Abstract
Ultrafine nano-silica, with its smaller particle size compared to conventional nano-silica, is typically synthesized via gas-phase methods that require complex raw materials and processes. Here, we report optimized conditions for preparing ultrafine nano-silica via a precipitation reaction between fluosilicic acid waste and aluminum hydroxide. The optimal conditions determined were as follows: reverse feeding, surfactant addition at 3.0%, aluminum-to-silicon ratio of 2.00, reaction time of 30 min, reaction temperature of 90 °C, and stirring rate of 250 rpm. Under these conditions, ultrafine nano-silica with a uniform particle size of approximately 11 nm was successfully synthesized. Characterization of samples prepared by different feeding methods showed that the reverse feeding method yielded ultrafine nano-silica with superior performance indicators compared to the forward feeding method. Analysis of system changes during synthesis indicated that the feeding method influenced the acidic or alkaline environment of the substrate, affecting SiF4 hydrolysis and the subsequent dehydration and condensation of Si-(OH)4 monomers, leading to variations in silica particle agglomeration. Further, an analysis of the effects of alkyl chains in surfactants on the surfaces of silica particles, including -OH substitution or Si-O-Si bond cleavage, provided insights into the action mechanism of surface groups on ultrafine nano-silica particles.
1. Introduction
Ultrafine nano-silica, represented as SiO2∙nH2O, is an amorphous white powder characterized by a particle size of less than 20 nm. It offers advantages over conventional nano-silica, including a smaller particle size, larger specific surface area, stronger adsorption capacity, and enhanced thermal stability.1,2 Due to these unique properties, ultrafine nano-silica is widely used as an inorganic filler and reinforcing agent in various applications, such as papermaking, rubber, and coatings.3,4 Currently, gas-phase methods are the predominant approach for synthesizing ultrafine nano-silica, yet these methods involve complex processes and incur high costs for raw materials.
Fluorosilicic acid waste, primarily generated as a by-product of wet-process phosphoric acid production 5 or as a waste stream from integrated circuit manufacturing, 6 has seen increasing attention due to the growth of the phosphate and semiconductor industries. The disposal of fluorosilicic acid waste presents an environmental challenge, making it advantageous to repurpose this waste as a raw material for silica production, thereby recovering silicon. 7 Xue et al. achieved an average silica particle size of approximately 790 nm by gradually adding aluminum hydroxide to preheated fluorosilicic acid. 8 Wang et al. enhanced this method by adding a mixture of aluminum hydroxide and surfactant to preheated fluorosilicic acid, obtaining silica particles in the range of 550-670 nm. 9 Li et al. examined the impact of forward and reverse addition methods of ammonia water on the aggregation of nano-silica particles, finding that forward addition produced an irregular spatial network of aggregated nanoparticles, while reverse addition resulted in spherical nanoparticle aggregates. 10
Overall, the feeding method significantly influences both particle size and the aggregation state of synthesized nano-silica particles. 11 In view of this, this study utilizes fluorosilicic acid waste and aluminum hydroxide as the precipitant, with a focus on examining the effects of feeding method and surfactant on the particle size of silica samples produced via precipitation reaction. Through this approach, ultrafine nano-silica with an average particle size of approximately 11 nm was prepared, addressing the high material costs and process complexities associated with conventional synthesis methods. Finally, analyses using Fourier transform infrared (FT-IR) spectroscopy and energy dispersive X-ray spectroscopy (EDS) were conducted to investigate the mechanistic role of the feeding method and surfactant on particle size and aggregation in the resulting ultrafine nano-silica.
2. Experimental
2.1. Chemicals
Fluorosilicic acid (industrial grade, w(H2SiF6) = 18.7%), Aluminum hydroxide (AR, McLean reagent), surfactant (home-made, similarly hereinafter), deionized water.
2.2. Sample preparation
Samples were prepared with and without the addition of surfactant using two different feeding methods: forward and reverse.
2.2.1. Forward feeding
The fluorosilicic acid waste solution (with or without surfactant) was heated in a water bath to the target temperature under constant stirring. A measured amount of aluminum hydroxide was then added, and the reaction proceeded for a specified duration. The resulting white powder sample was obtained through filtration, washing, and drying.
2.2.2. Reverse feeding
Aluminum hydroxide (with or without surfactant) was mixed with deionized water and heated in a water bath to the target temperature under constant stirring. The fluorosilicic acid waste solution was subsequently added, and the reaction proceeded for a specified duration. The resulting white powder sample was obtained through filtration, washing, and drying.
2.3. Characterizations
The synthesized nano-silica samples were characterized using several analytical techniques. Fourier Transform Infrared Spectroscopy (FT-IR) on a Thermo Nicolet IS50 IN10 spectrometer was employed to identify functional groups on the particle surface, while X-ray Diffraction (XRD) using a PANalytical X-Pert PRO MPD diffractometer provided information on the crystallographic structure. The surface morphology and particle size were examined with a Hitachi Regulus 8230 high-resolution Field Emission Scanning Electron Microscope (FE-SEM), and the internal structure was observed in detail using High-Resolution Transmission Electron Microscopy (HRTEM) on a Talos F200X G2 microscope. Additionally, the Micromeritics ASAP 2460 Automatic surface area and porosity analyzer from United States was used to analyze pore volume of samples.
3. Results and discussion
3.1. Determination of precipitation reaction process conditions
To optimize the particle size of the synthesized silica, various parameters were examined, including feeding method, surfactant addition (mass ratio of surfactant to theoretical silica yield, %), aluminum-silicon molar ratio, reaction time, reaction temperature, and stirring rate.
3.1.1. Surfactant addition
Figure 1(a) shows the relationship between surfactant addition and the average particle size of silica samples, comparing reverse and forward feeding methods under controlled conditions: aluminum-to-silicon ratio of 2.00, reaction time of 30 min, reaction temperature of 90 °C, and stirring rate of 250 rpm. The data show that reverse feeding consistently produced smaller particle sizes than forward feeding. When surfactant addition was below 3.0%, particle size decreased as surfactant addition increased. At 3.0% surfactant, the smallest particle size was achieved (∼11 nm) with reverse feeding. Beyond 3.0%, particle size increased as surfactant concentration rose. This trend is likely due to insufficient surfactant coverage below 3.0%, leading to particle bridging and aggregation, while excess surfactant (>3.0%) increases solution viscosity, hindering particle mobility and promoting agglomeration.
12
Therefore, a surfactant addition of 3.0% was determined as optimal. Effect of different reaction conditions on the average particle size of silica samples: surfactant addition (a), aluminum-to-silicon ratio (b), reaction time (c), reaction temperature (d), stirring rate (e).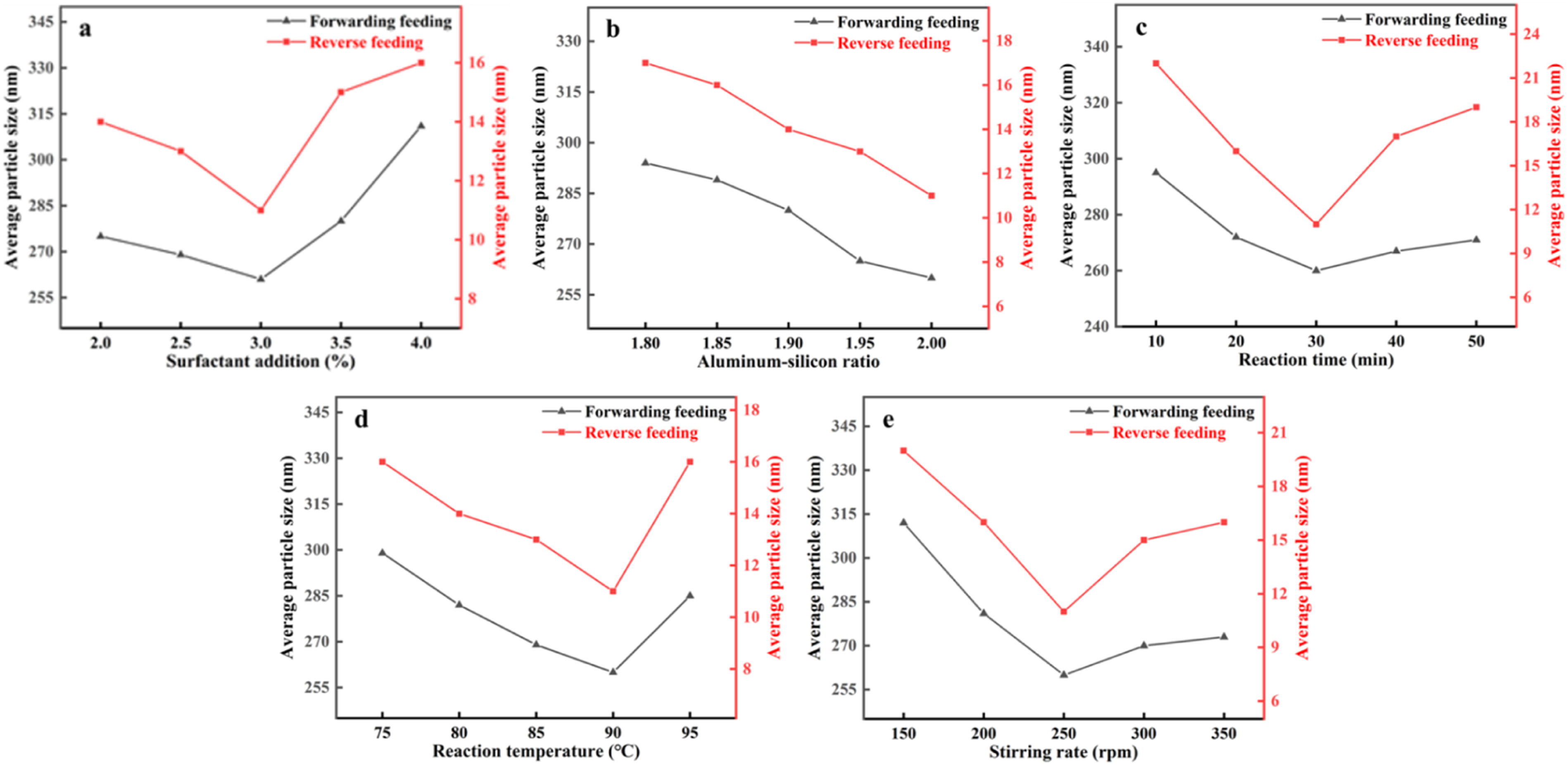
3.1.2. Aluminum-to-silicon ratio
Figure 1(b) shows the relationship between the aluminum-to-silicon ratio and the average particle size of silica samples for different feeding methods under controlled conditions: surfactant addition of 3.0%, reaction time of 30 min, reaction temperature of 90 °C, and stirring rate of 250 rpm. The results indicate that reverse feeding consistently produces smaller particle sizes than forward feeding. As the aluminum-to-silicon ratio increases, the average particle size decreases, reaching a minimum of approximately 11 nm at a ratio of 2.00 with reverse feeding. Higher aluminum-to-silicon ratios may compromise silica purity, making 2.00 the optimal ratio.
3.1.3. Reaction time
Figure 1(c) illustrates the relationship between reaction time and the average particle size of silica samples under the conditions: 3.0% surfactant addition, aluminum-to-silicon ratio of 2.00, reaction temperature of 90 °C, and stirring rate of 250 rpm. The data show that reverse feeding consistently produced smaller particles than forward feeding. When the reaction time was less than 30 minutes, particle size decreased as time increased, reaching a minimum of approximately 11 nm at 30 minutes with reverse feeding. Beyond 30 minutes, particle size increased, likely due to particle aggregation and the precipitation of β-AlF3·3H2O, which affects both product quality and particle size. Therefore, 30 minutes was identified as the optimal reaction time for achieving minimal particle size.
3.1.4. Reaction temperature
Figure 1(d) shows the effect of reaction temperature on the average particle size of silica samples under conditions of 3.0% surfactant addition, aluminum-to-silicon ratio of 2.00, reaction time of 30 min, and stirring rate of 250 rpm. The results indicate that reverse feeding consistently yielded smaller particles than forward feeding. Below 90 °C, particle size decreased as temperature increased, reaching a minimum (∼11 nm) at 90 °C with reverse feeding. However, at temperatures above 90 °C, particle size increased due to enhanced Brownian motion, which promotes particle collision and agglomeration. 13 Thus, 90 °C was identified as the optimal reaction temperature.
3.1.5. Stirring rate
Figure 1(e) displays the effect of stirring rate on the average particle size of silica samples, under conditions of 3.0% surfactant addition, aluminum-to-silicon ratio of 2.00, reaction time of 30 min, and reaction temperature of 90 °C. The data indicate that reverse feeding produced smaller particles than forward feeding. At stirring rates below 250 rpm, particle size decreased as the rate increased, reaching a minimum (∼11 nm) at 250 rpm with reverse feeding. At rates above 250 rpm, particle size increased, likely due to enhanced concentration gradients at the stirring center, which promotes aggregation. 14 Thus, 250 rpm was identified as the optimal stirring rate for minimizing particle size.
According to the these experiments, the optimal conditions for preparing ultrafine nano-silica are: reverse feeding, 3.0% surfactant addition, aluminum-to-silicon ratio of 2.00, reaction time of 30 min, reaction temperature of 90 °C, and stirring rate of 250 rpm.
3.2. Characterization and analysis of the samples
3.2.1. XRD pattern of the sample
Figure 2 presents the XRD patterns of samples prepared under optimized conditions using both forward and reverse feeding methods. In both cases, a broad diffraction peak appears at 2θ = 23°, confirming that the samples are composed of amorphous silica.
15
XRD patterns of samples prepared by different feeding methods under optimized conditions: forward feeding (a), reverse feeding (b).
3.2.2. FE-SEM, TEM images, and EDS analysis
Figures 3 and 4 display the FE-SEM and HRTEM images of silica samples prepared under the optimized conditions via both forward and reverse feeding methods respectively. Significant differences in particle size were observed between the two methods. The sample prepared by forward feeding (Figure 3(a)) had an average particle size of approximately 300 nm, with uneven particle distribution and pronounced agglomeration (measured by Nano Measurer software). In contrast, the sample prepared by reverse feeding exhibited ultrafine, irregular particles with an average size of about 11 nm, along with some small, attached particles. The larger size and agglomeration seen in forward feeding are likely due to a high local SiO2 concentration, leading to surface hydroxyl interactions and particle aggregation.
16
Reverse feeding, by introducing fluorosilicic acid waste into the aluminum hydroxide slurry, maintained a lower local SiO2 concentration, thus reducing particle size. Furthermore, a comparison with Figure 3(b) demonstrates that surfactant addition significantly reduced particle size. FE-SEM images of silica samples prepared by different feeding methods under optimized conditions: forward feeding (a), reverse feeding (b). HRTEM image of silica samples prepared by reverse feeding.

Figure 5 presents EDS spectra of silica samples prepared by different feeding methods. The spectra show that the silica sample prepared by reverse feeding has significantly higher surface contents of carbon (C) and oxygen (O) compared to the sample prepared by forward feeding, while the silicon (Si) content remains relatively constant. This indicates that more surfactants adhere to the surface of the silica particles in reverse feeding, effectively reducing surface tension, inhibiting agglomeration, and resulting in smaller particle sizes.
17
EDS plots of silica samples prepared by different feeding methods under optimized conditions: forward feeding (a), reverse feeding (b).
3.2.3. Pore volume analysis of the samples
Figure 6 displays the pore volume distribution of silica samples prepared by forward and reverse feeding methods under optimized conditions. The pore volume of the forward feeding sample was calculated to be 0.0251 cm3·g-1 during adsorption and 0.0228 cm3·g-1 during desorption, whereas the reverse feeding sample exhibited much higher values of 0.997 cm3·g-1 and 0.994 cm3·g-1, respectively. This difference is likely due to the aggregation of particles in forward feeding, which encapsulates surfactant internally and blocks the pore structure.
18
In contrast, the smaller, less aggregated particles in the reverse feeding sample (as seen in Figure 3(b)) allow the surfactant to act primarily on the particle surface, preserving the pore structure and resulting in a significantly higher pore volume. Pore size distribution of silica samples prepared by different feeding methods under optimized conditions. Forward feeding (a), reverse feeding (b).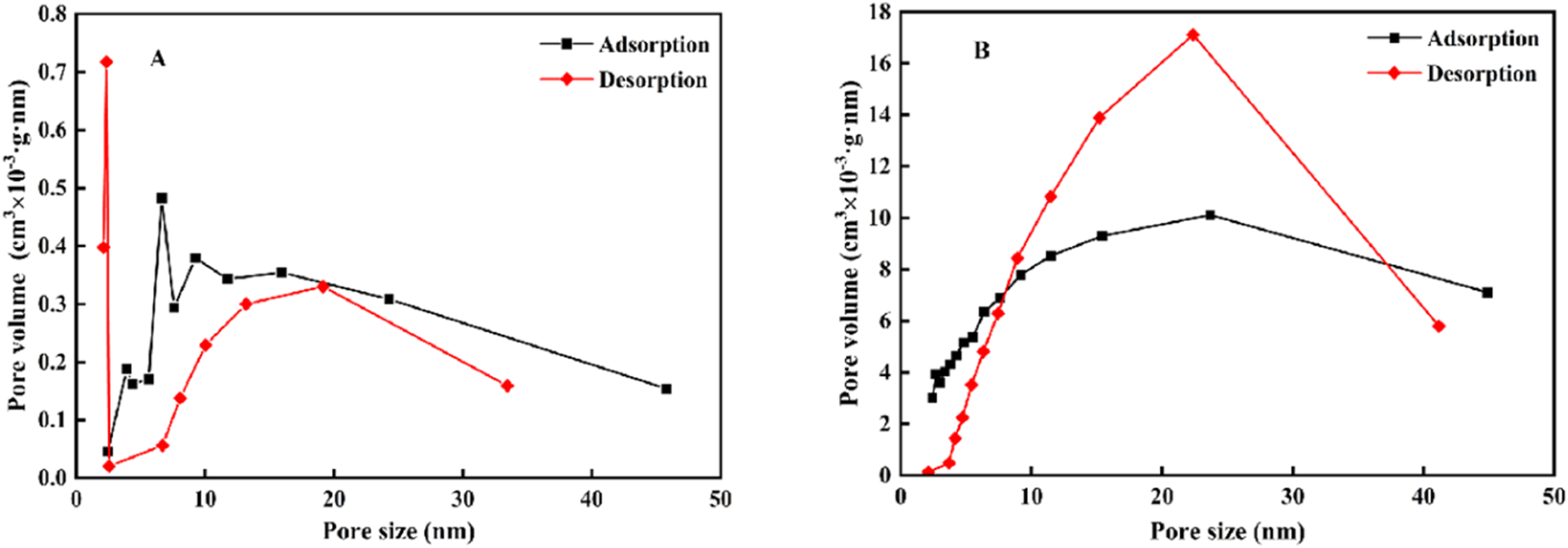
3.3. Research on the formation mechanism of ultra fine silica
3.3.1. Mechanism of forward and reverse feeding action
In fluorosilicic acid, silicon is bonded to fluorine in an octahedral structure with six covalent bonds.
19
The overall reaction for preparing silica from fluorosilicic acid and aluminum hydroxide is:
This process can be broken down into three reactions
20
:
Initially, three molecules of H2SiF6 react with two Al(OH)3, forming the intermediate Al2(SiF6)3 (containing three SiF62- and two Al3+ ions). This unstable SiF62- undergoes hydrolysis,21,22 where electron density shifts towards fluorine atoms, leading to the decomposition of the six-coordinated SiF62- into the four-coordinated SiF4 and two F– ions. The unstable SiF4 then reacts with H2O to form Si-OH tetrahedra and HF, which further reacts with Al(OH)3 to produce AlF3. These Si-OH tetrahedra subsequently undergo dehydration and condensation, forming silica (Figure 7). Total reaction process of H2SiF6 with Al(OH)3.
The hydrolysis of the SiF4 intermediate and the condensation of Si-(OH)4 tetrahedra are crucial in determining the particle size and uniformity of the resulting silica. Variations in feeding methods influence the pH and concentration of SiF62- in the system, thereby impacting the hydrolysis of SiF4 and the subsequent dehydration-condensation of Si-(OH)4 tetrahedra. 23
In forward feeding, the substrate liquid is acidic fluorosilicic acid waste, which contains a high concentration of H+ ions. These H+ ions initially attack the fluorine atoms in SiF4, protonating them and shifting electron density towards fluorine, creating a reactive site on the silicon nucleus. Water molecules then attack the silicon atom, leading to SiF4 hydrolysis and forming F3Si-OH. Due to electronegativity differences, the electron cloud on silicon shifts toward fluorine, and further hydrolysis forms F2Si-(OH)2, which subsequently converts into Si-(OH)4 monomers. During the dehydration and condensation of Si-(OH)4 monomers, the high H+ concentration protonates one of the oxygen atoms, making the Si-O group positively charged. This attracts nearby Si-OH groups, removing H3O+ and forming Si-O-Si bonds, eventually producing -(Si-O-Si)n-polymers. As the reaction proceeds, H+ ions react with Al(OH)3, gradually raising the pH, which weakens the condensation reaction. The remaining Si-(OH)4 monomers aggregate due to -OH attraction, resulting in larger particle sizes and severe agglomeration in the final silica sample (Figure 8). Reaction process during forward feeding.
In reverse feeding, the substrate is an alkaline aluminum hydroxide slurry, rich in OH– ions. This process, illustrated in Figure 9, begins as the small ionic radius of OH– enables it to attack the silicon nucleus nucleophilically,
24
creating a negatively charged, 5-coordinated transition state. The resulting charge repulsion shifts electron density towards fluorine atoms, breaking the Si-F bond and initiating hydrolysis to form F3SiOH. Further hydrolysis generates weakly acidic Si-(OH)4 monomers. Reaction process during reverse feeding.
In the alkaline environment, these Si-(OH)4 monomers undergo dehydrogenation, forming a strong base that facilitates nucleophilic attacks on other silicon nuclei. This promotes efficient dehydration and condensation, leading to cross-linking between Si-(OH)4 monomers and the formation of silica particles. The OH– ions promote SiF4 hydrolysis and the condensation of Si-(OH)4, reducing -OH content and resulting in smaller particle sizes with better dispersion. 10
3.3.2. Mechanism of surfactant action
Based on experimental results and sample analyses, the role of surfactants in forming ultrafine nano-silica appears to operate through two primary mechanisms:
(1) As shown by the EDS results in Figure 5, in reverse feeding, the addition of fluorosilicic acid to the alkaline aluminum hydroxide solution creates a lower local concentration of silicon in the initial reaction stage, leading to greater surfactant adhesion on the silica particle surfaces, and resulting in the formation of smaller silica crystals.
The surfactants attachment mechanism is illustrated in Figure 10
22
The adsorption of surfactant molecules onto the surface of nascent silica particles drastically reduces their surface free energy,
25
This reduction in surface energy, in turn, significantly weakens the primary driver of particle aggregation-the van der Waals attraction. By creating both a steric and an electrostatic barrier, the surfactants inhibit the coalescence and Ostwald ripening of the particles. This suppression of further growth mechanisms directly results in the formation of smaller, more discrete final silica particles. Schematic of surfactants attachment mechanism.
(2) Figure 11 shows the FT-IR spectra of silica samples prepared by different feeding methods. Both samples exhibit stretching vibration of Si–OH groups at 943 cm-1, and characteristic Si-O-Si vibrations: bending at 472 cm-1, symmetric stretching at 806 cm-1, and asymmetric stretching at 1103 cm-1.26,27 A broad peak near 3430 represents the -OH asymmetric stretching vibration, while the peak around 1632 cm-1 corresponds to the H-O-H bending vibration.
28
Additionally, the methylene stretching peaks at 2921 cm-1 and 2848 cm-1 indicate that the surfactant is grafted onto the silica surface, regardless of whether forward or reverse feeding is used.29,30 FT-IR plots of silica samples prepared by different feeding methods under optimized conditions: forward feeding (a), reverse feeding (b).
Comparing the FT-IR spectra of silica samples prepared by two methods in Figure 11, the sample prepared by reverse feeding shows lower intensities of the bending of Si–OH at 943 cm-1, and the Si-O-Si peak at 472 cm-1 which overlapping with the Si-OH bending at 473 cm-1.31 These results suggest fewer hydroxyl groups and Si-O-Si bonds on the silica surface in the reverse-fed sample, indicating a greater replacement of hydroxyl groups and partial disruption of Si-O-Si bonds. 32
The mechanism of the replacement and disruption might be schematically illustrated as Figure 12. The R-OH groups in the surfactant’s alkyl chain can form strong hydrogen bonds with the oxygen atoms of highly reactive dangling Si-O- bonds on the silica particle surface, or undergo a coupling reaction with them, eliminating one molecule of water to form R-O-Si bonds. This results in the grafting of the surfactant’s alkyl chains onto the particle surface. When multiple surfactant molecules are densely adsorbed in a specific region of the surface, mutual repulsion between the alkyl chains and thermal motion generate steric hindrance and mechanical tension.33,34 This subjects the stressed Si-O-Si bonds to additional stretching or twisting, lowering their activation energy for cleavage. Under attack by ambient H2O, these bonds undergo hydrolytic cleavage and may form new R-O-Si bonds with R-OH groups
35
The saturation of high-energy dangling bonds and the surface grafting of the surfactant increase the lipophilicity of the particle surface, thereby reducing the surface energy. This effectively weakens interparticle agglomeration, thus facilitating the formation of smaller and more dispersed SiO2 particles. Schematic diagram of the action mechanism of surfactants on the substitution of hydroxyl and the breaking of Si-O-Si bond.
4. Conclusions
In summary, using fluorosilicic acid waste as the raw material and aluminum hydroxide as the precipitant, this study investigated the effects of feeding method and surfactant on silica particle size, identifying reverse feeding as optimal. The process was optimized with surfactant addition at 3.0%, an aluminum-to-silicon ratio of 2.00, reaction time of 30 minutes, reaction temperature of 90 °C, and stirring rate of 250 rpm, yielding silica particles with an average size of approximately 11 nm, uniform distribution, and good dispersion. FT-IR and EDS characterizations provided insights into the formation mechanism of ultrafine nano-silica. The alkaline environment under reverse feeding promotes SiF4 hydrolysis and the condensation of Si-(OH)4 monomers, reducing aggregation and particle size. Additionally, the adhesion of surfactant molecules onto the particle surface facilitates the substitution of Si-OH groups by the surfactant’s alkyl chains. This saturates the high-energy dangling Si-O- bonds to form new R-O-Si bonds, thereby enhancing the surface lipophilicity. Consequently, the surface energy is reduced, leading to a further decrease in particle size.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.




