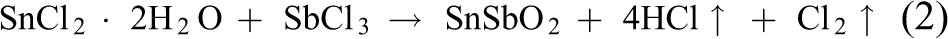Abstract
The searches for advanced battery materials are endless. Ferrites have drawn much attention as a potential anode material in Li-ion battery. Nanocrystalline transition metal ferrite MFe2O4 (M = Ni, Co and Cu) thin films were deposited by spray pyrolysis technique over antimony doped tin oxide substrate. The phase and surface morphology were studied by X-ray diffraction and scanning electron microscope measurements, respectively. Magnetic property, film thickness and electrochemical performance of the materials are studied. The result shows that the films are of single phase, with cubic spinel structure for NiFe2O4 and CoFe2O4 and tetragonal structure for CuFe2O4. Magnetization studies reveal that the films are ferrimagnetic in nature. Electrochemical measurement reveals NiFe2O4 and CuFe2O4 are having good recyclable nature, which can be used as potential anode material for Li-ion batteries.
Introduction
Advanced novel materials are important for different applications, where nanosize and tailored properties play a vital role. Subsequently, synthesis and characterization of nanostructured materials have become imperative for the advancement of science and technology. The manipulation of physical and chemical residences of the materials is one of the challenging areas in physics, chemistry and materials science. The specific properties of the substances including structural, electric, optical, magnetic and electrochemical properties especially depend on the size and shape of the nanoparticles. Therefore, the particle size and shape are considered as crucial parameters for the nanomaterials in novel applications. Currently, synthesis and application of magnetic nanocomposite is a subject of intense studies because of their particular properties, which make them very appealing from both the scientific view of understanding their properties and the technological significance of improving the performance of recent materials. Nanocomposite is an innovative material compared to the simple and substituted ferrites. It is a new class of material that has emerged in very recent years. The usual carbon-based electrode materials have low ‘charge–discharge’ capacity (theoretical value: 372 mAh g1) and thus cannot match the growing demand for high-capacity secondary batteries. Various types of ferrite-based composites had been studied which includes magnetic ferrites composed with nonmagnetic oxides and polymers. Transition metallic oxides and binary metal oxides are promising anode electrode materials for rechargeable lithium-ion batteries (LiBs). Their benefits encompass high theoretical capacities, protection-in-operation, environmental friendliness, mild expenses and so on. 1 –5 Nanoscaled spinel ferrites for LiBs were intensively developed as anode materials because of their novel Li-ion storage mechanism. The mechanism differs from the classical Li insertion/extraction and Li alloying/dealloying approaches. It entails the formation and decomposition of lithium oxide, accompanying the discount and oxidation of metal nanoparticles. 6 –12 The search for new advanced materials for the batteries in portable devices and power tools is endless. LiB serves as the best choice in the portable energy storage devices. 13,14 Abundant research is being done in search for electrode material with higher charge capacity and good recyclable nature. Among various nanostructured materials, transition metal oxides and mixed transition metal oxides have received much attention owing to their relatively higher charge–discharge capacity (i.e. 400–900 mAh g1) in LiBs with more beneficial safety concern for practical use. 15
Ferrites having the general formula MFe2O4 (M = transition metal ion, e.g. Ni, Co, Cu and so on) are one of the most attracting class of materials as electrode in LiBs for their intriguing properties such as various redox states, electrochemical stability, pseudo capacitive behaviour and so on. 7,16,17 Oxide ferrites such as NiFe2O4, CoFe2O4 and CuFe2O4 in powder are active anode materials in LiBs. 18 –21 But the poor electrical conductivity, severe volume expansion and high oxidation potential restricted their practical applications. Moreover, the drop in the reversible capacitance and poor cyclability are major drawbacks. 22 Recently, NuLi and Qin 7 reported that these materials deposited over a conducting substrate gives better electrochemical performance compared to powders.
Spray pyrolysis is a process for preparing particles or films by forming droplets from a precursor solution, then evaporating and decomposing them in a reactor. This process has proven to be quite useful for the preparation of multifunctional particles, with many reports into the effect of the main variables on particle formation. In recent studies, various structures and composites for electrode materials have been reported that are aimed at enhancing the electrochemical performance in LIBs by means of spray pyrolysis. 23
In the present work, thin films of NiFe2O4, CoFe2O4 and CuFe2O4 have been deposited by spray pyrolysis technique over conducting antimony doped tin oxide (ATO) coated over quartz to serve as anode material in LiBs. ATO substrate can be annealed at temperatures greater than 500°C, which is best suitable material for ferrites formation. ATO is of low cost and good crystallinity compared to other commercially used substrates such as indium tin oxide (ITO). The phase and surface morphology were studied using X-ray diffraction (XRD) and scanning electron microscope measurements (SEM), respectively. The magnetic property, film thickness and electrochemical studies are performed using vibrating sample magnetometer, XP-1 surface profiler and cyclic voltammetry (CV) to understand their response and suitability for use in LiBs.
Experimental procedure
Deposition of conducting ATO electrode over quartz substrate
In the deposition of thin films, substrate cleaning is very important. Commercially available quartz (2.5 cm × 2.5 cm × 1 mm) was used as substrate. The process used before deposition is that the substrates were first ultrasonically cleaned by de-ionised water and finally dried in air. Thin film was deposited using homemade spray pyrolysis where tin chloride and antimony chloride are dissolved in 350 ml of ethylene glycol, 150 ml of water and 5 ml of HCl is added to maintain the pH. The substrate was maintained at 400°C as measured by a K-type thermocouple. Compressed and moisture filtered air is used as the carrier gas and the substrate to nozzle distance is fixed at 35 cm. The precursor solution was sprayed onto the preheated substrate at a pressure of 40 kg/cm2 through a spray gun of inner nozzle diameter 0.3 mm. The spray time is fixed at 0.5 s with a 30-s interval between the sprays. The aerosol on reaching the hot substrate undergoes a pyrolytic decomposition to form clusters, which then develop into a continuous film in the presence of water as follows:
The prepared ATO substrates show a resistance value less than 40 Ω (5 mm × 5 mm) and are comparable with that of the commercially available ITO substrates (∼ 20 Ω).
Deposition of NiFe2O4, CoFe2O4 and CuFe2O4 films over ATO substrate
The solution decomposes pyrolytically and the reactions involved are as follows:
The deposited samples were annealed at 600°C for 2 h in air. The optimal deposition parameters for NiFe2O4, CoFe2O4 and CuFe2O4 films are given in Table 1.
Precursors and substrate temperature to deposit NiFe2O4, CoFe2O4 and CuFe2O4 films.
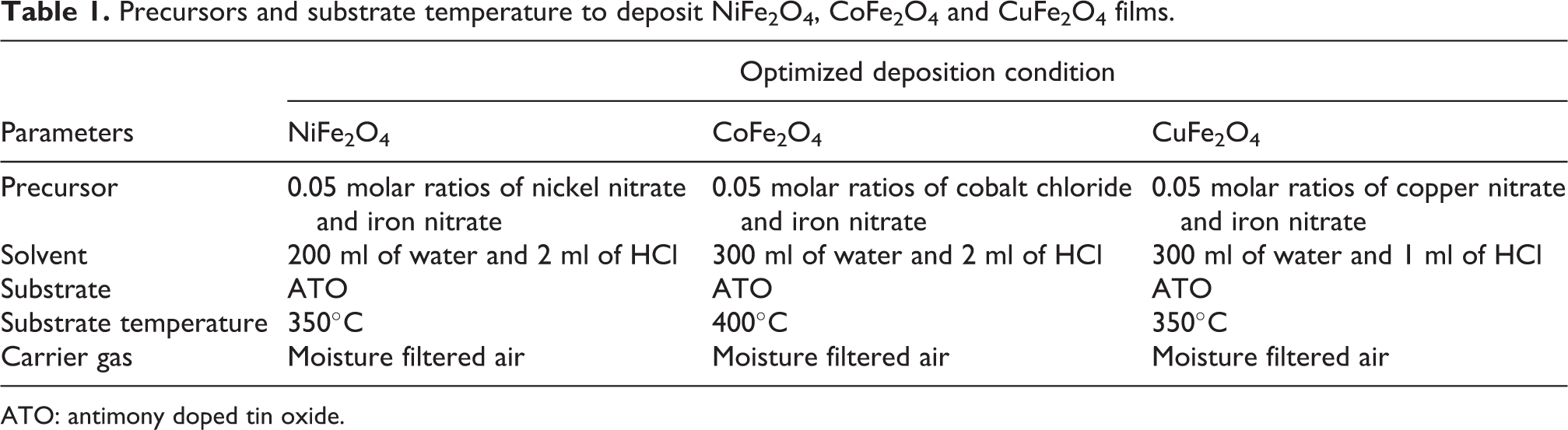
ATO: antimony doped tin oxide.
Characterization techniques
The phase of the deposited films was studied by XRD (INEL PSD MODEL) using Co–Kα (λ = 1.789 Å) radiation. The surface morphology of the films are analysed using SEM (JEOL, Japan JSM 840-A) and atomic force microscope (AFM) in dynamic mode (SPI 3800 N). Magnetic measurements were done in a vibrating sample magnetometer. The film thickness was estimated using a XP-1 Surface Profiler of Ambios technology, stylus profilometer Inc. Electrochemical performance of the thin films was carried out using CV (model: CHI Instrument-CHI1100A).
Results and discussion
Structural analysis
Figure 1 shows the XRD patterns of NiFe2O4, CoFe2O4 and CuFe2O4 films on ATO substrate. It is observed that in addition to ATO peaks, the other peaks in Figure 1(a) and (b) confirm the cubic phase formation of NiFe2O4 (JCPDS card no. 88-0287) and CoFe2O4 (JCPDS card no. 22-1086) with lattice parameters a = b = c = 8.309(0) Å and a = b = c = 8.203(3) Å, respectively. In Figure 1(c), apart from the ATO peaks, the other peaks indicate formation of tetragonal CuFe2O4 (JCPDS card no. 34-0425) with lattice parameters a = b = 5.842(5) Å and c = 8.682(1) Å. The average crystallite sizes, calculated using Scherrer’s formula, were 22 nm, 19 nm and 18 nm for NiFe2O4, CoFe2O4 and CuFe2O4, respectively.

XRD patterns of spray deposited and annealed (600°C/2 h in air). (a) NiFe2O4, (b) CoFe2O4 and (c) CuFe2O4 films on ATO substrate. XRD: X-ray diffraction; ATO: antimony doped tin oxide.
SEM analysis
The SEM images are shown in Figure 2. It can be seen that CoFe2O4 and CuFe2O4 films (Figure 2(b) and (c)) consist of relatively dense, compact and continuous morphology, whereas NiFe2O4 film (Figure 2a) is composed of spherical particles with pores in-between. The morphology of CoFe2O4 and CuFe2O4 films are uniform with few large sized particles. The average particle sizes were calculated as 32 nm, 26 nm and 28 nm for NiFe2O4, CoFe2O4 and CuFe2O4, respectively.

SEM images of (a) NiFe2O4, (b) CoFe2O4 and (c) CuFe2O4 films. SEM: scanning electron microscope.
AFM analysis
Figure 3 shows the AFM images of the films. The surface of NiFe2O4 (Figure 3(a)) has hillocks over the entire scanned region. The AFM images of CoFe2O4 and CuFe2O4 films (Figure 3(b) and (c)) are relatively smooth compared to that of NiFe2O4. The root mean square roughness values are 1.4 nm for NiFe2O4, 4.2 nm for CoFe2O4 and 20 nm for CuFe2O4 films.

AFM images of (a) NiFe2O4, (b) CoFe2O4 and (c) CuFe2O4 films. AFM: atomic force microscope.
Magnetic properties
The magnetization result at room temperature is shown in Figure 4. All the samples show prominent hysteresis loop due to ferrimagnetic behaviour. NiFe2O4 and CuFe2O4 films show soft magnetic property with the coercivities (HC) of 281 and 562 Oe and saturation magnetization (MS) values are 0.005 in emu/g and 0.0066 in emu/g, respectively. CoFe2O4 is one among the very few ‘hard’ ferrites. The magnetization result of CoFe2O4 shows high HC of 770 Oe and low magnetization value of 0.0012 emu compared to soft magnetic NiFe2O4 and CuFe2O4 films.

Magnetization curves of NiFe2O4, CoFe2O4 and CuFe2O4 thin.
CV analysis
Cyclic voltammetry is one of the most versatile electroanalytical techniques for the study of electroactive species. The aim of this study is to test the electrochemical performance of the spray deposited NiFe2O4, CoFe2O4 and CuFe2O4 thin films. The conventional three electrode cell was used with NiFe2O4, CoFe2O4 and CuFe2O4 thin films as working electrodes. The spectrum was recorded between the range −1.2 V and +1.2 V at a potential sweep rate of about 0.1 V/s in the electrolyte, 0.01M LiClO4 + propylene carbonate (Figure 5). A platinum wire and saturated calomel electrode were used as counter and reference electrodes to study the electrochemical performance of the films. During the negative half cycle, Li+ ions gets intercalated into the film from the electrolyte causing an oxidation reaction (colored state). For CuFe2O4, an anodic peak appeared at 120 mV in the voltammogram while for NiFe2O4 peak occurs at 680 mV. Voltammogram for all the films are different from the other, that is, magnitude of the peak current is different for all films. This may be because of the difference in mobility of the charged particles inside the films. The porous NiFe2O4 film has larger loop area with slight asymmetry; however, the smaller value of current observed in the loops suggests a non-faradaic process. 24 The electrochemical measurement reveals the films are stable, and no peeling off of films was observed during 50 cycles. The diffusion coefficient was determined from the Randles-Sevcik equation and was found to be 0.0004 cm2/s. The charging and discharging property measured from the electrochemical study concludes that the NiFe2O4 and CuFe2O4 ferrite thin films could be used as promising anode material for LiBs.
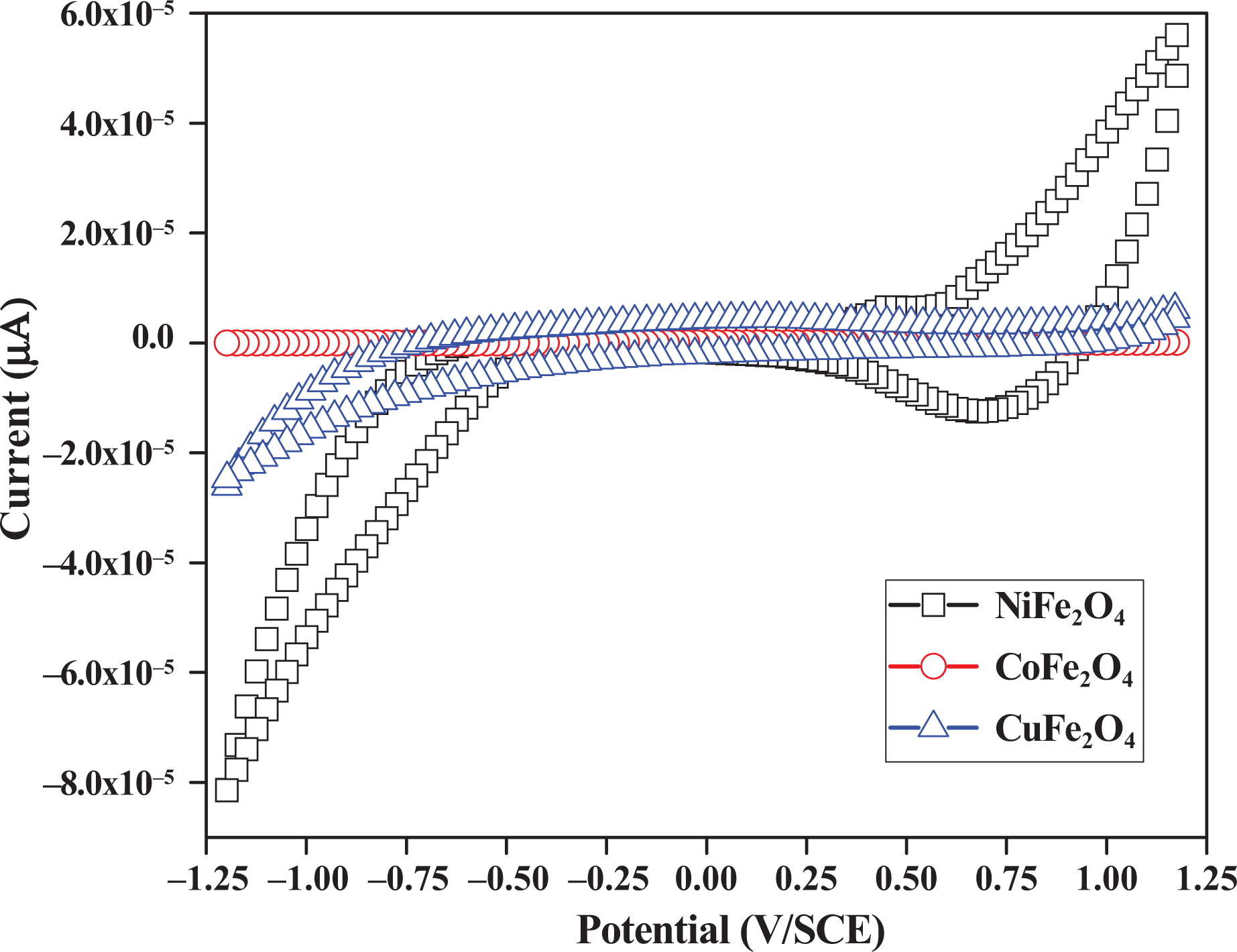
Cyclic voltammograms of NiFe2O4, CoFe2O4 and CuFe2O4 thin films.
Conclusion
Nanocrystalline NiFe2O4, CoFe2O4 and CuFe2O4 ferrite thin film was deposited over the ATO substrate by spray pyrolysis. The XRD result confirm single phase without any evidence of secondary phases. The SEM and AFM study reveal that the NiFe2O4 film surface consists of spherical particles with porous nature when compared to CoFe2O4 and CuFe2O4. The magnetization result clearly shows hysteresis behaviour due to ferrimagnetic nature. The electrochemical data implies that the NiFe2O4 and CuFe2O4 ferrite thin films can be used as promising anode material in LiBs, when compared to CoFe2O4. Further optimization of NiFe2O4 and CuFe2O4 film may lead to the development of low-cost and high-efficient anode material for LiB.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.