Abstract
Purpose:
To explore the occurrence and risk factors of intraoperative hypotension during children’s Marfan syndrome scoliosis surgery and summarize the associated hemodynamic features and handle measures.
Methods:
Twenty-two Marfan syndrome scoliosis patients who underwent spinal surgery at Beijing Children’s Hospital were retrospectively reviewed between January 2001 and January 2020. Intraoperative hypotension is defined as the minimum mean arterial pressure ≤ 60 mm Hg. The patients were divided into the hypotension group and the control group. Clinical, radiographic, and operative data were compared between the two groups. The risk factors, hemodynamic features, and handle measures for intraoperative hypotension in Marfan syndrome scoliosis surgery were analyzed and summarized.
Results:
Twenty-two patients were included in the study, with a mean age of 11.4 years at initial surgery. The follow-up period ranged from 24 to 152 months. Intraoperative hypotension occurred in 14 cases, with an incidence of 63.6%. The proportion of pulmonary dysfunction in the hypotension group was higher than in the control group (100.0% vs 50.0%, p < 0.05). The spinal flexibility was significantly lower in the hypotension group (28.3% ± 14.2% vs 46.5% ± 11.5%, p < 0.05). Fourteen patients with intraoperative hypotension had decreased intraoperative systolic blood pressure 21.0%–50.0% compared with baseline. One patient had a transient decrease in the muscle strength of the lower limbs. No complications were observed during the follow-up.
Conclusion:
The incidence of intraoperative hypotension in Marfan syndrome scoliosis children who underwent surgery was 63.6%. The risk factors included preoperative pulmonary dysfunction and poor spinal flexibility. Comprehensive preoperative evaluation and effective hemodynamic handling measures should be undertaken to prevent further complications in children with Marfan syndrome scoliosis.
Introduction
Marfan syndrome (MFS) is an inherited connective tissue disorder with a mutation in the FBN-1 gene, which involves the skeletal, cardiovascular, respiratory, and ophthalmic systems, with an incidence of approximately 0.02%–0.03%.1,2 Scoliosis is observed in 60% of patients with skeletal system involvement. The conservative treatment approaches for Marfan syndrome scoliosis (MSS) are poor, with most patients needing surgical treatment.3,4 Moreover, MFS is often complicated by severe cardiovascular disease, thoracic deformities, and pulmonary dysfunction. MFS associated with hemodynamic instability is common during corrective spinal surgery. In recent years, many researchers have reported the orthopedic effect of MSS and its postoperative complications.4–6 According to our clinical observations, many MSS patients may developed intraoperative hypotension (IOH) during surgery. However, few studies have focused on the occurrence and control of IOH during MSS surgery.
In this study, we reported our outcomes of MSS patients, explored the risk factors associated with IOH in MSS surgery, and summarized its features and management measures. We hypothesized that preoperative pulmonary dysfunction and poor spinal flexibility may lead to the IOH during the MSS surgery.
Materials and methods
Patients
Twenty-two patients who underwent corrective spinal surgery at Beijing Children’s Hospital were retrospectively reviewed between January 2001 and January 2020. The inclusion criteria were as follows: (1) confirmed diagnosis of MSS 2 and (2) age < 18 years. The exclusion criteria were (1) loss of clinical or radiographic data, (2) history of cardiac surgery, and (3) follow-up of fewer than 24 months. Informed consent was obtained from the guardians of the patients, and the Institutional Ethics Committee approved the study of the Beijing Children’s Hospital, Capital Medical University (IEC-C-006-A04V.06, 2021).
During the MSS surgery, a standardized anesthetic protocol was used for all patients. Children were routinely ventilated using mask oxygen, connected to pulse oximetry, non-invasive blood pressure, and electrocardiography (ECG). Dexamethasone 0.1 mg/kg, atropine 0.01 mg/kg, propofol 2–3 mg/kg, sufentanil 0.5–0.6 μg/kg or fentanyl 1–2 μg/kg, cisatracurium 0.1–0.2 mg/kg or rocuronium 0.5 mg/kg were used to intravenous induction. Anesthesia was maintained with total intravenous anesthesia (TIVA) or combined intravenous and inhalation anesthesia (CIIA). 7 All children were given CIIA (11 cases) before 2016, while the other children were given TIVA after 2016. Tracheal intubation was performed, and volume-controlled ventilation was set to maintain end-tidal carbon dioxide between 35 and 45 mmHg. Children were monitored for esophageal temperature and Bispectral Index. Radial artery catheterization was used to monitor the systolic blood pressure (SBP) and diastolic blood pressure (DBP), and data were logged at 5 min intervals, including heart rate, oxygen saturation, and electrocardiogram.
Data collection
All the included patients’ clinical and radiographic data were reviewed and analyzed. The patients’ general data included sex, age, height, weight, and comorbidities associated with other systemic diseases. The body mass index (BMI) was calculated as weight/height 2 (kg/m2). Spinal parameters included the curve type of scoliosis, the apical vertebra (AV) location, and preoperative and postoperative Cobb angles. Spinal flexibility was calculated as (Cobb angle on the anteroposterior image − Cobb angle on the convex bending image)/Cobb angle on the anteroposterior image × 100%. The Haller index (HI) is the ratio of the transverse to anteroposterior chest wall diameters determined from a single axial computed tomography scan at the point of maximal pectus deformity (Figure 1). The AV deviation was calculated as (distance from the midpoint of the AV to the convex thorax)/(distance from the midpoint of the AV to the concave thorax) (Figure 2). The correction rate of the Cobb angle was calculated as (preoperative Cobb angle − postoperative Cobb angle)/preoperative Cobb angle × 100%. All patients underwent echocardiography before surgery, and the aortic sinus diameter was measured.
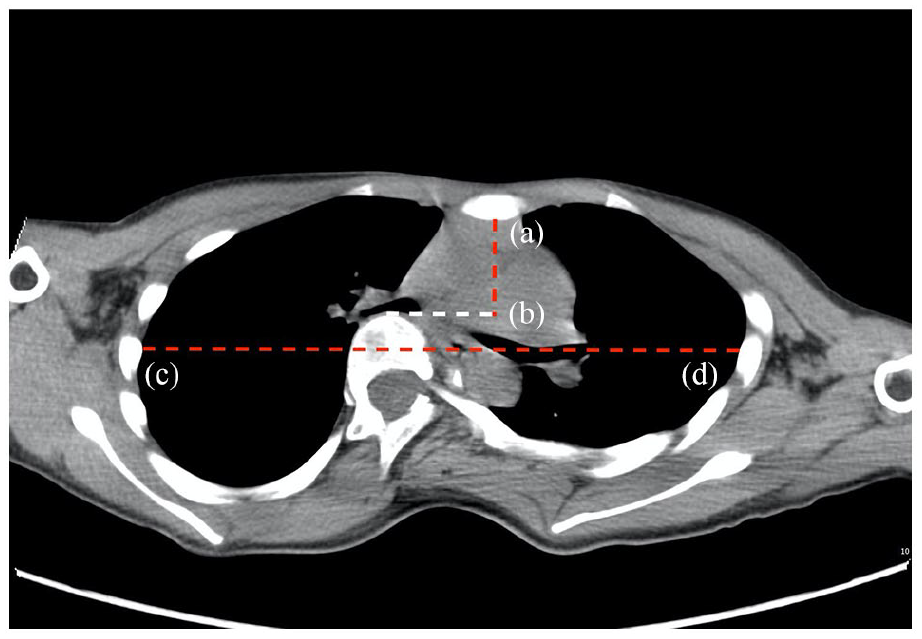
Haller index (HI) is the ratio of the transverse to anteroposterior chest wall diameters, determined from a single axial computed tomography scan at the point of maximal pectus deformity transverse diameter of the chest (line cd) to the distance between the posterior aspect of the sternum and the anterior portion of the vertebra (line ab): HI = cd/ab.
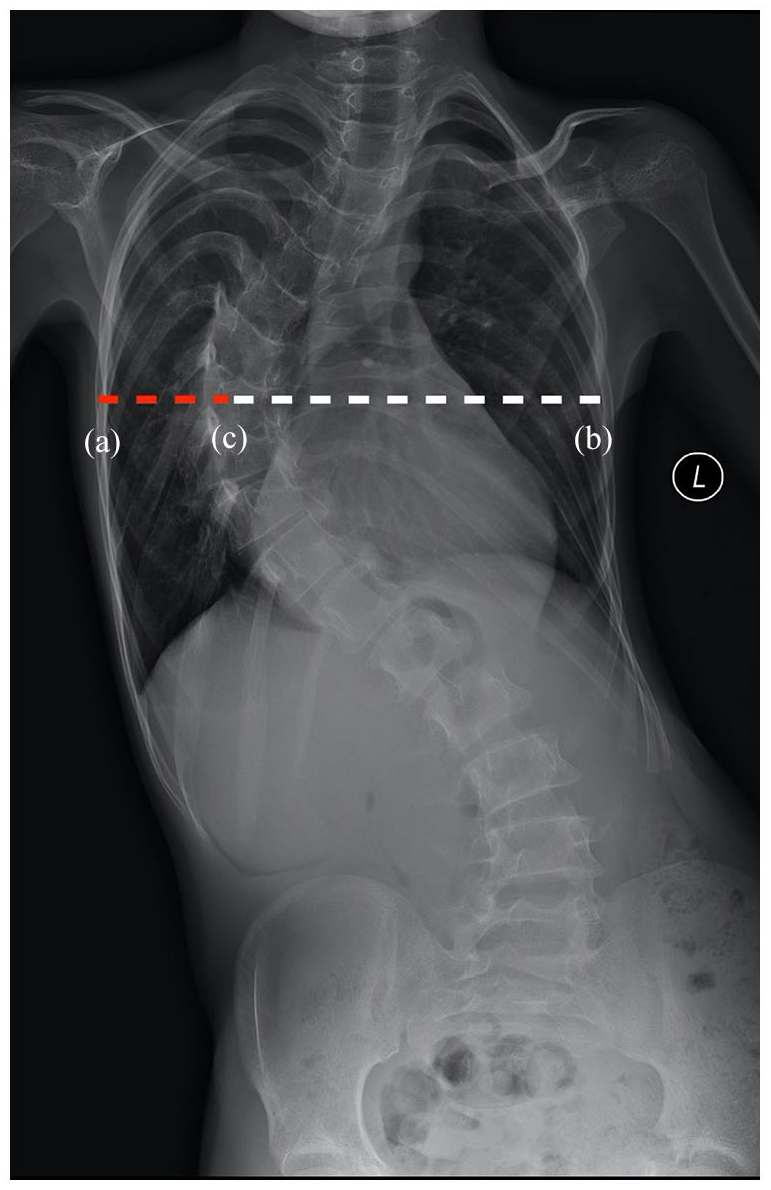
The ratio of apical vertebral (AV) deviation was calculated as the distance from the midpoint of the AV to the convex thorax (line ac)/the distance from the midpoint of the AV to the concave thorax (line bc): ac/bc.
Hemodynamic parameters were recorded, including the duration of anesthesia, American Society of Anesthesiologists (ASA) grade, pre-anesthesia SBP, maximum SBP, minimum SBP, pre-anesthesia DBP, maximum DBP, minimum DBP, intraoperative blood loss, and volume of infused crystalloids, colloids, red blood cells, plasma, autologous blood, and urine. The reduction of SBP from baseline was calculated as (pre-anesthesia SBP − minimum SBP) / pre-anesthesia SBP × 100%. The mean arterial pressure (MAP) was calculated as (SBP + DBP × 2) / 3. All the procedures were performed during the intraoperative neurophysiological monitoring (IONM) after 2016. The IOH was defined as a minimum MAP of ≤ 60 mm Hg.8,9 All 22 patients enrolled in the study were divided into hypotension group and control group according to the occurrence of IOH. The patients were followed up at 3, 6, and 12 months postoperatively and yearly. Neurological, cardiovascular, and cerebrovascular complications were recorded during surgery and at follow-up.
Statistical analysis
All data were analyzed using SPSS (version 24.0; IBM, Chicago, USA). Normal distribution and homogeneous variance tests were performed for all data. The normal distribution and homogeneity variance tests are presented as mean ± SD and compared using Student’s t-test. Non-normal distribution or inhomogeneity variance tests were presented as medians and interquartile ranges and compared using the Mann–Whitney U test. The counting data were expressed as percentages (%) and processed using the chi-square test or Fisher’s exact probability method. Differences were considered significant when the p-value was < 0.05.
Results
Characteristics of MSS patients
Twenty-two patients were included in the study with a mean age of 11.4 ± 3.3 years at initial surgery (9 boys and 13 girls). The follow-up period ranged from 24 months to 152 months. IOH occurred in 14 cases, with an incidence of 63.6%. Fourteen patients were in the hypotension group and eight in the control group. Twelve patients (85.7%) in the hypotension group and four (50.0%) in the control group underwent spinal fusion, respectively. The general preoperative data of the patients in the two groups are presented in Table 1. There were no differences in age, weight, height, or BMI between the two groups (p > 0.05). The proportion of pulmonary dysfunction in the hypotension group (100.0%) was higher than that in the control group (50.0%) (p < 0.05). In spinal fusion patients, 12 patients (100.0%) in the hypotension group and 2 patients (50.0%) in the control group had pulmonary dysfunction, respectively. There were no differences in the proportion of cardiac structural anomalies and thoracic deformities between the two groups (p > 0.05). Moreover, the diameter of the aortic sinus and preoperative HI were not significantly different between the two groups (p > 0.05). Seven patients (50.0%) in the hypotension group and four patients (50.0%) in the control group used TIVA, respectively, while the other patients used CIIA.
Comparison of the general data between the two groups.
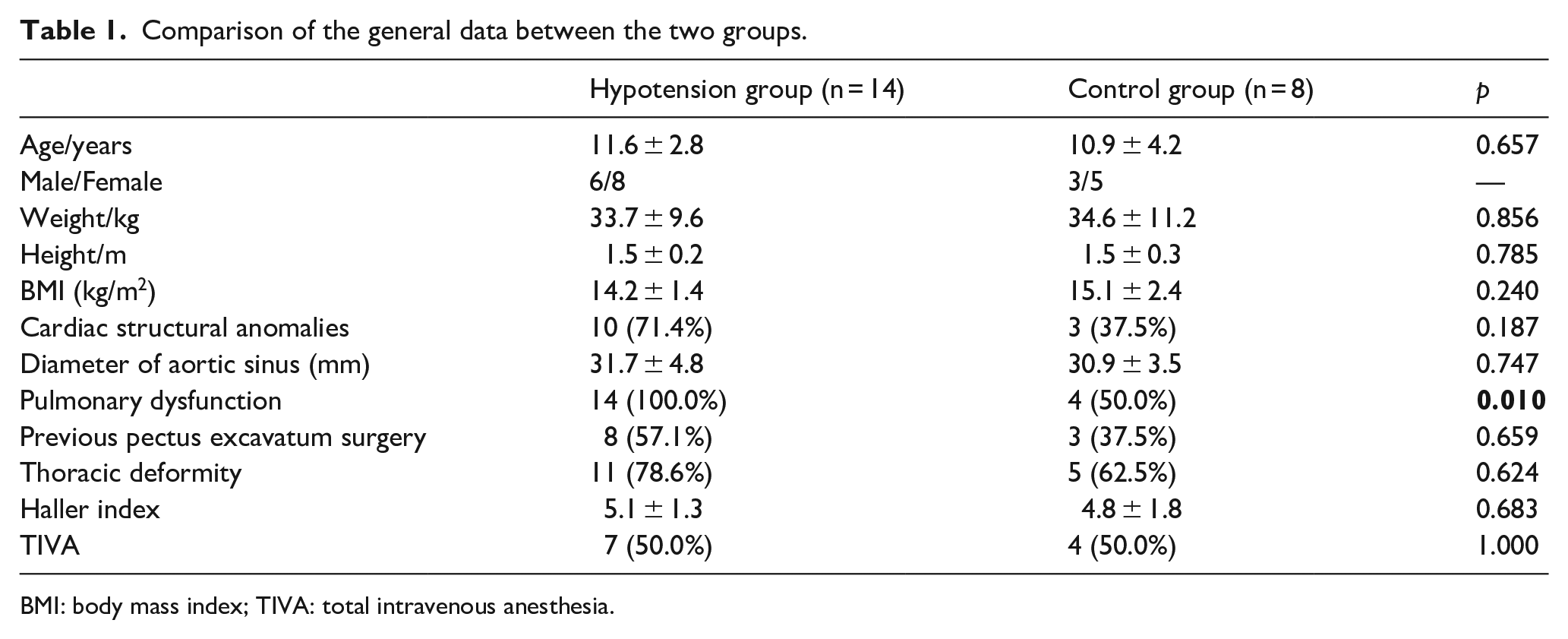
BMI: body mass index; TIVA: total intravenous anesthesia.
Radiographic and operative data of patients
Sixteen patients underwent posterior spinal fusion, and six underwent traditional dual growing-rod surgery. The radiographic and operative data of spinal fusion patients are presented in Table 2. The AVs were identified in the T4–T12 region, and the scoliosis curve type was double thoracic in both groups. The preoperative Cobb angle in the hypotension group (66.9° ± 22.8°) was greater than that in the control group (55.1° ± 38.0°), but there was no difference between the two groups (p > 0.05). The flexibility in the hypotension group (28.3% ± 14.2%) was lower than that in the control group (46.5% ± 11.5%; p < 0.05).
Radiographic and operative data of spinal fusion patients.
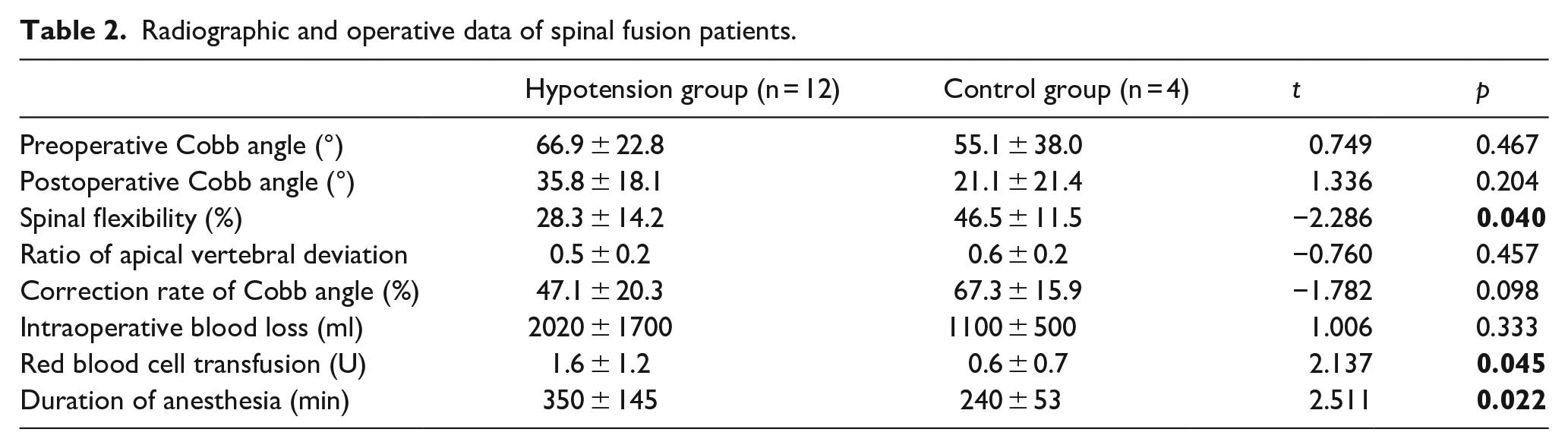
The frequency of red blood cell transfusion in the hypotension group (1.6 ± 1.2 U) was higher than in the control group (0.6 ± 0.7 U; p < 0.05). The duration of anesthesia in the hypotension group (350 ± 145 min) was longer than that in the control group (240 ± 53 min; p < 0.05).
Parameters of intraoperative hemodynamics in the hypotension group
The parameters of intraoperative hemodynamics in the 14 patients with IOH are presented in Table 3. The minimum duration of IOH is 5–10 min. Two patients underwent growing-rod surgery (case 1, 3), while the other 12 underwent posterior spinal fusion. The preoperative ASA was grade 2 in seven patients and grade 3 in another seven patients. Twelve children (85.7%) developed IOH when placing the rods during surgery, two children developed IOH after changing positions. Vasopressors were used in eight patients, including one patient with epinephrine (case 10), one patient with dopamine and epinephrine combined (case 5), and the other seven with dopamine. The SBP of 14 patients with IOH decreased by 21.0%–50.0% compared to the baseline, while their minimum MAP was 37–59 mm Hg. The intraoperative blood loss in 14 patients was 500–5800 mL, all receiving allogeneic blood. The longer duration, larger SBP decrease range from baseline, and lower minimum MAP were found in the spinal fusion patients compared with the growing-rod patients. One patient received allogeneic blood exclusively (case 10), while the other 13 received a combination of allogeneic and autologous blood transfusion. One patient had a transient decrease in lower limb muscle strength (case 9), which improved with the administration of methylprednisolone, nutritional supplements, and neurological drugs. No cardiovascular or neurological complications occurred in any of the patients during follow-up.
Surgical data of MSS patients in hypotension group.
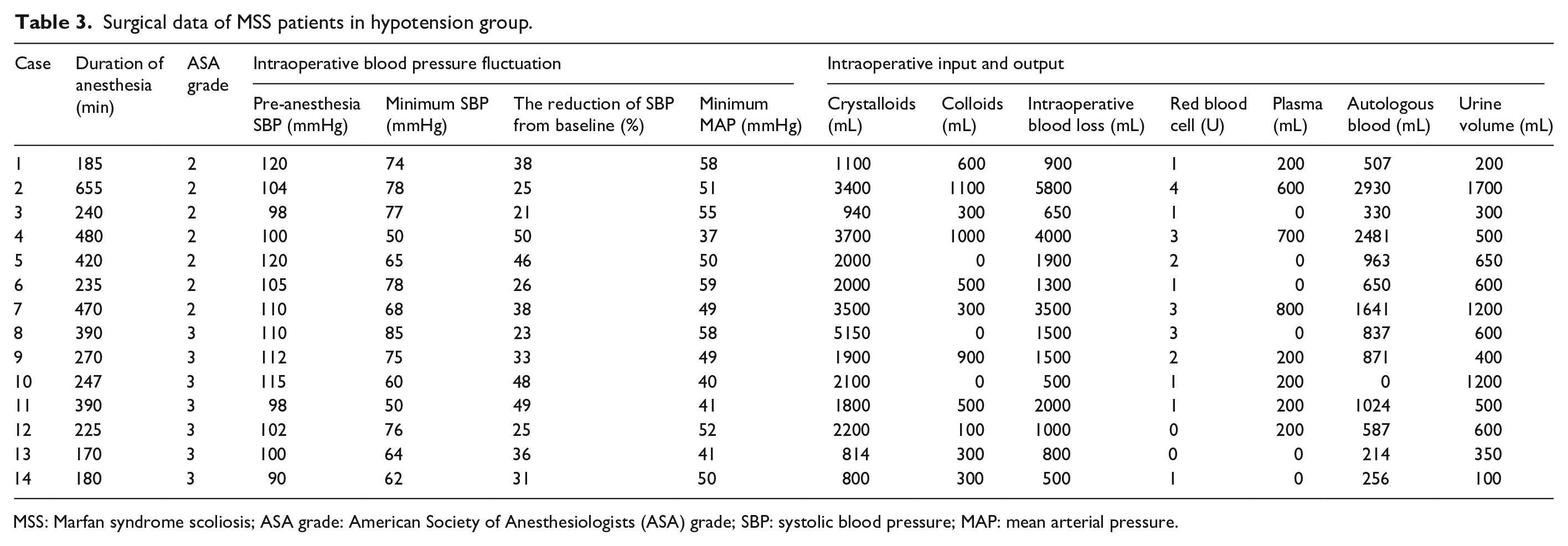
MSS: Marfan syndrome scoliosis; ASA grade: American Society of Anesthesiologists (ASA) grade; SBP: systolic blood pressure; MAP: mean arterial pressure.
Discussion
MFS is an autosomal dominant disorder that usually has defects in the synthesis, secretion, and incorporation of fibrin. It is best known for its associated cardiovascular abnormalities. It is now understood to affect many other systems, including the ophthalmologic and pulmonary systems. Musculoskeletal symptoms include scoliosis, generalized ligamentous laxity, chest deformity, and low bone mineral density. 3 Scoliosis is the most common spinal deformity associated with MFS. For mild scoliosis, brace treatment is the primary option for preventing curve progression. Studies have shown that conservative treatment, such as braces, cannot effectively control MSS progression, and early stable posterior fusion is key to preventing the progression of scoliosis. However, cases of MSS are rare and often combined with heart and thoracic deformities, making surgery difficult.
Previous studies have reported the outcomes and postoperative complications in patients with MSS. In clinical practice, we found MSS patients are more likely to develop the IOH during the surgery. However, few studies have focused on the incidence and control of IOH during MSS surgery,4–6,10,11 with few revealing that severe IOH may lead to cerebral hypoperfusion and subsequent irreversible damage.12,13 Moreover, IOH can cause spinal cord ischemia during corrective spinal surgery, leading to postoperative motor and sensory dysfunction. 14 Studies on adult scoliosis correction surgery indicated that IOH might lead to postoperative delirium, prolong hospital stays and decrease the survival rate.15,16 In this study, the incidence of IOH was 63.6%, and the recorded intraoperative MAP went as low as 37 mm Hg, while the SBP fluctuated by 50%. Three patients underwent intraoperative rescue due to IOH, and one experienced transient neurological complications after surgery. Although no severe cardiovascular or cerebrovascular complications were observed postoperatively, paying attention to IOH control is still necessary.
Patients with MSS often have poor cardiopulmonary function owing to severe cardiovascular diseases. During corrective surgery for scoliosis, patients are always placed in a prone position, and the prolonged chest wall compression may cause significant changes in the cardiovascular system, including decrease in stroke volume and cardiac index, ultimately resulting in IOH. 17 In this study, patients with cardiac structural anomalies and pulmonary dysfunction were more likely to develop IOH. In spinal fusion cases, all patients in the hypotension group had pulmonary dysfunction, while only half were in the control group. Results are the same in the growing-rod implantation cases. Thus, in the study, children with pulmonary dysfunction before surgery are more likely to have IOH when undergoing spinal fusion or growing-rod implantation surgery. Therefore, cardiologists and anesthesiologists should conduct a comprehensive preoperative evaluation for MSS patients with cardiac structural anomalies. In addition, corrective surgery for scoliosis is not recommended in patients with MSS that have complications such as untreated aortic dissection aneurysm, severe mitral valve prolapse, aortic insufficiency, or ascending aortic dilatation (diameter ≥ 6 cm). 18 Furthermore, blood pressure and heart rate should be monitored after patients are placed prone before surgery. The position of the chest pad should be adjusted in time if the blood pressure drops and the heart rate fluctuates significantly. Surgery can be continued after the patient’s vital signs have stabilized. Moreover, few researchers have proposed that transesophageal echocardiography can closely monitor cardiac function during surgery. 19
Previous studies have reported that 30%–60% of MFS cases are complicated due to pectus excavatum. 20 Moreover, the compression of the heart and lungs can lead to a considerable decline in cardiopulmonary compensatory capacity, which is an important reason for IOH. Multiple studies have suggested that a prone position in patients with scoliosis and the presence of pectus excavatum may be associated with IOH. Galas et al. 21 reported a pectus excavatum and scoliosis case that led to severe IOH during spinal fusion surgery, with transesophageal echocardiography revealing sternal compression of the right heart. They argued that the right ventricular wall is much thinner and more vulnerable to direct compression during surgery, resulting in severe IOH. Tauchi et al. 22 demonstrated that de-rotation during scoliosis surgery leads to a decrease in the anteroposterior diameter of the thorax and an increase in HI, which may compress the heart. An increase in the HI leads to cardiac compression and displacement. Furthermore, a larger ratio of AV deviation may require a higher magnitude of correction, thus placing more pressure on the heart. In this study, 16 children had thoracic deformities, including eleven patients who had undergone corrective pectus excavatum surgery. Uvodich et al. 23 reported that a patient with MSS developed IOH after corrective surgery for scoliosis. They found that IOH was due to the severe pectus excavatum, prone position, and decreased chest diameter after surgery. The severity of pectus excavatum should be noted before surgery, and the HI should be evaluated after surgery to reduce the risk of heart and lung compression. IOH was usually found in the correction of scoliosis, especially in the placement of the rods. Our study showed that 12 children (85.7%) in the hypotension group developed IOH when placing the rods during surgery, which may change the thoracic cavity, finally leading to IOH. The prone position has a greater impact on the child’s cardiopulmonary function due to the compression of the thoracic cavity by surgical manipulation, which can lead to traction on the outflow tract, lack of myocardial blood supply, and decreased blood pressure. In this study, the blood pressure of two children in the hypotension group were raised after changing positions, indicating that prone positioning in the MSS surgery may lead to IOH. In conclusion, assessing the severity of pectus excavatum and pulmonary function is essential both preoperatively and postoperatively in patients with MSS who present with thoracic deformities. Furthermore, surgical intervention for MSS should be performed in collaboration with thoracic surgeons. To reduce the risk of IOH, patients should perform adaptive exercises to improve the pulmonary function and cardiopulmonary compensatory capacity before surgery.
Scoliosis in MFS is characterized by an early onset and rapid progression in children, quickly developing into a severe rigid type. In addition, scoliosis associated with MSS is often complicated by dural ectasia, thin lamina, decreased bone mineral density, ligament instability, and other anatomical variations, making the surgery more difficult and riskier. 24 Complication rates among MFS undergoing spinal surgery are typically higher than those patients without MFS. 25 Previous studies have shown greater surgical difficulty in patients with MFS than in adolescent idiopathic scoliosis (AIS) patients. 26 Moreover, spinal deformity in MFS is usually seen in these younger patient populations, of which 84.6% have been diagnosed with MFS under 18 years of age. Severe scoliosis and fusion segments lead to a greater blood loss during MSS surgery and result in blood perfusion disorders and abnormal coagulation function.4,6,27 Few surgeons have applied anterior spinal release procedures combined with posterior spinal fusion to achieve good correction of scoliosis.28,29 However, Qiao et al. 4 proposed that the posterior spinal fusion procedure has a shorter surgery time, less blood loss, and more stable intraoperative hemodynamics than the anterior spinal release procedure. Of the 16 children who underwent spinal fusion surgery included in this study, the preoperative Cobb angle was more significant, and spinal flexibility was poorer in patients in the hypotension group than that in the control group. Patients in the hypotension group had more fusion segments and lower Cobb angle correction rate. In the hypotension group, the longer duration, larger SBP decrease range from baseline, and lower minimum MAP were found in the spinal fusion patients when compared with the growing-rod patients. It can be concluded that patients who underwent spinal fusion surgery may be more likely to have IOH, and the condition may be more severe. More cases are needed in the future to confirm this point. The minimum duration of IOH in this study was 5–10 min. After hypotension was detected by intraoperative monitoring, drugs or changing position were performed to raise patients’ blood pressure immediately, thereby avoiding the occurrence of serious complications. It was demonstrated that IOH was associated with longer duration, greater intraoperative blood loss, and frequent erythrocyte transfusion.
This study showed that a large amount of blood loss could occur quickly in patients with hypotension, and the use of dopamine and repeated positional changes can help maintain the stability of vital signs. Kurucan et al. 30 found a significantly higher rate of blood loss in pediatric patients. Unlike previous studies, this study showed that children in the hypotension group received significantly more blood transfusion than those in the control group, but there was no statistical difference in intraoperative blood loss. To some extent, a larger amount of red blood cell transfusion may reflect more intraoperative blood loss. Both red blood cell transfusion and intraoperative blood loss were associated with weight, age, and other factors among the children. In addition, IOH could be related to many factors, such as position, thoracic deformity, and spinal flexibility. Intraoperative blood loss was not convincing as one of the independent risk factors for IOH because of the limited sample size. Moreover, MSS patients with IOH possess a risk for spinal cord ischemia; therefore, performing an intraoperative wake-up test or intraoperative electrophysiological monitoring is necessary. Active monitoring to elucidate the cause of abnormal results with the timely administration of glucocorticoids to reduce the spinal cord edema and inhibit inflammation is essential.
It is worth mentioning that intraoperative anesthesia protocols may also be associated with IOH. In the past two decades, the intraoperative anesthesia protocols mainly included inhalation anesthesia (IA), TIVA, and CIIA. In recent years, IONM has been widely used in spinal surgery. IA makes motor evoked potentials (MEPs) difficult to induce on IONM, especially in young and lean children. MEP amplitudes are smaller, show a higher coefficient of variation, and require higher intensity to obtain with IA compared with TIVA. 31 Moreover, IA can also lead to postoperative nausea and vomiting, with a longer postoperative recovery time in spinal surgery.7,32 Therefore, TIVA and CIIA were used in this study. Due to the application of IONM, all children were given CIIA (11 cases) before 2016, while the other children were given TIVA (11 cases) after 2016 in this study. Hemodynamic parameters were compared between the two different anesthesia modalities and were not statistically different. Further study will expand the sample size to verify whether there is a necessary connection between anesthesia and IOH.
For patients with MSS with risk factors for IOH, formulating proper hemodynamic management with anesthesiologists is imperative. Studies have shown that propofol or sevoflurane can prevent the increase in stress hormones and help maintain hemodynamic stability during surgery. 33 Due to the limited myocardial contractility in children, cardiac output mainly depends on the heart rate. The cardiac output drops significantly when the heart rate decreases and affects cerebral perfusion. Anesthetic drugs can cause myocardial depression and decrease the heart rate. Few studies have demonstrated that an appropriate amount of atropine and timely rehydration can increase the heart rate, antagonizing the partial effects of the anesthetic drugs. 34 The relief of abdominal compression and adequate blood pressure control is essential to reduce intraoperative blood loss. In addition, a small amount of epinephrine can be injected around the incision to reduce local bleeding.
Furthermore, multiple studies have shown that intrathecal injection of morphine after anesthesia can significantly reduce intraoperative blood loss by inhibiting sympathetic nerve activity, maintaining intraoperative hemodynamic stability, and facilitating postoperative analgesia. However, it can lead to respiratory depression, and controlling the dosage and monitoring the complications is essential. Autologous or allogeneic blood transfusions should be administered when blood loss is significant. Anesthesiologists should monitor the colloidal osmotic pressure and coagulation to provide timely plasma transfusion. Autologous blood transfusions are preferred because of the few complications associated with them. In this study, all patients with MSS were treated with autologous blood transfusion combined with allogeneic blood transfusion, and no transfusion-related complications were observed under strict monitoring during surgery.
Limitations should be considered in this study. First, it is a retrospective study and is inherently prone to selection bias and confounding factors. Second, MSS children with IOH have rarely been reported in the literature, we have included as many cases as possible to make the study more convincing. However, the study still involved a small sample size, which limited our conclusions. The sample size will continue to be expanded in the future to supplement the study, making the results more perfect and credible. Although the IOH rate seemed to be higher and more severe in spinal fusion surgery in this study, the relationship between the two kinds of surgery approaches and blood pressure fluctuation is still more complex and requires continued study with higher volume studies. Nevertheless, to the best of our knowledge, this is the first study to demonstrate the occurrence and risk factors of IOH in patients with MSS.
In conclusion, the incidence of IOH in children who underwent surgery for MSS is 63.6 %. The risk factors include preoperative pulmonary dysfunction and poor spinal flexibility. In children with MSS, comprehensive preoperative evaluation and effective hemodynamic handling measures should be undertaken to prevent further cardiovascular and cerebrovascular complications.
Footnotes
Author contributions
J.L. and M.Z. designed the study and analyzed the data. Z.Y. and Xu.Z. revised the article and supervised the study. Xi.Z. offered help in the anesthesiology. W.Z. helped in the grammatical revision of the article. All authors contributed to the article and approved the submitted version.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Beijing Municipal Science and Technology Commission (No. Z191100007619052 and Z201100005420027 to Xin Zhao), Beijing Hospitals Authority Clinical Medicine Development of special funding support (No. XMLX202145 to Xin Zhao), and Capital’s Funds for Health improvement and Research (No. 2022-2Z-2098 to Xin Zhao).
Ethics approval
This retrospective study of the Chinese cohort was approved by the ethics committee of Beijing Children’s Hospital, Capital Medical University, National Center for Children’s Health (IEC-C-006-A04V.06, 2021).
Informed consent
We have already signed the consent form with patient’s parents.
