Abstract
The combination of nanoparticles and polycarboxylate ether superplasticizers is expected to simultaneously improve the durability and mechanical properties of mortar and concrete. However, the uniform dispersion of nanoparticles in cement materials has always been a difficult challenge. In this paper, nanoparticles are modified with surfactants to provide a research direction for better dispersion of nanoparticles in cementitious materials. Modified allyl ether polycarboxylate superplasticizer with short-branched chains was synthesized from acrylic acid and allyl glycidyl ether. Nano-silica/polycarboxylate ether was obtained by grafting nano-silica onto polycarboxylate ether molecular chains through intermolecular dehydration, thus achieving a homogeneous dispersion of nano-silica in the cementitious material. The results show that nano-silica/polycarboxylate ether has better dispersibility in cement matrix and strong clay resistance, which compensates for the poor dispersion of polycarboxylate ether synthesized from conventional polyethylene glycol graft chains in the presence of clay impurities.
A short-chain allyl ether-based polycarboxylate superplasticizer modified by nano-silica is introduced in this paper. It provides an idea to solve the problem of conventional long-chain polyethylene glycol grafted polycarboxylate ether being sensitive to clay impurities. It makes an effective exploration for the difficulty of uniform dispersion of nano-silica in cementitious materials.
Introduction
Polycarboxylate ether (PCE) superplasticizer possesses outstanding characteristics, such as better slump adjustment, higher water reduction rate, and lower dosage.1,2 It is a new type of superplasticizer commonly used in advanced concrete in recent years. The mechanical properties of concrete can be improved and the strength and durability of hardened cement can be enhanced, which has attracted the attention of many researchers.3,4 The charge of the carboxyl groups in the backbone helps the PCE to adsorb onto the cement particle and the long side chains disperse the cement particles by steric effect improving the rheology of the paste with low water content.5,6 The common synthetic raw materials of PCE are polyethylene glycol (PEG) long-chain olefins, such as MPEG-Type PCE, APEG-Type PCE, HPEG-Type PCE, IPEG-Type PCE, and PAAM-Type PCE.7,8 These PCE show high sensitivity to clay impurities contained in concrete aggregate and limestone powder, which will weaken their dispersion ability. 9 The PEG side chains of conventional PCE occupy the interlayer space between the aluminosilicates of the clay. 10 The superplasticizer would be sequestered in the clay through this chemisorption (intercalation), thus would not be able to exert its steric dispersion effect. 11 It is assumed that the synthesis of PCE with short branched chains will reduce the intercalation, thus achieving the effect of PCE dispersion in the presence of clay impurities.
The compressive behavior and the stability of mortar and concrete are expected to be simultaneously enhanced by the integration of nanoparticles and PCE.12,13 Due to the good dispersibility of nano-silica (NS), they are expected to have a significant role in building materials. However, the uniform dispersion of NS in cementitious materials has always been a difficult problem, and the traditional ultrasonic dispersion method is not suitable for industrial applications. Studies have shown that NS can be grafted onto the molecular chain of PCE, that is to say, a novel type of polycarboxylate superplasticizer can be obtained by modifying PCE with NS.14,15 For example, Suárez et al. 16 investigated the effect of NS of different particle sizes and different types of superplasticizers on the rheological properties and compressive strength of cement slurries and found that coarser nanoparticles had better mechanical properties and compatibility. Huang and Wang 17 used PCE containing silane-coupling agent to modify the surface of NS particles under microwave irradiation to improve the distribution of NS particles. The compatibility of PCE with NS and the stability of NS in saturated calcium hydroxide solution were improved.
In this paper, novel-modified allyl ether-based polycarboxylate superplasticizers with short-branched chains were synthesized using acrylic acid (AA), allyl glycidyl ether (AGE), and vinyl triethoxysilane (VTES) as raw materials. Nano-silica/polycarboxylate ether (NS/PCE) with better dispersion can be obtained by doping NS onto PCE, and NS can be evenly dispersed in cementing materials. The molecular structure of the superplasticizer was characterized by Fourier Transform Infrared Spectroscopy (FTIR) and gel permeation chromatography (GPC). The particle size of NS/PCE and its dispersion behavior were investigated by dynamic light scattering (DLS) and transmission electron microscopy (TEM). Synthesis of PCE with short-branched chains provides a new idea for clay impurities resistance, and doping of NS on PCE provides a research direction for better dispersion of nanoparticles in cement matrix.
Results and discussion
Orthogonal experiment and results
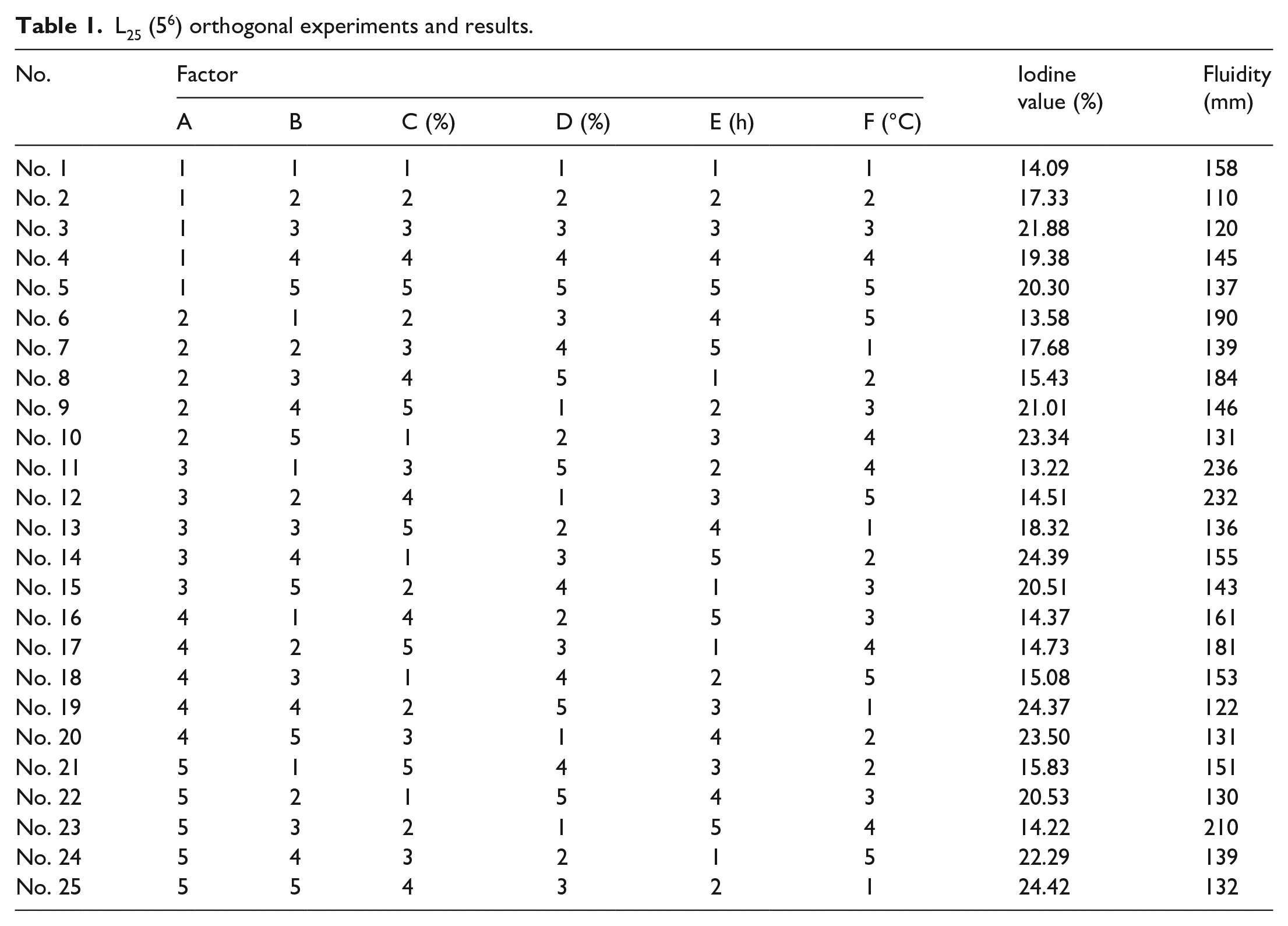
The results of the orthogonal experiments are shown in Table 1. The analysis of the orthogonal tests with the iodine value of PCE and the fluidity of cement paste as evaluation indexes are presented in Table 2 and Table 3, respectively.
L25 (56) orthogonal experiments and results.
Orthogonal experimental results with iodine value of PCE as evaluation index (range analysis).

Orthogonal experimental results with fluidity as evaluation index (range analysis).
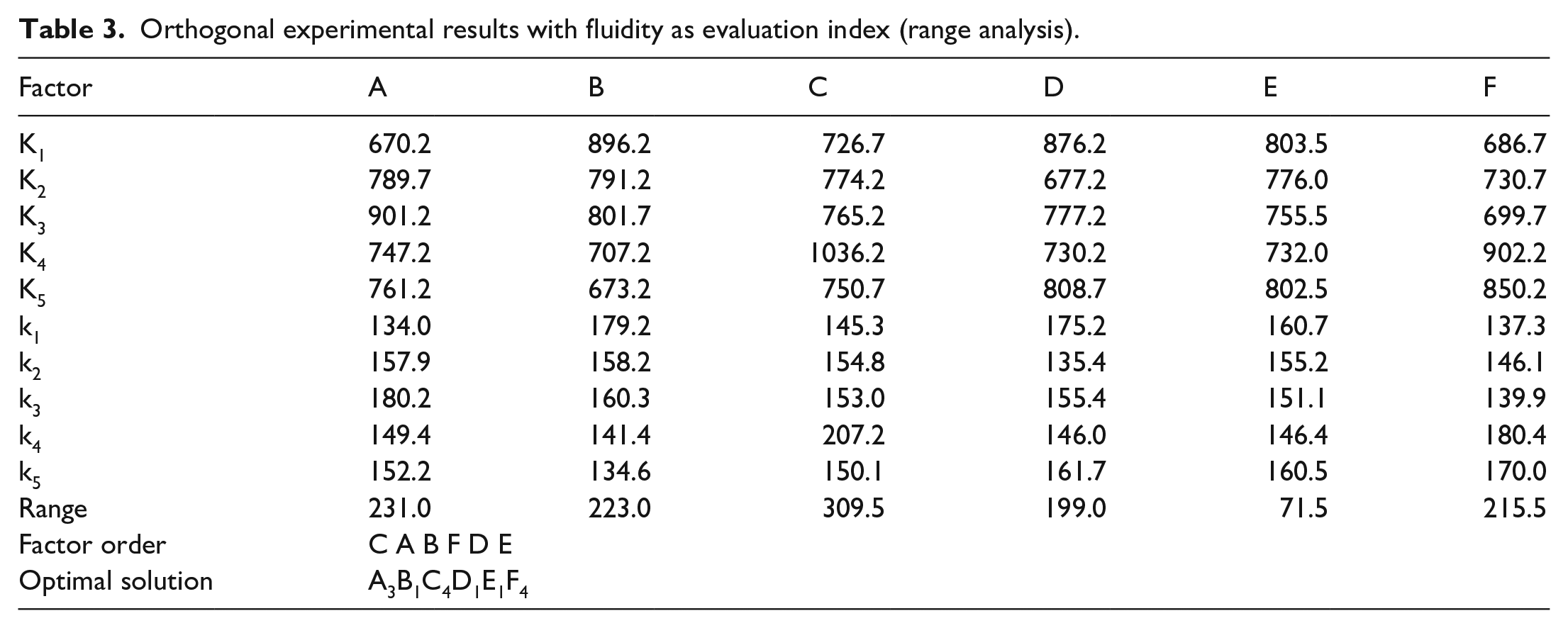
From Tables 2 and 3, it can be seen that optimal synthesis scheme is A3B1C4D1E1F4, whether iodine value or fluidity is used as the evaluation index. To be specific, the molar ratio of AGE to AA is 1:0.8, the molar ratio of AGE to VTES is 1:0.3, the mass of ammonium persulfate (APS) accounts for 2.5% of the total mass of polymerized monomer, the mass of 3-mercaptopropionic acid (MPA) accounts for 1% of the mass of polymerized monomer, the polymerization time is 4 h, and the polymerization temperature is 70°C.
It can be seen from Table 4 that under the optimum experimental conditions, the iodine value of the reaction product is 12.93%, the reaction conversion is 96.23%, and the fluidity of cement paste is 250 mm.
Conversion rate and fluidity under optimal reaction conditions.

Characterization of PCE and NS/PCE
FTIR spectra of the PCE are shown in Figure 1(a). Peaks at 1715 cm−1 are attributed to the stretching vibration of C=O. Peaks at 1272 and 1105 cm−1 correspond to the stretching vibration of C–O. Peaks at 1042 cm−1 are C–O–C stretching vibration, whereas the peaks at 1403 cm−1 are related to the deformation vibration of O–H. Given that there is no absorption peak of C=C at 1620–1680 cm−1, it indicates that each monomer has been successfully polymerized and PCE was obtained.

(a) FTIR spectra of PCE and (b) NS and NS/PCE.
FTIR spectra of the NS and NS/PCE are shown in Figure 1(b). However, 10% NS/PCE was chosen as representative due to the similarity of the FTIR spectra of NS/PCE. Peaks at 805 cm−1 in the spectra of NS and NS/PCE are related to the stretching vibration of Si–O–Si. Peaks at 1382 and 1113 cm−1 are attributed to the stretching vibration of Si–O. Peaks at 3448 cm−1 in both samples correspond to the stretching vibration of O–H, whereas the peaks at 1631 cm−1 are related to the bending vibration of O–H in water chemically bound to KBr crystal. 18 The spectra of 10% NS/PCE showed peaks of NS and PCE, indicating that NS/PCE had been successfully synthesized. 19
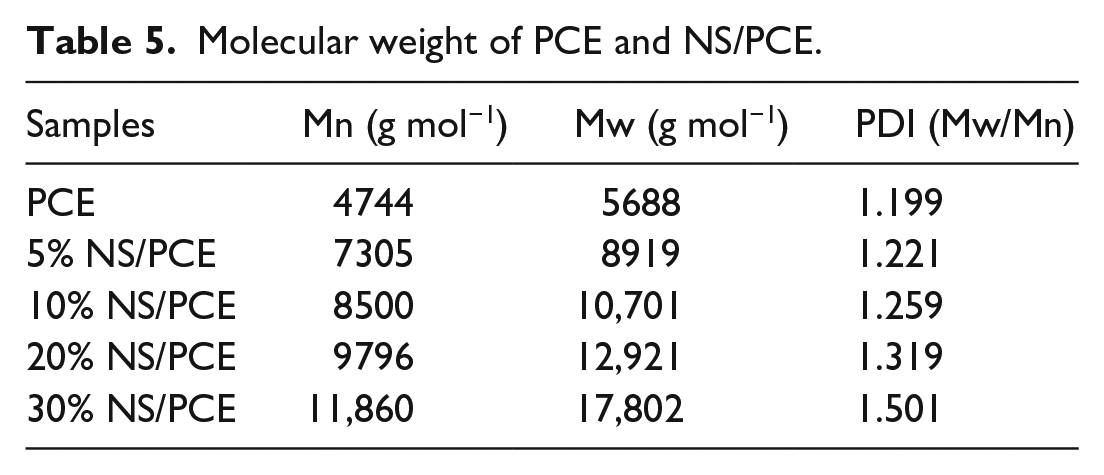
The number average molecular weight (Mn), mass average molecular weight (Mw), and polydispersity index (PDI) of PCE and NS/PCE were determined by GPC. The relative molecular mass and its distribution are one of the important parameters controlling the molecular structure of polycarboxylate superplasticizer. The relative molecular mass of all NS-doped PCE is larger than that of PCE alone, and the relative molecular mass of NS/PCE increases sequentially with increasing NS concentration, probably due to the successful grafting of NS on the PCE chain. When the PDI value is close to 1, the molecular weight distribution is more homogeneous and the polymerization is better. 20 As illustrated in Table 5, the PDI values of PCE, 5% NS/PCE, and 10% NS/PCE were close to 1. The PDI value of 30% NS/PCE was much greater than 1, which may be due to the excess of nanoparticles addition resulted in a strong cross-linking between NS and PCE. 21 It is found in Table 5 that the Mn of the superplasticizer increases progressively as the NS content increases and the molecular chain of the superplasticizer is grown, which can further improve the spatial site resistance effect of the superplasticizer.
Molecular weight of PCE and NS/PCE.
The zeta potential values for NS and NS/PCE are given in Figure 2. The absolute value of zeta potential for NS, 5% NS/PCE, 10% NS/PCE, 20% NS/PCE, and 30% NS/PCE increases and then decreases with increasing NS. This is probably due to the successful grafting of NS particles onto PCE molecules. The addition of appropriate amounts of NS increases the molecular chain, further increases the steric effect of water reducer, making the overall polymerization of NS/PCE good. However, when the content of NS is too high, it is easy to form cross-linked copolymer in the solution, which is not conducive to the dispersion of particles. The larger absolute value of zeta potential indicates the greater electrostatic rejection of the system, the higher bilayer repulsion of the particles, the more favorable the dispersion of the particles, and thus the better dispersion stability. 22 The results show that appropriate amounts of NS can improve the dispersion performance of PCE.
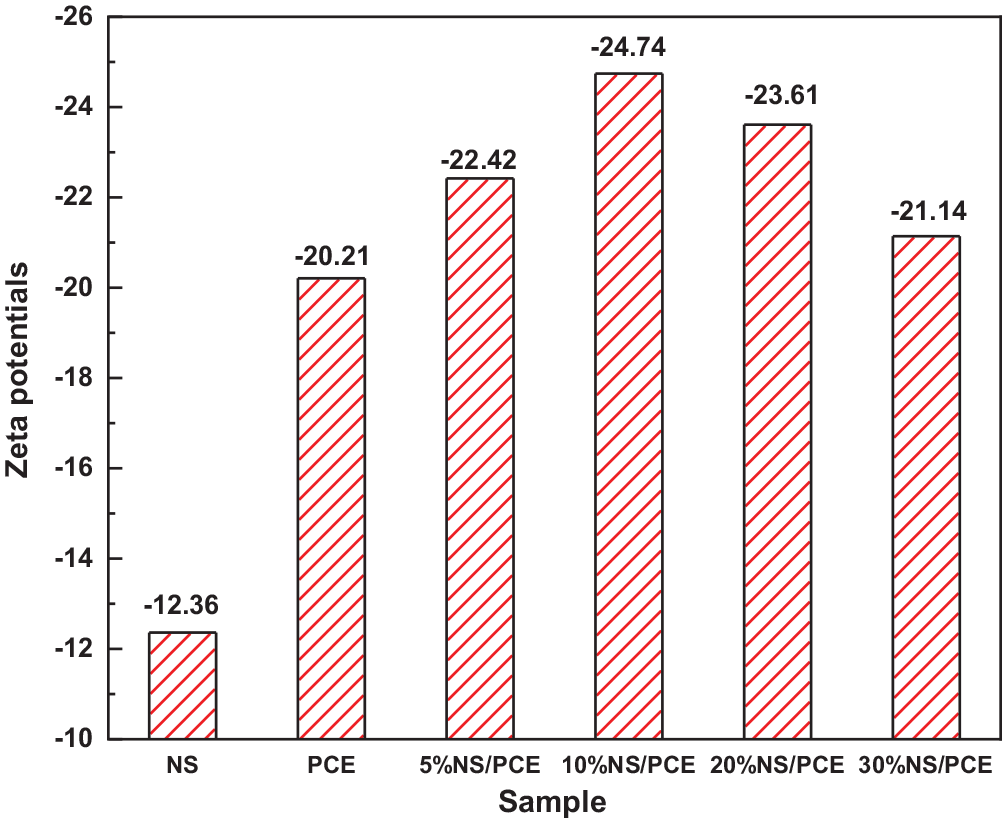
Zeta potentials of NS, PCE, and NS/PCE.
The dispersion properties of PCE chemistry combined with surface-modified NS were investigated by observing the sample morphology with TEM and measuring the particle size distribution of the samples by DLS. Figure 3(a) shows TEM graphs of unmodified and modified NS, where unmodified NS particles were observed to be clustered together, while NS/PCE particles were significantly dispersed. Figure 3(b) shows that the average diameter of pristine NS particles is 15.7 nm. With increasing shell-to-core ratio, the average diameter increases to 32.7 nm, which demonstrates that the effective steric hindrance is increased due to the existence of side chain of PCE.23,24
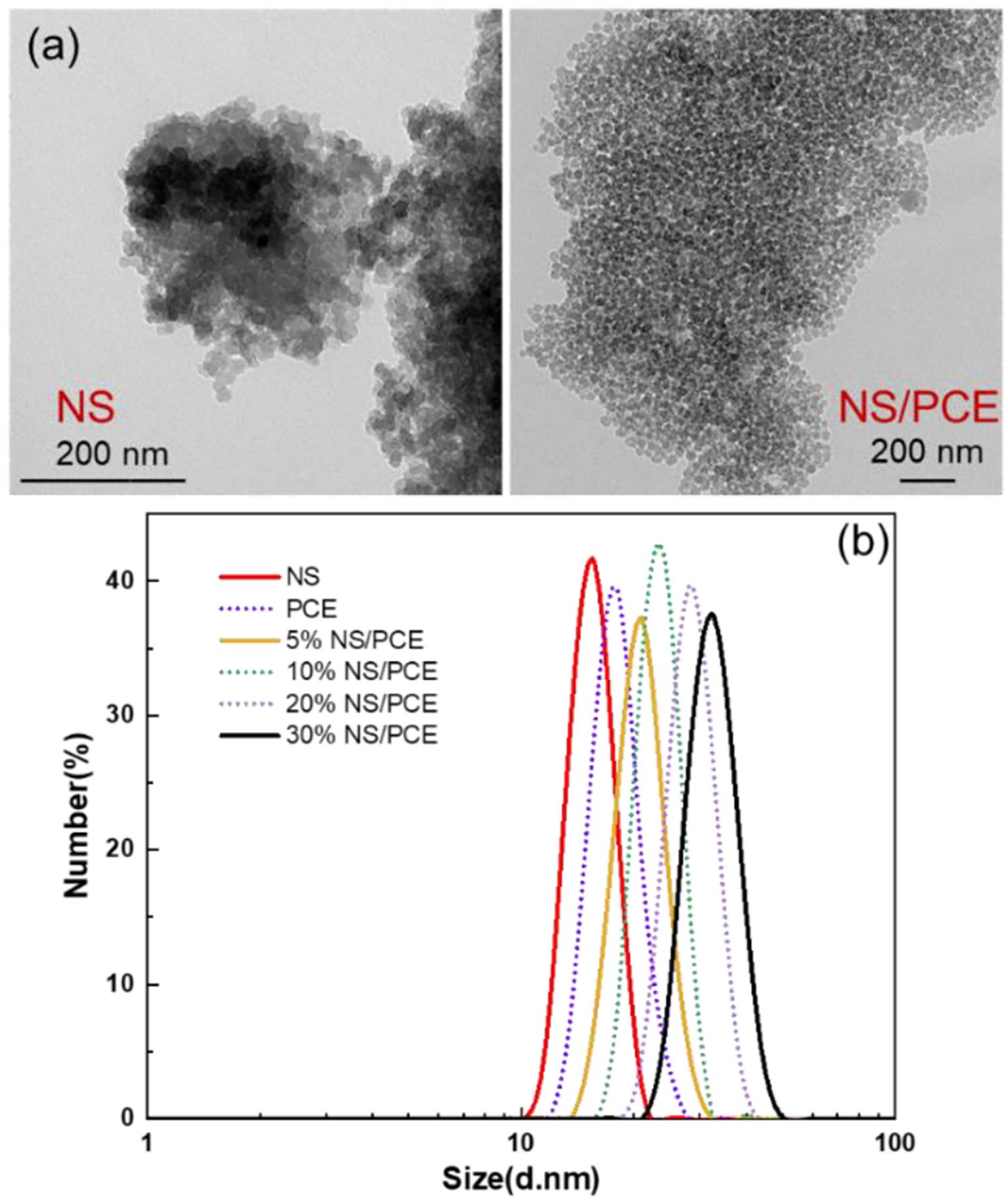
(a)TEM images of NS and NS/PCE and (b) particle size distribution of NS, PCE, and NS/PCE measured by DLS.
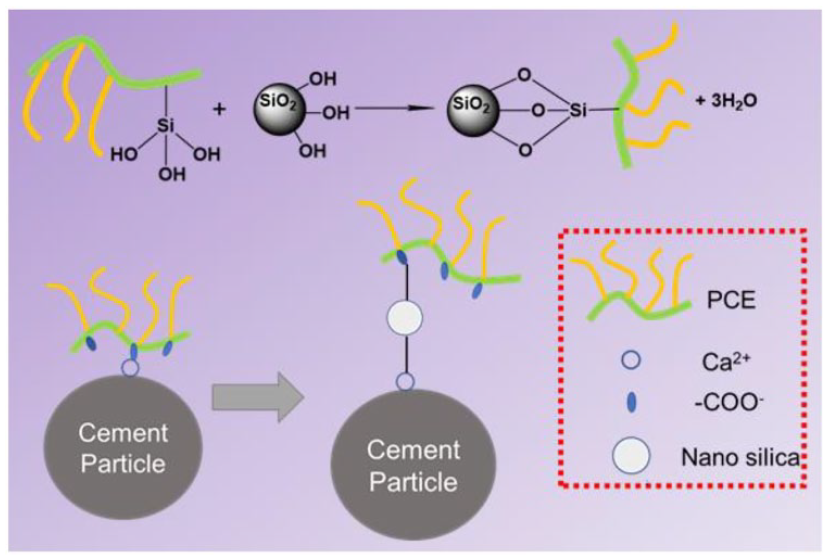
PCE is a class of cement dispersants with comb- or brush-like structures. These comb-like side chains improve the rheology of low-water-content slurries by dispersing the cement particles through a steric effect (Figure 4). Based on the above experimental results, a possible mechanism for the formation of NS/PCE is proposed. NS contains a large number of silicone hydroxyl groups on its surface, which are highly reactive and can be substituted by many functional groups. 25 The –OH in the carboxyl group of PCE undergoes dehydration condensation with the silica hydroxyl group in NS to form NS/PCE, as depicted in Figure 4, which is grafted on PCE molecular chain through Si–O– chemical bond, forming NS/PCE. Due to the introduction of NS, the molecular chain of polycarboxylate superplasticizer grows and the relative molecular weight increases, thus enhancing the spatial site resistance effect of superplasticizer molecules on cement particles and improving the dispersing ability of superplasticizer on cement particles.
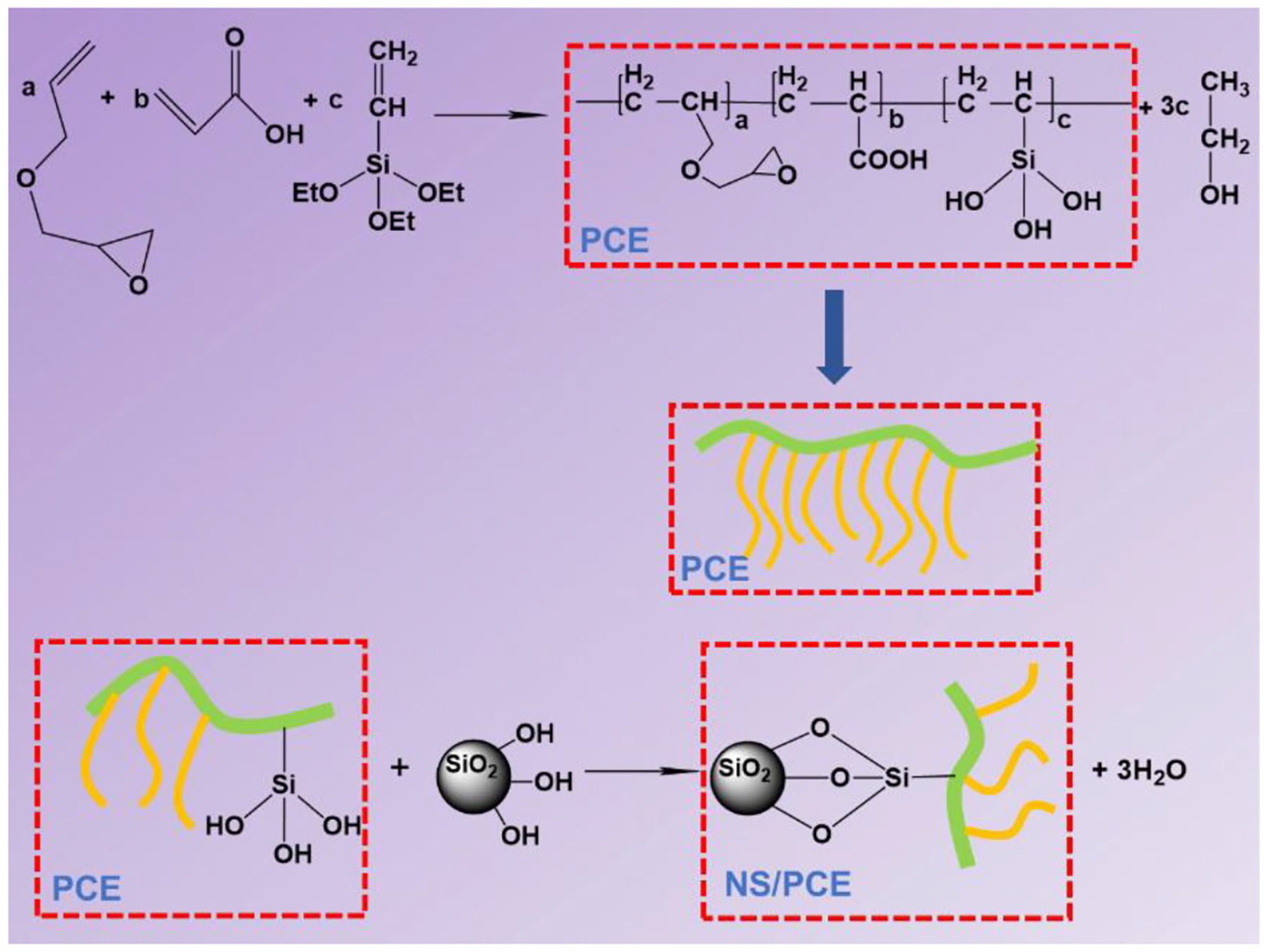
Schematic diagram of NS/PCE formation mechanism.
Effect of NS/PCE on properties of fresh cement pastes
To study the fluidity retention, the initial fluidity of cement pastes containing PCE or NS/PCE was compared with the fluidity after 1 h, as shown in Figure 5(a). Compared with the initial state, the fluidity after 1 h was all reduced, which may be due to the large amount of superplasticizer consumed by adsorption in the initial stage, leaving a small number of free superplasticizer molecules in the cement paste, resulting in a high loss of fluidity after 1 h. The fluidity of cement paste containing NS/PCE remains better compared with PCE, which may be due to silica hydroxyl groups that can be used as adsorption groups in the molecular structure of NS/PCE to increase the adsorption capacity and dispersion capacity.26,27 The cement fluidity of 5% NS/PCE, 10% NS/PCE, and 20% NS/PCE was greater than that of PCE, which may be because NS is successfully grafted onto PCE. The introduction of the silicone hydroxyl group transforms the original electrostatic adsorption into a chemisorption with a stronger adsorption force. 28 The fluidity with 30% NS/PCE has the smallest flow rate, which may be due to the fact that the cement surfaces are covered with NS groups, and the silica hydroxyl groups tend to condense NS to form cross-linked polymers in the alkaline cement slurry, which to some extent destroys the adsorption and dispersion properties of the superplasticizer on the cement particle, resulting in a lower fluidity. 29

(a) Initial fluidity and fluidity after 1 h of cement pastes and (b) fluidity of cement pastes with different clay contents.
To assess the dispersion stability of PCE and NS/PCE in clay, cement slurries containing montmorillonite were prepared. As shown in Figure 5(b), the fluidity of the cement paste decreased with increasing clay content. When 2 wt% of clay was added to the cement, the fluidity of PCE, 5% NS/PCE, 10% NS/PCE, 20% NS/PCE, and 30% NS/PCE decreased by approximately 20.1%, 17.5%, 16.3%, 15.0%, and 18.3% respectively. This indicates that PCE and NS/PCE containing short-branched chains interact weakly with clay impurities. Replacing the long chains of PEG with short-branched chains offers a viable way to mitigate the negative effects of clay on PCE. 30
The adsorption capacities of PCE and NS/PCE on the surface of cement particles are shown in Figure 6. The adsorption capacity of PCE or NS/PCE on cement particles increases with the increase in PCE or NS/PCE. When the content of PCE, 20% NS/PCE, and 30% NS/PCE is 6 mg·g−1 and the content of 5% NS/PCE and 10% NS/PCE is 12 mg·g−1, the adsorption capacity reaches the saturation point. The results showed that the adsorption capacity of 5% NS/PCE and 10% NS/PCE at saturation point was significantly higher than that of PCE, 20% NS/PCE, and 30% NS/PCE. This may be due to the high aggregation rate and grafting efficiency of NS in 5% NS/PCE and 10% NS/PCE, whereas when the content of NS is too high, it is easy to form cross-linked copolymer in the solution, which is not conducive to the adsorption of particles. 19

Adsorption amount of PCE and NS/PCE.
The adsorption mechanism of PCE and NS/PCE on the surface of cement particles is illustrated in Figure 7. For PCE, the adsorption behavior is predominantly through Ca2+ bridging on negatively charged mineral surfaces (e.g. C3S and C–S–H), or electrostatic adsorption between positively charged surfaces of cement particles (e.g. C3A and AFt) and carboxyl groups in PCE. 31 For NS/PCE, the silica hydroxyl groups in NS/PCE dehydrate and condense with the hydroxyl groups on the surface of the cement particles, creating adsorption to the cement particles through chemical bonding. Chemisorption is stronger than electrostatic adsorption, which has been confirmed by scholars. 32 Therefore, the adsorption capacity of NS/PCE is much larger than PCE. While the NS content is excessive, it creates adsorption inhomogeneity in the cement paste and the presence of excess silicone hydroxyl functional groups in the polymer, which tend to condense to form cross-linked copolymers in alkaline cement paste, as shown in Figure 7(c), which reduces the number of silicone hydroxyl functional groups that can provide effective chemisorption, thus greatly reducing the adsorption performance of NS/PCE on cement particles. 33 Therefore, the introduction of an appropriate amount of silica hydroxyl groups into the main chain of polycarboxylate superplasticizer is more helpful to improve the adsorption and dispersion performance of polycarboxylate superplasticizer.

Schematic diagram of dispersion and adsorption: (a) PCE, (b) NS/PCE with appropriate NS, and (c) NS/PCE with excessive NS.
As seen in Table 6, when the fluidity of cement mortar is controlled at 180 ± 5 mm, the water required for mixing cement mortar is obtained. Therefore, the water reduction rate of PCE (or NS/PCE) can be calculated according to the water demand under the same mortar fluidity.
Mix design of mortars and the water-reducing rate of NS/PCE.

Conclusion
To sum up, this work synthesized novel-modified allyl ether-based polycarboxylate superplasticizer from short-chain olefins, with iodine value of PCE and fluidity of cement paste as the main evaluation indicators in an orthogonal test. For the results of the different orthogonal experiments, the optimum reaction conditions were determined: n(AGE):n(AA):n(VTES) = 1:0.8:0.3, ω(APS) = 2.5%, ω(MPA) = 1%, polymerization time = 5 h, and the polymerization temperature = 70°C. The PCE with short-branched chains was successfully synthesized, which made up for the lack of high sensitivity of traditional PCE synthesized by PEG to clay. The NS was grafted onto the PCE molecular chain by free radical polymerization to obtain NS-doped polycarboxylate superplasticizer. The modification of nanoparticles by surfactants provides a research direction for better dispersion of nanoparticles in cementing materials and solves the difficult problem of uniform dispersion of nanoparticles in cement materials. The results showed that NS/PCE synthesized from short-chain olefins had better dispersion, adsorption to cement slurries, and stronger clay resistance.
Experimental section
Chemical and materials
AGE, AA, APS, MPA, VTES, 1,4-dioxane, sodium hydroxide, potassium bromide, sodium thiosulfate, potassium bromate, and starch used in this experiment were all analytically pure, and no further purification was required. NS dispersion with an average particle size of 15 nm and solid content of 30% by mass was provided by Shanghai AI.pure Biotechnology Co., Ltd. (Shanghai, China). The clay sample used in this study was sodium montmorillonite supplied under the trade name of KSF by Shanghai Yuanye Biotechnology Co., Ltd. (Shanghai, China) whose specific surface area is 35 m2 g−1 (Brunauer–Emmett–Teller (BET) method and N2 adsorption). Ordinary Portland cement P·I 42.5 was produced by Fushun Cement Co., Ltd (Sichuan, China), and chemical composition of the cement is listed in Table 7. Standard sand was produced by Xiamen ISO Standard Sand Co., Ltd (Fujian, China).
Chemical composition of cement (wt %).

Preparation
Preparation of PCE
Using 1,4-dioxane and deionized water as double solution system, 6.848 g AGE, 3.425 g VTES, and 1,4-dioxane were added to the three-necked flask. Then, we added 0.393 g APS and 0.236 g MPA and deionized water to the beaker and placed in the drip funnel after it completely dissolves. Dissolved 3.459 g AA in deionized water was placed in another drip funnel. When the reaction temperature reaches 70°C, the solution from the two droplet funnels is added within 30 min. On completion of the addition of solution from the two droplet funnels, the mixture was stirred for another 2 h. The yellow solution was obtained by neutralizing 30 wt% NaOH solution to pH value of about 8.
Preparation of NS/PCE
A series of NS/PCE were prepared with different NS-to-PCE mass ratios. In 5% NS/PCE, 10% NS/PCE, 20% NS/PCE, and 30% NS/PCE, ingredient mass proportions of colloidal NS/PCE were 5%, 10%, 20%, and 30%, respectively, while the same synthetic route was used. Take 5% NS/PCE, for example, 30 g of 30 wt% PCE solution was dispersed in a flask and reacted by adding drops of 1.5 g of 30 wt% NS solution at 60°C within 30 min. Finally, the mixture was stirred for a further 120 min and 5% NS/PCE was obtained.
Preparation of cement pastes, mortars, and cement pastes containing clay
According to the Chinese standard GB/T 8077-2012, 300 g of cement, 87 g of water (water–cement ratio of 0.29), and 0.9 g of PCE or NS/PCE were mixed and stirred to prepare cement slurries. Cement mortar was prepared by mixing 450 g of cement, 1350 g of standard sand, 1.35 g of PCE or NS/PCE, and water. Cement paste containing clay was prepared by mixing 300 g of cement, 3 g of clay (1 wt%), 87 g of water, and 0.9 g of PCE or NS/PCE.
Orthogonal experimental design
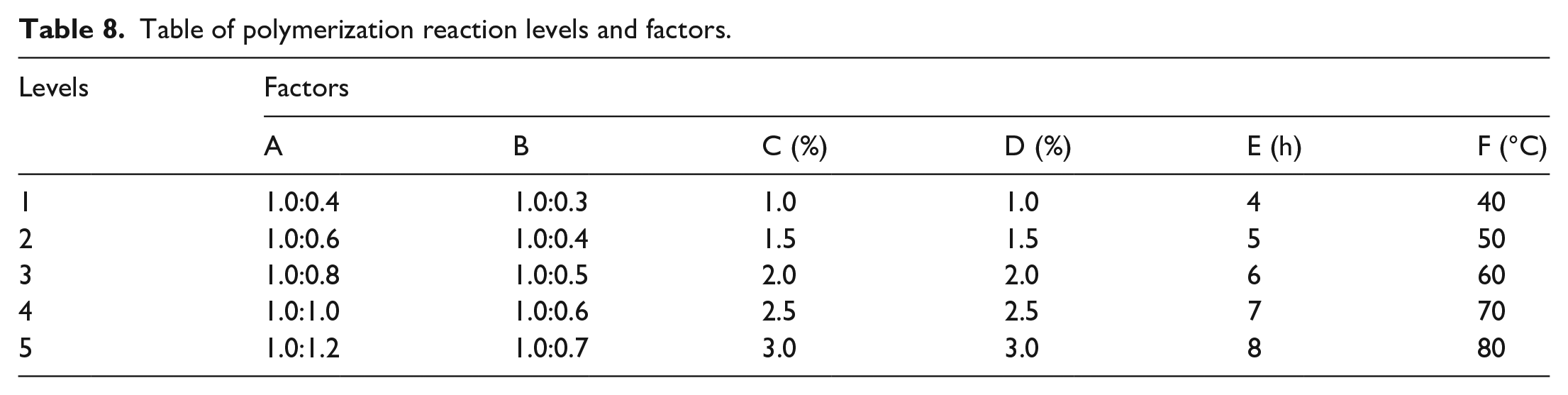
Orthogonal experiments were designed based on the mechanism of free-radical polymerization and microkinetic principles while referring to various literature reports. 34 Orthogonal experiment uses iodine value of PCE and fluidity of cement paste as dual evaluation indicators. The iodine value indicates an index of the degree of unsaturation in organic compounds. The lower the iodine value, the lower the –C=C– content in the product, and the higher the reaction conversion rate. The fluidity of cement paste is an indicator to evaluate the quality of concrete admixture, the higher the fluidity means that the better the compatibility and dispersion between superplasticizer and cement. Meanwhile, the orthogonal experiment selected six factors—the AGE was set at 6.848 g (0.06 mol) and the molar ratio of AGE to AA (A), molar ratio of AGE to VTES (B), mass of initiator of redox system (APS) as a percentage of the total mass of polymerization monomer (C), mass of chain transfer agent (MPA) as a percentage of the total mass of polymerization monomer (D), polymerization time (E), and polymerization temperature (F)—as influencing factors. Five levels of each factor were selected to design the L25 (56) orthogonal experimental table, and the arrangement of each factor and level variables are shown in Table 8.
Table of polymerization reaction levels and factors.
Test methods
The prepared PCE and NS/PCE solutions are directly tested for iodine value and fluidity. The prepared PCE and NS/PCE solutions were dialyzed in a semi-permeable (MD44-3500) membrane for 1 day to remove unreacted small molecules and freeze-dried by vacuum to obtain dried samples for FTIR, GPC, DLS, and TEM tests. The samples were ground to a powder together with KBr, pressed, and then the absorption wavelengths of the samples were determined by FTIR at 400–4000 cm−1. The samples were dissolved in water to form an aqueous solution of 1 mg mL−1 and then filtered through a 0.22 μm microporous membrane before selecting a 0.1 mol L−1 NaNO3 solution as the mobile phase for testing GPC, which was used to determine the relative molecular mass (Mn, Mw) and PDI (PDI = Mw/Mn) of PCE and NS/PCE. Samples were dissolved in 1 mg mL−1 of water for zeta potential and particle size distribution testing by DLS. The samples were dissolved in anhydrous ethanol and ultrasonically dispersed for 5 min, followed by dropwise addition of the sample solution to be tested onto a copper grid and observation of the microscopic morphological changes of the nanoparticles before and after modification using TEM. The amount of superplasticizer adsorbed on cement particles was measured by ultraviolet spectrophotometry (UV-vis), with specific experimental steps referenced in the literature. 35
The iodine value test was carried out in accordance with Chinese standard GB/T 1676-2008. Fluidity of cement paste and water-reducing rate was carried out in accordance with Chinese standard GB/T 8077-2012.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this paper.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this paper: This work was supported by the National Natural Science Foundation of China (grant nos. 52173158 and 32171725), Key Research and Development Program of Jiangsu Province (grant no. BE2021081), Postgraduate Research and Practice Innovation Program of Jiangsu Province (grant no. KYCX20_0125), and Fund Project for Transformation of Scientific and Technological Achievements of Jiangsu Province of China (grant nos. BA2019054 and BA2021044).
