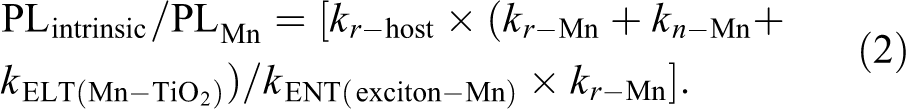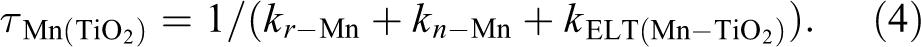Abstract
In this work, we improved the photocurrent of self-powered ultraviolet photodetector via doping manganese in CsPbCl3 perovskite nanocrystals light harvester. The doped manganese in nanocrystals has the following three features to assist electron transfer from CsPbCl3 nanocrystals to titanium dioxide: (i) the fast exciton-to-manganese energy transfer process benefits for competing electrons with perovskite exciton recombination, (ii) the charge carrier lifetime is very long for manganese d-states due to its spin and orbital forbidden transition, and (iii) the electrons can effectively transfer to the titanium dioxide layer from 4T1 of manganese d-states due to the smaller energy barrier. Based on the above, the self-powered photocurrent density of photodetectors has nearly twice enhancement from 0.08 mA·cm−2 to 0.14 mA·cm−2 and a high responsivity up to 7.3 mA·W−1 was achieved at 340 nm.
Introduction
Although the ozone layer of the atmosphere can block the majority of ultraviolet (UV)-C (100 ≤ λ < 280 nm) radiation according to the International Commission on Illumination, UV-A and B (280 ≤ λ < 400 nm) radiation can still reach the earth’s surface and be the culprit for many diseases. 1,2 For monitoring the UV index, it is important to develop UV photodetectors (PDs), especially for the self-powered UV PDs from the view of being energy saving, portable, and miniature. 3 –9 In the past few years, many self-powered UV PDs have been explored from materials to devices to extract the charges efficiently. 10 –19 However, the photocurrent responsivity of these PDs is still lower than the visible light PDs. The reasons are the great energy barrier in heterostructure interfaces and defect-induced fast electron trapping, which both are detrimental to electron extraction.
All inorganic lead (Pb) halide perovskite nanocrystals (NCs) (NCs indicate the crystals with at least one dimension in the scope of 1–100 nm) are in the research spotlight due to their high defect tolerance, solution-based processing, and low fabrication cost. 20 –26 In recent years, halide perovskite NCs have been widely used for solar cells (SCs), LED, lasers, PDs, and so on. 22 –25 For example, a remarkable power conversion efficiency of 24.2% has been achieved for halide perovskite SCs. 22 In LED field, an external quantum efficiency exceeding 20% have been achieved. 23 Besides pure halide perovskite NCs, impurities-doped halide perovskite NCs have also attracted numerous attentions. Various dopants, such as Mn2+, Cd2+, Zn2+, and Sr2+ ions, have been used for improving the stability, the emission quantum yield, the emission wavelength, and the toxicity of perovskites. 27 –30 In 2016, Klimov group reported the manganese (Mn)-doped CsPbCl3 (Mn: CsPbCl3) perovskite NCs, which have long photoluminescence (PL) lifetime originating from the spin and orbital forbidden transition of Mn d-states (4T1–6A1). 31 –33 It is believed that the long-lived charge carriers by Mn doping are advantageous to improve the efficiency of SCs. 34 –40 The fast exciton-to-Mn energy transfer (ENT) can suppress the electrons trapped by defects and benefit for collection of electrons by Mn dopant beforehand. 41,42 . The 4T1 energy level of Mn is suitable for ENT to conduction band (CB) of titanium dioxide (TiO2) due to their smaller energy barrier.
Since PDs have similar working principle (departure of electrons and holes from light harvester layer) to that of SCs, we expect that the doping of Mn inside CsPbCl3 can be also helpful to improve the photocurrent responsivity of self-powered UV PD. Inspired by this idea, we fabricated the carbon-based fully printable self-powered perovskite UV PDs with improved performance by doping Mn in CsPbCl3 NCs light harvester. As-prepared PDs have three following main advantages: (i) the Mn: CsPbCl3 NCs work as UV co-absorbers to offset the low absorption coefficient of TiO2 in TiO2/zirconium dioxide (ZrO2)/carbon triple-layer mesoscopic scaffold (TMS); (ii) the electrons can be efficiently extracted from CsPbCl3 NCs to TiO2 via Mn-assisted electron transfer (ELT). The photocurrent of self-powered UV PDs was improved from 0.08 mA·cm−2 to 0.14 mA·cm−2 and a high responsivity up to 7.3 mA·W−1 was obtained at 340 nm; (iii) the PDs in this work are made by silk screen printing technology, which is not only convenient but also compatible with low-cost carbon counter electrodes.
Experimental
Materials
(II) chloride (PbCl2; 99.9%, Aladdin,Shanghai, China), cesium chloride (CsCl; Aladdin, ≥99%), n-octylamine (≥99%, Aladdin), oleic acid (OA; 90%, Alfa Aesar, Tianjing, China), manganese(II) chloride (MnCl2) tetrahydrate (99%, Sinopharm Chemical Reagent Co., Ltd, Shanghai, China), N,N-dimethylformamide (DMF; 99.5%, Kelong, Chengdu, China), TiO2 (Degusssa p25, 20–50 nm), ZrO2 (<100 nm, Aladdin), terpineol (99%, Macklin, Shanghai, China), ethyl cellulose (EC) (99%, Aladdin), ethanol (99.7%, Kelong), n-hexane (97%, Kelong), acetone (99.5%, Kelong), diethyl ether (99%, Kelong), toluene (99%, Sinopharm Chemical Reagent Co., Ltd). TiO2 (or ZrO2) paste: 8 g TiO2 (or ZrO2) and 4 g EC were put into 30 mL terpineol and 15 mL ethanol, followed by stirring using ball milling for 2 h. Carbon paste: 2 g carbon black powder was mixed with 6 g graphite powder in a 30-mL terpineol solution and then 1 g ZrO2 powder and 1 g EC were added into the solution, followed by stirring using ball milling for 2 h.
The fabrication of carbon-based fully printable TMS
The fluorine-doped tin oxide (FTO) glass substrates (NSG, Shanghai, China) were etched by laser and cleaned by ultra-sonication with detergent, Deionized (DI) water, acetone, and ethanol. The TiO2, ZrO2, and carbon films were screen printed on the substrates layer by layer. The TiO2 and ZrO2 layers were sintered at 500°C for 40 min, and the carbon layer was sintered at 350°C for 1 h, forming the mesoporous triple layer-based scaffold. Herein, ZrO2 acts as an insulating space layer between TiO2 layer and carbon layer to prevent the short circuit.
The synthesis of Mn: CsPbCl3 NCs
The “aqueous phase” cesium (Cs)-precursor was prepared via dissolving 1 mmol CsCl in 1 mL DMF. The Pb-precursor was prepared via dissolving 1 mmol PbCl2 in 1 mL DMF. The “oil phase” was prepared by mixing 8 mL OA and 0.8 mL n-octylamine in 30 mL n-hexane. Then the “aqueous phase” Pb-precursor solution was added dropwise into the “oil phase” solution. After 5 min, the “aqueous phase” Cs-precursor solution was added into the above solution. After mixing, emulsion was formed and the color of solution turned from clear to slight white. Demulsion and purification processes were proceeded by adding 20 mL acetone. Then the mixture was centrifuged at 6500 rpm for 15 min to obtain precipitates. For Mn: CsPbCl3 NCs, the different MnCl2 amount was added to the Pb-precursor solution before adding to the “oil phase.”
The Mn: CsPbCl3 NCs sensitized TMS
Ten microliters of 5 wt% Mn: CsPbCl3 NCs in toluene was dipping on the carbon electrode. To ensure the NCs penetrate into the bottom of the TMS, 3 µL DMF and 10 µL toluene were dropped circularly via the dissolution and recrystallization process. Due to the porous structure of TMS, a continuous phase of Mn: CsPbCl3 is filled between carbon and FTO/TiO2 electrodes.
Characterization
UV-visible (UV-vis) absorption spectra were recorded with a Shimadzu 3600 UV-vis near-infrared spectrophotometer (Shimadzu, Japan). PL spectra were recorded with a Shimadzu RF-5301 PC spectrofluorometer excited at 350 nm. The time-resolved PL spectra were obtained by a streak camera (Hamamatsu C5680, Hamamatsu). The ultrafast pump pulses were from a femtosecond laser (Legend-F-1k, Coherent, 1 kHz, 100 fs, American). Excitation pulses at 400 nm were obtained by doubling the fundamental wavelength in a β-barium borate crystal. X-ray powder diffraction investigations were carried out using the D/max-2500/PC diffractometer (Rigaku) with copper Kα radiation (λ = 1.541 Å). X-ray photoelectron spectroscopy (XPS) was investigated using a PHI550 (Perkin–Elmer) spectrometer with magnesium Kα excitation (1253.6 eV). The inductively coupled plasma optical emission spectrometer (ICP-OES) was measured via Varian 720-ES spectrometer (Agilent). The scanning electron microscope (SEM) and mapping tests were performed on a field-emission SEM (FEI Inspect F50). For current–voltage (J-V) measurement, a solar simulator (model 66902) from Newport, which has a 300-W xenon lamp with an irradiance of 100 mW·cm−2 at the surface of the SC, was used. For comparing the self-power current, the current density is directly plotted with voltage. For evaluating the performance of PD, the logarithm of current density is used to plot with voltage. Moreover, J-V measurements were also used for evaluating the reproductivity and experimental errors of the as-prepared PDs. The incident photon-to-current conversion efficiency (IPCE) was measured with a 300-W xenon lamp and a lock-in amplifier (Oriel) in the wavelength range of 300–800 nm. The photodetection was tested via home-built system integrated into a monochromator and Keithley 2400 SourceMeter (Keithley).
Results and discussion
Figure 1(a) and (b) is the device structure and fabricating processes of the self-powered Mn: CsPbCl3-based UV PDs. The TiO2, ZrO2, and carbon films in TMS were screen printed onto an FTO glass substrate layer by layer (Figure S1). The Mn: CsPbCl3 NCs are filled into the TMS directly by dip-coating method. From the cross-sectional images in Figure 1(c), the mesoporous TiO2, ZrO2, and carbon layers are found to have a thickness of 0.53, 1.54, and 5.26 μm. The Mn: CsPbCl3 NCs have an average size of approximately 10 nm with a tetragonal phase structure (Figure 1(d) and Figure S2). 43 NC compositions of Mn 2p, Pb 4f, Cs 3d, and chlorine (Cl) 2p were measured via XPS and the corresponding chemical states were shown to be Mn2+, Pb2+, Cs+, and Cl− (Figures S3 and S4). To overcome the poor penetrability of the ligand-capped NCs and facilitate their diffusion to the bottom of TMS, the recrystallization property of perovskite NCs was utilized via circularly dropping DMF and toluene. 44,45 The DMF firstly take away the ligand from NCs surface and then lead to a slight and instantaneous dissolution. This will prompt the diffusion of NCs. Then, toluene was quickly dropped on TMS to make the solution supersaturated and recrystallize to new NCs.

The schematic illustration of Mn: CsPbCl3-sensitized carbon-based fully printable TMS (a) and the crystal structure of Mn: CsPbCl3 NCs (b). (c) SEM image of TMS used for self-powered UV PDs; (d) transmission electron microscope (TEM) for used Mn: CsPbCl3 NCs. The scale bars are 5 µm and 20 nm for SEM and TEM, respectively. Mn: CsPbCl3: manganese (Mn)-doped CsPbCl3; NC: nanocrystal; TMS: triple-layer mesoscopic scaffold; UV: ultraviolet; PD: photodetector.
Figure 2(a) is the current density–voltage (J-V) curves of the UV PDs under AM1.5 (100 mW·cm−2) illumination. The performance of self-powered UV PDs is obviously dependent on Mn concentration (measured by ICP-OES in Table S1). (i) When increasing the Mn/Pb molar ratios from 0% to 3.5%, the self-powered current (JSC) at voltage of 0, which is a main index for evaluating whether a PD can work by self-power, enhances from 0.08 mA·cm−2 to 0.143 mA·cm−2. It indicates more electrons have been extracted from CsPbCl3 NCs to TiO2 after Mn doping. From the PL spectra in Figure 2(b), besides CsPbCl3 intrinsic emission around 411 nm, the doping of Mn brings a new emission band around 573 nm, 31 –33 which attributes to d-d recombination transition at Mn 4T1 energy level after exciton-to-Mn ENT as shown in Figure 3. By increasing Mn doping concentration in NCs, more excitons will transfer to Mn d-states as suggested by increased Mn emission at 573 nm and quenched intrinsic emission of perovskite at 411 nm. Theoretically, the transfer of excitons to Mn should lead to decreased excitons in the host CsPbCl3 CB and hence leading to a decreased Jsc directly from CsPbCl3 CB to TiO2 (process 1 in Figure 3). However, the measured Jsc has a reverse change compared with PDs using undoped NCs. A reasonable explanation is that the electrons transferred to Mn 4T1 energy level will subsequently transfer to TiO2 (process 2 in Figure 3). Moreover, this new ELT process 2 is more efficient than the previous one directly from CsPbCl3 CB to TiO2 (process 1 in Figure 3). Only in this case, the measured Jsc will increase after Mn doping. (ii) When enhancing the Mn amount, the open circuit voltage (Voc) has a slight decrease from 0.78 V to 0.64 V. The Voc is calculated by the potential difference between the Fermi energy levels. 46 From the absorption spectra in Figure S5, the absorption peak position doesn’t have an obvious change. The decreased Voc is not attributed to the band gap change but attributed to the doped Mn. The d-states of Mn can work as electron storage center due to the fast exciton-to-Mn ENT and long-time charge carriers (up to 0.5 ms). This leads to the decrease in Fermi energy level. The photo-physical processes in Mn: CsPbCl3/TiO2 heterojunction interface were proposed in Figure 3. The photo-generated excitons can take place in the following pathways: (1) band edge carrier transfer, (2) Mn-assisted ELT, (3) Mn d-states transition, (4) intrinsic band edge emission, and (5) defect-induced electron trapping.

(a) J-V curves and (b) PL spectra for the Mn: CsPbCl3 NCs sensitized TMS with different Mn/Pb molar ratios. The PL (c) and TRPL decay spectra (d) for Mn: CsPbCl3 NCs films without/with TiO2 film. (e) and (f) The corresponding transient PL recorded by streak camera. Mn: CsPbCl3: manganese (Mn)-doped CsPbCl3; NC: nanocrystal; TMS: triple-layer mesoscopic scaffold; TRPL: time-resolved PL; PL: photoluminescence; J-V: current–voltage; Mn/Pb: manganese/lead; TiO2: titanium dioxide.

The photo-physical processes of Mn: CsPbCl3/TiO2 heterojunctions. The k is the rate constant of the processes (1–5). Due to continuous phase Mn: CsPbCl3 being filled into porous ZrO2 insulating layer, electrons are transferred by perovskite rather than ZrO2. Thus, the energy band of ZrO2 is not shown.
To demonstrate the mechanism further, the PL properties of Mn: CsPbCl3 NCs on TiO2 film were explored (Figure 2(c) to (f)). For the Mn: CsPbCl3 NCs, the excitons have three main recombination routes, namely, processes 3, 4, and 5 in Figure 3. The ratio of PLintrinsic and PLMn can be written as equation (1). The detailed calculation is shown in supporting information.
After depositing Mn: CsPbCl3 NCs on the TiO2 film, the electrons on CsPbCl3 CB would have extra transferring processes 1 and 2 in Figure 3 due to the lower CB of TiO2. Therefore, the intensity of both CsPbCl3 intrinsic emission and Mn emission would reduce in Figure 2(c). Remarkably, the Mn emission at 577 nm has 81% attenuation, which is greater than the intrinsic emission at 415 nm (38%). The reason for much higher (lower) attenuation of Mn (intrinsic) emission can attribute the extremely long (short) PL lifetime at Mn (perovskite CB), making ENT more (less) competitive than recombination. In other words, the increased ratio of PLintrinsic/PLMn after deposition of perovskite NCs on TiO2 film confirms the existence of more efficient Mn-to-TiO2 ELT (process 2) than perovskite CB-to-TiO2 ELT (process 1). If so, based on the equations (3) and (4), the extra transfer route for Mn (KELT(Mn-TiO2)) will lead to a faster fading of PL lifetime.
From the PL lifetime of Mn, the deposition of Mn: CsPbCl3 NCs on the TiO2 film exactly leads to the decreased PL lifetime from 0.668 ms to 0.553 ms in Figure 2(d) and Figure S6, confirming the presence of Mn-to-TiO2 ELT (process 2). The corresponding transient PL spectra measured by a streak camera under the same conditions are shown in Figure 2(e) and (f). The results also show a rapid quenched emission of Mn around 580 nm after the deposition of Mn: CsPbCl3 NCs on the TiO2 film, confirming the ELT from Mn to TiO2.
The UV light-sensing behaviors of the PDs are showed in Figure 4. The current of PDs increases remarkably from 0.013 mA·cm−2 (dark condition) to 0.143 mA·cm−2 under AM1.5 illumination at 0 V bias and hence points toward the feasibility of using it under a self-powered mode (Figure 4a). The ratio between photocurrent (Ilight) and dark current (Idark) is estimated to be approximately 10, suggesting a good photosensitivity as a self-powered device. From the IPCE in Figure 4(b), the response range is between 300 nm and 400 nm. This matches well with the band gaps of CsPbCl3 and TiO2. 47 –49 The responsivity at 340 nm is up to 7.3 mA·W−1. Compared with the similar TiO2-based heterojunctions used for self-powered UV PDs (the maximum response wavelength is below 400 nm), this is nearly the highest value in comparison to the reported self-powered PDs (Table S2). The photocurrent of PDs exhibited a good linear dynamic range with the light intensity from 1 mW·cm−2 to 65 mW·cm−2 (inset in Figure 4(b) and Figure S7). The time-dependent current curve is measured with periodical on/off light (340 nm) at 0 V bias as shown in Figure 4(c). High stability and repeatability were achieved with several cycles of switching on and off upon light (Figure 4d). The rise and decay times of PD are 30 and 20 ms (the rise time is defined as the time for the current to raise to 90% of the peak value and the decay time is defined as the time for the current to decay to 10% of the peak value), which are faster response time than most of the reported self-power UV PDs (Table S2).

Light-sensing tests of Mn: CsPbCl3-based self-power UV PDs: (a) J-V curves under dark and AM1.5 in a log coordinate, (b) IPCE spectra. The inset is photosensitivity linearity under light intensity from dark to 65 mW·cm−2. (c) Time-dependent photocurrent curves excited at 340 nm. (d) Rise and decay time of PDs. UV illumination (λ = 340 nm, power = 65 mW·cm−2) measured for light-on and light-off states at 0 V bias. Mn: CsPbCl3: manganese (Mn)-doped CsPbCl3; UV: ultraviolet; PD: photodetector; J-V: current–voltage; IPCE: incident photon-to-current conversion efficiency.
The reproducibility of as-prepared self-power UV PDs is confirmed by Figure S8 in the supporting information. Four PDs, which prepared by adding the same batch of Mn: CsPbCl3 NCs to four zones of TMS array on the same FTO substrate, were monitored by J-V measurements. As can be seen, the measured J-V curves are quite similar, indicating the good reproducibility for the PDs prepared at the same substrate of TMS array. The experimental error is below 4% for the self-powered current density at a voltage of 0 V as listed in detail in Table S3 in the supporting information. It should be mentioned that when different batches of TMS array are used, the measured J-V curves are slightly different. However, it is no doubt that the doping of Mn can enhance the performance of CsPbCl3-based self-powered UV PDs since TMS array on the same FTO substrate has been used for investigating the effect of Mn in Figures 2 and 4. In addition, as-prepared self-powered UV PDs show good stability in a vacuum desiccator. The performance of PDs shows little change in a week (Figure S9 in the supporting information).
Conclusions
In summary, we developed CsPbCl3 NCs-based self-powered UV PDs and improved the carriers’ separation efficiency via the Mn-mediated d-states. Compared with the PDs using undoped CsPbCl3 NCs, the photocurrent has a great enhancement from 0.08 mA·cm−2 to 0.14 mA·cm−2. Moreover, a high responsivity up to 7.3 mA·W−1 was obtained at 340 nm. Due to the use of carbon-based and fully printable properties of TMS, the fabrication of PDs is low-cost and facile.
Supplemental material
Supplemental Material, Supporting_Information - Carbon-based fully printable self-powered ultraviolet perovskite photodetector: Manganese-assisted electron transfer and enhanced photocurrent
Supplemental Material, Supporting_Information for Carbon-based fully printable self-powered ultraviolet perovskite photodetector: Manganese-assisted electron transfer and enhanced photocurrent by Shuhong Xu, Guangguang Huang, Chunlei Wang, Haibao Shao and Yiping Cui in Nanomaterials and Nanotechnology
Footnotes
Author contributions
S. Xu designed the experiments, analyzed the data, and wrote the manuscript. G. Huang participated in designing the experiments and analyzed the data and conducted the experiments. Wang and Shao gave some advices in writing the manuscript, participated in the analysis of experimental data, and revised the whole manuscript. Y. Cui reviewed the discussed the results. All authors reviewed the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by Natural Science Foundation of China (61535003), Natural Science Foundation of China (Grant Nos. 61875037, 21875034, 61704093), the Foundation of Jiangsu Province for Outstanding Young Teachers in University (Grant No. BK20180064), and the Fundamental Research Funds for the Central Universities.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.