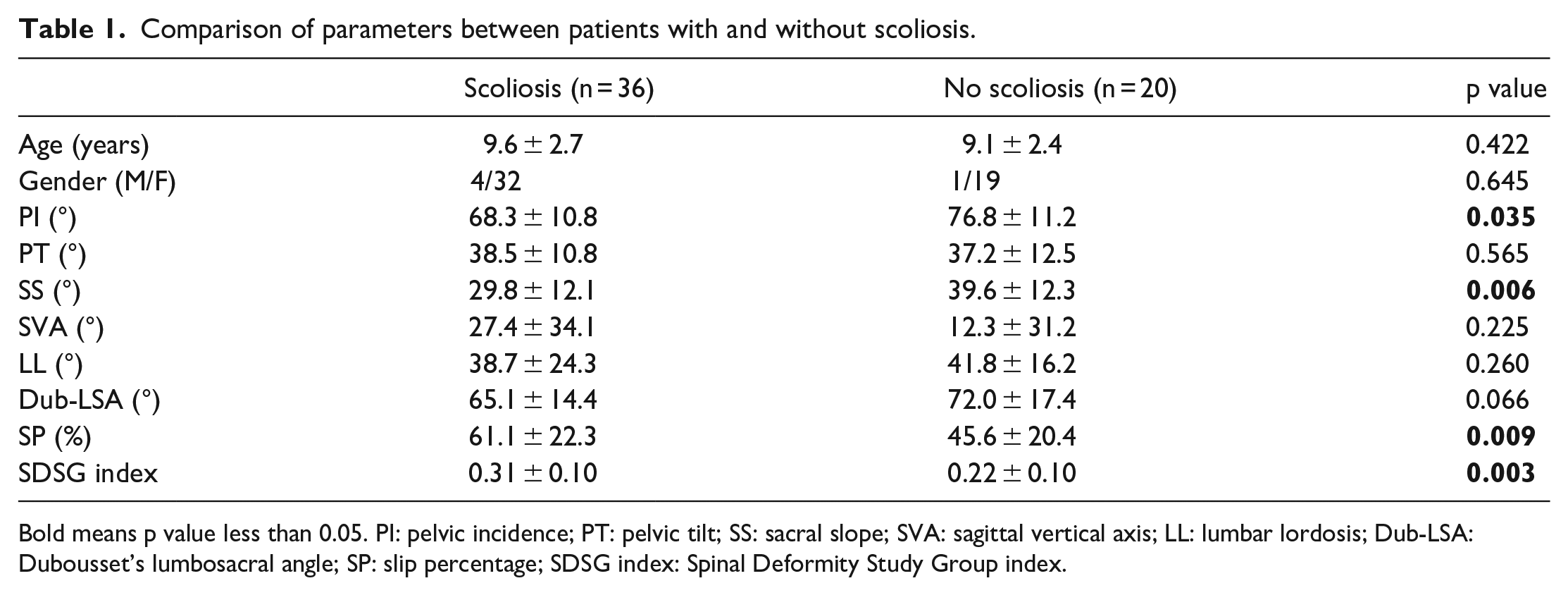
Bold means p value less than 0.05. PI: pelvic incidence; PT: pelvic tilt; SS: sacral slope; SVA: sagittal vertical axis; LL: lumbar lordosis; Dub-LSA: Dubousset’s lumbosacral angle; SP: slip percentage; SDSG index: Spinal Deformity Study Group index.
Univariate logistic regression analysis showed that SP (odds ratio [OR]: 1.037; p = 0.018), SDSG index (OR: 1.094; p = 0.006), SS (OR: 0.929; p = 0.021), and PI (OR: 0.926; p = 0.024) were significantly associated with scoliosis (Table 2). In the multivariate logistic regression analysis, only SDSG index (OR: 1.082; p = 0.042) was an independent risk factor for scoliosis (Table 3). The ROC analysis showed that the optimal SDSG index cut-off value was 0.288 (sensitivity, 54.5%; specificity, 82.4%; area under the ROC curve, 0.733; p = 0.004). In other words, the risk of scoliosis significantly increased with SDSG index >0.288 (Figure 2).
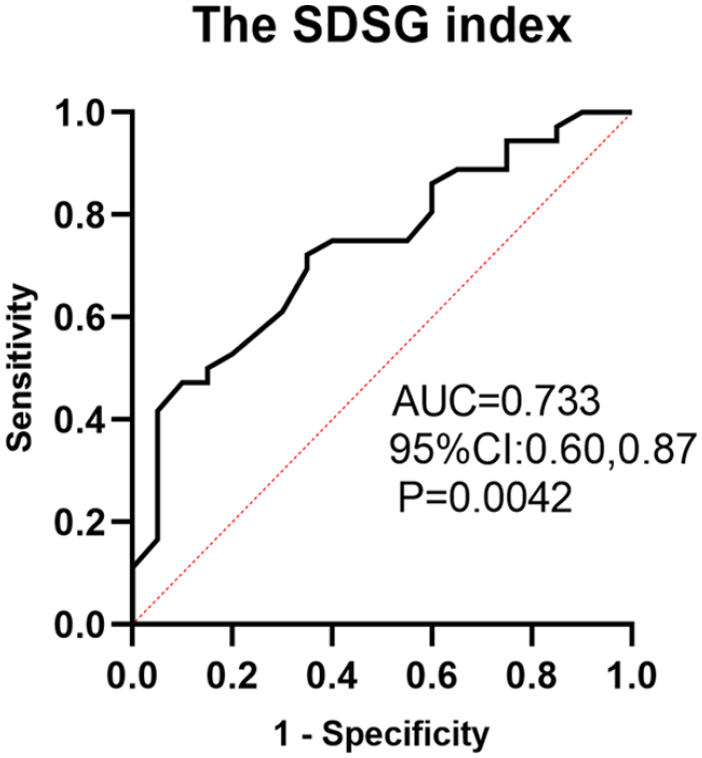
Receiver operating characteristic curve for SDSG index.
Univariate logistic analysis of risk factors for coexisted scoliosis in spondylolisthesis.
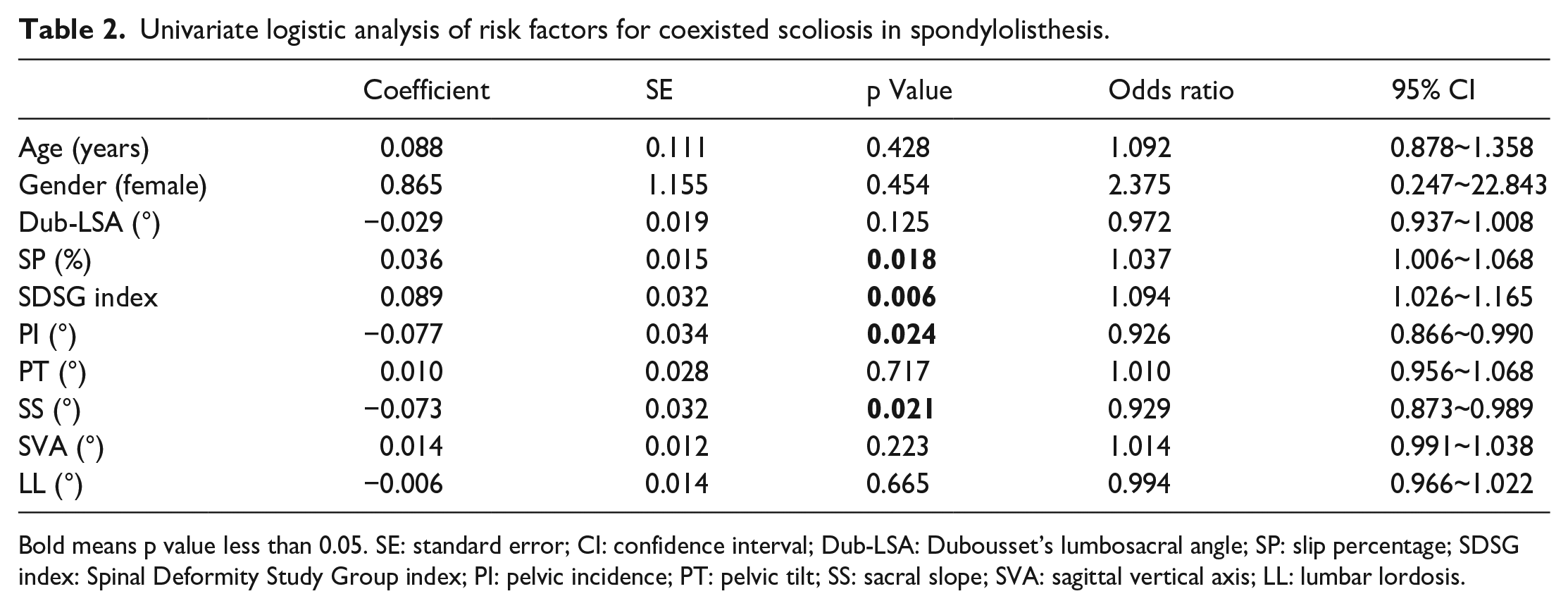
Bold means p value less than 0.05. SE: standard error; CI: confidence interval; Dub-LSA: Dubousset’s lumbosacral angle; SP: slip percentage; SDSG index: Spinal Deformity Study Group index; PI: pelvic incidence; PT: pelvic tilt; SS: sacral slope; SVA: sagittal vertical axis; LL: lumbar lordosis.
Multivariate logistic regression analysis of risk factors for scoliosis.
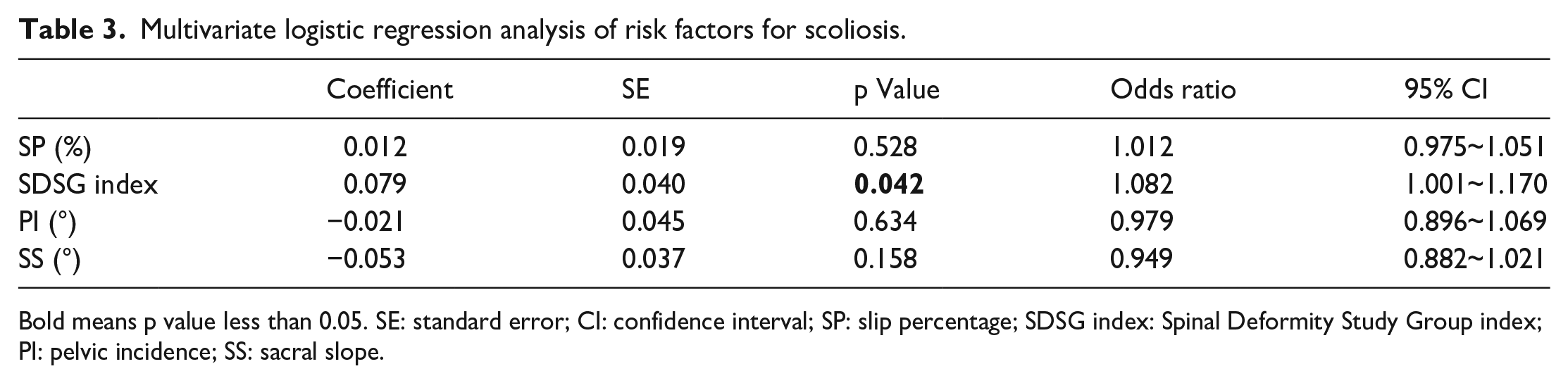
Bold means p value less than 0.05. SE: standard error; CI: confidence interval; SP: slip percentage; SDSG index: Spinal Deformity Study Group index; PI: pelvic incidence; SS: sacral slope.
Patients were followed up clinically and radiographically for a minimum of 24 months. Mean follow-up time was 68.5 ± 36.4 months (range: 24–158). As shown in Table 4, SP and sagittal parameters showed significant improvement postoperatively. SP decreased from 55.6% ± 22.8% preoperatively to 16.3% ± 14.0% at last follow-up (p < 0.001), SVA decreased from 24.2 ± 36.3 mm to 6.4 ± 21.6 mm (p < 0.05), and Dub-LSA increased from 65.7° ± 14.2° to 95.8° ± 14.5° (p < 0.001).
Variations of sagittal spine-pelvic parameters at preoperative and last follow-up.
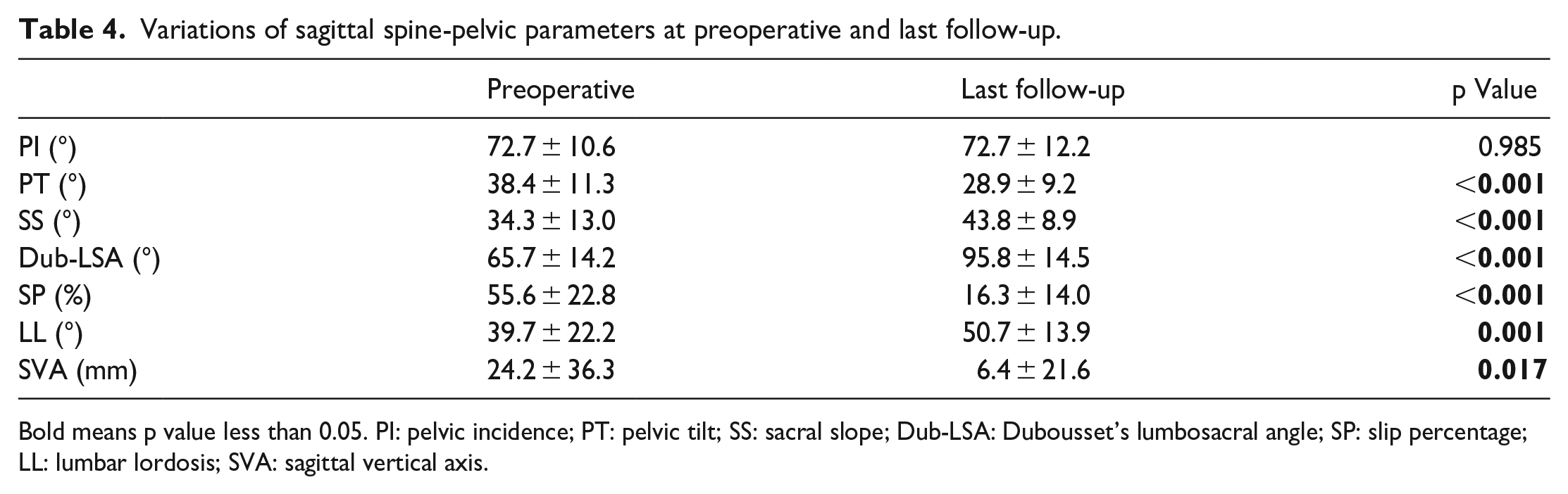
Bold means p value less than 0.05. PI: pelvic incidence; PT: pelvic tilt; SS: sacral slope; Dub-LSA: Dubousset’s lumbosacral angle; SP: slip percentage; LL: lumbar lordosis; SVA: sagittal vertical axis.
Among the 36 patients with scoliosis, the mean Cobb angle decreased from 20.3° ± 8.8° before surgery to 8.5° ± 8.9° at last follow-up, exhibiting an average scoliosis correction rate of 59.3%. Meanwhile, the median Cobb angle was reduced from 17.4° (range: 11.0°–47.8°) before surgery to 8.2° (range: 0°–40.6°) at last follow-up (Figure 3). In the scoliosis group, there are nine patients with thoracic curves, 14 with thoracolumbar curves, and 13 with lumbar curves. The mean scoliosis correction rate of thoracic patients was 65.5%, thoracolumbar patients was 62.4%, and lumbar patients was 36.5%. Cobb angle was less than 10° at last follow-up in 22 of the 36 patients (61.1%) in the scoliosis group (Figure 4).
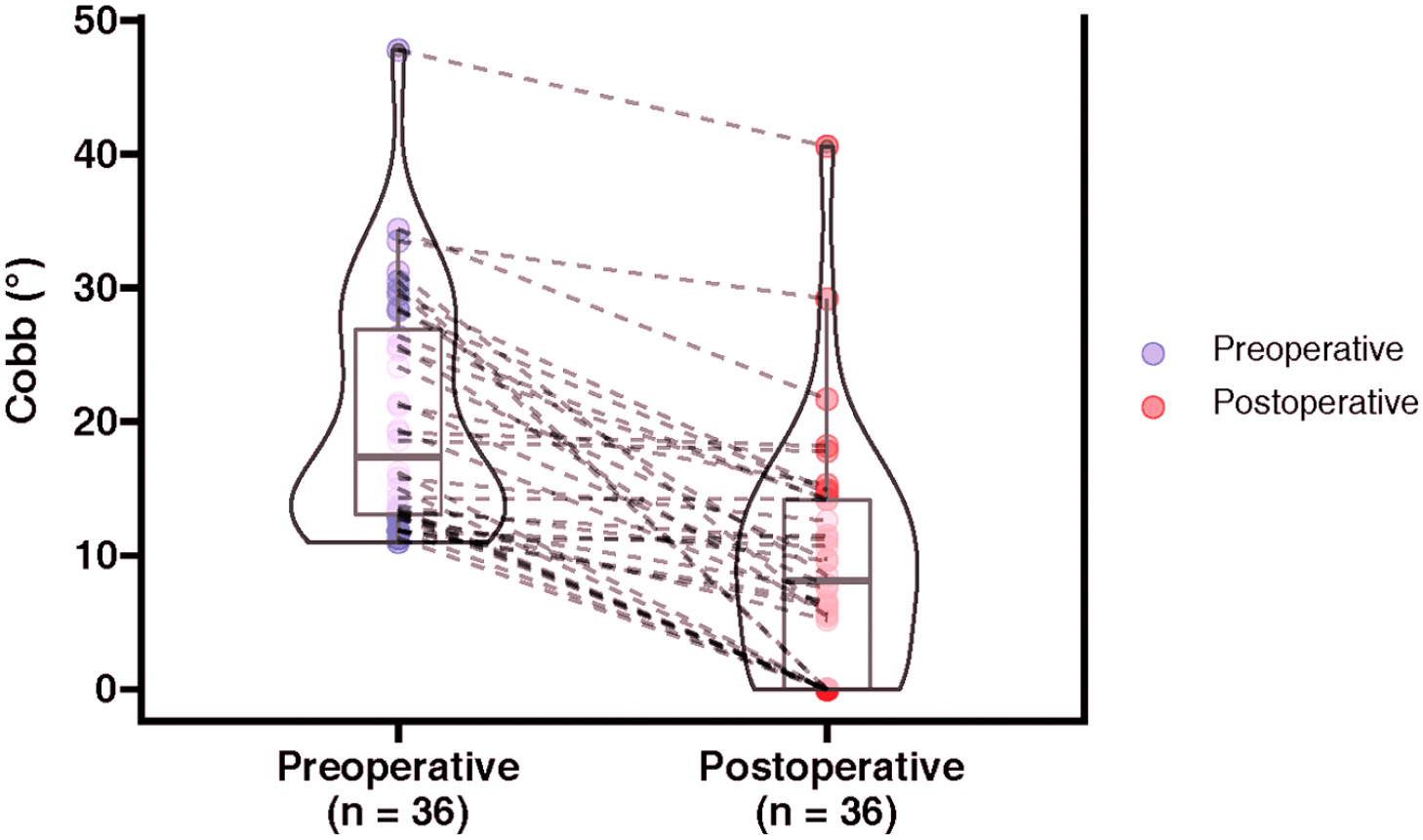
The variation of Cobb angle from preoperative to postoperative. Each point represents a patient with scoliosis, and the line connected the variation within the same patient.
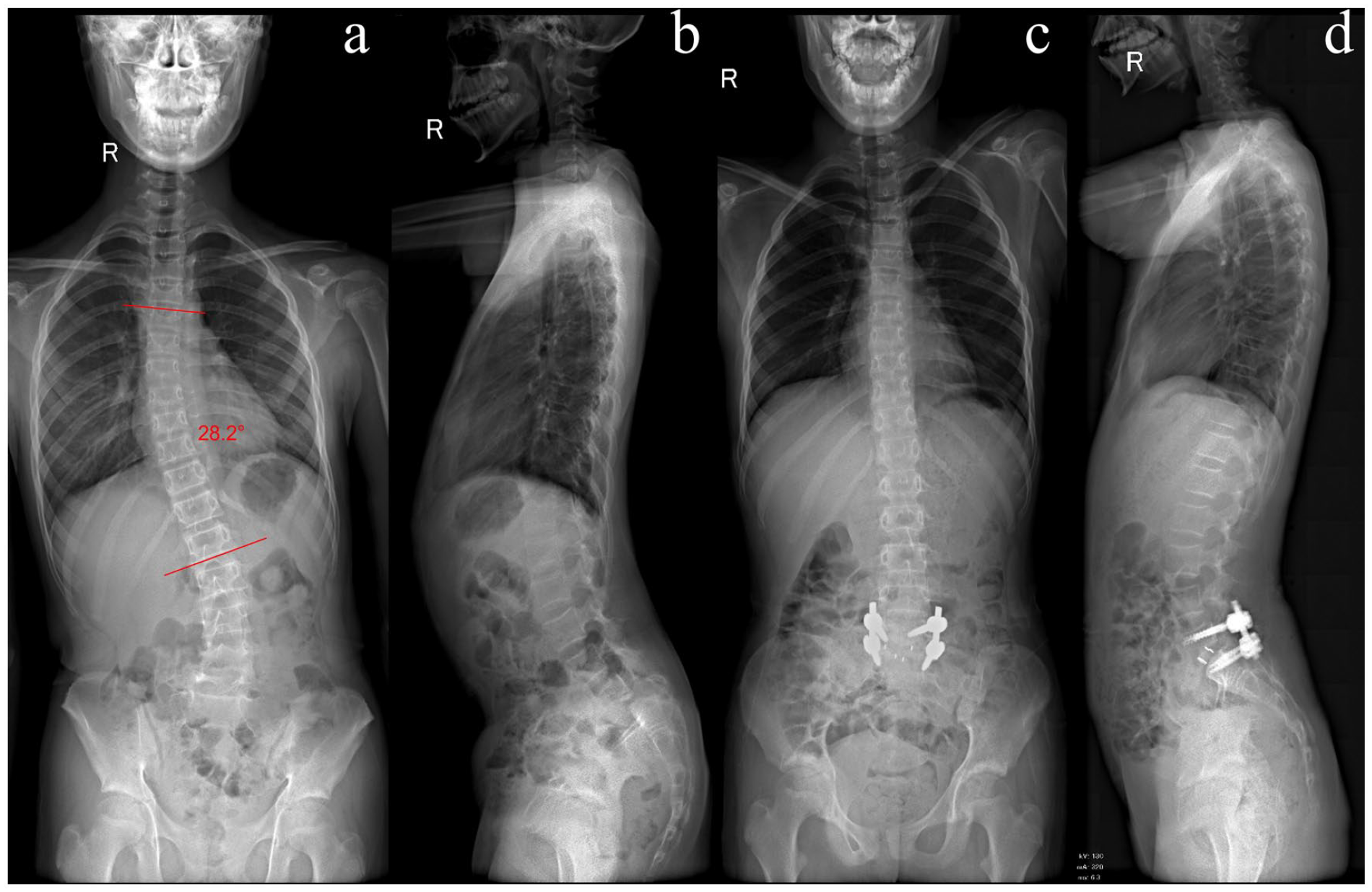
An 11-year-old female with dysplastic spondylolisthesis. (a) Preoperative posteroanterior radiography shows a small and long curve (28.2°). (b) Lateral radiography shows a grade IV spondylolisthesis. (c) Postoperative posteroanterior radiography shows complete scoliosis correction. (d) Postoperative lateral radiography shows significant improvement of the spondylolisthesis.
Complications occurred in six patients (10.7%), lower limb weakness in three, wound infection in two, and cerebrospinal fluid leakage in one. All complications resolved after treatment within 1 month of surgery. No permanent complications occurred.
Discussion
In this study of 56 children with dysplastic spondylolisthesis who underwent surgery for spondylolisthesis reduction and kyphosis correction with internal fixation, 36 patients (64.3%) also had scoliosis. This rate is higher than previously reported ones. Libson et al. 7 reported a scoliosis incidence of 43.1% in patients with symptomatic spondylolisthesis. In a study of 190 adolescents with lumbar spondylolisthesis, Seitsalo et al. 5 noted that 48% of the patients also had scoliosis, a rate significantly higher than that of the general population. Another study reported that incidence of scoliosis was higher in patients with dysplastic spondylolisthesis than in those with the isthmic type (47% vs 25%). 6 In contrast to previous studies, we intentionally limited our analysis to children with dysplastic spondylolisthesis. The characteristics of dysplastic spondylolisthesis combined with the developmental growth that occurs in children may be the reason for our study’s higher incidence of scoliosis.
In our study, severe S1 dysplasia was a risk factor for scoliosis. Previous studies have pointed out that asymmetrical rotation of the olisthetic vertebra or isthmic defect creates an unstable foundation, which may contribute to development of scoliosis.11,12 This theory was first explained by Tojner 13 in 1963: the asymmetric foundation causes lateral shift of the olisthetic vertebra, which in turn causes the vertebra above the slip to rotate into torsional scoliosis. By analyzing computed tomography images of spondylolisthesis patients with scoliosis, Peterson and Wenger 14 confirmed that rotatory change at the L5 level might be the cause of scoliosis in children with spondylolisthesis. Du et al. 15 studied radiography and computed tomography images of 36 adolescent patients with both spondylolisthesis and scoliosis and reported that patients with scoliosis have a more obvious L5 tilt and rotation. S1 dysplasia results in increased susceptibility to asymmetric rotation, which leads to scoliosis. In our study, patients with scoliosis had a higher degree of sacral dysplasia than those without scoliosis. This association was confirmed in our multivariate logistic regression analysis. Therefore, severe S1 dysplasia appears to be a risk factor for scoliosis based on our study and previous ones.
Severe spondylolisthesis usually provokes more severe clinical symptoms. Patients with scoliosis in our study had a higher SP than those without scoliosis. Previous studies have reported an association between clinical symptoms and scoliosis in spondylolisthesis patients. In a large cohort study of 1743 patients with spondylolisthesis, Libson et al. 7 reported that the incidence of scoliosis was significantly higher in those with low back pain than in those who were asymptomatic. They hypothesized that muscle spasms related to low back pain may contribute to development of scoliosis. This hypothesis has become widely accepted by other researchers 11 and may also explain our findings. Patients with a higher SP tend to have more severe symptoms, such as lower back pain, which can cause muscle spasms that lead to scoliosis.
We also observed lower PI and SS in patients with scoliosis. A possible explanation for this is that patients with dysplastic spondylolisthesis have a sagittal spine imbalance.16,17 Some researchers believe that coronal malalignment is associated with sagittal imbalance. 18 Imbalance may cause gait and posture abnormalities that result in scoliosis. This suggests that sagittal imbalance is a contributing cause of scoliosis in patients with dysplastic spondylolisthesis. However, further research is required to confirm.
Most cases of spondylolisthesis-induced scoliosis can be corrected or alleviated by spondylolisthesis surgery. Scoliosis improvement was achieved in all patients in our study, which is consistent with previous studies. In several case reports, scoliosis correction was achieved by surgical reduction of the spondylolisthesis.19 –22 These authors recommended observation for postoperative scoliosis rather than surgery, even for non-flexible curves or those with a large Cobb angle. Curve resolution may have occurred because of muscle spasm relief and correction of the unstable lumbosacral foundation with reduction and internal fixation. In our study, 61.1% of patients with scoliosis had a Cobb angle of less than 10° at last follow-up. In the remaining patients, degree of scoliosis correction was lower, possibly because rotation of the slipped vertebra was not corrected. Notably, none of the patients in our study had idiopathic scoliosis, which should be managed as a separate entity. 11
This study had several limitations. First, selection bias may have been present because of its single-center retrospective design and relatively small sample size. Moreover, some patients were followed for only 2 years. Future large-scale multicenter prospective studies are warranted to further clarify the association between dysplastic spondylolisthesis and scoliosis.
Conclusion
Approximately 64.3% of patients with dysplastic spondylolisthesis had concurrent scoliosis. Severe S1 dysplasia and high SP may be risk factors for developing scoliosis. The probability of scoliosis was high in patients with SDSG index >0.288. Scoliosis resolved spontaneously after spondylolisthesis reduction and fixation in most patients.
Footnotes
Author contributions
J.J. designed the study and analyzed the data. Z.Y. and X.Z. revised the paper and supervised the study. All authors contributed to the article and approved the submitted version.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This retrospective study of the Chinese cohort was approved by the ethics committee of Beijing Children’s Hospital, Capital Medical University, and National Center for Children’s Health (IEC-C-006-A04-V.06, (2022)-E-232-R).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Beijing Natural Science Foundation (L222095), Beijing Hospitals Authority Youth Program (QML20211209), and Beijing Talents Fund (2018000021469G275).
Informed consent
Consent forms were signed by the patient’s parents.
