Abstract
Background:
There are no clearly defined guidelines for the management of distal radial physeal injuries. We aimed to identify the risk factors for patients with distal radial physeal trauma for the risk of deformity, physeal closure, and revision procedure and develop a predictive model.
Methods:
The retrospective study included patients less than 16 years old with displaced distal radial physeal injuries treated between 2011 and 2018 across five centers in the United Kingdom. Deformity was defined as a volar angulation of >11°, dorsal angulation of >15°, a radial inclination of <15° or >23°, or positive ulnar variance. Presence of a bony bar spanning the physis was considered physeal closure.
Results:
This study comprised of 479 patients. In that, 32 (6.6%) patients had a second procedure. Also, 49 (10.2%) patients had closure of physis, and 28 (6%) patients had deformity at the end of follow-up. The occurrence of deformity had a strong correlation with age (p = 0.04) and immobilization duration (p = 0.003). Receiver operating characteristic analysis showed that age >12.5 years (p = 0.006) and sagittal angulation of >21.7° (p = 0.002) had a higher odd of deformity. Immobilization for <4.5 weeks (p = 0.01) had a higher revision rate. The nomograms showed good calibration, with a sensitivity of 70% and specificity of 75%.
Interpretation:
The nomograms provide accurate, pragmatic multivariate predictive models. Anatomical reduction is recommended in patients >12.5 years of age with >22° of dorsal angulation with cast immobilization for no less than 4.5 weeks. Any revision procedure should be performed within 11 days from the date of injury to reduce the risk of physeal damage.
Introduction
Distal radial physis (DRP) fractures are among the most common injuries in children; 80% occur after 10 years of age.1–6 The DRP contributes to 75% of the longitudinal growth of the radius. It carries a vital role in remodeling following injury. Despite substantial growth potential, remodeling varies among children. Larsen and his colleagues2,6 found that the remodeling potential for correction decreases with increased angulation and age more than 10 years.
Controversy exists regarding the optimal management of DRP injuries, and there are no universally accepted guidelines for managing Salter-Harris type distal radial fractures. Some investigators recommend operative intervention of these injuries over the age of 10 years.7–11 On the other hand, most authors recommend non-surgical intervention. 6 There is paucity of evidence about the percentage apposition or the angular deformity threshold (which is considered acceptable and will remodel).1,12 Also, while some authors note that these injuries heal without complication for the most part, 13 others describe adverse outcomes.1,8,14,15 Finally, there is no consensus regarding the incidence of growth arrest; some suggest infrequent premature closure while others advocate the opposite.16,17 Given the importance of achieving satisfactory outcomes, defining the maximum acceptable amount of displacement/translation and angulation would act as a useful guide to help patients and surgeons make a shared decision. 6
In summary, there are no studies predicting the radiological outcome of these injuries nor are there any studies investigating the correlation between poor radiological outcome and function. Nonetheless, the standard practice of assessment of these fractures with radiographs and the heightened parental anxiety following a radiological deformity cannot be ignored. In addition, the emotional impact of a repeat procedure and the financial, medical, and social implications that follow a second procedure cannot be emphasized enough. Hence, we believe that a study predicting the radiological outcome and the risk of second procedure would be able to address these issues and aid in possible evolution of consent/treatment process and shared decision-making.
We aim to identify factors associated with the risk of deformity at the end of follow-up, risk of second procedure, and apparent physeal closure. Our secondary aim was to develop a prediction model for patients with DRP injuries to aid shared decision-making. Drawing from the model, we can define a minimum acceptable standard for reduction and immobilization and propose management guidelines.
Methods
Following individual institution approval, we conducted this retrospective cohort study at five centers—Newcastle upon Tyne Hospitals NHS Foundation Trust, Northumbria Healthcare NHS Foundation Trust, South Tees NHS Trust, University Hospitals of Leicester, and Royal Stoke University Hospital.
Study population
The study population comprised of patients less than 16 years of age with isolated DRP injuries presenting between January 2011 and April 2019. We used a database from local coding departments to identify these patients.
Study design
Data were gathered retrospectively using online medical records. The information included were primary demographic data, date of admission, time to treatment, type of treatment, duration of immobilization, type of immobilization, duration of follow-up, and complications.
Radiographs were evaluated by investigators (S.K., H.H., A.F., G.M., B.E., and H.L.) at respective sites. To minimize the interobserver variability, all authors were trained by the lead author (S.K.) to perform radiological assessment and alignment in a uniform manner. Alignment in the sagittal and coronal planes was assessed both pre-operatively and post-operatively. In addition, we also recorded the pre-operative displacement and post-operative displacement. The percentage of displacement was calculated by measuring the width of uncovered radial metaphysis divided by the total width of radial metaphysis in sagittal plane on the lateral view. We also evaluated the radiographs for the presence of partial or complete premature closure of the physis. Finally, the length of follow-up was determined by reviewing clinic letters and radiographic follow-up.
Defining outcome variables
Our outcome variables were malalignment, premature closure of physis, and revision treatment. Malalignment was defined as a volar angulation of more than 11°, dorsal angulation more than 15°, a loss of radial inclination less than 15° or more than 23°, or positive ulnar variance.18–20 Since multiple databases showed that ulnar variance was typically negative in the pediatric population, we did not routinely obtain plain radiographs of the unaffected wrist. 21 Any second procedure performed to correct the loss of alignment following the first procedure was considered revision treatment. Finally, physeal closure was categorized as partial or complete. The presence of a bony bar surrounded by an otherwise normal physis on anteroposterior (AP) and lateral radiographs was considered partial physeal closure, whereas a bony bridge spanning the whole of the physis was considered complete physeal closure (Figure 1).
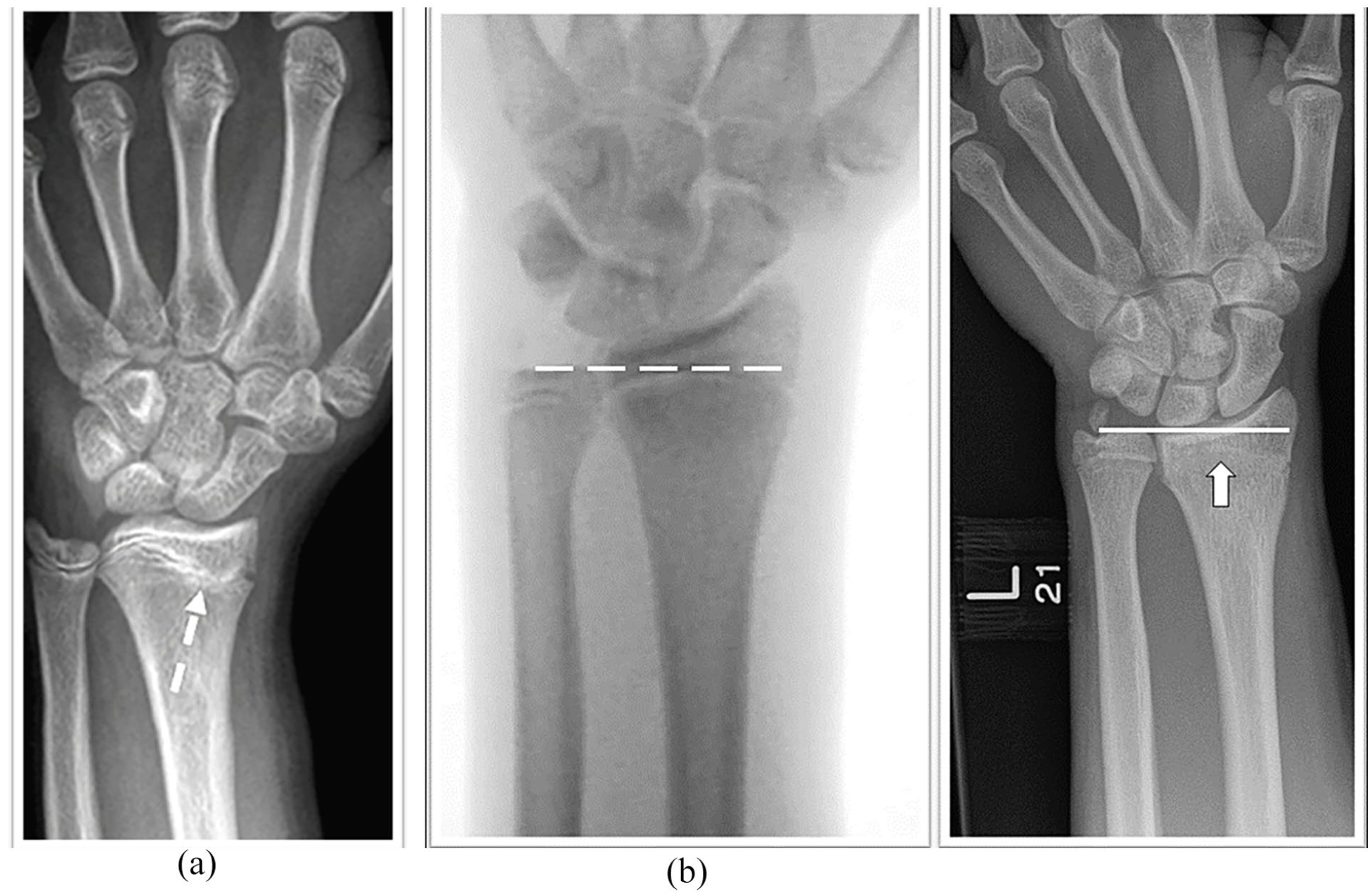
Radiographic example of physeal arrest in pediatric distal radius: (a) partial arrest with bony bar (white dotted arrow) and (b) intra-operative plain radiograph shown with negative ulnar variance. At 1-year follow-up, presence of neutral ulnar variance with complete physeal arrest (white solid arrow).
Statistical analysis
All statistical analyses were performed using R version 3.5.0 software. 21 Categorical variables were all expressed as absolute numbers and percentages. We used mean, median, and standard deviation (SD) for continuous variables. Significance level for the hypothesis tests was set at p < 0.05.
Logistic regression was performed to assess the effect of various prognostic factors on premature physeal closure, revision surgery, and distal radius deformity. Bootstrap with 5000 resamples was supplied, and a 95% confidence interval was used. Also, the Hosmer-Lemeshow goodness of fit test was used to calibrate the model. Receiver operating characteristic (ROC) curve was used to define the minimum acceptable standards for guidelines.
Building a predictive model
We provided predictions for the occurrence of distal radius deformity, risk of a revision procedure, and risk of apparent physeal closure based on significant clinical and radiological factors from the binary logistic regression model. The prediction model was built in the form of nomogram. 22
Using the bootstrapping method, the data were taken and repeatedly resampled to produce numerous simulated samples of the same size as the original dataset.
Each of these simulated samples has unique characteristics, such as a mean and other metrics. For these resamples, the nomogram calibration was then plotted.
Perfect predictions are referred to as 45° lines. Nomogram overprediction was indicated by points estimated below the 45° lines, whereas nomogram underprediction was indicated by points estimated above the 45° lines. The nomogram prediction can therefore be calibrated for resamples through boot strapping.
To justify the use of this prediction model in clinical practice, the predictive performance of each model was assessed using the area under the ROC curve.
Using the nomogram
Each nomogram consists of two scales. The first scale (Score) is the point assigned for each variable. Above this (Score) scale, the nomogram consists of rows corresponding to each variable included in the model. Each variable is assigned a point value (upper scale, Score) based on the patient’s clinical and radiological characteristics. A vertical line is made between the appropriate variable and the scale (Score). The assigned score for all variables is summed, and the total is located on the bottom scale labeled as the “Total score.” Once the total is located, a vertical line is made between total score and probability.
Results
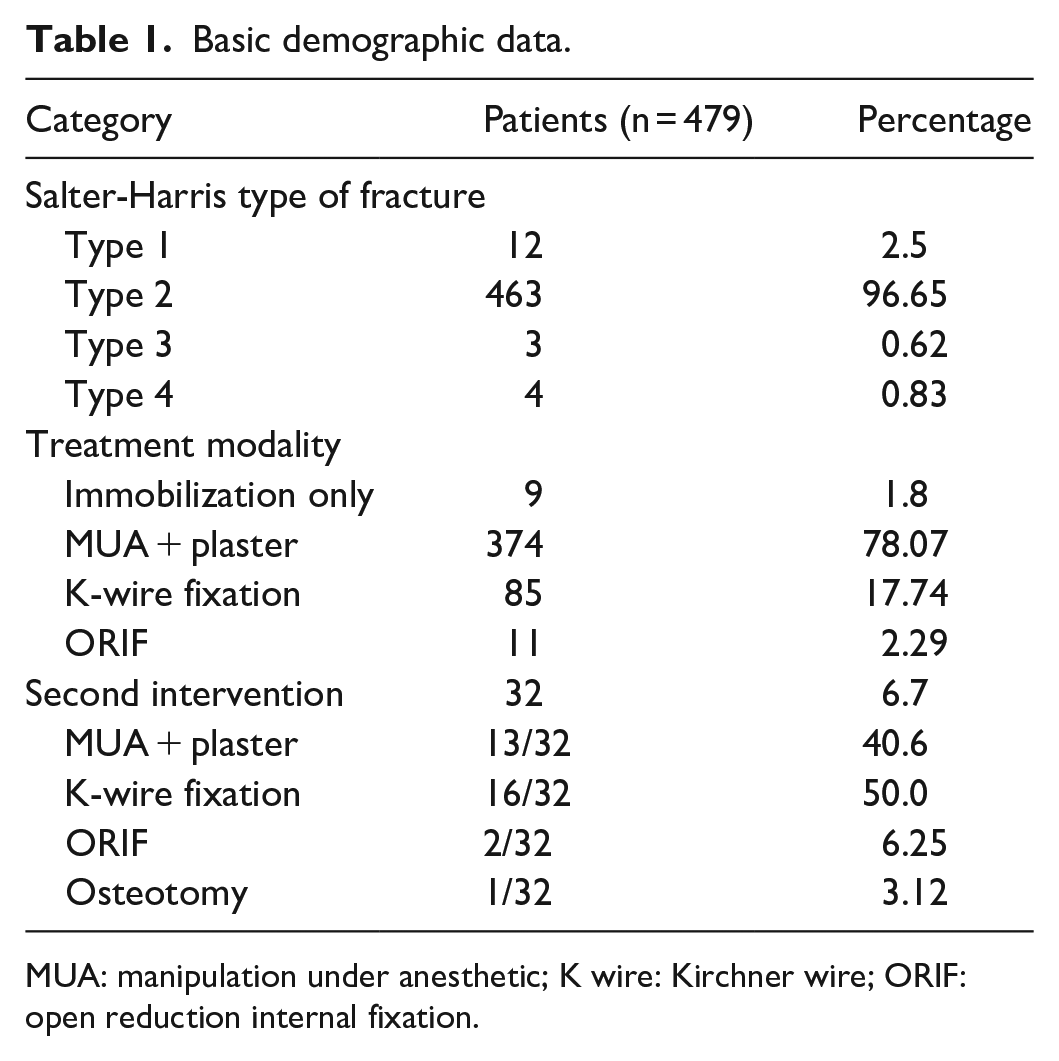
Our study comprised of 479 patients presenting with isolated DRP injuries with a median age of 13 years (9–15); 96% (463/479) of the patients had a Salter-Harris Type II injury. In all, 78% (374/479) of patients were treated with manipulation and immobilization in below elbow plaster. The median duration of immobilization was 4 weeks (2–6 weeks). The median follow-up duration was 9 weeks (6–205 weeks); the rest of the data is summarized in Tables 1 and 2.
Basic demographic data.
MUA: manipulation under anesthetic; K wire: Kirchner wire; ORIF: open reduction internal fixation.
Clinical and radiological demographic data.
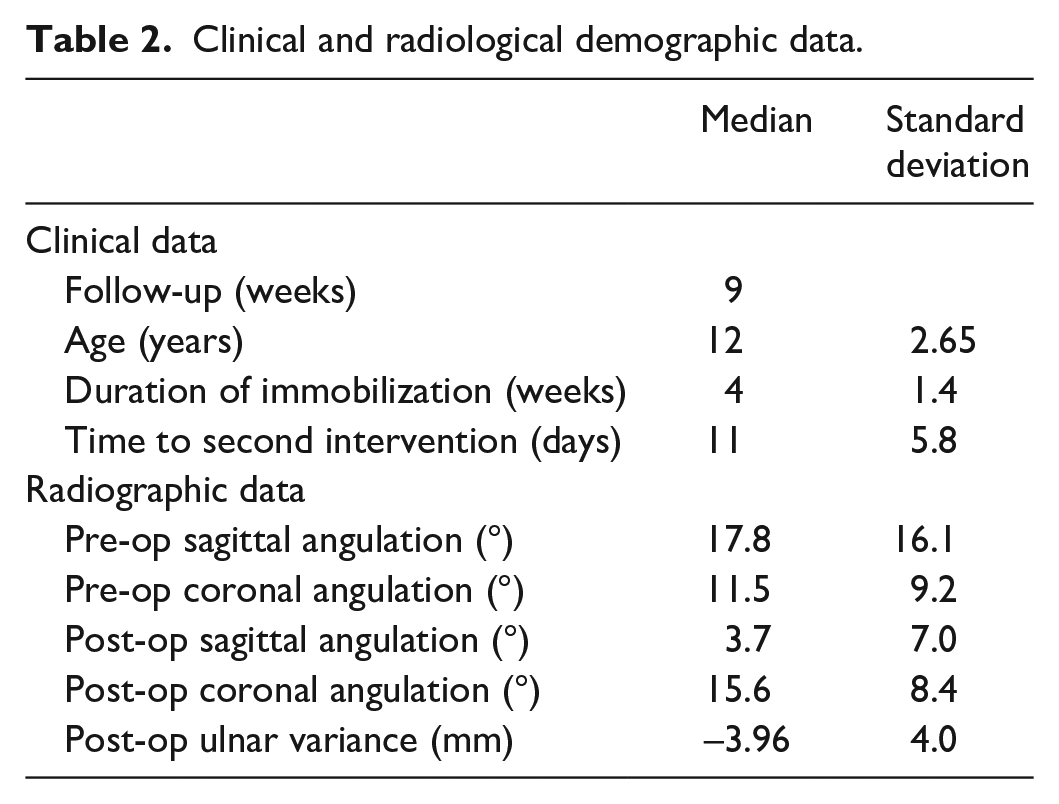
The incidence of second intervention due to loss of alignment after the first treatment was 6.6% (32/479). The median time to the second intervention was 11 days (SD = 5.8); 50% of these patients (16/32) were treated with K-wire fixation, whereas 40.6% (13/32) of patients underwent further manipulation and re-casting and 6.3% (2/32) had open reduction internal fixation (ORIF). Furthermore, 3.1% (1/32) needed osteotomy.
In the cohort who had second procedure, 78% of patients had malalignment as per criteria mentioned previously. Following manipulation, median lateral angulation was 2° volar (range = 10° dorsal to 12° volar) and median radial inclination was of 18° (range = 12–24). Following manipulation, there was a mean 9° (SD = 12.5°) change in lateral angulation and 4° (SD = 5°) change in AP angulation, following second procedure.
The deformity incidence was 5.8% (28/479). Loss of radial inclination (<15° (9/28)) and persistent distal radial dorsal angulation (>15° (5/28)) were the most common deformities (Table 3). The mean follow-up in the cohort of patients with deformity was 30 weeks (SD = 6.4).
Unsatisfactory radiological outcomes.
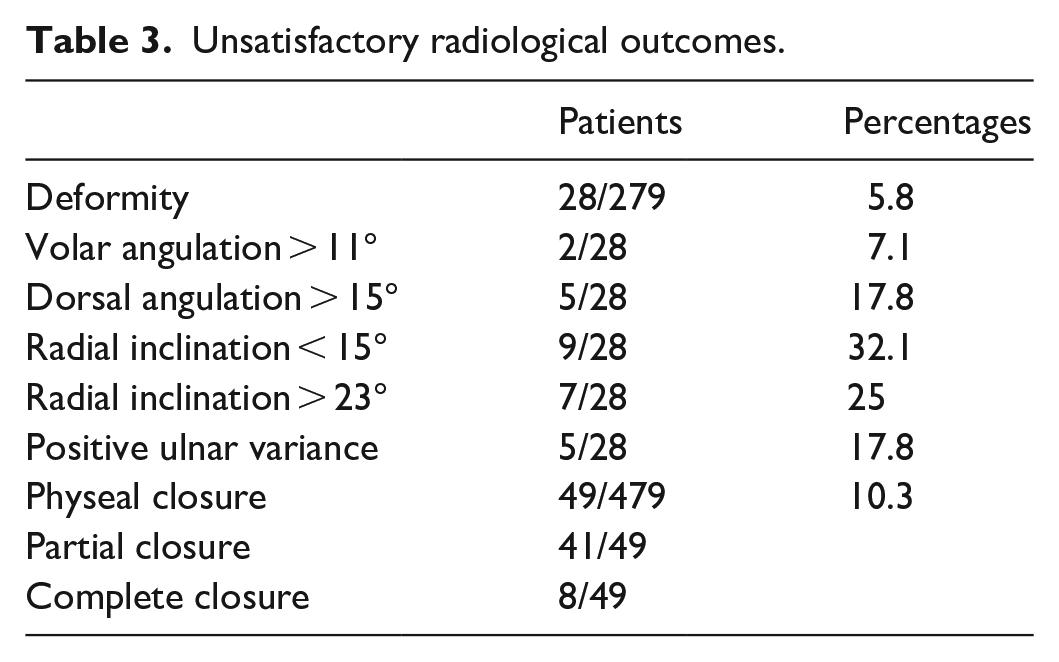
The overall incidence of physeal closure was 10.2% (49/479) at the end of follow-up. Among these, 41 (8.5%) patients had partial physeal closure and 8 (1.7%) patients had complete physeal closure. In all, 50% (4/8) of patients with complete physeal closure developed deformity, whereas only 1 out of 41 patients with partial physeal closure developed deformity (Table 3). Individual patient data in this subgroup were listed in Table 4.
Demographic, treatment, and radiological data of patients with physeal injuries.
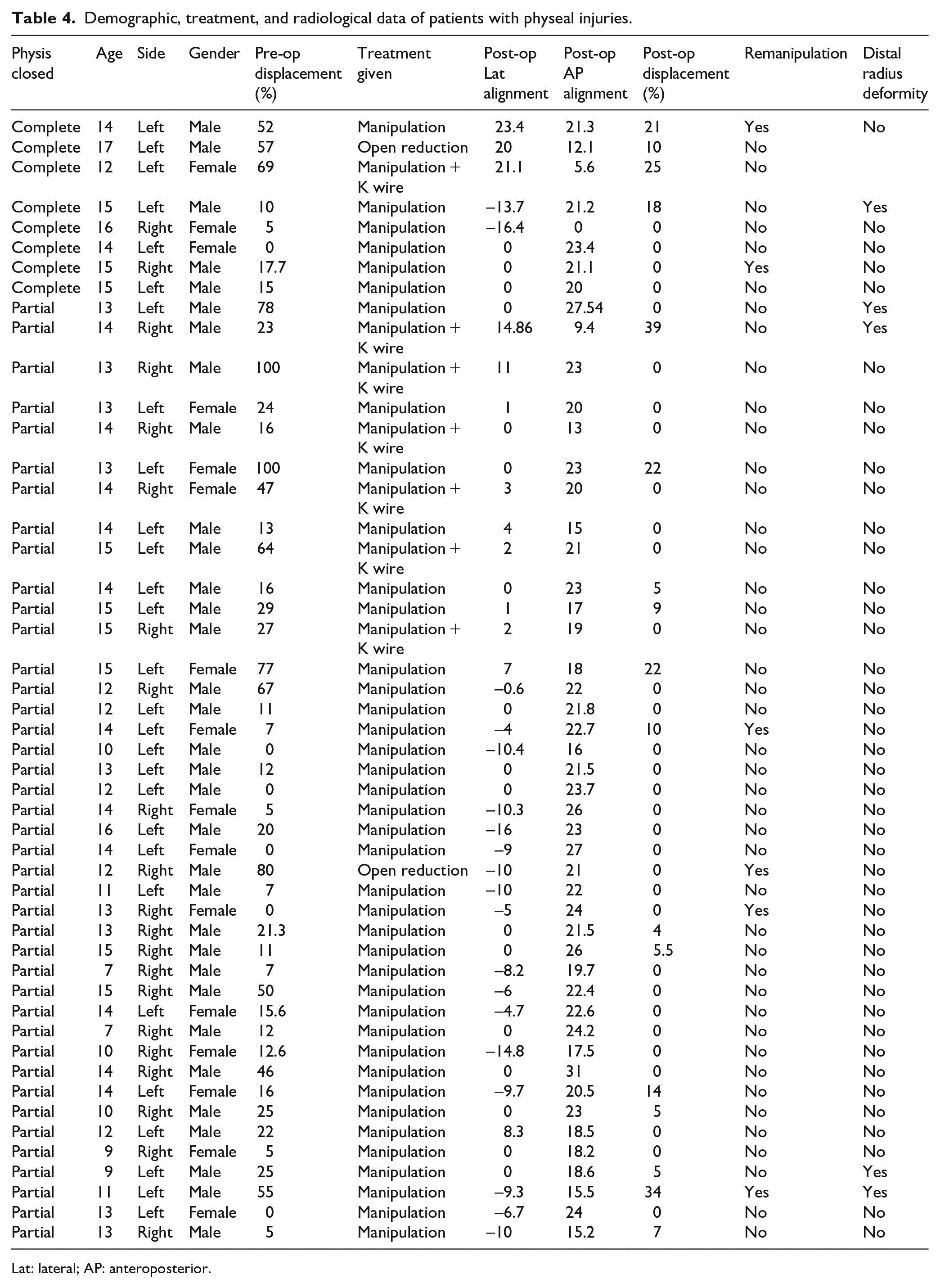
Lat: lateral; AP: anteroposterior.
In our study, most of the patients had dorsal displacement (88.3% (423/479)); 8.8% (42/479) of patients had volar displacement and 2.9% (14/479) had neutral tilt. Incidence of deformity was 1.25% (26/423) in dorsally displaced group and 2% (1/42) in volar displaced group. Remanipulation was higher in volar displaced group, 11.9% (5/42), compared to 5.9% (25/423) in dorsally displaced group. Similarly, incidence of physeal damage was 8.7% (37/423) in dorsally displaced group and 19% (8/42) in volar displaced group. However, our analysis showed that the direction of deformity had no impact on occurrence of deformity (p = 0.6), physeal damage (p = 0.7), and remanipulation (p = 0.1).
Distal radius deformity
We found that higher odds of distal radius deformity were associated with age (p = 0.03) and post-operative translation (p = 0.02). Thus, post-operative reduction, as evidenced by post-operative lateral angulation and translation, was an important factor associated with higher distal radius deformity odds. Immobilization duration had no significant association with deformity.
Revision treatment
The odds of having a second intervention were higher with a greater post-operative translation (p = 0.0001). Thus, a poor reduction was associated with increased risk of further displacement necessitating a second intervention.
Risk of physeal damage
On analyzing the impact of various factors on parts of physeal closure, we identified that age (p = 0.0001), time to management (p = 0.01), and pre-op displacement (p = 0.001) were associated with higher odds of physeal closure.
Nomogram construction and validation
Table 5 lists the logistic regression models’ results for predicting all three outcomes (i.e. deformity, physeal injury, and risk of revision treatment).
Multivariable logistic regression model for each outcome.
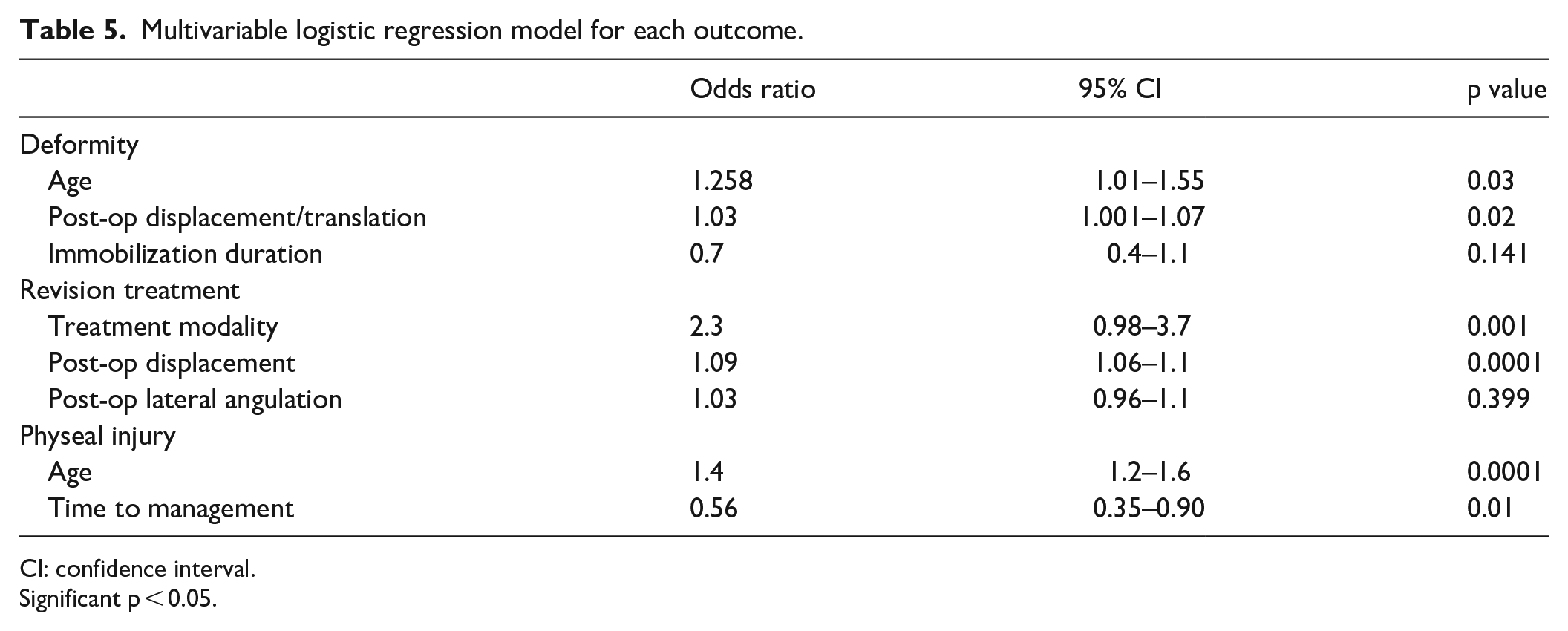
CI: confidence interval.
Significant p < 0.05.
Three nomograms based on these models were developed and appear in Figures 2–4. Below each nomogram is its corresponding predictive accuracy calculated using the area under the ROC curve. The area under the ROC curve measuring the model’s overall predictive accuracy was 0.80 for the nomogram predicting the risk of distal radius deformity, 0.87 for the nomogram predicting the risk of revision treatment, and 0.80 for the nomogram predicting complete physeal closure. Figure 5 shows calibration plots for risk of revision treatment, distal radius deformity, and physis closure. The calibration plots show that points scattered close to diagonal line indicate good calibration.
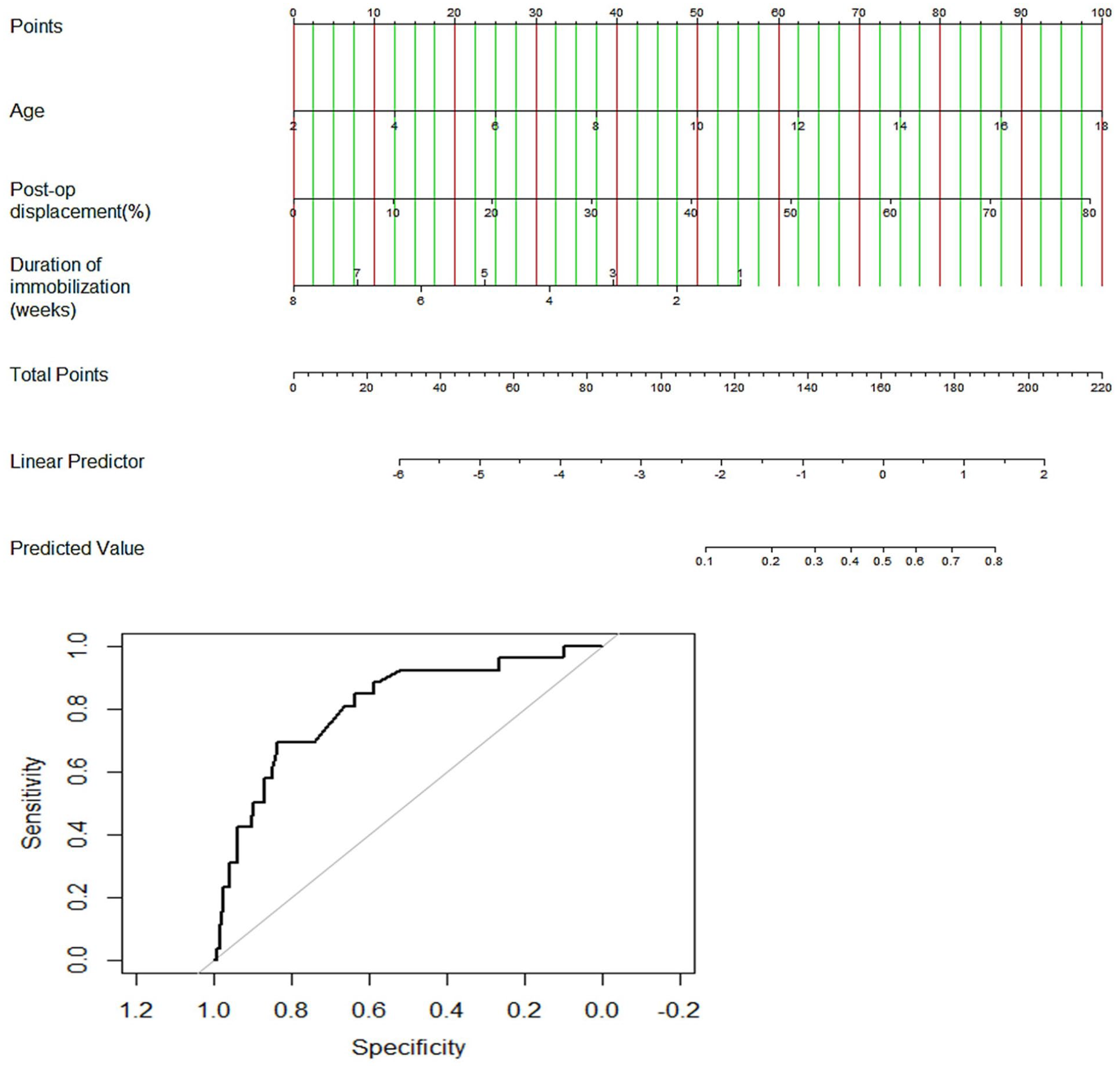
Nomogram to predict likelihood of distal radius deformity.
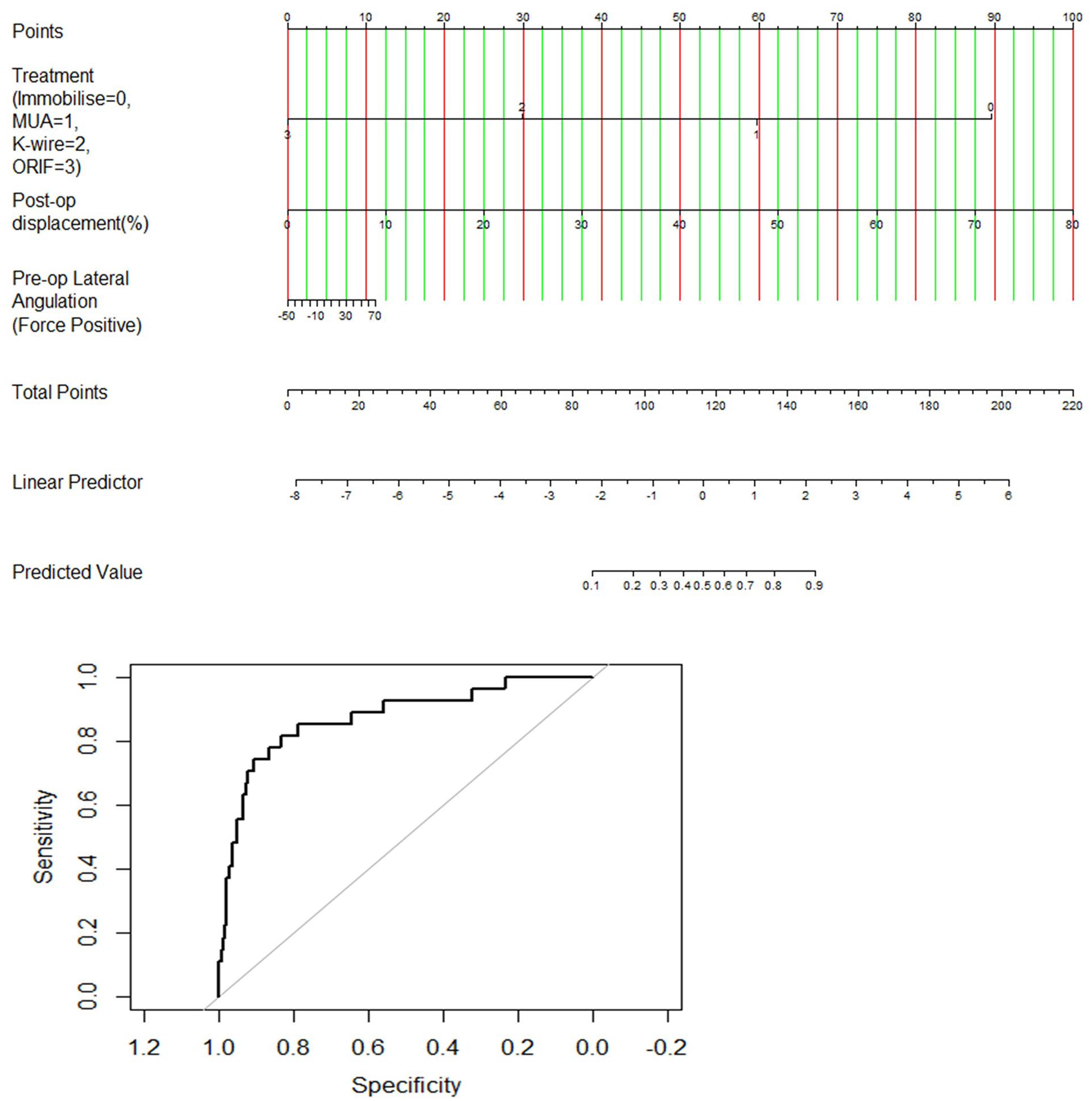
Nomogram to predict likelihood of revision treatment.
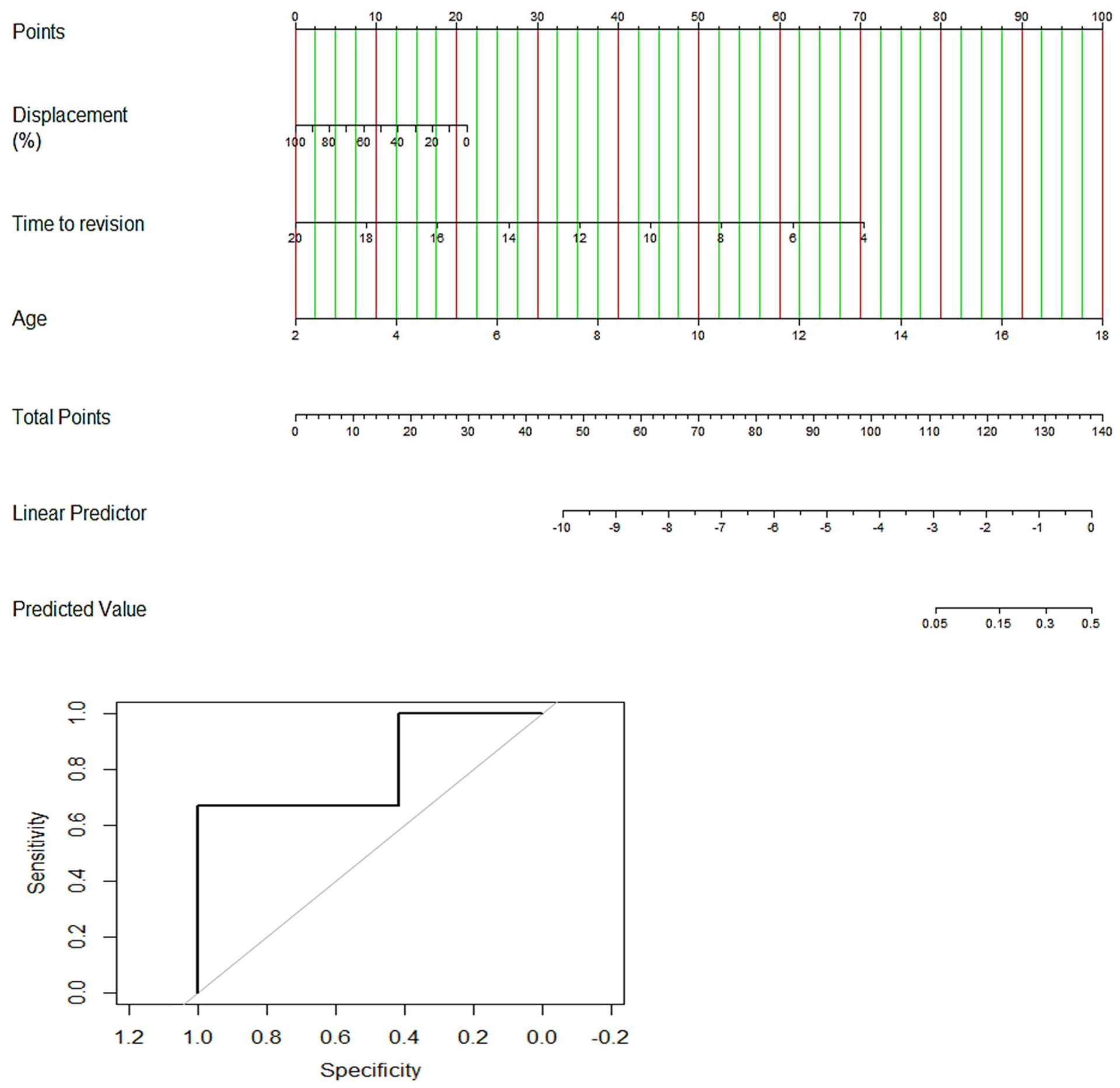
Nomogram to predict likelihood of physeal injury.
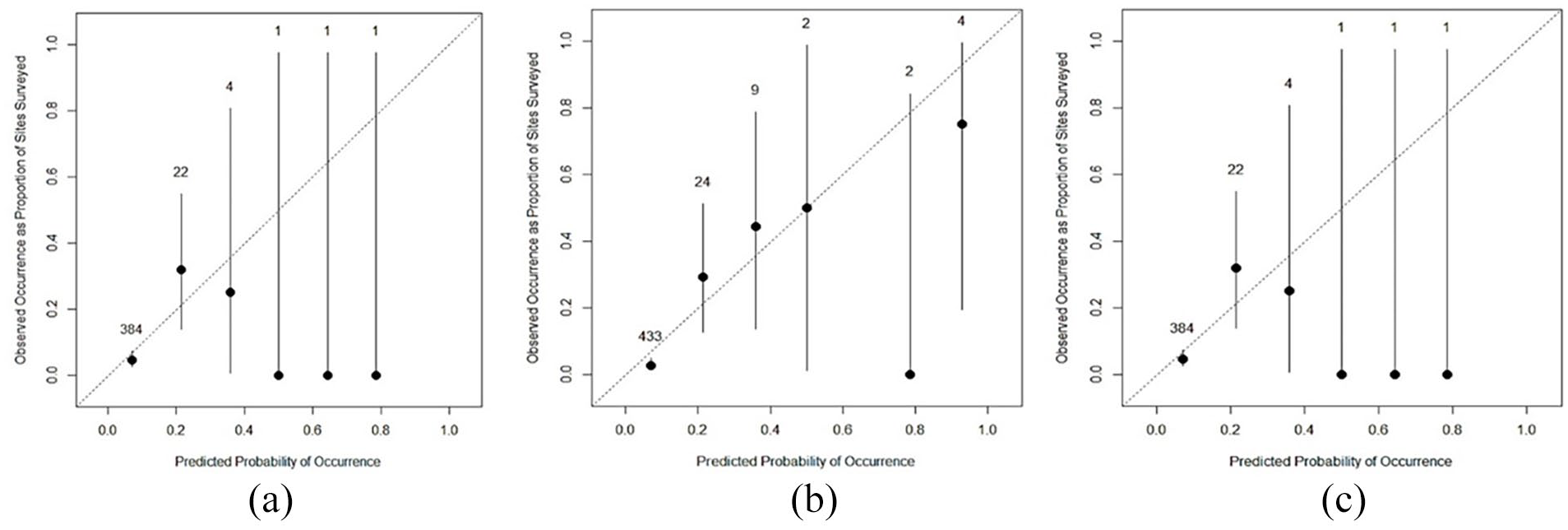
Calibration plots for (a) distal radius deformity, (b) revision treatment, and (c) physeal closure: observed probabilities plotted against predicted distal radius deformity, revision treatment, and risk of physeal closure, respectively.
Defining minimum acceptable standards and guidelines
We have used the ROC curve to identify minimum acceptable standards of the prognostic factors (identified through logistic regression) associated with the outcome variables.
Distal radius deformity
We found that age more than 12.5 years (sensitivity: 80%; specificity: 60%, p = 0.006) or pre-operative sagittal angulation of 21.7° (sensitivity: 70%; specificity: 60%, p = 0.01) were associated with higher odds of developing deformity.
Risk of revision treatment
Our analysis showed that immobilization for less than 4.5 weeks (sensitivity: 80%; specificity: 70%, p = 0.001) had an association of revision treatment, and post-operative translation of more than 5% (sensitivity: 72%, specificity: 82%) was associated with a higher rate of revision surgery. We would like to clarify here that this includes cohort of patients in whom cast had deliberately been applied for a shorter length of time, excluded from the analysis are patients who had them removed from their casts early (11–12 days) to have the revision procedure.
Risk of physeal closure following revision surgery
According to our analysis, a revision procedure after 11 to 12 days from injury was associated with a higher risk of physeal closure (sensitivity: 78%, specificity: 77%).
Discussion
There is no consensus regarding optimal management of DRP injuries, and there is significant conflict regarding acceptable alignment and length of immobilization. The unknown remodeling potential of these injuries necessitates identification of factors predicting/preventing the risk of treatment failure and provides guidelines to aid the decision-making process.
Deformity may complicate DRP injuries.1,6,12 The incidence of the distal radius deformity in this cohort was 5.8%. Nietosvaara et al. 1 stated that 48% of Salter-Harris 1 and 2 distal radius fractures healed in malunion, albeit usually mild. However, their study did not provide specific radiographic criteria for mild malunion. Nevertheless, they mentioned that 13% of the cases healed with more than 20% displacement and 14% healed with at least 10° angulation. Even though we report a lower incidence of deformity, this is because we have specific radiographic criteria to define malunion in contrast to Nietosvaara et al. By comparison, Lee et al. 12 state that the deformity rate was 7%—more in keeping with the findings of this study.
Our analysis showed that higher odds of developing distal radius deformity were associated with older age (p = 0.03), inadequate reduction as evidenced by post-op sagittal alignment (p = 0.002), and post-operative translation (p = 0.02). Our findings are similar to Larsen et al. 6 and Houshian et al., 2 who found that the potential for correction is decreased with increased angulation and age over 10 years. Although boys tend to have longer growth remodeling potential in comparison to girls of same age, this does not reflect in our analysis as a significance risk factor.
Loss of alignment following initial treatment is a recognized complication following a DRP injury. Miller et al. 23 found that the incidence of displacement following closed reduction could be as high as 39%. Furthermore, Houshain et al. 2 have described that a pre-operative displacement of greater than 50% has a higher risk of loss of position and needs to be monitored closely in the post-operative period. In our study, the incidence of loss of alignment leading to the second procedure was 6.6%. Our statistical analysis showed that the odds of displacement were higher with inadequate reduction (p = 0.001). Similarly, McQuinn and Jaarsma 14 concluded that pre-operative displacement of more than 50% and the inability to achieve anatomical reduction were significant risk factors for loss of reduction. With regard to anatomical reduction, our analysis suggests that anatomical reduction is a significant factor in preventing re-displacement.
Physeal arrest may complicate DRP injury. Larsen et al. 6 reported the risk of premature physeal arrest to be up to 4.3%. In this study, the incidence of partial physeal closure at the end of follow-up (mean = 9 weeks) was 10.2%, and complete physeal closure was 1.6%. This study uniquely categorizes physeal closure into partial and complete. In addition to this, our analysis showed that the risk of physeal closure was associated with age (p = 0.0001), delayed time to management (p = 0.017), and severe pre-op displacement (p = 0.001).
Larsen et al. 6 commented that no studies had analyzed factors associated with physeal arrest that could be altered to prevent this. Our analysis showed that any revision procedure performed more than 11 days after injury had a higher risk of physeal damage (sensitivity 78%, specificity 77%). We believe our study uniquely identifies variables associated with physeal closure, identifies patients at a high risk of physeal closure, and provides guidelines at the time of management to prevent the risk of physeal injury/closure.
When caring for children with DRP, to date, there is no model to facilitate patient-specific decision-making. Hence, we have developed a prediction model which we believe uniquely presents clinicians with the possibility to predict the risk of deformity, apparent physeal injury, and risk of revision treatment. The models’ predictive accuracy has been assessed, and all three prediction models performed well. Our predictive models use readily available clinical variables, which allows for an easy use. In addition, our retrospective study clinicians reflect the prevalent variation practice such as duration for cast immobilization. This ensures that the findings are applicable to the majority of settings.
In our model, age, duration of immobilization, and post-op displacement were prognostic variables for distal radius deformity. An awareness of the three variables should serve as a guide for the physician treating these injuries regarding acceptable reduction and immobilization duration. Similarly, for patients who have had a loss of position following initial treatment, the model uses the combination of pre-op displacement and time to revision surgery to give the risk of physeal closure. This would guide the surgeon to time the second procedure and aid shared decision-making. Finally, for patients undergoing initial or revision treatment, the prediction model can act as a guide to choosing the modality of treatment and acceptable standards of reduction to reduce the risk of further revision treatment.
A potential limitation to this study is that most of the patients included in this study had a Salter-Harris Type II injury, leading to a bias toward predicting and managing this specific subtype of injuries. However, this reflects everyday occurrence, where Salter-Harris Type II injuries are the most common subtype. Second limitation is findings based on radiological outcomes and lack of patient-reported outcome measures. Nonetheless, the need to avoid a second procedure cannot be overemphasized due to medical, social, and financial repercussions, not to mention the patient dissatisfaction of having a poor radiological outcome or the need for revision surgery. 14 In addition, the deformity assessment was made at the end of follow-up rather than end of growth. This, though, reflects the current practice of not following the patients until the end of growth, hence provides a pragmatic guide to physicians to counsel the parents regarding the expected outcome at the time being discharged from care. Finally, this study has utilized radiographs physeal closure assessment instead of computed tomography (CT) or magnetic resonance imaging (MRI), hence indicate apparent physeal closure rather than true physeal arrest. However, we believe a nomogram based on radiographs would provide a better practical tool for identifying at-risk patients in an outpatient clinic who may benefit from enhanced follow-up or further investigations.
Conclusion
Our study shows that older age of occurrence was associated with occurrence of distal radius deformity, whereas younger age was associated with increased odds of physeal damage. Similarly, the quality of post-op reduction was a factor associated with higher odds of further displacement and is a significant risk factor for need for second intervention and occurrence of deformity. Finally, other factors associated with physeal damage were time to management and pre-op displacement.
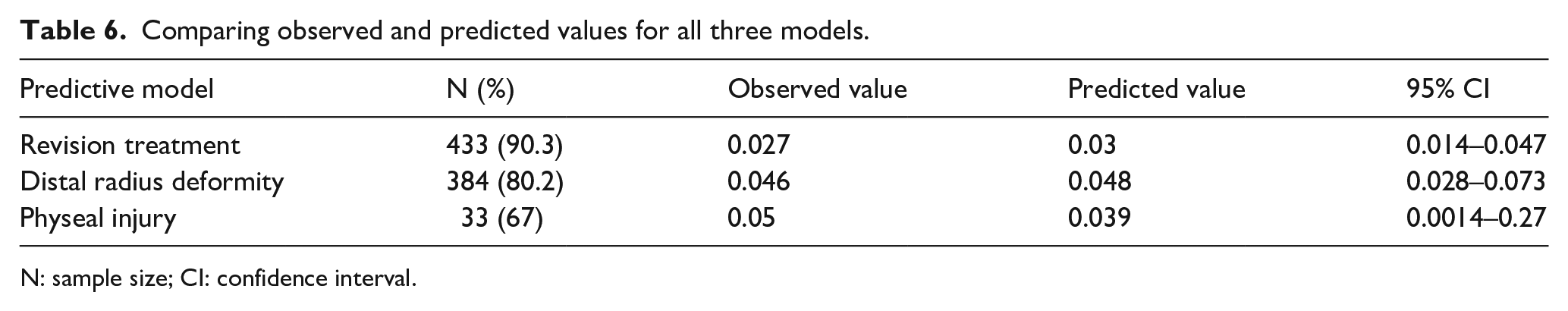
The models have displayed a reasonable predictive accuracy (Table 6). The predictive accuracy of models is between 80% and 87% (as assessed by areas under curve). These nomograms provide accurate, accessible, multivariate predictive models that should be validated prospectively in a large independent study. Furthermore, the study’s prediction model shows the possible evolution of a shared decision-making process.
Comparing observed and predicted values for all three models.
N: sample size; CI: confidence interval.
Summary
Based on our findings, we recommend achieving an anatomical reduction (<5% displacement/translation) in patients over 12.5 years of age with more than 22° of dorsal angulation with cast immobilization for no less than 4.5 weeks. Any revision procedure should be performed as soon as possible and within 11 days from the date of injury to reduce the risk of physeal damage.
Footnotes
Author contributions
K.S., E.W., V.S., A.A., and H.P. contributed to study concepts. K.S., C.H.H., A.A., and H.P. contributed to study design. K.S., C.H.H., F.A., M.G., E.B., and L.-W.H contributed to data acquisition. K.S., C.H.H., E.W., V.S., A.A., and H.P. contributed to quality control of data and algorithms. K.S. and C.H.H. contributed to data analysis and interpretation. K.S. and C.H.H. contributed to statistical analysis. K.S. contributed to manuscript preparation. K.S., C.H.H., F.A., M.G., E.B., and L.-W.H. contributed to manuscript editing. Manuscript review: K.S., C.H.H., F.A., M.G., E.B., L.-W.H., E.W., V.S., A.A., and H.P. contributed to manuscript review. K.S. is the principal investigator.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
This study was reviewed and approved by our institutional review board, and it was determined that written consent was not required for this study.
