Abstract
In this study, nano-carbonated hydroxyapatite (n-CHAp) was successfully synthesized with abalone shells (
Introduction
The main mineral phases in human bones and teeth as identified through X-ray diffraction (XRD) testing have an apatite structure in the form of calcium hydroxyapatite (Ca10(PO4)6(OH)2).
1
The ions forming the calcium hydroxyapatite structure are calcium (Ca2+) and phosphate (PO43-).
2
Nano-hydroxyapatite (n-HA) is an alternative material newly used in orthopedic applications because it can support bone tissue’s ability to regenerate itself.
3
HA has the lattice parameters of
The presence of carbonate ions in biological apatites has been shown to increase metabolic tissue activity. 10 The substitution of carbonate ions in the HAp lattice structure can form three types of CHAp, namely, A-type, B-type, and AB-type. Biomedical applications for biological apatites use the B-type CHAp, which are carbonate ions replacing phosphate ions in the HAp lattice structure. 11 The presence of B-type carbonate ions in the biological apatite structure has been shown to reduce the degree of crystallinity with the implication of increasing solubility. Therefore, it can increase the interaction ability between bioceramics as implant material and implant’s target. 1 Thus, CHAp has been developed as a biomaterial alternative, especially in bone tissue engineering applications. In recent years, it was therefore very important in repairing damaged bones and regenerating bone function. 12
As the bioceramics for bone tissue engineering application, CHAp must have similar characteristics to natural bone and teeth where the particle size must be in nanometer size range. The material with nanometer size range can have significantly different physicochemical properties. 13 Importantly, the physical and chemistry structure of the nanostructure can influence characteristic of cell in contact with the surface. 14 Therefore, to determine whether the sample synthesized was nanometer size, it was necessary to characterize using transmission electron microscopy (TEM). Transmission electron microscopy analysis can be used to determine the morphology and particle size distribution of the n-CHAp.
A variety of methods to synthesize CHAp have been developed, such as hydrothermal and precipitation methods. Precipitation methods have been shown to produce nanometer-sized material with a simple process.15–18 The characteristics of bioceramics material can be analyzed based on its bioactivity ability, depending on several synthesis parameters, such as the deposition or aging time of the solution. Increasing the main diffraction peaks’ intensity along with the extension of solution time indicated that the crystallinity structure of CHAp increased. 19
The n-CHAp synthesis process takes a source of calcium (Ca2+), phosphate (PO43-), and carbonate (CO32-), which can have either natural or chemical ingredients. Several studies show that the production of bioceramics apatite can use natural sources of calcium, such as corals, sea shells, eggshells, 20 crab shells, 21 snail shells, 22 and green mussel shells.7,23 In this study, the n-CHAp was synthesized using abalone shells as the natural ingredient and source of calcium because of the higher content of calcium carbonate (CaCO3) in abalone shells, which was 90–95% 24 and is easily found in Indonesia.3,25
This work explores the potency of abalone shells
Materials and methods
The fabrication is divided into two main stages: preparation of calcium oxide (CaO) from abalone shells waste and synthesis of nano-CHAp. The schematic methods for this study are shown in Figure 1(a). (a) Schematic of methods to fabricate and characterize of n-CHAp and (b) crystallographic analysis of n-CHAp with aging time variations: (a) aging 0 h, (b) aging 24 h, and (c) aging 48 h. n-CHAp: nano-carbonated hydroxyapatite.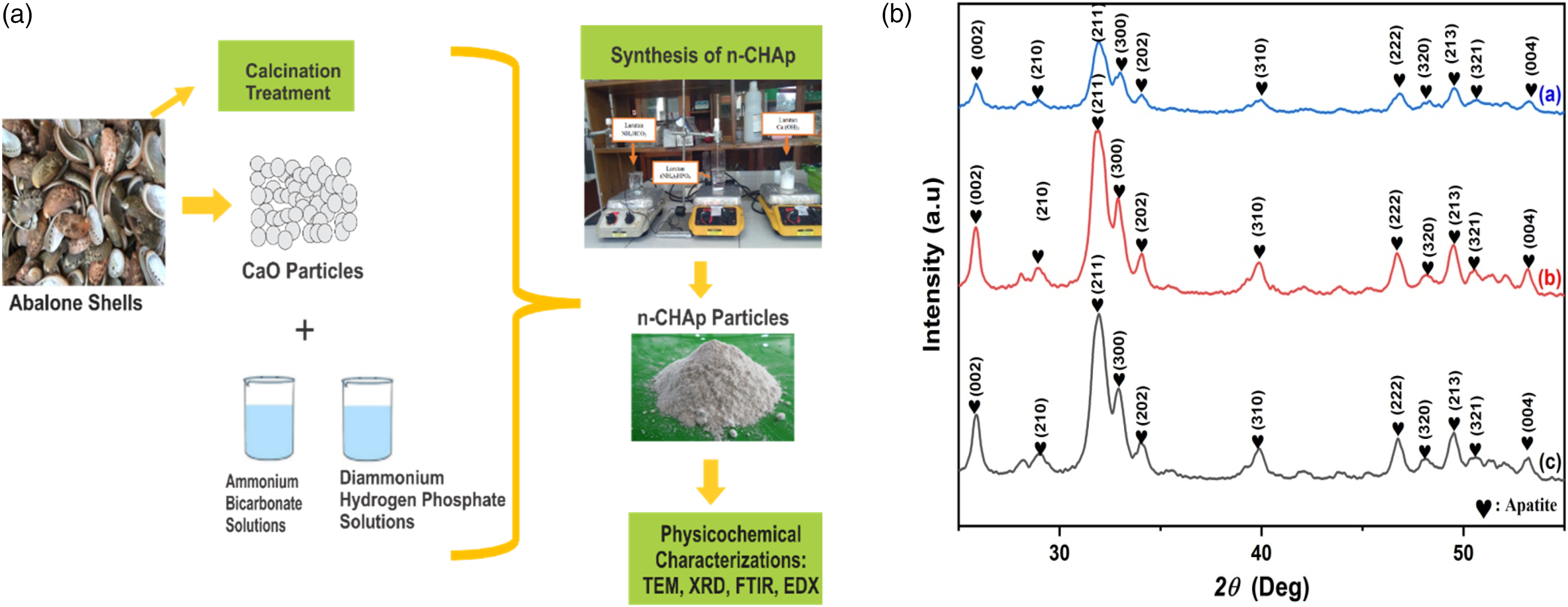
Materials
The abalone mussel shells used as a source of CaCO3 were taken from Bali, Indonesia. The precursors of diammonium hydrogen phosphate ((NH4)2 HPO4), ammonium bicarbonate (NH4HCO3), and ammonium hydroxide (NH4OH) 25% solution were purchased from Merck (USA).
Preparation of CaO
Preparation of CaO was adopted based on the previous research. 26 It began with washing the abalone shells with water and continued with the removal of water content using an oven at 100°C for 4 h, followed by the milling process to obtain abalone CaCO3 powder. Then CaCO3 was decomposed into CaO through the calcination process. The furnace was used at a temperature of 1000°C for 4 h and sieved using the 170 mesh sieve to obtain CaO powder with a smaller grain size.
Synthesis of the n-CHAp
Based on the previous research, 26 this step began with the imposition of carbonate solutions from 5.1192 g of NH4HCO3 and 50 mL of distilled water into phosphate solutions from 8.5536 g (NH4)2HPO4 and 70 mL of distilled water in alkaline conditions (addition of 15 mL NH4OH) to run out until the carbonate solution formed a carbonate–phosphate solution. This was followed by a carbonate–phosphate solution being dropped into the calcium solution made from 6.048 g of CaO and 60 mL of distilled water with a penetrating speed of 1 mL/minute until it ran out while kept under agitation with the hotplate stirrer with a stirring speed of 350 rpm. The synthesized solution was stirred for 1 h with a temperature control of 60°C, and then the solution continued to settle with time variations, namely, 0, 24, and 48 h. The resulting solution was filtered at room temperature for 24 h, washed using a centrifuge for 20 min at 4000 rpm, dried at 90°C for 24 h, and sintered at 650°C for 2 h.
Characterization of the n-CHAp
The n-CHAp sample of the synthesized product was characterized using XRD Panalytical XRD Type Pro to identify the crystalline phase and to analyze the crystallographic properties of the sample, such as lattice parameters, crystallite size, and degree of crystallinity. The formula to determine the lattice parameters of 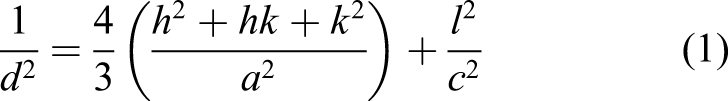

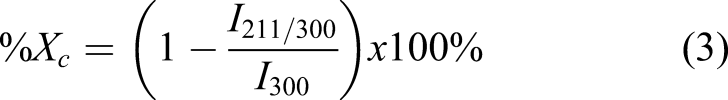
The characterization using TEM Jeol JEM 140 was used to determine the morphology, particle size distribution, polydispersity, and aspect ratio of the n-CHAp with all variations in aging time based on morphology data. Characterization using FTIR Thermo Nicolet iS10 was used to identify the functional groups characteristic of the n-CHAp, namely, PO43-, CO32-, and OH-, and the percentage of mineral components of the samples was characterized using EDX JSM-6510LA.
Results and discussion
Crystallographic analysis
The crystallographic properties of n-CHAp powders were determined by XRD. This analysis was used by matching it with JCPDS No. 09-0432 with a range of 20°–60° and analyzing the crystallographic properties of the n-CHAp through the calculation of lattice parameters, crystallinity degree, and crystallite size. Figure1(b) shows the diffraction pattern resulting from the characterization using the XRD of the n-CHAp samples synthesized with aging time variations.
Based on the result of XRD analysis, the main diffraction peak was formed at the position of the diffraction angle 31.93°, 32.98°, and 34.08° with the diffraction plane (211), (300), and (202), such as the result conducted by previous research
28
JCPDS No. 09-0432 reveals the HAp phase has a main diffraction peak at the position of diffraction angle 31.78°, 32.90°, and 34.09°, so there is a shift in the position of the main diffraction angles from the sample to the value of the greater diffraction angle. The shift in the position of the diffraction angle in the diffraction plane—(211), (300), and (202)—toward the greater diffraction angle value is due to the substitution of carbonate ions in the HAp lattice structure, which causes the cell contraction of the apatite unit on the
Calculation analysis of the nano-carbonated hydroxyapatite crystallographic properties with aging time variations.
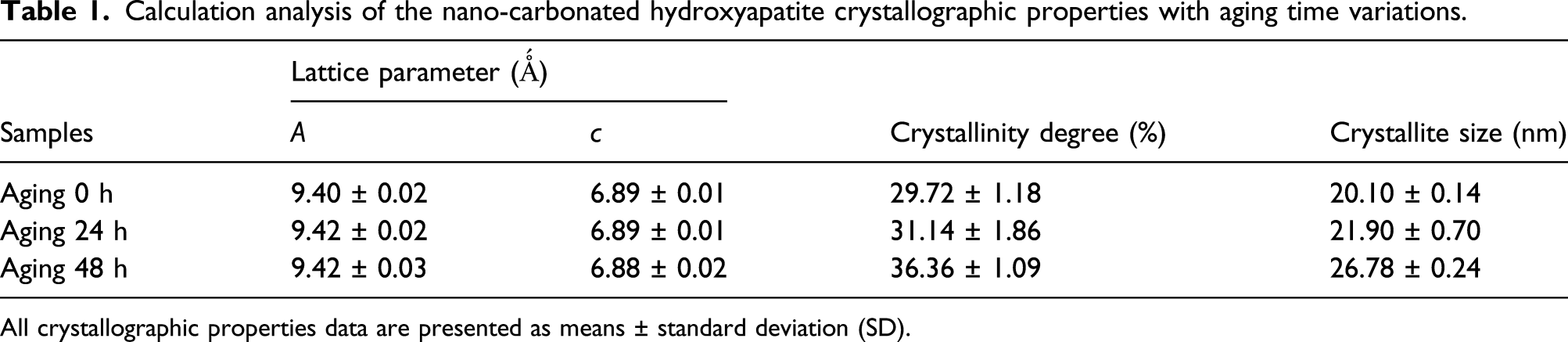
All crystallographic properties data are presented as means ± standard deviation (SD).
Nanostructures and functional group of n-CHAp analysis
The particle size distribution, polydispersity, and aspect ratio of the n-CHAp with all variations in aging time based on morphology data from TEM results were calculated based on the measurements of 30 randomly selected particles using ImageJ software. As shown in Figure 2(a)–(c), it can be seen that the shape of the sample grains was almost the same, namely, an irregular circle.33,34 The whole CHAp looked irregular.
35
Morphology of the n-CHAp with aging time variations: (a) aging 0 h, (b) aging 24 h, and (c) aging 48 h, and (d) FTIR spectra of n-CHAp with aging time variations. n-CHAp: nano-carbonated hydroxyapatite.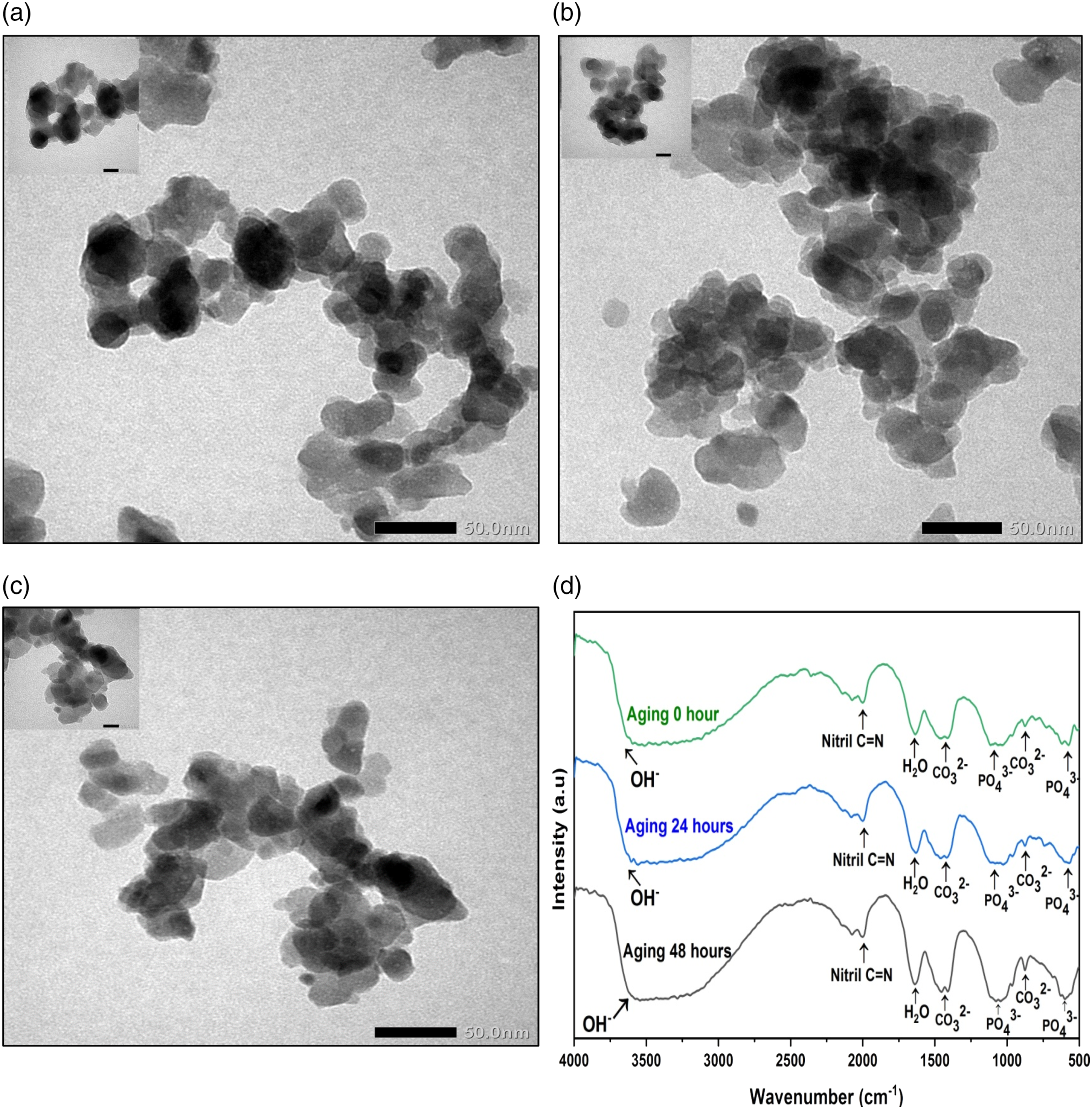
Particle size distribution, polydispersity, and aspect ratio (length/width) analysis of carbonated hydroxyapatite with aging time variations.
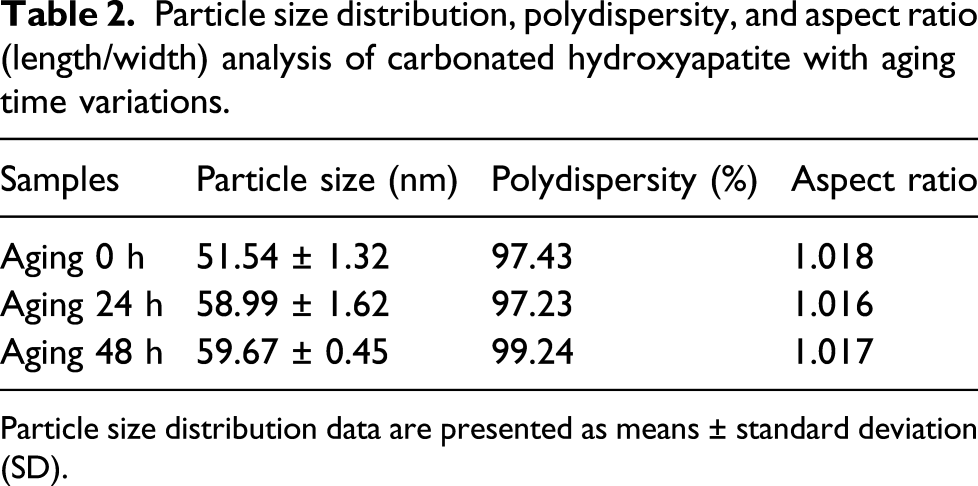
Particle size distribution data are presented as means ± standard deviation (SD).
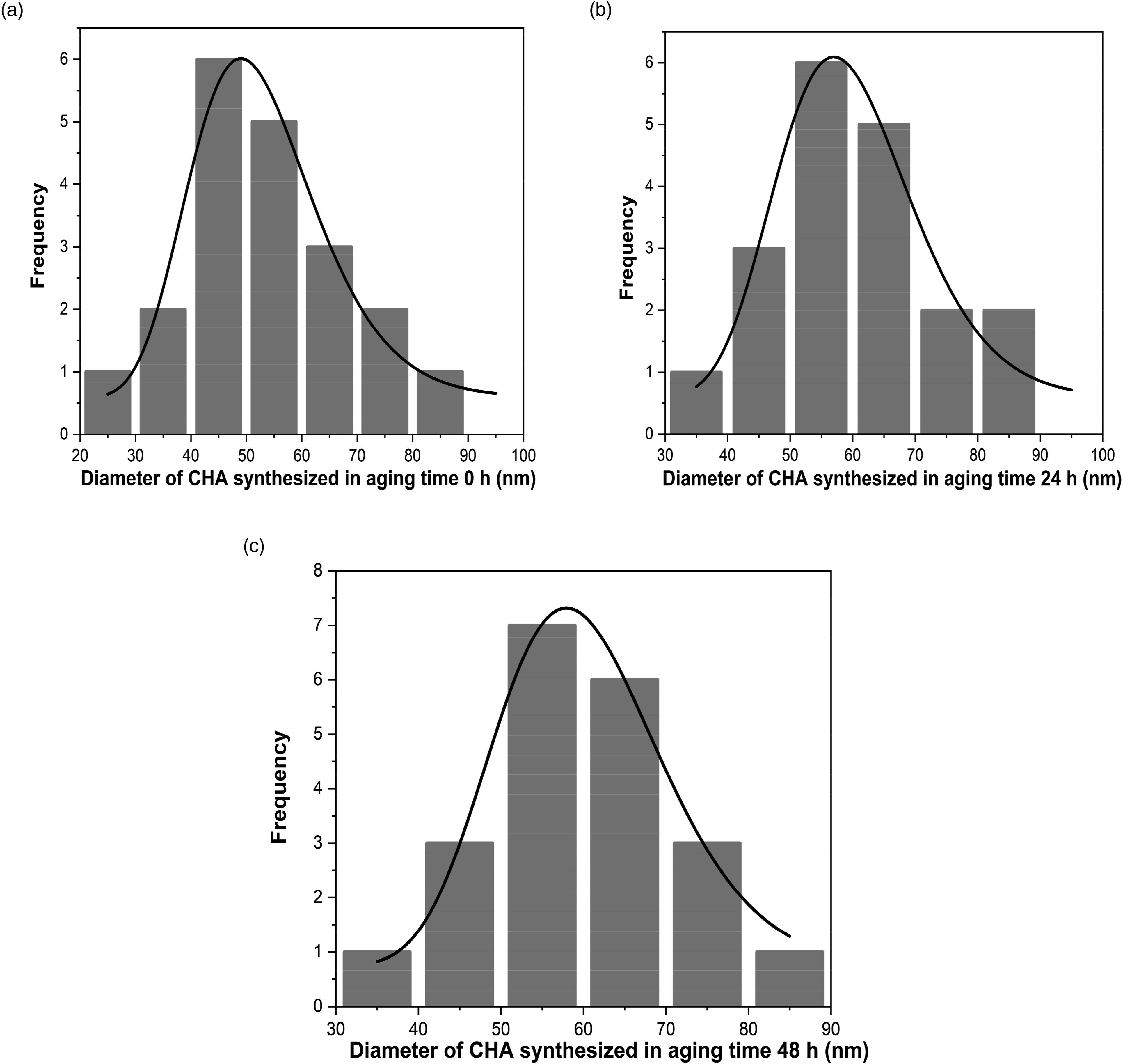
Particle size distribution of the nano-carbonated hydroxyapatite with aging time variations: (a) aging 0 h, (b) aging 24 h, and (c) aging 48 h.
The length and width of 30 particles were measured to obtain an average value, and this was then computed to obtain the ratio of length to width (aspect ratio). As shown in Table 2, the aspect ratio tended to decrease due to the increased aging time in the n-CHAp. It has been suggested that the decrease in the aspect ratio was caused by the higher substitution of carbonate into the phosphate site in the cell lattice (B-type CHAp). 34
FTIR analysis is used to identify characteristic n-CHAp function groups, namely, PO43, CO32-, and OH-. Figure 1(d) shows the absorption band pattern resulting from the characterization using FTIR for the n-CHAp samples synthesized with aging time variations. From these data, it can be seen that all characteristic functional groups of the n-CHAp have been formed for all variations of aging time, such as the result conducted by previous research. 26 In this study, the spectrum of PO43- functional groups showed the increasing sharp as the aging time proceeds. This result caused the presence of increasing phosphate ions with the implication of the increasing crystallinity degree. The hydroxyl and phosphate functional groups can be characteristics of HAp crystallinity, where by both hydroxyl and phosphate spectra get sharper with the variations carried out; it can be said that the samples are increased crystalline. 19 In the spectrum of characterization, the CO32- functional group is also identified, which indicates that the carbonate ion has been substituted into the HAp lattice structure, that supports the EDX data. The CO32- function group is formed at 1438 cm−1 and 878 cm−1, which is the characteristic of B-type carbonates. 29
Element’s composition in the n-CHAp Analysis
Element’s composition in the nano-carbonated hydroxyapatite with variations in aging time.
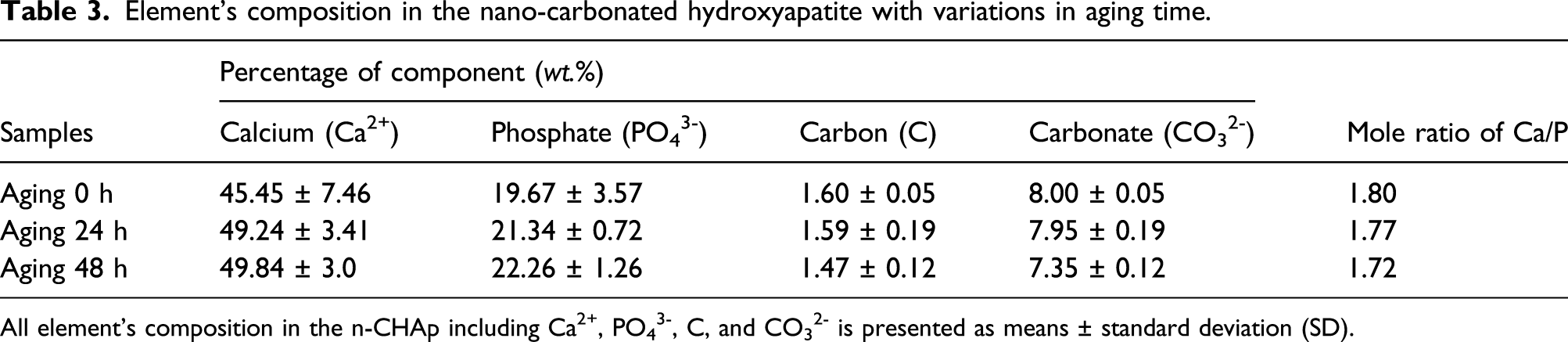
All element’s composition in the n-CHAp including Ca2+, PO43-, C, and CO32- is presented as means ± standard deviation (SD).
Conclusion
The characteristics of the n-CHAp samples are influenced by the varied aging time. Based on the data analysis, the formation of the XRD spectrum shows the characteristic of the n-CHAp, with the shift in the HAp diffraction angle due to the substitution of carbonate ions for all samples with variations in aging time. The calculation of lattice parameter values and identification of functional groups demonstrate the characteristic of B-type CHAp, with the increased crystallite size and crystallinity degree. Seeing whether the FTIR spectrum is formed sharply, the longer the aging time occurs in the phosphate uptake group, the mole ratio of Ca/P decreases with morphological changes due to agglomeration. These data indicate that grain size increases. Based on these characteristics, all B-type CHAp samples based on abalone shells
Footnotes
Acknowledgments
The authors would like to thank Apri I. Supii for his support of the sample procurement. The authors acknowledge the facilities and technical assistance from the Material Physics and Electronics Laboratory and staff of LPPT UGM, Indonesia.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are immensely grateful for the Directorate of Research, Universitas Gadjah Mada through Riset Kolaborasi Indonesia (RKI) Program 2020 (814/UN1/DITLIT/DIT-LIT/PT/2020) for financially supporting this research.
