Abstract
Ethyltriethoxysilane-functionalized graphene oxide/polyimide composite films were synthesized. Eighty percent improvement in Young’s modulus, high thermal stability at 800°C, and 3.46-fold increase in dielectric constants compared to polyimide with a dielectric loss of only 0.035 were exhibited by these composite films.
Introduction
Polyimide (PI) exhibits excellent physicomechanical properties in a broad temperature range, exceptionally high radiation resistance, and superior semiconductor properties, which predetermine the main PI applications in electronics, electrical engineering, and aviation. 1 To extend potential applications of PI, its reinforcement with inorganic nanofillers has attracted considerable attention.
Polymer nanocomposite has made integration of various types of materials into one system with enhanced performance or newly acquired function. Due to the nano effect of nanofillers, polymer nanocomposites exhibit outstanding properties with small amount of nanofillers. 2,3 Compared with the traditional nanofillers, graphene, a planar monolayer of carbon atoms arranged into two-dimensional (2-D) honeycomb lattice with carbon–carbon bonds, 4 reunites a variety of intriguing properties never observed in a single material, such as high electrical conductivity(6000 S/cm), 5 exceptional thermal conductivity (5000 W m−1 K−1), 6 and superior mechanical properties with ultimate strength of 130 GPa and Young’s modulus of 1 TPa 7 making graphene the ultimate nanofiller for improving the properties of polymer matrix composites. Insolubility in aqueous media and tendency to form agglomerates are the two major problems encountered while dealing with graphene as nanofiller, which result in weakening interfacial interaction between graphene and polymer interface. 8 –11 Graphene oxide (GO) being a precursor of graphene has overcome these shortcomings of graphene. Various functional groups are present on basal plane and edges. GO results in its swelling and homogeneous dispersion. 12 Surface modification of nanomaterial with some organic functionality also helps in avoiding agglomeration by improving the dispersion of nanomaterials in polymer matrices. One of the useful coupling agents for the modification of nanomaterials is silane. Silane coupling agents are grafted to the polar surfaces of inorganic nanomaterials; this modification by grafting improves the dispersion ability of nanomaterials in various organic media.
Various types of silane were reported by different research groups as reinforcement in PI-based composite. Alkyl-functionalized graphene oxide (AGO)/PI nanocomposites were synthesized by Do Hoon Lee and his coworkers via in situ polymerization, with improved tensile strength and modulus. 13 Peijiang Liu and his coworkers synthesized 3-aminopropyltriethoxysilane (APTS)-functionalized GO/PI nanocomposites, with improved tensile strength, Young’s modulus, and thermal stability. 14
In this study, authors have functionalized the GO with ethyltriethoxysilane (ETEOS) by a known method; subsequently, this ETEOS-functionalized graphene oxide (EGO) was used for composite formation with PI. To the best of our knowledge, EGO was used for the first time as a reinforcement filler in PI. These composites were then characterized to study the effect of EGO on the properties of PI.
Experimental
Materials
GO was synthesized from natural graphite powder having approximately 70 µm particle size, 99.9% purity, and 2.25 g/cm3 density. ETEOS, hydrochloric acid (HCl), sulfuric acid (H2SO4, 99.9%), dimethylacetamide (DMAc, 99.8%, water content < 0.005%), pyromellitic dianhydride (PMDA, 99.2%), 4,4′-oxydianiline (ODA, 97%), and ethanol (C2H5OH) were purchased from Sigma-Aldrich, USA. Sodium nitrate (NaNO3, >99.5), and N,N-dimethylformamide (DMF) were purchased from Merck.
Synthesis of GO and EGO
Modified Hummer’s method was used for the synthesis of GO. 15 ETEOS (600 mg) solution in 90 ml ethanol was added in ultrasonic dispersion of GO (300 mg) in 300 ml deionized water which was taken in 500 ml three-necked flask. Subsequently, HCl (0.5 ml) was added slowly into this flask; mixture was stirred and heated at 70°C for 2 h. Later on, acid was removed by washing the mixture with deionized water followed by filtration. Physically adsorbed ETEOS was removed by ultrasonically dispersing and subsequently filtering the filtered powder. This procedure was repeated thrice (Figure 1). Resulting powder was dried in vacuum oven at 60°C until a constant weight. 16

Synthesis of EGO. EGO: ETEOS-functionalized graphene oxide; ETEOS: ethyltriethoxysilane.
Synthesis of GO/PI and ETEOS/PI (EGO/PI) nanocomposites
For EGO/PI nanocomposites, PI was synthesized by thermally imidizing the poly(amic acid) (PAA). For the synthesis of PAA, 100 ml DMAc was taken in a flask kept in a glove box under anhydrous conditions and at about 5°C. ODA (8.56 g, 42.74 mmol) was added to this flask and dissolved by mechanically stirring for 30 min; later, PMDA (10.25 g, 47.48 mmol) was also added in it with vigorous stirring. Polymerization resulted in the increase in the viscosity of the solution which hindered the stirring. To facilitate stirring, additional 133 ml of DMAc was added to this solution. Presence of the anhydride groups at the chain ends was ensured by adding extra quantity of PMDA to the reaction mixture after 6 h. Solution was stirred for further 18 h continuously so as to complete the polymerization reaction. Flexible and free-standing hybrid films were cast from this solution by evaporating the solvent at 70°C for 12 h.
Nanocomposites with different contents of EGO were synthesized by solution blending method, in which ultrasonic dispersion of EGO in N,N-DMF was mixed with PAA by stirring and sonication.
For thermal imidization of PAA into PI, PAA/EGO composite films were heated at 100°C, 200°C, and 300°C for 1 h each to get EGO/PI nanocomposites’ films (Figure 2). GO/PI nanocomposite films were prepared similarly.
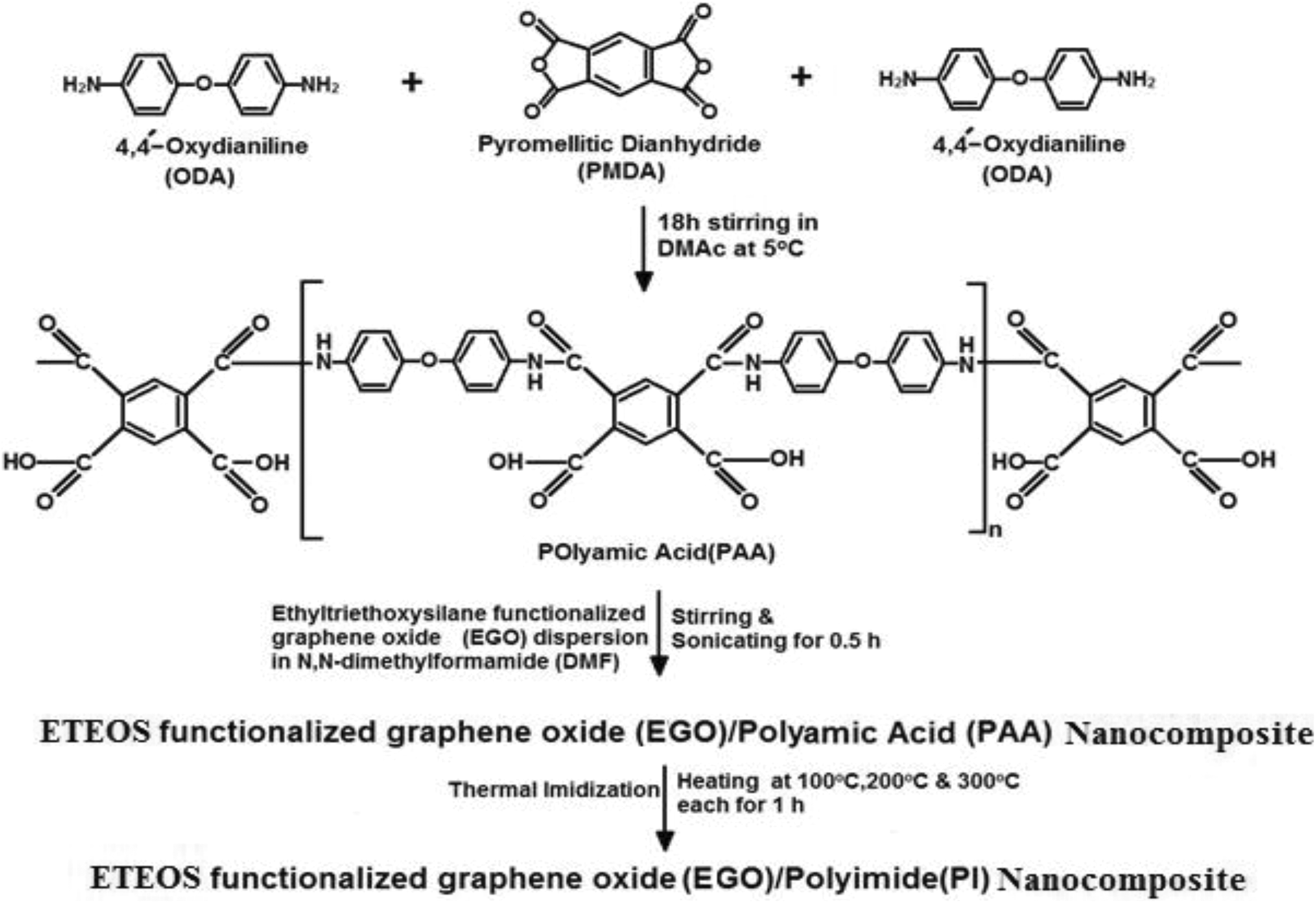
Nanocomposite synthesis.
Characterization
VG scientific ESCA Lab 2201-XL equipped with a hemispherical electron analyzer was used for X-ray photoelectron spectroscopy (XPS) analysis. Rigaku D/A X-ray diffractometer was used for X-ray diffraction (XRD) studies. Thermo Scientific Nicolet 6700 was used to record Fourier transform infrared (FTIR) spectra. Samples were dried in vacuum at 45°C for 24 h before examination. Measurements were made at room temperature at a scanning range of 4000–500 cm−1. NETZSCH TG 209 F1 instrument was used for determining the thermal stability of the nanocomposites. LEO 1530 VP field emission scanning electron microscope (LEO. Co., Germany) was used for scanning electron microscopy (SEM). Mechanical properties of the composites were measured by Instron 5565 Electronic Universal Tensile Strength. Dielectric constant and loss were measured by Agilent Technologies Precision Impedance Analyzer (Santa Clara, California, USA) in the frequency range of 1–103 kHz.
Results and discussion
Characterization of EGO
Functionalization of GO with ETEOS is confirmed by XPS. By comparing XPS survey scan of GO (Figure 3(a)) and EGO (Figure 3(b)), it was found that the appearance of two new peaks of Si2p and Si2s at 100 and 150 eV in the XPS pattern of EGO confirmed the functionalization of GO with ETEOS. These results are in agreement with the previous work. 17 C1s spectral analysis is used to have evidence of the reaction between ETEOS and GO. From the Figure 4(b), it appears that after functionalization of GO with ETEOS, the peak corresponding to the hydroxyl group in the C1s spectrum of GO (Figure 4(a)) weakened significantly with the appearance of two new peaks at 283.7 eV and 285.4 eV that corresponds to C–Si and C–O–Si, respectively, indicating the reaction between hydroxyl groups of GO and ETEOS.

XPS survey scan of (a) GO and (b) EGO. XPS: X-ray photoelectron spectroscopy; EGO: ETEOS-functionalized graphene oxide; ETEOS: ethyltriethoxysilane; GO: graphene oxide.

C1 s spectrum of (a) GO and (b) EGO. EGO: ETEOS functionalized graphene oxide; ETEOS: ethyltriethoxysilane; GO: graphene oxide.
For the determination of the degree of exfoliation of functionalized GO in the nanocomposite, XRD is used. Normally, the disappearance of the characteristic peak of GO sheets in the GO/polymer nanocomposite can demonstrate that sheets are fully exfoliated in the matrix. 18 –21 Figure 5 shows the XRD results, in which peak centered at 7° exhibits XRD patterns of EGO, whereas peak corresponding to GO appears at 12.5°. This clearly indicates that functionalization of GO with ETEOS results in an increase in interlayer spacing due to the introduction of ETEOS on the surface of GO. EGO/PI nanocomposite exhibits a peak centered at 15.25°; this peak distinguishes from the peaks of GO, EGO, and PI indicating their exfoliation. The increase of EGO content intensity of nanocomposite peak confirms the presence of EGO in the nanocomposite.
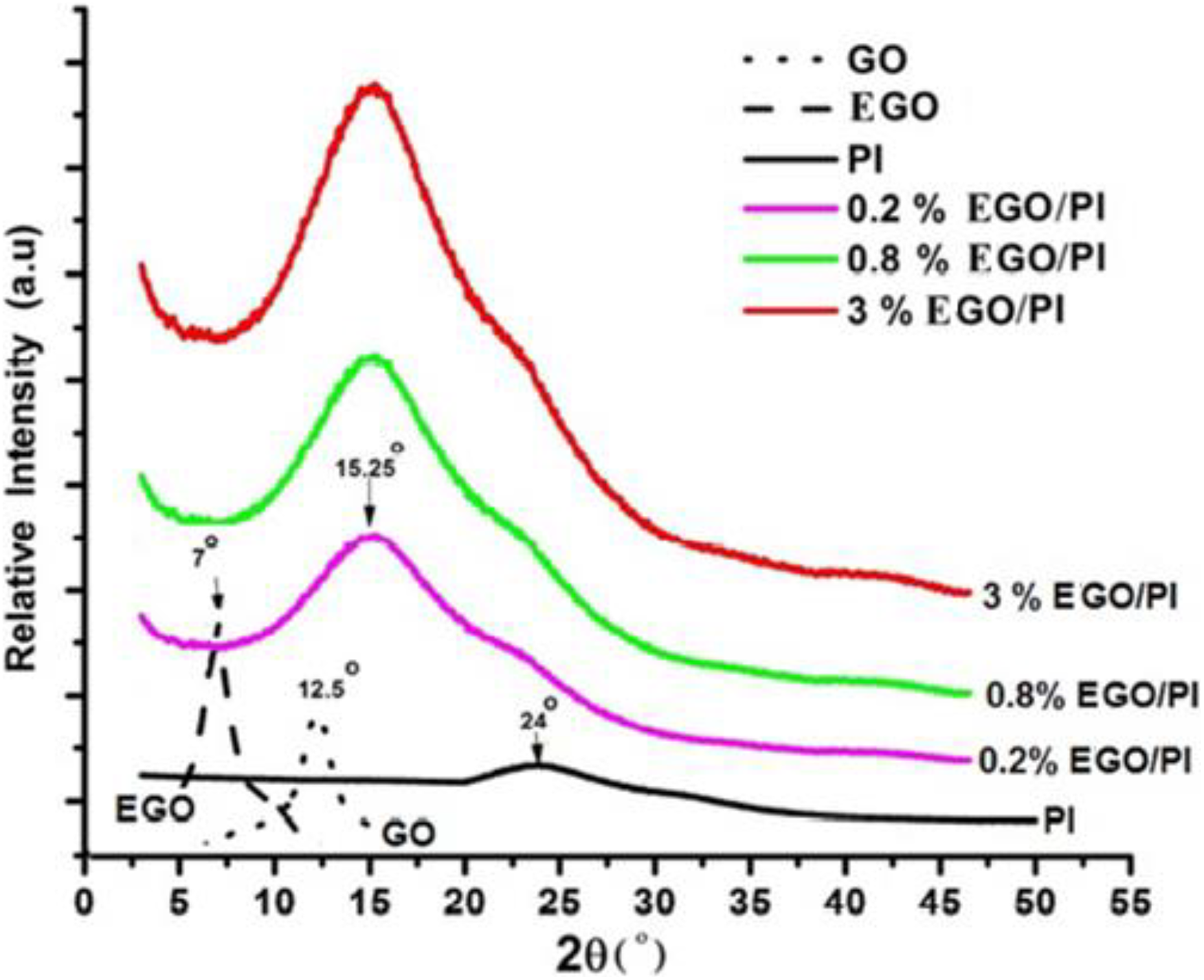
XRD analysis of EGO/PI nanocomposites. GO: graphene oxide; XRD: X-ray diffraction; EGO/PI: ETEOS-functionalized graphene oxide/polyimide; ETEOS: ethyltriethoxysilane.
FTIR spectrum (Figure 6) shows peak at 2962 cm−1 is due to CH3 stretching vibration, whereas peak at 2850 cm−1 appears due to CH2 groups of ETEOS. At 971 cm−1, peak arises due to stretching of Si–O–C bond, indicating the successful incorporation of ETEOS on the surface of GO via chemical bonding. Emergence of the peak at 1012 cm−1 is due to stretching of Si–O–Si, indicating the formation of Si–O–Si network. These peaks are in agreement with the previous work. 16 For imide absorption, bands appear at 1778, 1723, 1380, and 723 cm− 1, indicating the thermal imdization of PAA into PI. 22

FTIR spectra of the EGO, PI, and EGO/PI nanocomposites. FTIR: Fourier transform infrared; EGO/PI: ETEOS-functionalized graphene oxide/polyimide; ETEOS: ethyltriethoxysilane.
Dispersion of EGO in PI matrix
EGO’s excellent dispersion is proven by the SEM images (Figure 7) of fractured surfaces of nanocomposite films. Nanocomposite’s fracture surface is rough, whereas it is rigid for the nanocomposite which is the result of 2-D geometry of graphene sheets.

SEM images of EGO/PI nanocomposite. SEM: scanning electron microscopy; EGO/PI: ETEOS-functionalized graphene oxide/polyimide; ETEOS: ethyltriethoxysilane.
Tensile properties of EGO/PI nanocomposites
Table 1 summarizes tensile properties, signifying the improvements in tensile properties of EGO/PI films. There is 56.48% increase in young’s modulus for 0.2 wt% of EGO/PI nanocomposite as compared to 31.89% for 0.5 wt% of GO/ PI nanocomposite. There is 71.06% and 79.61% increase in Young’s modulus for 0.8 wt% EGO/PI and 3 wt% EGO/PI nanocomposite, respectively, as compared to PI. Fine dispersion of EGO may be the cause of this reinforcement. Final properties of nanocomposites are decided by the extent of the bonding between the polymer matrix (continuous phase) and filler (discontinuous phase). 23 Improvement in mechanical properties in this particular work is better as compared to the previous works. 24,25
Summary of tensile properties.

PI: polyimide; EGO: ETEOS functionalized graphene oxide; ETEOS: ethyltriethoxysilane; GO: graphene oxide.
Thermogravimetric analysis of EGO/PI nanocomposites
To examine the effect of EGO incorporation on the thermal stability of EGO/PI nanocomposites, thermogravimetric analysis (TGA) was carried out in the presence of nitrogen and in the 25–800°C temperature range. From the Figure 8, it appears that decomposition of PI and EGO/PI nanocomposites is completed at 800°C, thermal stability of the EGO/PI nanocomposites is higher than PI, and it increases with the increase of EGO content. Temperature corresponding to 5% weight loss is usually used for evaluating the thermal stability of nanocomposite. In this typical case for 5% weight loss, temperature of 0.2 wt% EGO/PI nanocomposites increases by 64°C and 19°C as compared to PI and 0.5 wt% GO/PI nanocomposite. Whereas for the same, 5% weight loss temperature increases by 80°C and 101°C for other two weight percentages, that is, 0.8 wt% EGO/PI and 3 wt% EGO/PI as compared to PI. This indicates thermal stability of EGO/PI nanocomposites.TGA data of EGO/PI nanocomposites are summarized in the Table 2. Results indicate that with the increase of EGO loadings from 0.2 to 3 wt%, charred residue of the nanocomposite also increases as represented by decrease in the weight loss. From this table, it appears that weight loss decreases from 32.50% to 30.30% as EGO content increases from 0.2 to 3 wt%. This indicates that the introduction of EGO into the nanocomposites enhances the char formation on the polymer surface, which consequently reduces the decomposition rate of the nanocomposites. This enhanced thermal stability can be attributed to the high heat resistance and structural changes that originate from the strong interfacial interaction between ETEOS groups of EGO and PI.
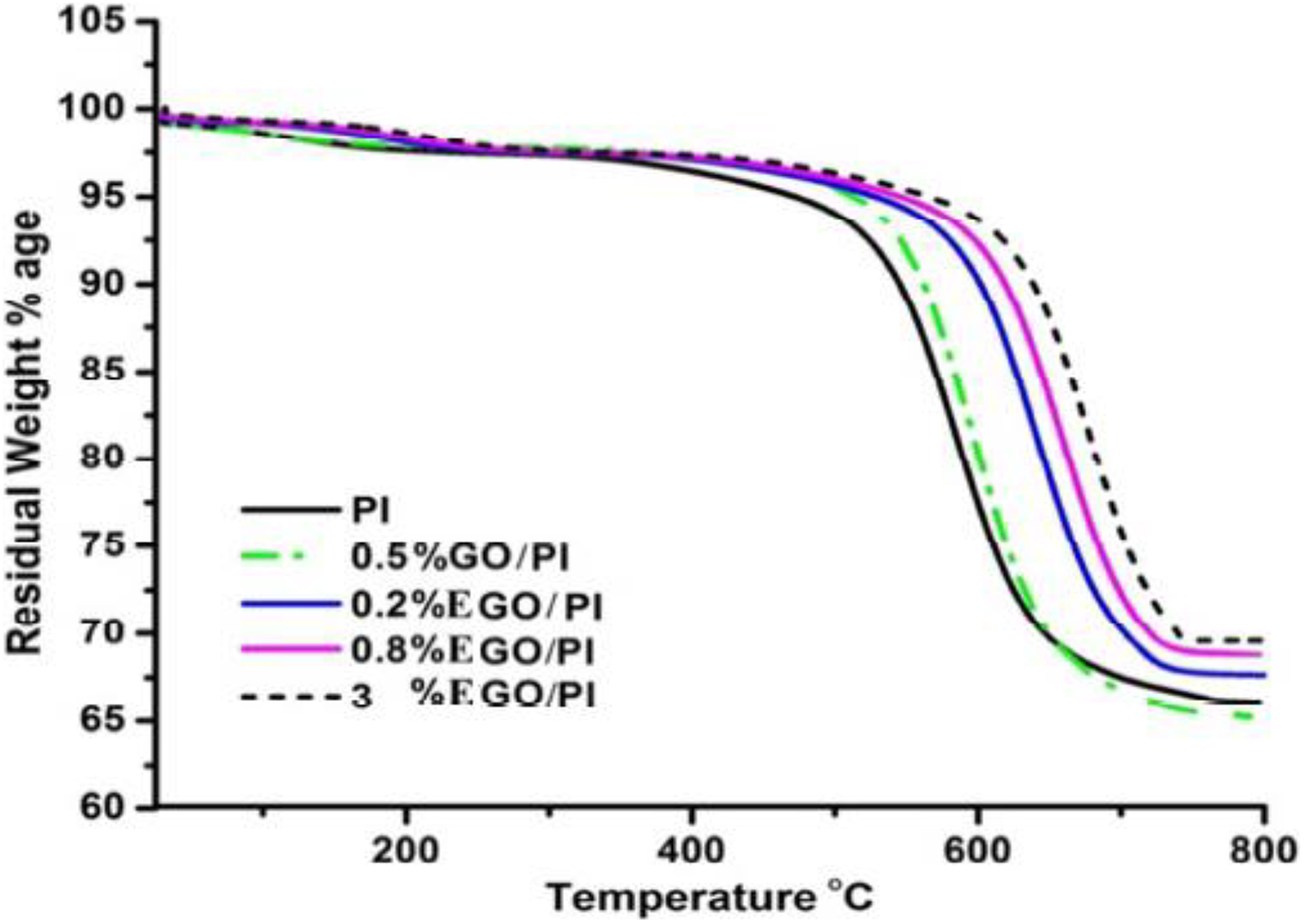
TGA curves of EGO/PI nanocomposites in comparison with PI and GO/PI nanocomposites. TGA: thermogravimetric analysis; EGO/PI: ETEOS functionalized graphene oxide/polyimide; ETEOS: ethyltriethoxysilane; GO: graphene oxide; PI: polyimide.
TGA data of PI and EGO/PI nanocomposites.

PI: polyimide; EGO: ETEOS-functionalized graphene oxide; ETEOS: ethyltriethoxysilane; GO: graphene oxide; TGA: thermogravimetric analysis.
Dielectric properties of EGO/PI nanocomposites
Capacitance method was used to measure the dielectric properties of the nanocomposites. Following equation was used to calculate capacitance

Dielectric properties of PI and EGO/PI nanocomposites as a function of frequency ranges from 1 kHz to 1 MHz at room temperature are presented in Figure 9. Dielectric constants of all the EGO/PI nanocomposites have relatively weak frequency dependence. The dielectric constant of 3 wt% EGO/PI nanocomposite decreases from 12.1 to 11.7 as the frequency increases in the studied frequency range of 1 kHz–1 MHz, while the dissipation factors are still less than 0.13, suggesting that it is a promising EGO-based dielectric stable composite. For 3 wt% EGO, dielectric constant increases to 12.1 at 1 kHz, whereas it was 3.5 for PI. More importantly, the dielectric loss tangent (Figure 10) of the nanocomposites is as low as 0.035.This increase in dielectric permittivity can be explained by interfacial polarization, 26 in which charge carrier originating from the external electrode accumulates at the interface, resulting in an increase in dielectric constant. Dielectric properties are also influenced by the dispersion of nanofillers in matrix, leading to higher dielectric constant and lower dielectric loss.

Dielectric constant of nanocomposites and PI. PI: polyimide; EGO/PI: ETEOS functionalized graphene oxide/polyimide; ETEOS: ethyltriethoxysilane; GO: graphene oxide.

Dielectric loss of nanocomposites and PI. PI: polyimide; EGO/PI: ETEOS-functionalized graphene oxide/polyimide; ETEOS: ethyltriethoxysilane; GO: graphene oxide.
Conclusion
An effective approach for preparing EGO/PI nanocomposites with good performances via solution blending polymerization has been demonstrated in this study. XPS studies have confirmed the successful functionalization of GO. Exfoliation of functionalized GO is confirmed by XRD. Mechanical properties of the nanocomposite exhibited improvement, and nanocomposite containing 0.2 wt% EGO demonstrated 56% increase in Young’s modulus as compared to PI. Thermal stability of the nanocomposite also exhibited minor improvement as compared to PI. Decrease of dielectric constant from 12.1 to 11.7 with the increase in frequency from 1 kHz to 1 MHz with dissipation factor less than 0.13 suggested that it is a promising dielectric stable nanocomposite. Consequently, this method provides an effective approach to develop a nanocomposite material for an extensive variety of applications.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of following financial support for the research, authorship, and/or publication of this article: This work was financially supported by Higher Education Commission, Pakistan.
