Abstract
This work aimed at exploring a new kind of purification catalysts for fuel ethanol engine exhaust. Platinum loaded on Ce0.5Zr0.5O2 was prepared by impregnation method and characterized by X-ray diffraction, Brunner–Emmet–Teller measurements (BET), temperature-programmed reduction of hydrogen, scanning electron microscope, and X-ray photoelectron spectra. The three-way catalyst platinum/ceria–zirconia/fw is prepared with the paste ball mill coating technique. And the catalytic performance is evaluated under simulated fuel ethanol engine exhaust gas condition. The catalysts not only show excellent low-temperature performances and several-way catalytic activities for carbon monoxide, hydrocarbons, nitrogen oxides, and acetaldehyde but also have better high-temperature resistance. The addition of sulfur dioxide to feedstream degrades the performances of the catalysts.
Introduction
Fuel ethanol produced from biomass presents an excellent substitute for gasoline because of its renewability, local production, low cost, and similar logistic infrastructure as fossil fuels. 1,2 Blending ethanol, a kind of renewable and clean oxygenated fuels, with gasoline (ethanol lower than 50% by volume) can be applied in gasoline engines directly without modifications and can reduce criteria air pollutants, such as total hydrocarbons (HCs) and carbon monoxide (CO). In China, over 27 cities have been using E10 as engine fuel since 2003, where E10 refers to the blending of 10% denatured fuel ethanol into gasoline. 3 According to findings from the Argonne National Laboratory in the United States, compared with gasoline engine, the concentration of conventional pollutants HC and CO is significantly lower from fuel ethanol engine, and nitrogen oxides (NOx) concentration is substantially in an order of magnitude, but organic pollutants concentration, due to the incomplete combustion ethanol, is increased significantly. The main components of these specific organic pollutants, such as acetaldehyde (CH3CHO) and formaldehyde, are important factors that could cause photochemical smog.
At present, the major researches for fuel ethanol engine focus on the combustion characteristics of the ethanol–gasoline blended fuel and the exhaust component. 3 –6 The three-way catalyst (TWC) has been used widely in the fuel ethanol engine. To the best of our knowledge, there has been no report on purification of the exhaust from this kind of engine.
Since the TWC would be run in the harsh working environment, including the continuously changed air speed, wider temperature range, higher water vapor concentration, and the presence of poisons, sometimes it would be exposed to a higher temperature above 1000°C. So the high-performance catalyst for fuel ethanol engine should not only have the fundamental properties of TWC but also have the better catalytic purification ability.
CexZr1−xO2 solid solution is the major constituent for TWC, 7 –9 and a large amount of highly active noble metals (NM) including platinum (Pt), palladium, and rhodium are supported on metal oxides with high surface areas to meet the stringent automotive emission regulations. 10 –12 On the other hand, NMs are the highly active catalysts for HCs. 13,14
In this article, Pt-loaded ceria–zirconia (CZ) solid solution was coated on honeycomb and then subjected to high-temperature hydrothermal treatments. Moreover, the catalytic performance for simultaneous purification of HC, CO, NO, and CH3CHO was investigated for the simulated exhaust, and the resistances to sulfur dioxide (SO2) poisoning performance were also researched.
Experimental
Materials preparation
Ce0.5Zr0.5O2 catalyst was prepared by coprecipitation of aqueous Ce(NO3)3 and ZrO(NO3)2 solutions with NH3·H2O as a coprecipitation agent. 15 The precipitation was performed at room temperature and at pH 10 with vigorous stirring for 1 h. After being aged at room temperature for 20 h, the precipitate was filtered and washed. To obtain the so-called fresh sample, the precipitate was first dried at 100°C overnight, and then calcined in the air at 700°C for 4 h.
Pt-loaded catalyst was prepared by incipient-wetness impregnation method with hexachloroplatinic acid as precursor. 16 The prepared fresh Pt/CZ power, gamma alumina (Al2O3) power with large special surface, Al2O3 sols solution, and water were mixed together with a certain mass ratio and grinded by ball milling with 1000 r·min−1 for 30 min. The slurry was washcoated onto a cordierite honeycomb ceramic with wall thickness as 400 CPSI (Corning Incorporated Co., Ltd, Corning, New York, USA) by the conventional dipping method. To increase the uniformity of coating, the compressed air was used to sweep the honeycomb. The loaded honeycomb was dried at 110°C for 4 h and calcined at 550°C for 4 h, which was labeled as Pt/CZ/fw-fresh. The as-received fresh samples were aged at 1050°C in 10% water vapor–air steam for 10 h, named Pt/CZ/fw-aged.
Materials characterizations
Temperature-programmed reduction of hydrogen (H2-TPR) was performed for the catalysts on a conventional TPR apparatus equipped with a thermal conductivity detector (TCD). In general, the sample (100 mg) was inserted in a quartz tube and sandwiched between two quartz wool plugs. Prior to each TPR run, the oxide catalyst was heated to 700°C in nitrogen (N2) for 30 min and then cooled to room temperature. This pretreated catalyst was then heated to 700°C at a ramp of 10°C·min−1 and reduced in a reducing environment (10% H2 in N2 at a flow rate of 40 mL·min−1). During heating, H2 consumption was monitored by the TCD.
The X-ray diffraction (XRD) patterns of the samples were measured on a powder X-ray diffractometer (X7000; Rigaku Co., Ltd, Japan) using copper
The special surface of the xPt/CZ samples was analyzed by N2 adsorption/desorption at liquid N2 temperature, using a Quantachrome NOVA-1200 (Quantachrome Co., Ltd, USA) gas absorption analyzer. The specific surface area was calculated with the BET equation.
The morphology for catalyst was observed in S-3500 N scanning electron microscope (SEM; Hitachi Co., Ltd, Japan).
Oxygen storage capacity (OSC) was determined from the change in weight as evaluated using a thermo-balance (Q600; TA Co., Ltd, Newcastle, Delaware, USA). Typically, 0.03 g of xPt/CZ sample was employed and the gas flow rate was 50 mL·min−1. The quantity of oxygen (O2) stored and released was evaluated by measuring the difference in weight between oxidizing step (50% O2/N2) and reducing step (20% H2/N2) and calculated in μmolO2·mol−1.
X-ray photoelectron spectra (XPS) were carried out on a VGESCALAB MKII spectrometer (VG Instruments Co., Ltd, UK) with a hemispherical electron analyzer and magnesium
Activity evaluation
The self-made activity evaluation system was shown in Figure 1.
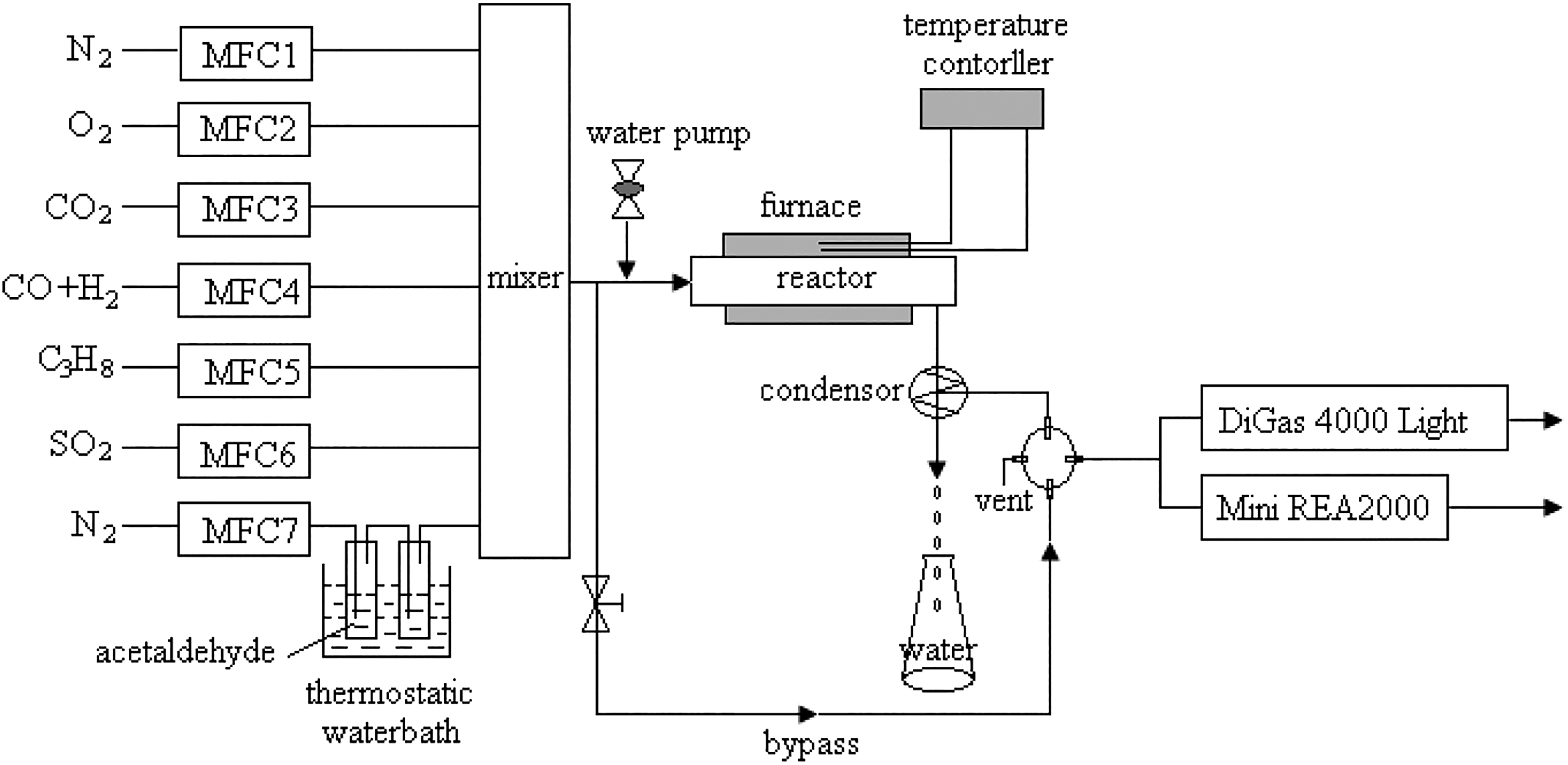
The temperature-programmed oxidation (TPO) experiment apparatus.
The catalysts were placed in a tube micro-reactor and reacted with a gas mixture of simulated exhaust from fuel ethanol engine. The simulated exhaust contained a mixture of CO (2%), H2 (1%), carbon dioxide (CO2; 10%), propane (C3H8; 0.05%), NO (0.06%), water (10%), CH3CHO (60 ppm, V/V
The curves of the relationship between conversion and temperature were obtained. T50 (the temperature at which the conversion of a certain reactant reached 50%) and the final conversion were used to show the catalytic activity of the catalysts.
Results and discussion
BET and OSC analysis
The textural properties of the samples are summarized in Table 1.
The basic characters of the catalysts.

OSC: oxygen storage capacity; Pt: platinum; CZ: ceria–zirconia.
The surface area of Ce0.5Zr0.5O2 solid solution is 132 m2·g−1, and the OSC value is 11.36 μmolO2·mol−1. When Pt was loaded on Ce0.5Zr0.5O2, the surface area of Pt/Ce0.5Zr0.5O2 decreased to 87 m2·g−1. Pt can increase the transfer rate of the surface O2 and lower the activation energy for H2 reduction, 17 so the OSC of Pt/CZ-fresh reached 22.09 μmolO2·mol−1. Due to the surface agglomeration and the crystal growth during the course of high-temperature hydrothermal treatments, the specific area of Pt/CZ-aged sharply dropped to 15 m2·g−1, and the OSC is 15.37 μmolO2·mol−1, which is still higher than the CZ sample.
XRD analysis
The XRD patterns of the three samples are shown in Figure 2.
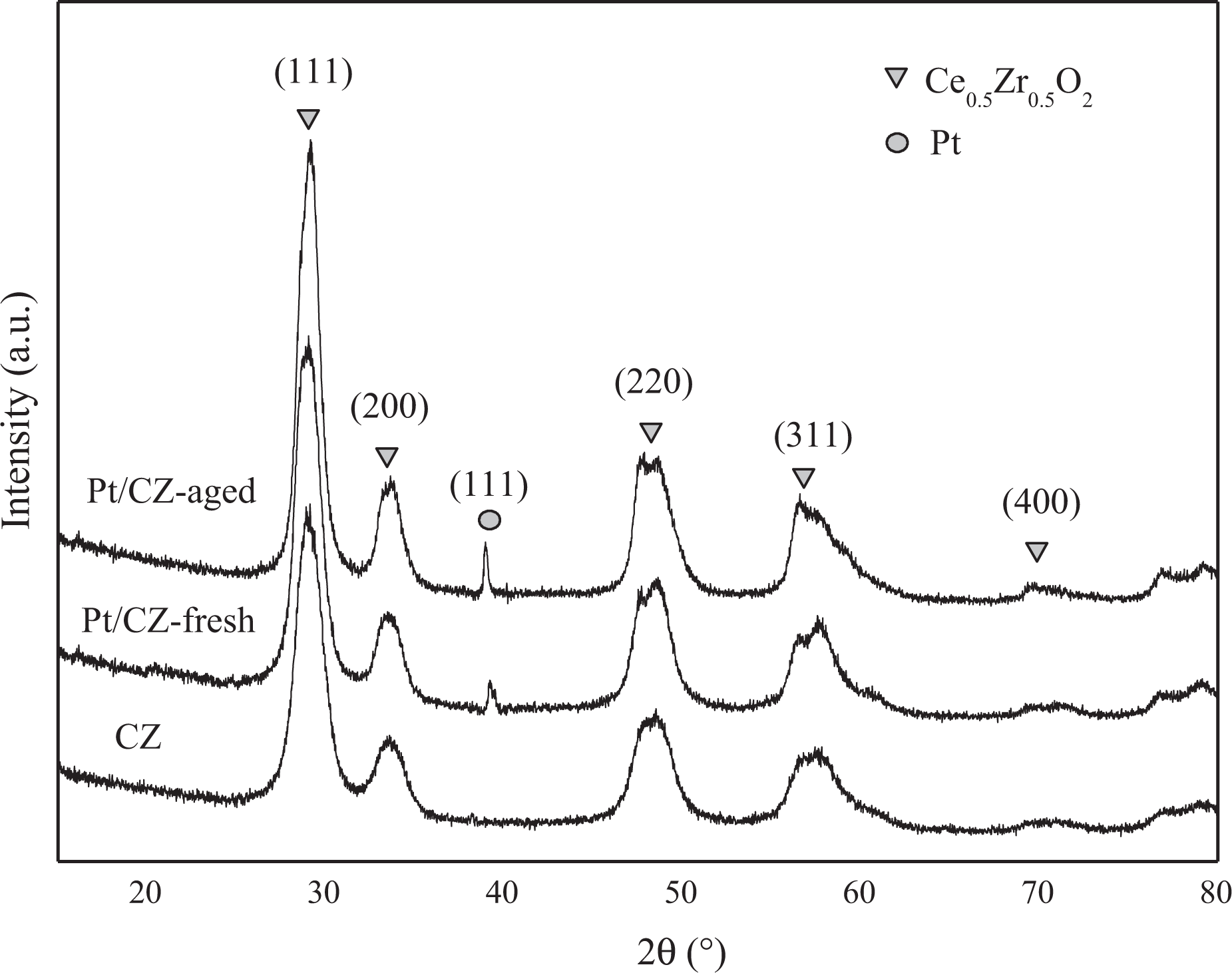
XRD spectra for the samples. XRD: X-ray diffraction.
The diffraction lines at 29.2°, 33.2°, 47.8°, and 56.2° (2
After Pt was loaded on Ce0.5Zr0.5O2, besides the main peaks for Ce0.5Zr0.5O2, the characteristic peaks for Pt0 (111) at 39° is detected,
18
which convinces us that the Pt exists mainly in the form of metal no matter whether the sample is aged. The characteristic diffraction peaks of XRD patterns of platinum oxide (PtO2; 2
Compared with Pt/CZ-fresh sample, the diffraction peaks of Pt/CZ-aged exhibit higher intensity and symmetry, which maybe mainly related to the sintering of Pt particles during the treatments course, and the crystal sizes of the (111) plane increased from 51.817 nm to 78.239 nm according to the calculation based on Scherrer equation.
XPS analysis
Figures 3 and 4 show the high-resolution XPS spectra of Ce 3d and O 1 s on the Pt/CZ/fw-fresh sample before and after TPO experiments for the simulated exhaust without SO2. All XPS spectra are splitted to get the relative content of each element in different valence states and the results are presented in Table 2.

Ce 3d XPS spectra of the sample. (a) Pt/CZ/fw-fresh sample. (b) Pt/CZ/fw-fresh sample after TPO reaction (no SO2). XPS: X-ray photoelectron spectra; Pt: platinum; CZ: ceria–zirconia; SO2: sulfur dioxide.

O1 s XPS spectra of the sample. (a) Pt/CZ/fw-fresh sample. (b) Pt/CZ/fw-fresh sample after TPO reaction (no SO2). XPS: X-ray photoelectron spectra; Pt: platinum; CZ: ceria–zirconia; SO2: sulfur dioxide.
Quantitative aspects of analyses of sample by XPS.

XPS: X-ray photoelectron spectra; Pt: platinum; CZ: ceria–zirconia; SO2: sulfur dioxide.
a The simulated exhaust without SO2.
b The lattice oxygen.
c The adsorbed oxygen or the surface hydroxyl species.
The Ce 3d XPS spectra in Figure 3 present several peaks, which contributed to Ce 3d 5/2 and Ce 3d 3/2, respectively. This imply that both status of Ce4+ and Ce3+ coexisted on the sample surface. Ce4+/Ce3+ ratio on the Pt/CZ/fw-fresh sample is higher than that on the reacted sample, which indicates that part of Ce4+ is converted to Ce3+ in the course of TPO reaction. It is reported that Ce3+ ions could help to facilitate the formation of defect O2 on a CeZrO2 surface. So a higher concentration of Ce3+ and higher defect O2 sites occur in the reacted sample, which would be further proved by the O 1 s spectra. 19
The O 1 s spectra show the coexistence between lattice oxygen (OI) and adsorbed oxygen (OII). In order to distinguish the two types of O2, the spectra for the O 1 s ionization feature are numerically fitted with two Gaussian–Lorenz features, as shown in Table 2 and Figure 4. 12
Compared with the fresh sample, the OII% in the sample after TPO experiment is decreased from 28.85% to 18.03% and the Ce4+% is changed from 62% to 51% accordingly. These results indicate that the OI takes part in the reaction and part of O* are offered by deoxidizing CeO2 to Ce2O3.
SEM analysis
The morphology of the fresh and aged samples of Pt/CZ catalyst was investigated by SEM (Figure 5).

SEM images of samples. (a) Pt/CZ-fresh. (b) Pt/CZ-aged. SEM: scanning electron microscope; Pt: platinum; CZ: ceria–zirconia.
Compared with the SEM image of Pt/CZ-fresh sample, the larger particle and nonuniform surface can be observed for Pt/CZ-aged sample, and the maximum particle may reach 40 µm or so. It can be implied that obvious sintering occurred in the aging treatment, triggering the agglomeration and the crystal growth, which agreed with XRD results. Meanwhile, the grain boundary of cerium zirconium solid solution can be seen for the two samples, and the structure was loose for fresh sample and was smooth for aged sample.
H2-TPR analysis
The reducibility of the three samples is determined by the TPR tests, as shown in Figure 6.
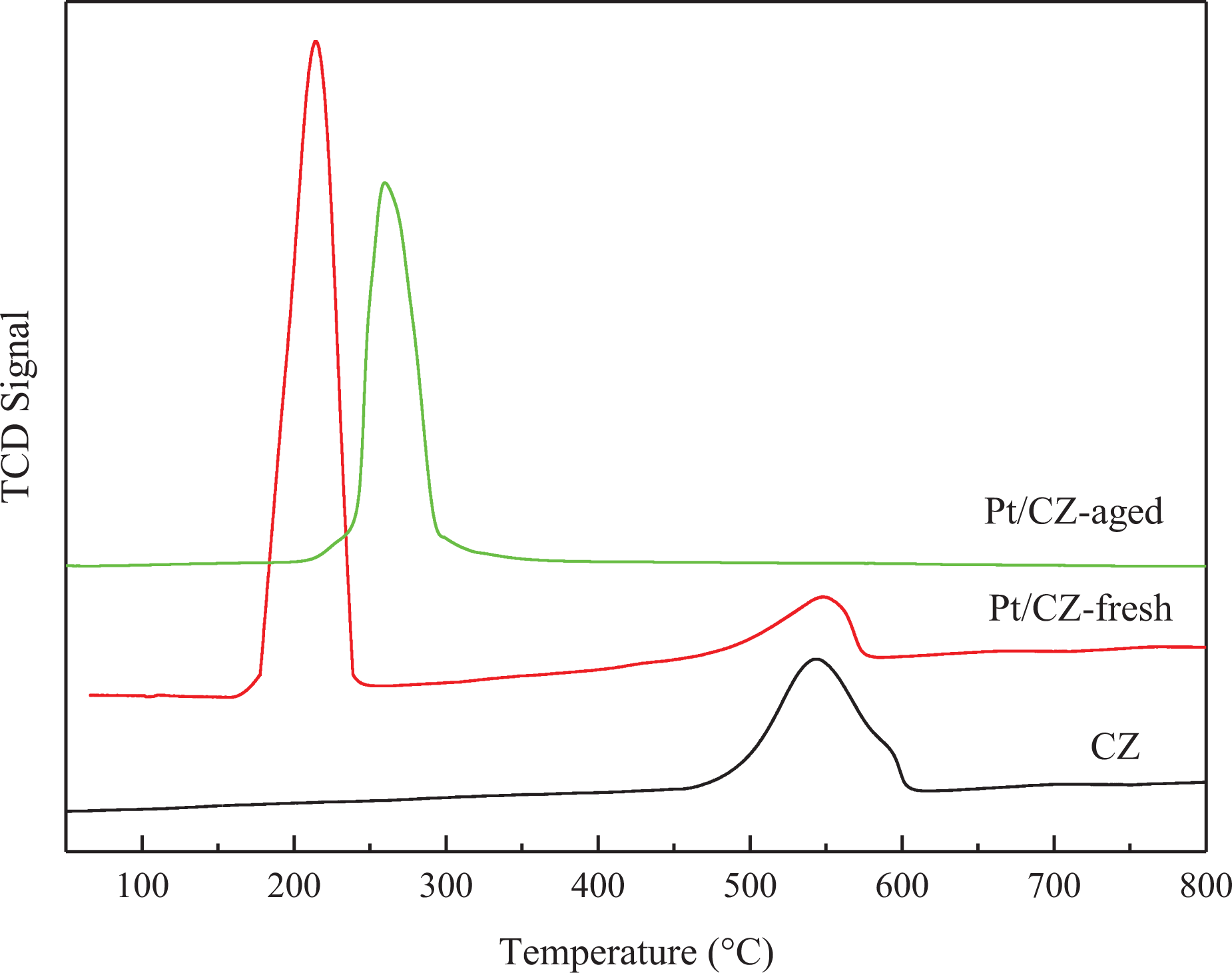
H2-TPR profiles of the samples. H2-TPR: temperature-programmed reduction of hydrogen.
The O2 release of CZ support starts at 400°C. Almost all Ce4+ is reduced to Ce3+ below 650°C, so the reduction peak (400–650°C) in the TPR profile can be ascribed to the reduction of subsurface and surface O2 of the CZ, and the two peaks are overlapped. The reduction temperatures are considerably lower than that of CeO2 (above 700°C), which is due to the increased mobility of bulk O2 in CZ solid solutions after the introduction of Zr4+ into the ceria lattice. 20 Two kinds of peaks, including the sharp peaks at 200°C and the broad peaks at 550°C, are detected in Pt/CZ catalysts. The mechanism of O2 release is suggested by several researchers. 21,22 The first peak can be associated with the reduction of PtO2 particles from the vicinity of CZ surface, which indicates that the loading of Pt can effectively promote the reduction performance of Ce0.5Zr0.5O2. The intensity of the second peak is a little weaker than that of CZ sample, which may correspond to the surface O2 on the unprompted Ce0.5Zr0.5O2. The O2 release temperature decreases with the Pt loadings. Thus, Pt loaded on Ce0.5Zr0.5O2 can improve the performance of active O2 in the sample and can increase the overflow rate of these active O2 transfers to CZ surface. So it can be revealed that the O2 release from CZ support is accelerated by Pt species. For the Pt/CZ-aged sample, the peak owning to the reduction of the Pt-promoted Ce0.5Zr0.5O2 solid solution rises to about 260°C, and the peak in the higher temperature disappears.
Activity analysis
The activity of the catalyst is represented by the ignition temperature (
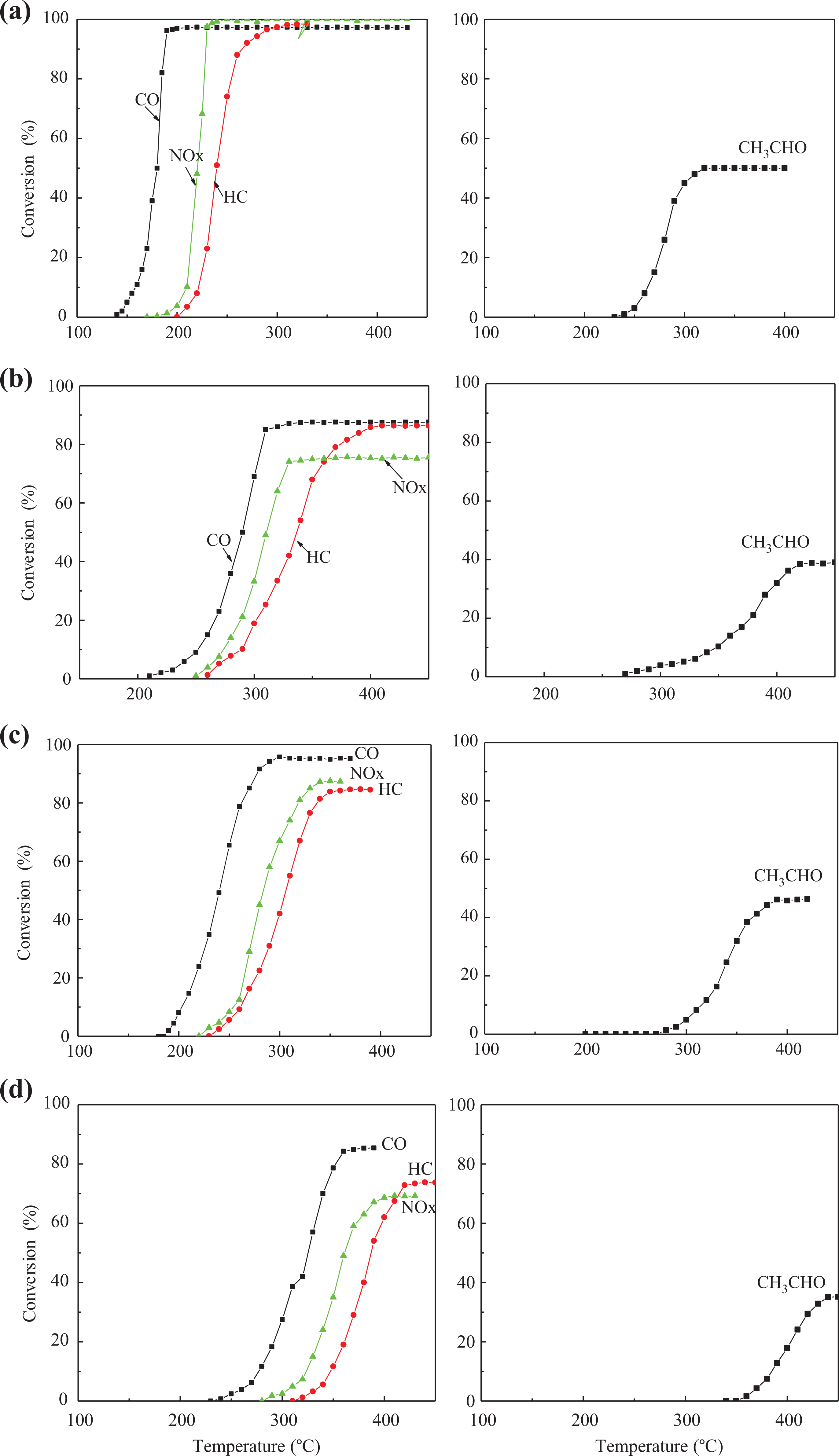
The dependence of conversion on reaction temperature for catalyst. (a) and (b) Fresh catalysts. (c) and (d) Aged catalysts. (a) and (c) No SO2. (b) and (d) In the presence of 0.003%SO2. SO2: sulfur dioxide.
The
Light-off temperature and SO2 poison on the catalysts.

SO2: sulfur dioxide; HC: hydrocarbon; CO: carbon monoxide; NOx: nitrogen oxides; CH3CHO: acetaldehyde; Pt: platinum; CZ: ceria–zirconia.
From Figure 7(a) and (b), it can be seen that the higher temperature is, the higher conversions of fresh catalyst Pt/CZ/fw to HC, CO, and NOx are, and the conversion for CH3CHO also exceeds 50%. For the Pt/CZ/fw-aged sample, the catalytic activity shows degradation. The T50 over Pt/CZ/fw-aged catalyst shifts to higher temperature with an increase of 53°C for CO, 56°C for HC, and 54°C for NOx, respectively. The maximum conversions to HC and NOx are also decreased by 13%, and the values to CO and CH3CHO only decreased slightly.
These results imply that the aged catalyst still exhibits higher catalytic oxidation activity, which also demonstrates that the prepared sample has low-temperature light-off property and high-temperature anti-aging performance. It may be attributed to the larger surface area of Ce0.5Zr0.5O2, which facilitates the uniform dispersion of precious metals on the support. A shell–core structure model proposed by Fan et al. 23 can explain this phenomenon, which describes the diffusion of ions and O2 vacancies as well as the possible surface structure serving as the carrier of Pt0 site which has impact on the catalytic activity remarkably.
From Figure 7(c) and (d), it can be clearly seen that in the presence of 0.003% (V/V) SO2, the deactivation occurs no matter fresh catalyst or aged catalyst is involved. The onsets of HC, CO, NOx, and CH3CHO conversions are all retarded and the conversion curves are about the same. The T50 over Pt/CZ/fw-fresh catalyst shifts to higher temperature with an increase of 105°C for CO, 84°C for HC, and 77°C for NOx, and the conversion of NOx decreases from 100% to 75.63%. The similar results can also be found in Salvador et al.’s research. 24 As the reaction temperature rises, the sulfate or sulfur species would aggregate on the catalyst surface, which leads to the accelerated deactivation of the TWC. In order to further improve the performance for anti-SO2 poisoning of the catalyst, the special metal components may be added in the O2 storage material, such as titanium, lanthanum, and so on.
Figure 8 gives the relationship between the

Curves of conversion versus
Conclusions
The synthesized Pt/CZ/fw catalysts present the higher catalytic performance for purification of HC, CO, NOx, and CH3CHO simultaneously from the fuel ethanol exhaust. After the hydrothermal aging treatment, the Pt/CZ/fw-aged catalyst remained the favorable catalytic oxidation properties; the combustion temperature was increased about 55°C. Although sulfur species aggregated on the catalyst surface, the SO2 in the atmosphere degraded the catalytic activity. The work of this article may provide useful design considerations for further work on the engine exhausts purification catalysts and other classes of catalysts.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from Support Project of High-Level Teachers in Beijing Municipal Universities in the Period of 13th Five-year Plan—Beijing Municipal College “Great Wall” Scholars Projects (CIT&TCD20190314).
