Abstract
This study aims to investigate the efficiency of a pilot prototype system comprising coagulation/flocculation, filtration, and nano-bimetallic iron/copper (Fe/Cu) degradation and adsorption units for the removal of chemical oxygen demand (COD), biological oxygen demand (BOD), color, total nitrogen (TN), total phosphorus (TP), and TSS from real textile wastewater. The total removal efficiencies of the system were 96, 98, 82, 69, 88, and 97%, respectively, using 0.5 g/L ferric chlorides as a coagulant under an optimum adsorption condition of pH 6.0, nano-dosage 1.4 g/L, contact time 80 min, and stirring rate 250 r/min at room temperature. Adsorption isotherms indicated that the removal of COD and TP obeys both Koble–Corrigan and Freundlich adsorption models, removal of color obeys both Koble–Corrigan and Hill adsorption models, and removal of TN and TSS obeys Koble–Corrigan and Khan models, respectively. Avrami kinetic models adequately describe the adsorption data for COD, BOD, TN, and TSS, while pseudo-second-order and intraparticle models described the removal mechanism of color and TSS, respectively. An artificial neural network (ANN) with r2-value exceeding 0.98 is accurate and can be used with confidence in predicting removal efficiencies of the targeted parameters. Sensitivity analysis results showed that the initial concentration was the most influential parameter for TSS removal with relative importance greater than 25%, while the bimetallic Fe/Cu dosage was the most influential factor for all other studied parameters with relative importance greater than 40%. The total treatment cost of the proposed system per m3 after scaling up was found to be US$4.5 for reuse of the treated water for the irrigation of forest trees.
Keywords
Introduction
Effluents from the textile industry usually contain high concentrations of unfixed dyes which can either be degradable or non-degradable. 1 Those toxic chemicals can be derived from fixing agents, surfactants, salts, and organic and inorganic chemicals and characterized by having high chemical oxygen demand (COD), biological oxygen demand (BOD), total suspended solids (TSS), pH, and temperature levels (at least 40°C), as well as having a strong color.2–4 The dyes in water may cause severe health impacts such as dermatitis, nausea, hemorrhage, severe irritation of the skin, and ulceration of the skin. 5 Additionally, high suspended solids in water can lead to the clogging of fish gills and reducing light penetration which reduces the algae’s photosynthesis ability. 3 Organic substances in the textile effluents are difficult to degrade and can be reduced into carcinogenic agents under anaerobic conditions, while the presence of inorganic substances in textile effluents is harmful to aquatic life. 6
All these undesirable characteristics contribute to the priority of treating textile wastewater prior to discharging into the environment. 7 Additionally, finding an efficient cost-effective technique for treating textile wastewaters is obligatory. The mainstream methods for treating real textile wastewater include biological treatment,8,9 the coagulation-flocculation process,10,11 adsorption,12,13 electrochemical treatment, 14 and photocatalytic treatment.15–17 It was found that the biological treatment has low abilities in removing color due to the poor biodegradation of certain types of dyes. 18 Other potential factors that might affect the removal efficiency by biological treatment include organic load, dye concentration, microorganism load, and oxygen concentration. Besides that, the alteration of the temperature between winter and summer has a remarkable effect on the growth rate of the microorganisms that are capable of digesting and breaking down harmful organic and inorganic compounds. Moreover, the long treatment time is not fully consistent with the high effluent flow rates associated with textile industries. 19 Serious disposal problems of large quantities of sludge and by-products are frequently related to coagulation and flocculation techniques. 20 Electrochemical treatment drawbacks lie in the significant amounts of energy required and special equipment requirements. 21 Photocatalytic oxidation is a treatment process in which compounds are decomposed in the presence of light energy. 22 Nanophotocatalysts have a shape dependent feature and greater surface ratio which help enhance the reactivity of catalyst. 23 Furthermore, the nanophotocatalysts have a high ability to degrade water pollutants due to the production of oxidizing agents at the surface of the material.22,24 The photocatalytic process has proven to be efficient in treating wastewaters containing dyes; however, it requires high energy and substantial investment. 25 Adsorption is another common technique for the efficient removal of toxic dyes from industrial wastewater. It is characterized as being an inexpensive technology, has low sludge generation and by-products production, especially when using small adsorbent doses.26,27 Activated carbon (AC), nanoscale zero-valent iron (nZVI), and bimetallic iron/copper nanoparticles (Fe/Cu NPs) are the most commonly used adsorbents.28,29 Nano-bimetallic Fe/Cu particles are distinguished as having high porosity, high reactivity, and large surface area.30,31 The bimetallic Fe/Cu nanoparticles have a high tendency to degrade and adsorb a wide range of organic compounds, such as chlorinated hydrocarbons, chlorinated ethanes, polychlorinated biphenyls, and poly and mono aromatic hydrocarbons. 32
The removal of dye compounds using bimetallic Fe/Cu nanoparticles undergoes two main procedures: reductive-degradation and adsorption. Therefore, bimetallic nanoparticles are preferred for reduction or dechlorination followed by adsorption of most organic compounds. A study has reported a high reduction efficiency of more than 95% for the dyes using bimetallic Fe/Cu nanoparticles. 33 Therefore, the adsorption of dyes onto bimetallic Fe/Cu can be a viable research topic. Various environmental factors can influence the adsorption process in wastewater such as adsorbent dosage, pH, contact time, stirring rate, and concentration. 34 The artificial neural network (ANN) is used to demonstrate the effects of these factors on the adsorption process. ANN involves interconnected neurons which are generally used to simulate human nervous system functions. 35 ANN has revealed a promising potential in driving meaningful relationships between imprecise data by relating input data with each other and with the output data.
The current study was performed at a textile factory located in El-Obour City, Qalyubia Governorate-Egypt. The wastewater effluent flow rate from this factory is about 1000 m3/day. The wastewater influent is generated from the dyeing and finishing of cotton and polyester fabrics. The current quality of the treated effluent did not meet the Egyptian standards of sewage discharge networks or non-fresh waterways. Therefore, this study proposed a novel multistage treatment system that can efficiently treat real textile wastewater. The novelty of the proposed system lies in integrating a tertiary treatment unit based on the nano-concept to the conventional treatment units (coagulation and sand filtration) through use of nano-bimetallic Fe/Cu for improving the final effluent quality. The optimum operating parameters were also identified, especially for the tertiary treatment unit to act as a scale up guideline. The performance of experimental factors (i.e., contact time, nano-bimetallic Fe/Cu dosage, pH, stirring rate, and dye concentration) on the adsorption process and removal efficiency was determined by conducting prototype experiments. Most of the technical literature has focused only on color removal; however, in this study, the authors have investigated the removal efficiency of another five parameters, in addition to the color removal: biological oxygen demand (BOD), chemical oxygen demand (COD), total nitrogen (TN), total phosphorus (TP), and total suspended solids (TSS). These parameters are important for the investigation since the effluent is normally discharged to the sewer system. A kinetic study was conducted to fit the adsorption data. The experimental factors and removal efficiency’s relationship were represented by employing ANN. Additionally, the optimization experiments were used to design a continuous-feed adsorption system to treat real textile wastewater. Finally, a techno-economic assessment including the amortization and operational costs was conducted. Thus, this paper acts as a guideline for textile industries interested in applying the proposed treatment system.
Materials and Methods
Chemicals and Reagents
The following chemicals were used in the current study: ferric chloride (FeCl3•6H2O, 98.5% pure, Arabic Lab.), copper (II) sulfate pentahydrate (CuSO4•5H2O, 99.5%, WINLAB), sodium borohydride (NaBH4, 98% pure, CDH Company), ethanol (C2H5OH, 99%, World co. for sub and med industries), ammonium molybdate ((NH4)6Mo7O24), 99.5, Koch-Light Laboratory Ltd), silver sulfate (Ag2SO4, 99%, Fluka company), mercuric sulfate (HgSO4, extra Pure, Oxford laboratory reagent company), potassium dichromate (K2Cr2O7, 99.5%, Loba chemie), potassium orthophosphate (KH2PO4, 99% pure, ADWIC), stannous (II) Chloride (SnCl2, 99.5% pure, Loba chemie), phenol phthalein (C20H14O4, extra pure, Loba chemie), ammonium persulphate ((NH4)2S2O8, 95.5% pure, Oxford), sodium hydroxide (NaOH, 99% pure, Oxford Co.), sulfuric acid (H2SO4, 95–97%, Honeywell Co.), and standard chrome solution (Cr(NO3)3, 998±5 mg/L, Merck company).
Samples collection and analysis
Raw textile wastewaters samples were collected from the effluent of a textile factory located in El-Obour City, Qalyubia Governorate-Egypt. This factory operates in two 8-h shifts. Four raw wastewater samples were collected daily (every 4 h) for the period from the 22nd to the 26th of December 2019 to represent the effluent variation. A total of 20 samples were collected during this study. The wastewater samples were collected in brown glass bottles involving a 2 mL sulfuric acid/L sample for COD and TP sample analysis. Samples were also collected in plastic containers and a dark condition at 4°C for color, BOD, TN, and TSS analysis.
Treated wastewater samples were withdrawn using a 10-mL syringe by Whatman No. 42 filter paper with 0.2 μm pore size. The collected samples (raw and treated) were analyzed for the following parameters: pH, COD, BOD, color, TN, TP, and TSS. The medium pH was monitored using Multimeter PH/OXI 340 I (WTW 340i/05281018, Germany). The color was measured using HACH DR/2010 spectrometer set at a wavelength of 455 nm, and distilled water was used as a blank. The method applied for the color test is the APHA Platinum-Cobalt Method with the unit of platinum-cobalt (Pt-Co). The other studied parameters (COD, BOD, TN, TP, and TSS) were analyzed according to the Standard Methods for the Examination of Water and Wastewater. 36 The wastewater samples analyses were conducted in a chemical laboratory inside Housing and Building National Research Centre (HBRC) in Cairo, Egypt.
Preparation of nZVI
Approximately 1.08 g of ferric chloride (FeCl3•6H2O) was completely dissolved in a 60 mL 4/1 (v/v) ethanol/deionized water mixture. The reducing agent was prepared by dissolving approximately 0.76 g NaBH4 in 200 mL of deionized water. The reducing NaBH4 solution was poured in a burette and slowly dripped dropwise into the FeCl3 solution with a rate of 0.05 mL/2 s. The black precipitate immediately formed after the addition of the first drops of NaBH4 solution as described in equation (1). The chemical reduction between NaBH4 and FeCl3 was used to form black-colored nZVI. The mixture was stirred for another 10 min after adding the excess amount of NaBH4 to complete the FeCl3 reduction. Then, the precipitated iron nanoparticles were separated from the liquid solution using normal filtration technique. Finally, the chemically prepared nZVI was dried at 80°C for 3.0 h. For storage, the prepared nZVI were protected against oxidation by adding a layer of acetone
Preparation of bimetallic Fe/Cu nanoparticles
The preparation of bimetallic Fe/Cu NPs was carried out in a chemical laboratory belongs to the Housing and Building National Research Center (HBRC). The bimetallic Fe/Cu nanoparticles were prepared according to a study conducted by Zin et al. 37 Approximately 1.0 g of freshly prepared nZVI was added into a copper sulfate (CuSO4●5H2O) solution at a flow rate of 0.1 g per 60 s with powerful stirring. CuSO4●5H2O solution was prepared by mixing 0.1 g of CuSO4 with 100 mL ethanol/distilled water (DW) (1:1) at 60°C. Afterward, the solution was allowed to settle for 15 min. The color of black nZVI was changed into a coppery color. The solution was filtrated using two sheets of Whatman filter papers (41 circles, diameter 150 mm). After filtration, the bimetallic Fe/Cu nanoparticles were washed two times with 20 mL of absolute ethanol (99.99%, HPLC grade). The prepared Fe/Cu nanoparticles were dried in the oven for 5 h at 60°C and stored under nitrogen and covered with a thin parafilm layer. 32
Characterization of the prepared Fe/Cu nanoparticles
The prepared bimetallic Fe/Cu nanoparticles were investigated using X-ray powder diffraction (XRD) techniques. An X-ray diffractometer (PANalytical’s X’Pert PRO MRD, the Netherlands) with a voltage value of 40 kV and a current value of 30 mA was used for this purpose. The bimetallic Fe/Cu nanoparticles were placed in a sample holder made of stainless steel and then inserted into the X-ray diffractometer. The XRD patterns were recorded using Copper K-alpha radiation (wavelength 1.54 Å). The scan range was recorded from 10o to 60o with a step size of 0.02o to cover the copper, iron, and iron oxides. Scanning Electron Microscope (SEM) (Philips, Quanta 250 field emission gun (FEG), USA) at a voltage of 25 kV and total magnification × 16 k was used to identify the morphological characteristics of prepared bimetallic Fe/Cu nanoparticles. Energy-dispersive spectroscopy (EDS) connected to the SEM was also used to identify the chemical characterization of the Fe/Cu nanoparticles.
Experimental work
Prototype experiments
A prototype system was created which consisted of an equalization tank, coagulation/flocculation tank, sedimentation tank, sand filter unit, adsorption tank, and sand filter unit. A schematic diagram of the coagulation, sedimentation, adsorption, and filtration/system is shown in Figure 1. Supplementary Figure 1 contains a photograph of the experimental system. All units are made of polyvinyl chloride (PVC). The equalization tank has a diameter of 60 cm and a height of 90 cm and has one tap in the side of the tank to control the flow. Coagulation/flocculation and adsorption tanks have a diameter of 25 cm and a height of 30 cm. Each of these two tanks contains two taps (one on each side) to control the input and output flow. These tanks also contain mechanical motors to help achieve a full mixing of the wastewater and the coagulant or adsorption material. The sedimentation unit has dimensions of 90 cm length × 45 cm width x 45 cm height and the conical part at the bottom of the tank helps to collect the sludge. The sedimentation tank has three taps: one in each side to control the input and output flow, as well as a tap at the bottom of the tank for sludge collection, and two sand filters, each having dimensions of 25 cm height and 20 cm diameter. The water enters the sand filter from the top and a small hose is fixed at the end-side of the filter to collect the filtered water. Schematic diagram of prototype system for the treatment of real textile industrial effluent.
The tests conducted at different operating parameters from pH, sorbent dose, contact time, stirring rate and initial concentration.

Calculations of treatment performance
The removal efficiencies of the studied contaminants (%R) were calculated using equation (2)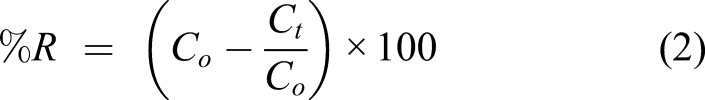
The adsorption capacity of bimetallic Fe/Cu NPs was estimated using equation (3)
Isotherm studies
Different isotherm studies were performed to describe the adsorption process onto bimetallic Fe/Cu NPs for monolayer and multi-layer nonlinear adsorption models: Freundlich, Langmuir, Toth, Sips, Hill, Redlich–Peterson, Khan, Koble–Corrigan, and Jovanovich. The nonlinear equations of these models were obtained from Mahmoud et al.. 32
Kinetic studies
To determine the suitable time for reaching the equilibrium state, the sample was placed in contact with bimetallic Fe/Cu NPs at different times, at room temperature. The amount removed at time t, Qt (mg/g), was calculated using equation (4)
To investigate the mechanisms of the kinetic process, the following kinetic techniques were applied: pseudo-first and second-order, intraparticle, Avrami, and Elovich. 32
Validation of adsorption isotherms and kinetics
A derivative of Marquardt’s Percent Standard Deviation (MPSD) error function equation was used to evaluate a better fit of nonlinear isotherm and kinetic models and to choose the most suitable model that can describe the treatment process, as listed in equation (5)
Artificial intelligence
An artificial neural network (ANN) model was built and tested to predict the removal efficiencies of the targeted parameters (COD, BOD, color, TN, TP, and TSS). The model has 3 main elements: (1) input layer which receives data from five experimental factors (pH, contact time, bimetallic Fe/Cu dosage, stirring rate, and concentration), (2) hidden layer which is comprised of ten neurons; their numbers are normally identified by trial and error, and (3) output layer which represents the removal efficiencies of the targeted parameters. Figure 2 shows the ANN structure which is expressed as 5–10–1. The input and target data were randomly separated into 20%, 20%, and 60% for testing, validation, and training procedures, respectively.
39
ANN of 5–10–1 structure used for the prediction of studied parameters removal efficiency.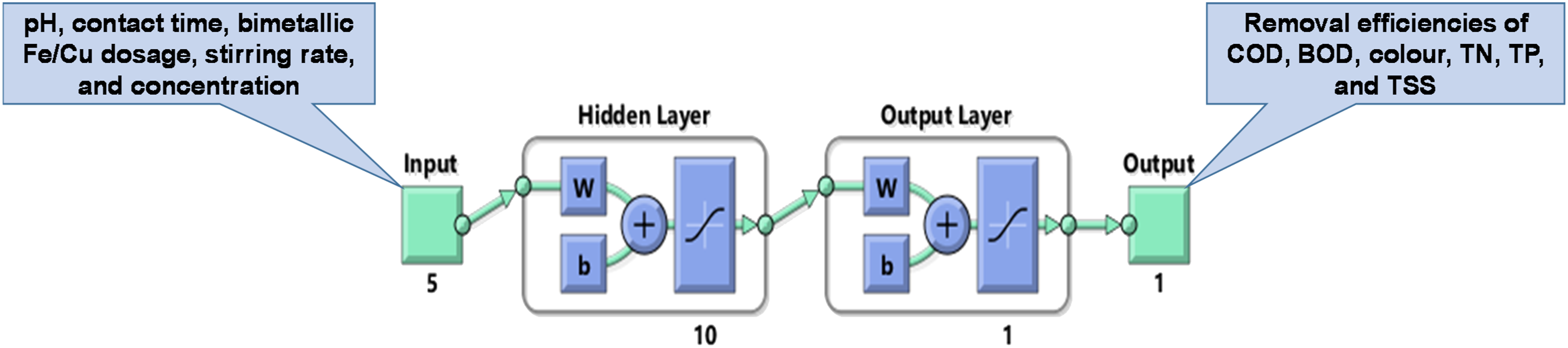
Equation (6) shows the “tansig” transfer function which was chosen for the hidden layer, while equation (7) shows the “purlin” activation function which was chosen for the output layer. A feedforward backpropagation algorithm was used for data classification. The comparison between the target and the output data was conducted by calculating the mean squared error (MSE). The MSE was also used to adjust both the weights and the biases, which occurs through passing back the MSE from the output node to the input layer (equation (8)). The training step was conducted using Levenberg–Marquartd method (trainlm) which is suitable for small and medium-size networks.
32



Sensitivity analysis was performed using multi-layer perceptron (MLP) nonlinear neural network regression model and linear regression analysis using an enter method to identify the relative importance of each operating parameter (contact time, nano-bimetallic Fe/Cu dosage, pH, stirring rate, and concentration) on the removal efficiency of the studied parameters (COD, BOD, color, TN, TP, and TSS).
Results and discussion
Characterization of bimetallic Fe/Cu
Figure 3(a) shows the XRD pattern of Fe/Cu nanoparticles with an angle (2θ) from 5° to 70°. The XRD pattern shows three peaks at (2θ) equal 43.3, 44.7°, and 50.8° for planes Cu (111), Fe (101), and Cu (200), respectively. A sharp peak at 2θ = 44.7° indicated a dominance of zero-valent iron (Fe0) in the prepared bimetallic Fe/Cu sample. Similar findings were previously observed in studies by Khosravi and Arabi
40
and Hamdy et al.
1
Figure 3(c) shows an EDX analysis of the prepared Fe/Cu NPs which indicates that the weight percent of all bimetallic Fe/Cu components. The EDX results showed the presence of C, O, Fe, and Cu elements with weight percents of 19, 10, 66, and 5%, respectively. The C signals may be due to the C-containing compounds shaped during the washing the prepared Fe/Cu nanoparticles with ethanol.
41
The O signals could be attributed to the formation of oxide(s) in the outer layer of nanoparticles. This formation normally occurs due to the reaction of nanomaterials with water or air during inspection and observation.28,41 Figure 3(c) and (d) shows an SEM image of the synthesized bimetallic Fe/Cu NPs samples and the results indicated that there is an irregular surface structure with particle size ranging between 20 to 30 nm. The SEM image showed that the prepared nanoparticles contain many pores that allows easier mass transfer and diffusion of molecules to the inner of the nanomaterial.1,42 The SEM image also showed that some nanoparticles agglomerated together to form larger nanoclusters. This likely occurs since the Fe/Cu nanoparticles are prepared by mixing freshly prepared nZVI, which is already agglomerated, with copper sulfate solution, as mentioned in the Preparation of bimetallic Fe/Cu nanoparticles section. The interconnection of iron nanoparticles with one another is mainly attributed to the static magnetism and the surface tension.28,43,44 Shih et al.42 and Hamdy et al.1 have also observed similar chain-like aggregates while preparing iron nanoparticles. Characterization of the nZVI: (a) XRD; (b) EDAX; (c) SEM 1 μm; (d) SEM 500 nm.
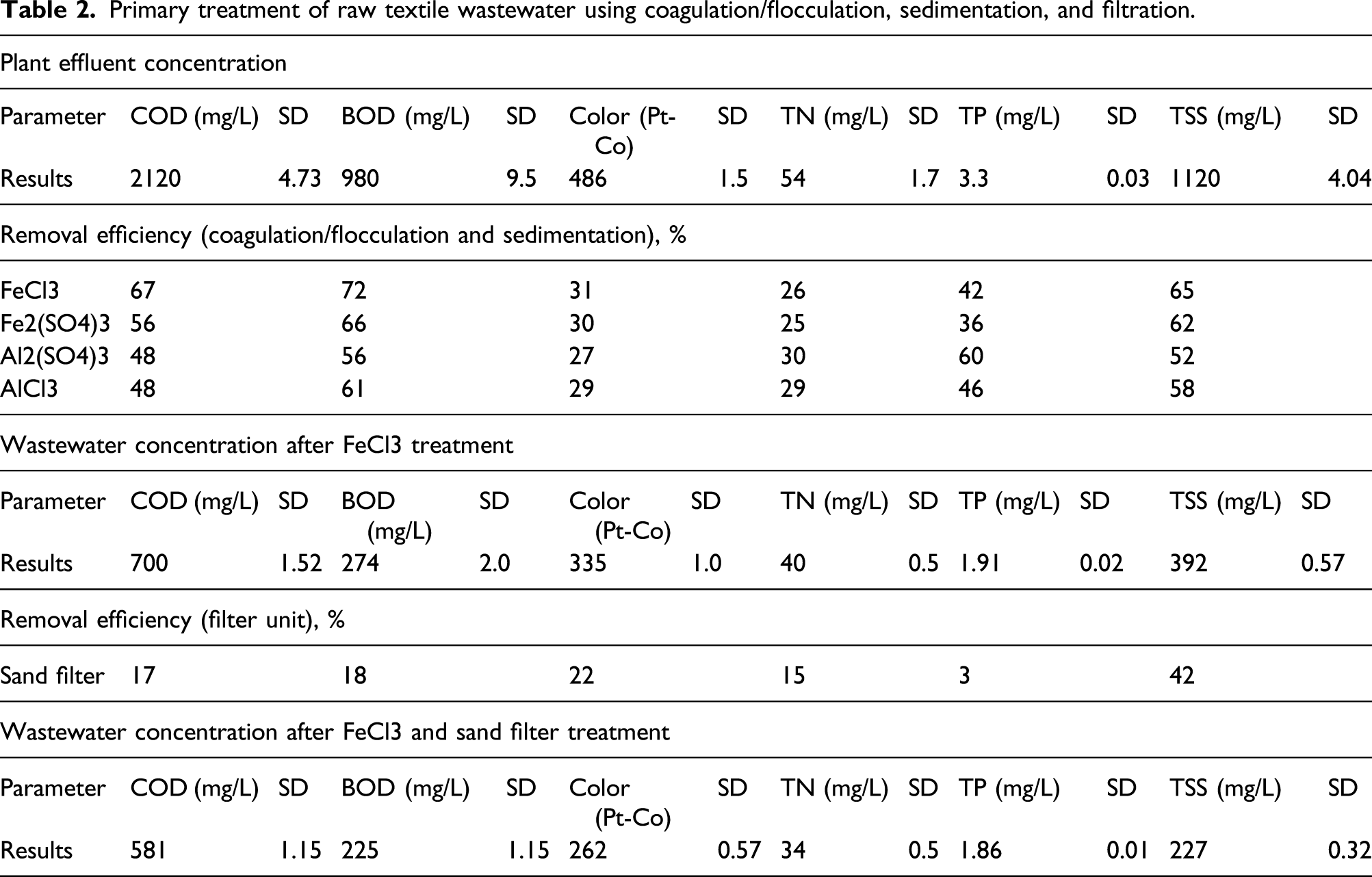
Primary Treatment Processes
Primary treatment of raw textile wastewater using coagulation/flocculation, sedimentation, and filtration.
Effects of operating parameters for the adsorption/filtration processes
Effect of pH
The effect of pH was studied in acidic, neutral and alkaline media at various pH values ranging from 2 to 10. The results indicated that the effect of pH is a significant parameter that affects the removal percentages as shown in Figure 4(a), where the maximum removal percentages were 87%, 91%, 66%, 50%, and 84% at pH 6 for COD, BOD, color, TN, and TSS removals, respectively. On the other hand, the phosphorus removal percentages gradually decreased with increasing pH values and the maximum removal percentages were 93% at pH 2 and reached 79% at pH 6.0. At acidic pH, considerable amounts of free electrons were exhausted to neutralize H+ in the acidic solution leading to a decrease in the degradation process of organic matter from textile effluent and the physical adsorption process takes place.
39
Also, significant amounts of bimetallic Fe/Cu doses were dissolved by the acid leading to a decrease in the vacant sites of sorbent material except for the case of phosphorous. The effect of the aeration process and the acid effect may transfer a significant amount of insoluble organic phosphorous into inorganic phosphate which may be suitable to chemisorbed phosphorus at nano-bimetallic surfaces. At pH 6 to 7, the nano-bimetallic Fe/Cu reaches the point of zero charges which is the most suitable case for the degradation process.
24
In alkaline media for pH greater than 7.0, the steric hindrance of negative OH− decreases the effect of free electrons for organic degradation, decreases chemisorption of charged negative molecules, and decreases the ability of the physical adsorption processes. Several studies have been conducted with textile wastewater treatment, and the results obtained indicated the effective pH was ∼ 6.0.
33
Effects of experimental factors on the removal of efficiency of COD, BOD, color, TN, TP, and TSS by Fe/Cu nanoparticles: (a) pH; (b) Fe/Cu nanoparticles dosage; (c) Contact time; and (d) stirring rate.
Effect of Fe/Cu nanoparticles dosage
Figure 4(b) shows the effects of Fe/Cu nanoparticles dose (0.2–3 g/L) at an initial pH of 6.0, room temperature 30°C, contact time of 80 min, and stirring rate of 250 r/min on the removal efficiency of all tested parameters. The removal efficiencies for all tested parameters were enhanced with an increase in the Fe/Cu nanoparticles dosage. For instance, when the dosage of Fe/Cu nanoparticles was increased from 0.2 g/L to 1.4 g/L, the removal efficiency increased from 61% to 87% for COD, from 70% to 91% for BOD, from 46% to 66% for color, from 38% to 50% for TN, from 56% to 79% for TP, and from 76% to 84% for TSS. This is mainly due to the increase in the number of vacant sites with increasing Fe/Cu nanoparticles dosage, 45 thereby entrapping significant amounts of molecular contaminants. However, less improvement in the removal efficiency was noticed with an increase in the Fe/Cu nanoparticles dosage greater than 1.4 g/L. This is could be due to the surplus of Fe/Cu nanoparticles dosage which may cause overlapping of the pore structure of the adsorbent, decreasing the interparticle distances, and shielding the binding sites from pollutants due to the increased boundary layer screening effect. 46
Effect of contact time
Effect of contact time on maximum uptake behavior for textile wastewater treatment using Bi Fe/Cu NPs.

Effect of stirring rate
Figure 4(d) displays the effects of stirring rate in the range of 50–300 r/min at an initial pH of 6.0, room temperature 30°C, Fe/Cu nanoparticles dose of 1.4 g/L, and contact time of 80 min on the removal efficiency of all the studied parameters. The optimum removal efficiencies for all tested parameters were recorded at a stirring rate of 250 r/min: 87% for COD, 91% for BOD, 66% for color, 50% for TN, 79% for TP, and 84% for TSS. Increasing the stirring rate to 250 r/min increased the chance of dye molecules to come in contact with Fe/Cu nanoparticles and thus improved the liquid−solid mass transfer.
Effect of initial concentration
Effect of initial concentration on maximum uptake behavior for textile wastewater treatment using Bi Fe/Cu NPs.

The efficiency of the system
Figure 5 summarizes the removal efficiency of the studied parameters after each treatment process. The removal efficiencies for COD, BOD, color, TN, TP, and TSS reached 73, 77, 46, 37, 44, and 80%, respectively, after using 0.5 g/L ferric chloride as a coagulant, followed by sand filtration. The total removal efficiency of the system after the adsorption unit followed by sand filter was 96, 98, 82, 69, 88, and 97%, respectively, with optimum adsorption conditions of pH 6, nanodosage 1.4 g/L, contact time 80 min, stirring rate 250 r/min at room temperature. The removal efficiency of the studied parameters after each treatment process.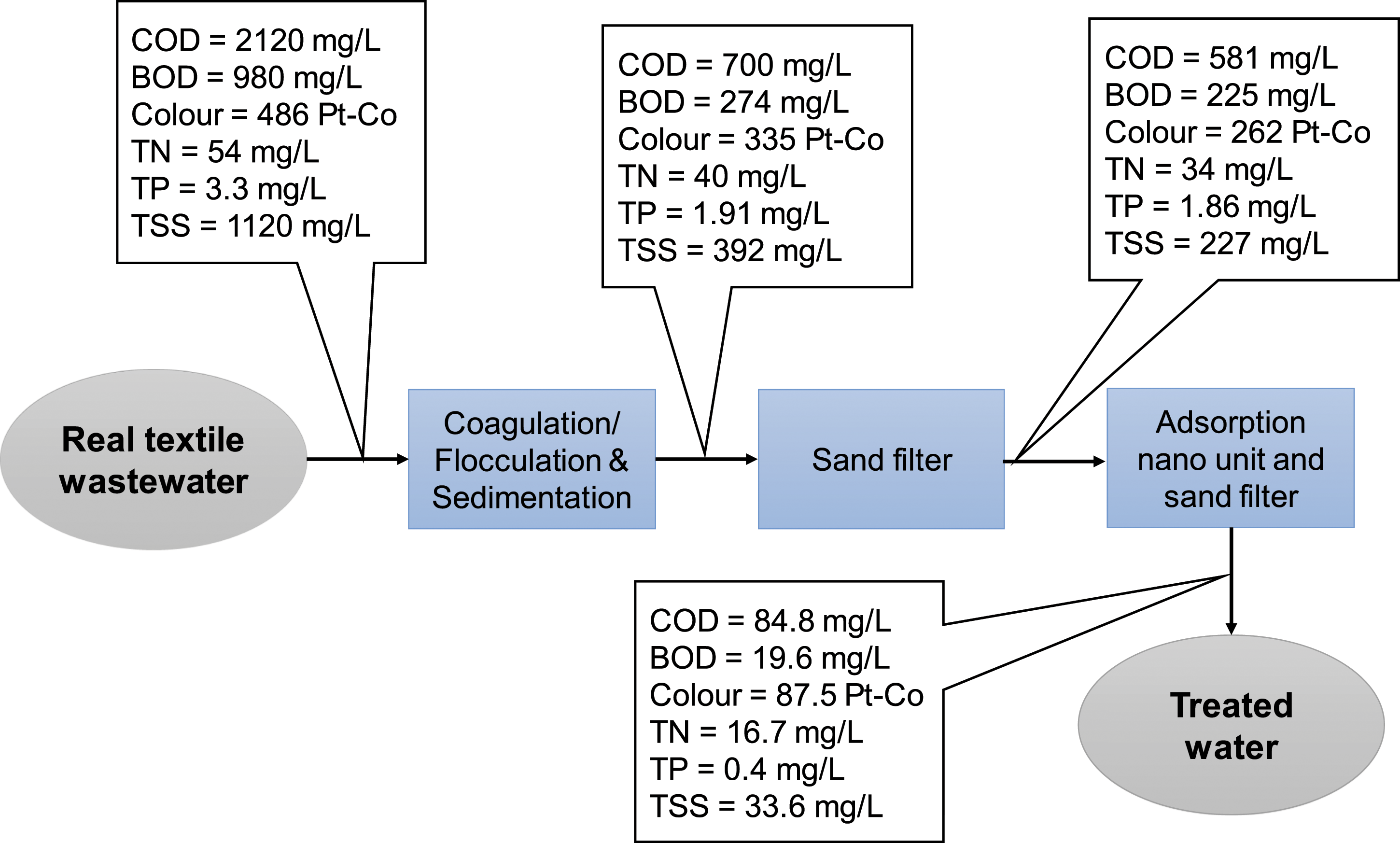
Adsorption isotherm studies
Coefficient of determination and constants of different adsorption isotherm models.
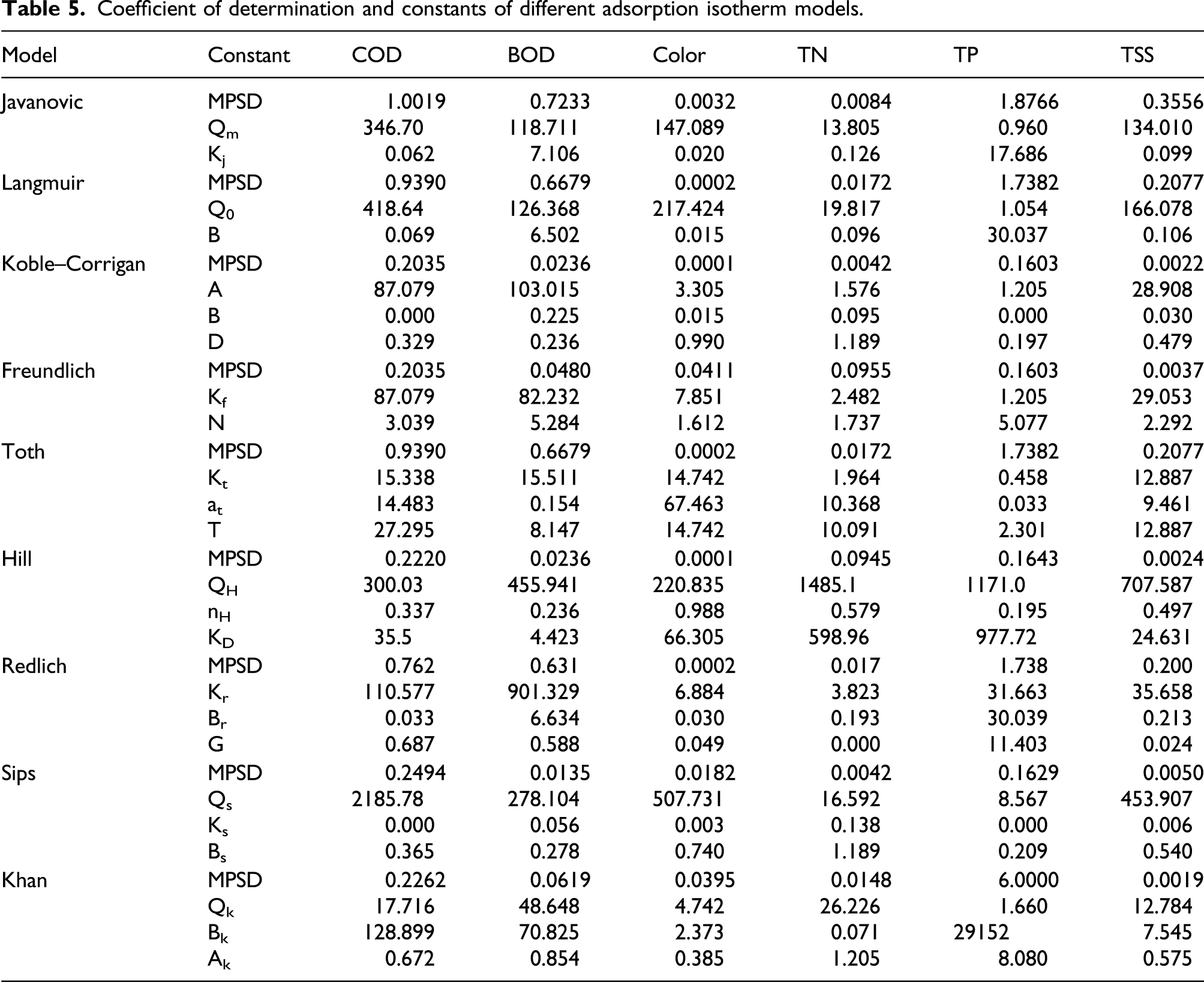
The color adsorption can be adequately described by the Koble–Corrigan and Hill models. Hill models assume formation of macromolecules in the bimetallic Fe/Cu NPs sites, so the adsorption mechanism act as cooperative phenomenon between color and bimetallic Fe/Cu NPs. The TN adsorption onto bimetallic Fe/Cu NPs obeys both the Koble–Corrigan and Sips models. Finally, the adsorption mechanism for TSS removal efficiency obeys the Khan model which can describe the maximum TSS uptakes onto Fe/Cu NPs. 52 Khan results indicated that the maximum uptake was 12.78 mg TSS/g Fe/Cu NPs which means there is another effective method that can affect the TSS removal efficiency, involving the degradation process. 53
Kinetic studies
Coefficient of determination and constants of different kinetic models.
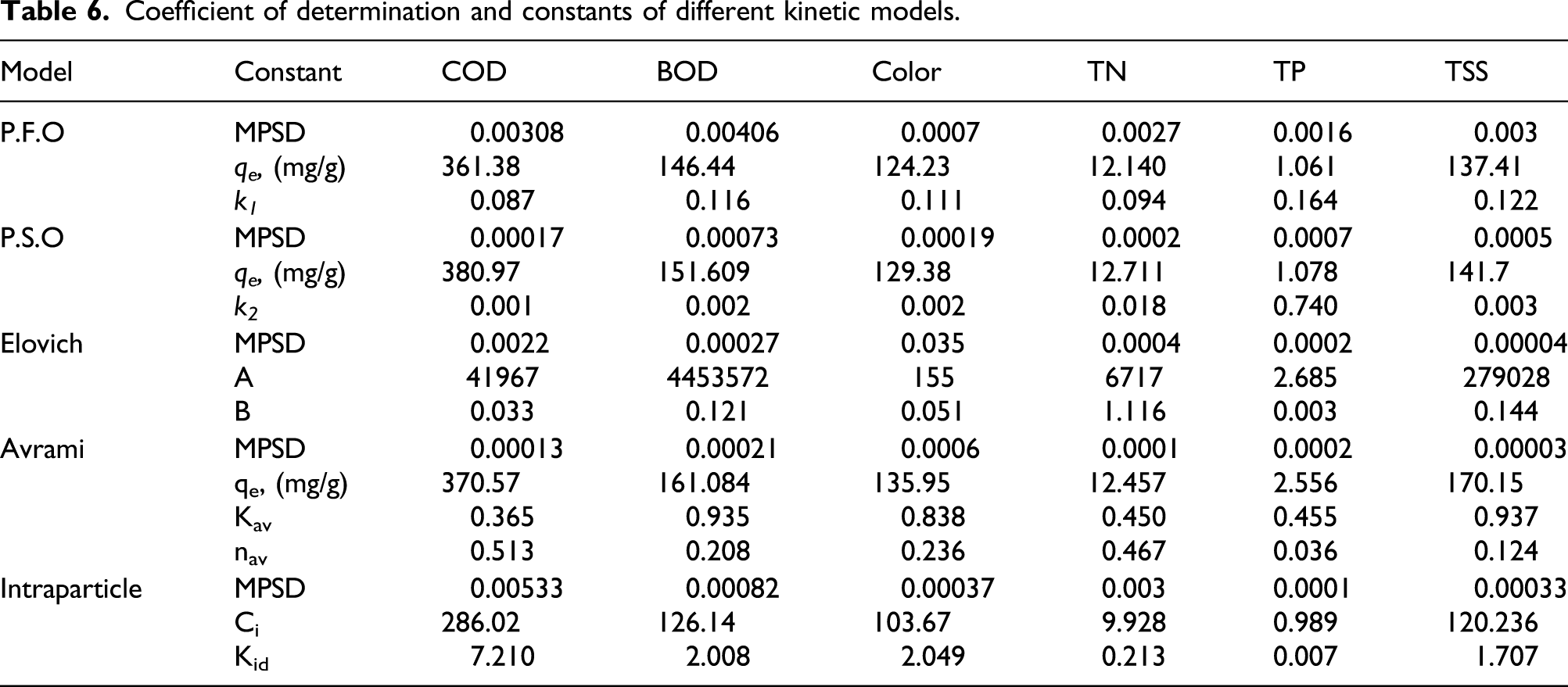
Real textile wastewater treatment by various adsorbents
Treatment of real textile wastewater using various adsorbents.

NA: Not available.
Artificial intelligence
Adjusted weights and biases
A weight matrix (W10×5) was generated as a result of the connection between each hidden layer neuron and each component of the input vector (P5×1). A net input (u10×1 = ΣW10×5.P5×1 + b10×1) was generated by adding the sum of the weighted inputs (ΣW10×5.P5×1) to a 10-length bias (b10×1). A “tansig” function was then used to transfer the calculated net input to the output layer. A weight matrix (W1×10) was generated as a result of the connection between the output layer single neuron and each hidden layer neuron (P10×1). A net input (u1×1= ΣW1×10.P10×1 + b1×1) was produced by adding the weighted inputs (ΣW1×10.P10×1) to 1-length bias (b1×1). This net input is then transferred using a “purlin” function to the output layer.
Training and validation performance
The magnitudes of the performance gradient for all tested parameters (COD, BOD, color, TN, TP, and TSS) ranged from 0.238 to 5.25, as shown in Figure 6. Since these values exceeded the least error level of 1 x 10– 5, the training process was terminated after 6 epochs. Figure 7 shows the mean square error (MSE) value at each iteration number. For all the tested parameters, the MSE for the training phase decreased with the increase in the number of epochs until reaching the lowest magnitude at epoch 6. This performance is common and does not pose a problem in the training phase. After epoch 0, the MSE for the validation step gradually increased, demonstrating that the created ANN model is overfitting the data. The same behavior was shared between the validation and test curves, where they increased in similarly with the number of epochs. The best validation performance was 0.404, 0.105, 0.174, 0.249, 0.211, and 0.212 at epoch 0 for COD, BOD, color, TN, TP, and TSS, respectively. Training performance: (a) COD; (b) BOD; (c) color; (d) TN; (e) TP; (f) TSS. Best validation performance: (a) COD; (b) BOD; (c) color; (d) TN; (e) TP; (f) TSS.

Regression plot
The coefficient of determination (r2-value) between the outputs and the targets of the generated network is presented in Figure 8. The solid line denotes the line of best fit, while the dashed line represents the line of perfect results. For all tested parameters, a good fit was obtained between the output and the target data for training, validation and test stages with r2-values ranged from 0.981 to 0.998 for the training step, 0.971 to 0.999 for the validation step, and 0.994 to 0.998 for the test step. The overall r2-value for all tested parameters ranged from 0.983 to 0.997, indicating the model’s accuracy and reliability. This indicates that more than 98% of the variations in the removal efficiency of the tested parameters (COD, BOD, color, TN, TP, and TSS) were explained by the tested environmental factors (pH, contact time, bimetallic Fe/Cu dosage, stirring rate, and concentration). Consequently, the developed ANN was able to accurately estimate the removal efficiency for the six tested parameters in the tested range of the five environmental factors. Regression plot between target and output: (a) COD; (b) BOD; (c) color; (d) TN; (e) TP; (f) TSS.
Sensitivity analysis
The relative and normalized importance of ANN operating parameters (contact time, nano-bimetallic Fe/Cu dosage, pH, stirring rate, and concentration) on the removal efficiency of the studied parameters (COD, BOD, color, TN, TP, and TSS) is shown in Figure 9. All the operating parameters had strong effects on the removal efficiency of the studied parameters. However, the bimetallic Fe/Cu dosage exhibited the most important factor among the operating parameter with a relative index greater than 40%, except with TSS removal, where the initial concentration was the most dominant factor followed by the bimetallic Fe/Cu dosage. Therefore, none of the studied operating parameters could be excluded from this study. These findings indicated that the developed network could express the performance of the adsorption process within the range of the operating environmental conditions. Sensitivity analysis of the relative and normalized importance of ANN operating parameters on the removal efficiency of the studied parameters.
Continuous-feed and scaling-up studies for textile wastewater treatment
A scaling-up design was performed for the proposed treatment system intended as a continuous-feed operation for a daily flow rate of about 1,000 m3.
57
The required volume in the cubic meter for each tank was calculated using equation (9).
Figure 10 shows a flow chart of the proposed treatment system. The proposed treatment system after scaling up comprises: (1) an equalization tank (1,000 m3) for textile wastewater storage, (2) coagulation/flocculation tank (10.0 m3) designed for a hydraulic retention time of 2 min and stirring rate of 100 r/min for the coagulation process and a hydraulic retention time of 10 min and a stirring rate of 50 r/min for the flocculation process, (3) sedimentation tank (10.0 m3) to allow flocs to settle out of the wastewater, (4) sand filter to remove suspended matter and any floating particles, (5) adsorption tank (85.0 m3) which has been designed based on the optimum contact time obtained from the prototype experiments of approximately 80 min, and (6) a sand filter to separate the treated water from the loaded nanoadsorbent. Flow chart of the proposed treatment system with the inputs and the outputs.
Reusing treated wastewater in irrigation
Since the treated wastewater is planned to be reused for the irrigation of forest trees, its quality must achieve the Egyptian regulatory limits listed in Ministerial Resolution No. 44 for the year 2000: COD, BOD, and TSS concentrations must not exceed 80, 40, and 40 mg/L, respectively. Based on the prototype experiments, COD, BOD, and TSS concentrations at the effluent could reach 75, 20.3, and 36.4 mg/L, respectively, at the following optimum environmental conditions: an initial pH of 6, room temperature 30°C, contact time of 80 min, stirring rate of 250 r/min, and Fe/Cu nanoparticles dosage of 1.4 g/L. Since the concentrations of these pollutants did not exceed the Egyptian regulatory limits, the treated effluent from the proposed pilot prototype system is safe for reuse for irrigation of forest trees.
Cost analysis of the proposed system
A detailed economic analysis was conducted for the proposed system for the scale up system. The annual capital expenditures (CAPEX) and operating expenses (OPEX) for each unit were estimated based on optimum environmental conditions obtained from the prototype experiments. The annual benefits expected from the construction of the system were also calculated based on the assumption that the treated wastewater will be used for irrigation purposes.
Annual capital expenditures
The annual capital expenditure represents the annual interest outflows of CAPEX over the lifetime of each treatment unit in the proposed system. It is calculated by dividing the CAPEX by the annuity factor (AF). The CAPEX for each unit was obtained from local suppliers based on the design volume of the tanks that can be obtained from the previous section. The AF depends on the expected lifetime of the treatment unit and the annual interest rate. The annual capital expenditure was computed using equation (10)
Operating expenses (OPEX)
The OPEX was calculated based on four main parameters: (1) coagulant and adsorbent materials (FeCl3 and bimetallic Fe/Cu NPs), (2) energy need to operate the system, (3) maintenance costs, and (4) labor cost.
58
The cost of the chemicals was calculated using equation (11)
C is chemical dosage (g/m3) (i.e., coagulation: 500 g FeCl3 per m3 and adsorption: 1,400 g bimetallic Fe/Cu NPs per m3)
P are the expenses per gram of chemical ($/g) (i.e., FeCl3: US$0.3/kg and bimetallic Fe/Cu NPs: US$4.35/kg)
The energy cost was calculated using equation (12)
E is the electricity cost per kilowatt-hour (kWh) ($/h) (i.e., US$0.1/kWh)
For the whole system, labor costs were calculated assuming 2 workers will be hired each of them working 60 h/week, and 4 weeks/month, with a salary of US$6/hour in Egypt. The maintenance cost (MC) was assumed as 2% of the CAPEX. 28
Total cost
CAPEX and OPEX of the proposed treatment system.

20 years-life-time and 6% interest.
Environmental considerations for disposal of loaded adsorbent
Safe disposal of loaded adsorbent is one of the most important issues facing industrial wastewater treatment using the adsorption technique. Nowadays, landfilling is the most widely used practice for textile sludge management and disposal. 59 Recent research has been published that addressed the reuse of incinerated textile sludge in the manufacture of some building materials, such as tiles, bricks, and ceramics.61–63 In other studies, textile and paint wastewater were used safely in the manufacture of the tiles and bricks,64,65 which may support the use of textile sludge after incineration in the building materials industry. However, sludge management and disposal must be carried out under strict conditions to ensure the safe processing of sludge and to avoid air pollution and the release of ashes containing hazardous volatiles and heavy metals. Hence, the safe management of textile sludge is still under examination.
Conclusions
This study investigated the efficiency of a pilot prototype system comprising coagulation/flocculation, filtration and nano-bimetallic iron/copper (Fe/Cu) degradation and adsorption units in the treatment of real textile wastewater. The removal efficiencies reached 73, 77, 46, 37, 44, and 80%, respectively, after using 0.5 g/L ferric chloride as a coagulant, followed by sand filtration. This study also demonstrated that using Fe/Cu nanoparticles is effective in treating real textile wastewater. The results of the prototype system showed that the optimum environmental condition for the reduction of COD, BOD, color, TN, TP, and TSS from real textile wastewater was contact time = 80 min, pH = 6, stirring rate = 250 r/min, and Fe/Cu nanoparticles dose = 1.4 g/L at room temperature. At the optimum condition, the achieved removal efficiency after passing through all the units of the system was 96, 98, 82, 69, 88, and 97%, respectively. Isotherm studies showed that not all tested parameters obey one single isotherm model. The Avrami kinetic model adequately describes the adsorption data for COD, BOD, TN, and TSS, while the pseudo-second-order and intraparticle kinetic models describe the removal mechanism of color and TP, respectively. Under the tested experimental conditions, the developed ANN model with a structure of 5–10–1 was able to accurately predict the removal efficiency of the tested parameters with r2 value ranged from 0.983 to 0.997. Sensitivity analysis showed that the operating parameters (i.e., contact time, nano-bimetallic Fe/Cu dosage, pH, stirring rate, and concentration) had considerable effects on the removal efficiency of the studied parameters. However, the initial concentration was the most influential factor for TSS removal with relative importance of greater than 25%, while the bimetallic Fe/Cu dosage was the most influential factor for all other studied parameters, achieving a relative importance of greater than 40%. The optimum conditions obtained from the prototype experiments were used to design the proposed system at a larger scale to treat about 1,000 m3 of real textile wastewater daily. The total treatment cost (including annual CAPEX and OPEX) per cubic meter of the proposed system was US$4.5 for the reuse of treated textile wastewater for the irrigation of forest trees. The present study indicated that the proposed novel multistage treatment system (coagulation followed by adsorption using Fe/Cu nanoparticles) can efficiently treat real textile wastewater at a reasonable price. The study also indicated that the designed ANN model is reliable and can be used to optimize the removal efficiencies of COD, BOD, color, TN, TP, and TSS from textile wastewater by Fe/Cu nanoparticles under various environmental conditions.
Future perspectives
Although the proposed multistage treatment system proved to be effective in treating textile wastewater, two main improvement can be applied on the system to increase its applicability. First improvement involves replacing the chemically prepared Fe/Cu nanoparticles by green synthesized nanomaterials in order to reduce the treatment cost. For example, Abdel-Aziz et al. 66 have synthesized Ficus Benjamina zero-valent Fe/Cu nanoparticles and used it for caffeine removal from aqueous solution. In another study, Farag et al. 67 have synthesized green nano-iron carbide and used it for phosphate removal from aqueous solutions. Fahmy et al. 68 provided a comprehensive review for different synthesis methods of iron oxide and zero-valent nanoparticles from several plant extracts and their possible application in dye removal.
Secondly, improvement involves entrapping the nanoparticles in a biopolymer material to allow the reuse of the nanomaterial. Entrapping the nanoparticles will also help avoid the toxic effect that could occur in the presence of nanoparticles with high concentration in water after treatment. Mahmoud et al. 69 have used the nano-iron entrapped in alginate biopolymer for removal of organochlorine pesticides from aqueous solutions. El-Shafei et al. 70 have used the same nanomaterial for removal of benzene, toluene, ethyl benzene, and xylene (BTEX) compounds. In another study, Liu et al. 71 have used the nano-iron entrapped in chitosan beads for removal of hexavalent chromium from wastewater.
Supplemental Material
sj-pdf-1-nax-10.1177_18479804211041181 – Supplemental Material for A prototype of textile wastewater treatment using coagulation and adsorption by Fe/Cu nanoparticles: Techno-economic and scaling-up studies
Supplemental Material, sj-pdf-1-nax-10.1177_18479804211041181 for A prototype of textile wastewater treatment using coagulation and adsorption by Fe/Cu nanoparticles: Techno-economic and scaling-up studies by Ahmed S Mahmoud, Mohamed K Mostafa and Robert W Peters in Nanomaterials and Nanotechnology
Footnotes
Acknowledgment
The authors would like to thank Al Wadi for Heavy Equipment Company for providing the materials needed to build the system. The authors would also like to extend their thanks to Eng. Mohamed Abdelsamea for helping in setting-up the system. Special thanks to Prof. Mostafa Kamal, the president of Badr University in Cairo (BUC), Prof. Ahmed Moussa, the dean of Faculty of Engineering and Technology at BUC and Associate Professor. Dalia Hamdy, the head of Civil Engineering Department at BUC for supporting this project by providing location to build up the system inside the BUC Campus and allowing the use of chemicals in the BUC’s chemical lab to run the system. The authors would like also to thank the Chemical lab at Housing Building Research Center (HBRC) for nano preparation, collecting the raw wastewater samples and performing the samples analysis. The authors extremely appreciate the continous collaboration between University of Alabama at Birmingham (UAB) represented by Prof. Fouad Fouad (Former Dean of Engineering School), BUC represented by Associate Professor. Dalia Hamdy, and HBRC represented by Prof. Khaled El-Zahabi (President) and Prof. Khalid M. Yousri (Vice-president). The authors also thank the reviewers for their hard work to improve the quality of this paper.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

