Abstract
Metal colloids in 2-mercaptoethanol using nanoparticles (NPs) of iron (Fe), cobalt (Co), and nickel (Ni) were prepared by chemical liquid deposition method. Transmission electron microscopy, electron diffraction, UV-VIS spectroscopy, and scanning electron microscopy with electron dispersive X-ray spectroscopy characterized the resulting colloidal dispersions. The NPs exhibited sizes with ranges from 9.8 nm for Fe, 3.7 nm for Co, and 7.2 nm for Ni. The electron diffraction shows the presence of the metals in its elemental state Fe (0), Co (0), and Ni (0) and also some compounds FeO (OH), CoCo2S4, and NiNi2S4.
Introduction
Recently, research on mesoscopic to nanoscopic scale materials has gained great momentum. Advances in interdisciplinary fields between chemistry, physics, biology, and various engineering applications have provided such development. The application of construction techniques of materials atom by atom (molecule by molecule) allows the synthesis of materials of the order of nanometers called “nanomaterials.” This type of investigations has a strategic importance which it concerns to the development of new materials of great functionality and for the integration of inorganic, organic, and biological materials.
By being able to handle and control the shape and size of nanomaterials, it becomes possible to obtain desired characteristics for different applications. Within materials at this scale, there are all kinds of nanoparticles (NPs), some built from their bulk and others by molecule-to-molecule technique. One of the most important characteristics of NPs is their large surface area that gives them exclusive chemical properties. Specifically, metallic NPs can enhance the properties of the raw metal and/or acquire new characteristics, depending on the size, shape, and the media, among others. Heavy metals have unique properties of electricity and paramagnetism, and for this, they are used in electrochemical processes, synthesis, and catalysis. These properties give the metallic NPs (MNPs) a great potential for application in areas such as linear optics, luminescence, electronics, optoelectronics, and catalysis. 1 –4 One of the most common presentations of metallic NPs (MNps) is that of the colloidal system which has been taking more and more interest in materials science. These metal colloids have a wide variety of applications in many fields of science, paint and dye industries, medicine, photography, heterogeneous catalysis, and agricultural chemistry.
Recently, nanoscale zerovalent iron (nZVI) has gained much attention for its use in a wide range of water treatment applications due to its unique properties, including extremely high surface area to volume ratio and an ability to be injected into the subsurface as a colloidal suspension. 5 –8 The high efficacy of nZVI for the removal of Cu2+ has been reported in several empirical studies 9,10 ; however, very little information exists on the physicochemical properties of the Cu NPs such as particles size, particle shape, or crystallinity when removed from solution by iron (Fe) zero NPs.
The cementation reaction between nZVI and aqueous Cu has been studied in a wide range of chemical conditions. More recently, some mechanistic studies were reported to understand the physicochemical composition of nanomaterial copper compounds. 11
On the other hand, although colloidal dispersions can be stable for long periods, sometimes years, they tend to settle slowly by the action of gravity, in prolonged rest. This occurs when suspended particles that are thermodynamically unstable form agglomerates of greater size and weight and finally flocculate. 12 In this way, it is understood that the stabilization of the colloid dispersions can be controlled from its synthesis to obtain the desired characteristics and to manage to take advantage of them and to maintain them in the time. Solvated metal atoms are used as precursors of new organometallic compounds but also as precursors of catalysts dispersed catalysts called SMAD (“Solvated Dispersed Metal Atom”). A characteristic of these new colloidal systems is their tendency to form films, when the solvent is evaporated. 13
In this work, to our knowledge is the first reports using this methodology to obtain several metal colloids prepared using 2-mercaptoethanol by chemical liquid deposition (CLD). 14 –16 These solutions were characterized by several techniques to describe composition, properties, and behavior in order to explain their stability based on the effects of the solution and the metal used. 17
Experiment
Colloid synthesis
The colloids of Fe, cobalt (Co), and nickel (Ni) in 2-mercaptoethanol synthesized by means of the method CLD. This method involves the co-deposition at 77 K (liquid nitrogen) of metallic vapors (Fe, Co, and Ni) with organic vapors of 2-mercaptoethanol in a metal atom reactor. 18
Solvent degasification
Previously, 2-mercaptoethanol was degasified by means of the technique freeze-pump-thaw to evacuate the air. This operation is repeated three times or until no longer there is gas loosening. The operations of degasification to dryness avoid that during the co-deposition reaction the metallic atoms are oxidized by the humidity or present oxygen previously in the solvent.
Co-deposition procedure
A typical condensation was carried out using the equipment specially designed, previously reported. 18,19 An alumina tungsten crucible was charged with around 80 mg of Co metal lumps. Distilled and dried solvent (e.g. 100 mL) were placed in a ligand inlet tube and freeze-pump-thaw degassed for five cycles. The reactor was pumped to 1 × 10−4 torr, while the crucible was warmed to red heat. The temperature in the W-Al2O3 crucible at red heat should be at the boiling point of Co. A liquid nitrogen (77 K) filled Dewar was placed around the vessel, and Co (1.35 × 10−2 moles) and 2-mercaptoethanol (60 mL) were co-deposited over 1 h period at rate of 0.65 mL min−1, approximately. The matrix was black at the end of a co-deposition. The matrix was allowed to warm slowly for 1 h to room temperature under vacuum by removal of the liquid nitrogen Dewar. Upon meltdown, a colorless dispersion was allowed to warm for another 30 min to room temperature. The sol was siphoned into a flask under nitrogen flow. Based on the amount of solvent consumed, the concentration of the colloid could be calculated.
Colloid characterization
UV-VIS spectrophotometry
For the studies of UV-VIS absorption, approximately 0.1 mL of colloidal dispersion was diluted in 3 mL of pure solvent avoiding the excessive agglomeration of colloidal particles and its high absorption. The diluted colloidal dispersions were poured in rectangular quartz cells of 1 cm of optical way and they analyzed in the rank 200–500 nm in spectrophotometer of double beam UV-2450 UV-Visible Shimadzu, Chemistry faculty of Universidad de Concepción. Pure solvent before each sample measured was used to reduce the noise signal.
Transmission electron microscopy conventional, scanning electron microscopy, and electron diffraction
For measurements of transmission electron microscopy (TEM), a drop of the colloidal dispersions just prepared was deposited on one copper grid of 150 mesh covered with coal (supplier Electron Microscopy Sciences, Universidad de Concepción) under a nitrogen atmosphere in a dry camera. The samples were analyzed in an EX-TEM JEOL-JEM 1200 II microscope (JEOL Technics Ltd, Tokyo, Japan) with a voltage of acceleration of 120 kV.
Kinetic stability
Stability for each of the colloidal dispersions studied through physical and chemical behavior of the same sample on time, at room temperature, and under inert atmosphere.
Electrophoresis
The electrophoresis measurements were performed ina a Zeta Meter 3.0 Model and 0.1 mL of colloid in 2-mercaptoethanol is placed in a cell, which applies a potential difference and the rate of migration of the particles is measured.
Results and discussion
The colloids of Fe, Co, and Ni were obtained by simultaneous co-deposition of these metals with the solvent 2-mercaptoethanol as shown in Figure 1. The excess solvent was added in order to achieve stabilization of the agglomerates or clusters of metal solvated.
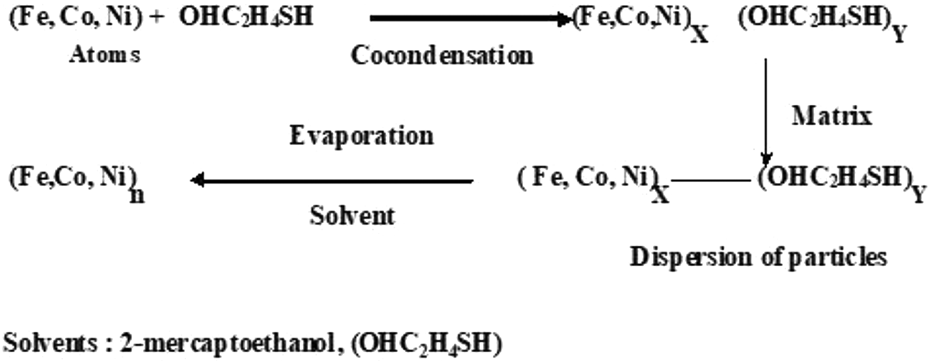
Reaction scheme.
With this technique were prepared monometallic colloids of Fe, Co, and Ni molar concentrations described in Table 1.
Stability of metal colloids in 2-mercaptoethanol.

Fe: iron; Co: cobalt; Ni: nickel.
Stability
For colloidal dispersions, the kinetic stability was measured in terms of its power of flocculation at room temperature. The colloid stability measured when 50% of the particles remains in suspension, this is measured by UV-VIS absorptions of the colloidal suspensions. 19 They have been studied the behavior of quantum dots of CdSe and relative colloidal stability in solution. They calculate the average NPs size theoretically, but in this system we obtained them by TEM. 20 Table 1 summarizes the stabilities of metal ions in the solvent 2-mercaptoethanol to each of the different concentrations prepared.
From the results, we can observe that for colloidal dispersions obtained from Co and Ni are quite stable in 2-mercaptoethanol, indicating that there is a strong solvation. In contrast, Fe is stable but no to the same extent as other metals due to the higher oxidation potential of Fe over Co and Ni. 19,21
The surface energy of a nano metal is different if their sizes are different. 21,22 This is due to quantum size effect, which is relevant in the nanotechnology materials. 23,24 The colloidal dispersions are systems that tend to produce clusters of low energy state. Henglein 25,26 defined the concept of electronic instability to make clear the clustering and coalescence of small particles. But if the particles are quite stable, it should be certain electronic instability when the energy Fermi level is arranged different in the particles.
The colloids of Fe-2-mercaptoethanol are less stable than reported in previous works using other organic solvents such as ethanol, 2-propanol, and 2-methoxyethanol probably due to a size effect. 27 Co colloids are considerably more stable at low concentrations (5.72 × 10−4 mol L−1) than what is reported, 24 which does not exceed 4 days, however here, are stable over 60 days. Much clearer is the case with Ni that compared to previous work 21 is significantly higher than the period of stability, whereas solvents have similar characteristics and used the same method of production. This is due to the difference in redox potentials of these metals.
However, in the case of Fe and Co, the stability of colloidal dispersions is dependent on concentration, where lower concentrations for colloidal dispersions are more stable than for those with greater concentration. In fact, it is attributed to stabilize less concentrated colloidal dispersions with lower energy needed for particles to remain dispersed in the reaction and improved the solvation power of the NPs.
The Ni suspension exhibit another behavior, indepent of the concentration of colloidal dispersions. Their stability over 60 days due to the strong interaction of the metal is observed with OH and SH bonds of the solvent.
Study of particle size
The particle sizes were determined by analysis of micrographs obtained in the transmission electron microscope JEOL JEM 1200 EXII with 4 Å resolution as shown in Figure 2, and these micrographs show the presence of agglomerated particles dispersed on a grid.

(a) Frequency histogram and dark-field micrograph of Fe-2-mercaptoethanol 1.7 × 10−3 (mol L−1), (b) frequency histogram and bright field micrograph Co-2-mercapoethanol 5.72 × 10−4 (mol L−1), and (c) frequency histogram and bright field micrograph for Ni-2-mercaptoethanol 7.45 × 10−4 (mol L−1). Fe: iron; Co: cobalt; Ni: nickel.
Fe particles have a considerably larger particle size than values reported in similar solvents, and Co particles have a size smaller than 4 nm that is lower than that reported by other systems before. 28
Table 2 summarizes the particle size of the metallic colloids in the solvent.
These colloids show similar stability to Au-2-propanol being also spherical and negatively charged particles. 29 The narrow particle size distribution becomes wider the time aging of the colloid. The values are summarized in Table 2. Also, Bi-2-propanol of Pd, Au, and Ag previously reported colloids just prepared exhibit a particle size of 5.1 nm 30 very similar to (PdSn)-2-propanol. This is probably due to the similarities in the dielectric constant of the solvent.
Particle size of colloidal dispersions of 2-mercaptoethanol.

Fe: iron; Co: cobalt; Ni: nickel.
* It was not possible to determine the particle size for Fe-2-mercaptoethanol at a concentration of 2.2 × 10−2 M.
According to TEM analysis, the clustering of particles is due to the association of several particles. Similar stability and behavior have been observed in other colloidal dispersions. 31 –35
Electron diffraction
This study was performed by electron diffraction (ED) for the systems Fe, Co, and Ni-2-mercaptoethanol in order to obtain diffraction patterns that have concentric rings or diffraction spots or in some cases both as shown in Figure 3.
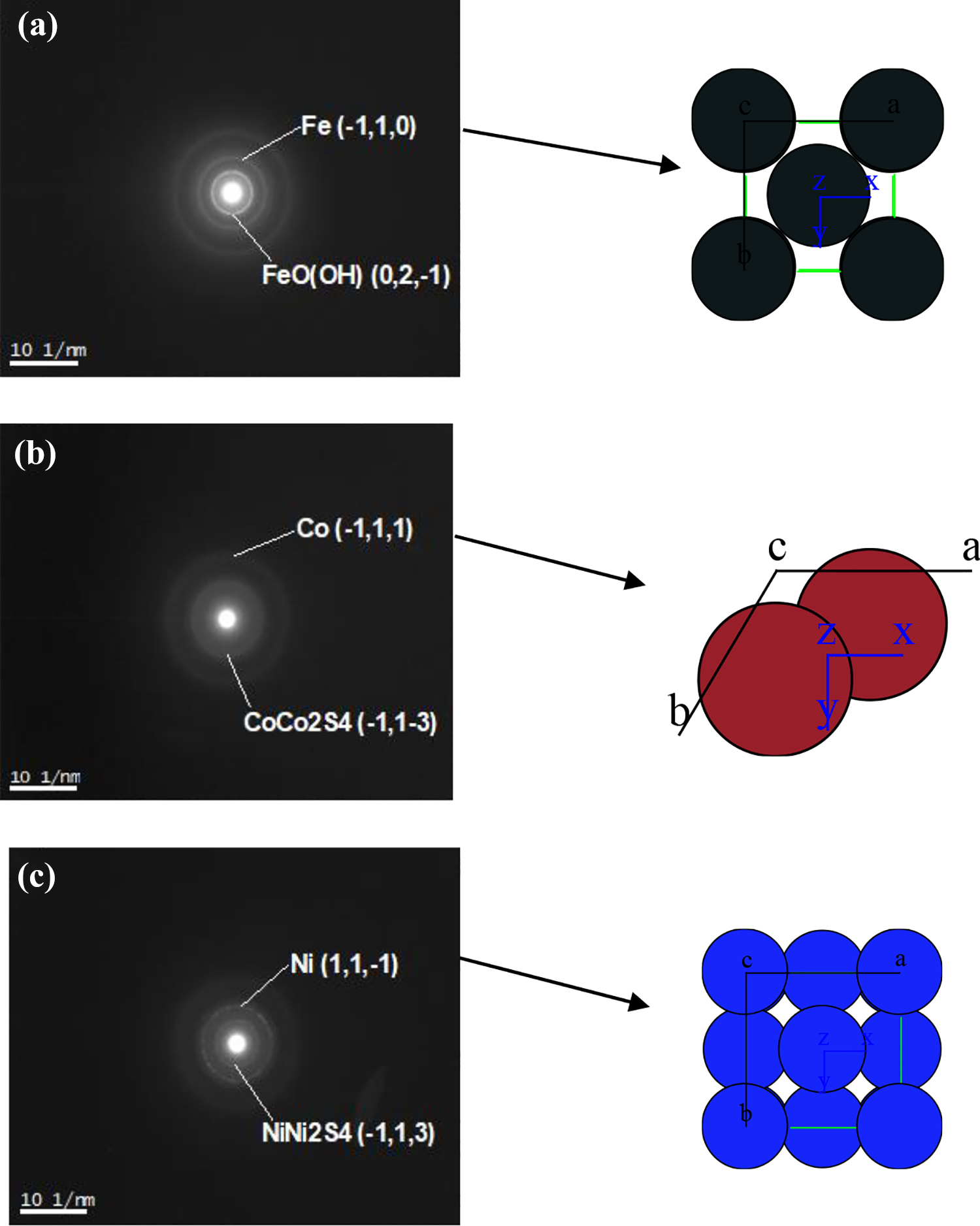
Electron diffraction for (a) Fe-2-mercaptoethanol, (b) Co-2-mercaptoethanol, and (c) Ni-2-mercaptoethanol. Fe: iron; Co: cobalt; Ni: nickel.
Table 3 presents a summary of the crystalline areas and their possible assignments of crystallographic planes for the most representative colloidal dispersions.
Crystal space and possible assignments for colloidal dispersions of Fe, Co, and Ni in 2-mercaptoethanol.

Fe: iron; Co: cobalt; Ni: nickel.
According to the results obtained from the diffraction measurements, the presence of metallic particles of each of the metals in its elemental state Fe (0), Co (0), and Ni (0) is observed. There is also the presence of Fe as FeO(OH), Co as CoCo2S4, and Ni as NiNi2S4. It is noteworthy that the particles thus formed are mainly amorphous in diffraction rings indicating that they have completely random oriented. Otherwise, it would be present as well-defined points.
One interesting point is the intensity of that form of the rings. If they are of very low intensity, it indicates that they are very small crystals that are in colloidal dispersions, which coincides with reports in the frequency histograms.
Scanning electron microscopy and electron dispersive X-ray spectroscopy
Scanning electron microscopy (SEM) revealed no differences in the morphology of the films obtained from the colloids synthesized Fe, Co, and Ni in 2-mercaptoethanol as shown in Figure 4.

SEM micrographs for (a) Fe-2-mercaptoethanol, (b) Co-2-mercaptoethanol, and (c) Ni-2-mercaptoethanol. SEM: scanning electron microscopy; Fe: iron; Co: cobalt; Ni: nickel.
SEM micrographs obtained for Ni show a scattered surface irregularity. In addition, electron dispersive X-ray spectroscopy (EDS) confirmed the presence of the metal surface with a rate of 65% in the films, as shown in Figure 5. This high value of metal embedded in the colloid can be explained by the higher affinity to form bonds through metal solvent –OH group of solvent molecules.

EDS spectrums for (a) Fe-2-mercaptoethanol, (b) Co-2-mercaptoethanol, and (c) Ni-2-mercaptoethanol. EDS: electron dispersive X-ray spectroscopy; Fe: iron; Co: cobalt; Ni: nickel.
In the case of Fe a rough surface atoms was observed, thet follow a certain order, these groups could correspond to growth of the agglomerates of colloidal particles. From EDS, it was confirmed the presence of metal with a low percentage (22.6%). Unlike the case of Co, the micrograph shows a uniform surface roughness, and the EDS analysis gave a rate of 47% metal surface.
Zeta potential
Electrophoretic measurements were carried out for the concentration of colloidal dispersion using a computer more stable Zeta Meter 3.0. Table 4 provides a summary of the values obtained experimentally from electrophoretic mobility, charge, and the estimated value of Zeta potential for more stable concentration of colloidal dispersions.
Electrophoretic mobility and zeta potential of colloidal dispersions.

Fe: iron; Co: cobalt; Ni: nickel.
* It was not possible to determine the zeta potential of suspensions of nickel as they present an anomalous behavior, independent of the concentration of the suspension.
The colloidal systems of Fe and Co particles in 2-mercaptoethanol have a positive nature, which is consistent with previous studies. 23,24 The difference is that the particles in 2-mercaptoethanol have the potential for a significantly lower charge than the one previously reported, and this could be due to the dielectric constant of the solvent which is less than other reported cases.
UV-VIS spectroscopy
Spectroscopic measurement is performed for colloidal dispersions of Fe, Co, and Ni in 2-mercaptoethanol in a range that covers from 200 nm to 500 nm as shown in Figure 6. The results of the experimental values are summarized in Table 5 of UV-VIS bands absorption for each colloidal dispersions. The values are getting close to the theoretical values reported by Creighton and Eadon. 32 Specifically, in the case of Fe away from its theoretical value due to its larger particle size. Table 6 summarizes the bands over 300 nm, and most of them are coincident.

UV-VIS spectrum for colloids (a) Fe-2-mercaptoethanol 1.7 × 10−3 M, (b) Co-2-mercaptoethanol, and (c) Ni-2-mercaptothanol. Fe: iron; Co: cobalt; Ni: nickel.
Experimental wavelengths for colloidal dispersions in 2-mercaptoethanol.

Fe: iron; Co: cobalt; Ni: nickel.
UV-VIS analysis results for Fe, Co, and Ni colloids.a

Fe: iron; Co: cobalt; Ni: nickel.
a Fe and Ni colloids are similar in size and they exhibit a couple of absorption bands in a similar range. But these values are not conclusive.
The Fe colloid possesses a band at a value very close to those reported, 19 and this would be because of the similarity of the solvent used for Co. A similar behavior occurs for colloids instead away from the values reported. 24 However, experimentally obtained values are close to those reported by Creighton et al. 22 The difference may be is due for the assumption of spherical shapes of the particles with and average size of 10 nm in aqueous solvent.
Conclusions
The synthesized colloids are stable, mainly Co and Ni, greater than 60 days, and the colloids of Fe are less stable (10–17 days). The values are reproducible since three samples were tested for each set of concentrations.
The TEM micrographs and frequency histograms reveal a particle size distribution for each metal, Co = 3 nm, Ni = 7 nm and Fe = 10 nm.
In EDs, the presence of the metal in the elementary state is confirmed for each of them Fe0 (−1,0,1), Co0 (−1,1,1), and Ni (1,1, −1), in addition to other species present FeO(OH) (0,2, −1), CoCo2S4 (−1,1,3), and NiNi2S4 (−1,1,3).
According to the SEM analysis, surfaces for irregular and dispersed Fe and Ni are shown, and for Co, a uniform distribution is observed. EDS identified metals on each of the surfaces analyzed, with a percentage of 22.59%, 47.0%, and 65.0% for Fe, Co, and Ni, respectively.
The colloids of Fe and Co have electric charge of character (+) with a ζ of 14.027 mV and 9.436 mV for Fe and Co, respectively; in Ni’s case, it was not possible to measure the zeta potential (ζ).
Colloids have continuous absorption into the visible spectrum and are increased in the ultraviolet with values for Ni, Co, and Fe of 216.20 nm, 219.20 nm, and 329.40 nm, respectively, which is coincident with the theoretical values except for Fe with a larger wavelength due to its larger particle size.
The active solids are thermally stable and have a decomposition at 100°C that could correspond to the presence of moisture in the samples. The solids in question have decomposition temperatures higher than 200°C, specifically Fe 222.45°C, Co 244.91°C, and Ni 288.29°C, respectively.
The results obtained by atomic absorption were different from one metal to another, Fe 22.7% Co 39.3%, and Ni 58.2%, but always similar content in each preparation.
Fourier transform infrared spectrograms reveal the interaction of metals with the solvent under study, and this is mainly carried out through the displacement to a longer wavelength of the link –OH, in addition to being of relevance is the disappearance of the link –SH which indicates that the interaction would be carried out in this place forming the link –S Metal.
Interaction with organic solvent are also observed in the results obtained from atomic absorption, where the results of the metals percentage are higher for Ni (64%) the Co (58%) and finally Fe with the lowest percentage (22%). Another indication is the particle size because being bound of the solvent the particles avoid agglomeration by repulsion of load between them so they do not agglomerate, obtaining particle sizes much smaller for Ni and Co than for Fe. Besides, the interaction with the solvent is fundamental corresponds to the stability of colloidal suspensions when the particles are more agglomerated. The colloidal suspension is less stable over time causing it to float, in this way the Ni suspensions (<60days) being the least stable Fe suspension (10-17 days).
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Fondecyt Grant # 1080704.
