Abstract
COVID-19 has lasting impacts on pharmaceutical companies, and it is crucial for managers to fully understand and monitor the impact areas to prepare for future organizational resilience building and transformation. However, the research on COVID-19 impacts is still discrete and anecdotal. Based on a textual analysis of 447 public pharmaceutical companies’ 10-K annual reports and statistical analysis of their financial data, this research systematically identifies major impact areas experienced by public pharmaceutical companies during COVID-19, the patterns of impacts (distribution and sentiment of impacts), and their association with firm characteristics. A topic modeling analysis reveals four impact areas, including new product development processes, sales and operations, COVID-19 treatment and prevention drug early development, and COVID-19 drug clinical trials. Companies reporting COVID-19 drug clinical trials are the most optimistic in the sample, while companies reporting sales and operations are the most pessimistic. Furthermore, firms exhibit heterogeneity in terms of the impacts they experience. Those whose primary business focus is on research and development are more likely to report impacts related to new product development processes, while those with diverse business focus tend to highlight issues on sales and operations. The impact areas uncovered in this research point out the domain for managers to watch for potential transformation, whereas the findings on firm heterogeneity guide managers to make efforts tailored to their firm characteristics.
Introduction
The COVID-19 pandemic has exerted a profound impact on societies and organizations, prompting continuous monitoring and assessment of its consequences for necessary business responses and transformation. 1 These effects vary across industries, necessitating industry-specific investigations. 2 This study explores the intricate impacts of COVID-19 on the pharmaceutical sector and the industry’s value chain activities, including drug discovery, development, manufacturing, and sales. 3 The sector witnessed both benefits and challenges during the pandemic, engaging in expedited launches of new COVID-related drugs while grappling with global supply chain disruptions, shifts in drug demand, and disrupted clinical trial activities.4,5
These intricate impacts and their aggregated implications for pharmaceutical businesses during the COVID-19 pandemic have not undergone systematic review and study, creating a gap in firm-level insights into the sector under pandemic disruption. Existing research on COVID-19 impacts focuses primarily on individual-level analysis. For instance, infoveillance studies concentrating on individual opinions and responses mine data from social media platforms such as Weibo and Twitter to discern top concerns related to COVID-19, 6 public attention to COVID-19 medications, 7 and emotional distress during COVID-19. 8 The limited scholarly research on the impact of COVID-19 on pharmaceutical companies centers on supply chain disruptions, 9 while discussions of other impacts remain anecdotal, primarily derived from industry reports and news articles.
Filling this void, this research aims to systematically investigate the business impacts of COVID-19 on pharmaceutical companies by examining firm annual reports and financial data. Key research questions include: (1) major impact areas narrated in firm annual reports; (2) patterns of firm impacts, considering the concentration and sentiment of impact areas; and (3) impacts on firms of various characteristics.
Analyzing 447 public pharmaceutical companies’ 10-K annual reports between April 2020 to March 2021, we employed topic modeling to identify major impact areas during COVID-19. Statistical analyses, including Tobit regressions and logistic regressions, were conducted to explore the association between company characteristics and specific impacts.
The topic modeling analysis revealed four common themes associated with COVID-19 in pharmaceutical firms’ annual reports: new product development processes, sales and operations, COVID-19 treatment and prevention drug early development, and COVID-19 drug clinical trials. Each of these topics represents a significant area of impact on pharmaceutical companies by the pandemic. A close examination of these topics revealed predominantly adverse effects, particularly in activities involving in-person interactions, such as new product development processes, clinical trials, and sales and operations. Notably, the disruption to planned new product development emerged as a common and severe issue within the industry. Sales and profit suffered a general reduction, although there were exceptions. The study further uncovered variations in the impacts across pharmaceutical companies, with larger firms reporting lower negative effects. Companies with a primary focus on research and development were more likely to report impacts related to new product processes, while those with diverse business focus tended to highlight issues on sales and operations.
This research contributes to the literature that studies the impact of COVID-19 pharmaceutical businesses by filling a gap in firm-level insights. It provides a comprehensive analysis of the pandemic’s effects on pharmaceutical companies using empirical data from 447 public firms’ 10-K annual reports, while prior studies primarily discuss specific impact areas of COVID-19 on pharmaceutical firms, such as supply chain disruption or clinical trial activities. From a methodological perspective, this research uses advanced machine learning, statistical analysis, and the 10-K reports as the dataset to provide a rigorous analysis of the impacts, adding to the scientific rigor compared to prior research that primarily relies on case studies for firm-level research. Furthermore, the analysis of differential impacts based on firm financial characteristics also contributes to the understanding of firm resilience during COVID-19. The study reveals that firms with more assets and financial leverage reported more areas of impact, but only those with high assets showed strong resilience to disruptions. Besides, larger firms experienced fewer negative effects and were more salient to COVID-19 disruptions. These insights offer key learnings for pharmaceutical business managers and prepare them to strategically navigate potential risks and opportunities amid business disruptions.
Literature review
The COVID-19 pandemic, characterized by its large-scale and highly disruptive nature, has significantly affected various aspects of businesses. A growing body of research has delved into understanding the intricate impacts of this adverse event, aiming to prepare businesses for future disruptive changes. This research stream has explored the repercussions of COVID-19 on different facets of business strategies and operations, encompassing human resources and employee well-being, 10 marketing, 11 consumer behavior,12,13 supply chain management,14,15 and competition. 16 Notably, industries such as education, 17 tourism, 18 hospitality, 19 and retail 13 have been focal points of this stream of research.
The impacts of COVID-19 are inherently complex and vary across industries, partly based on their reliance on in-person interactions and their resources to cope with the adverse effects. Different sectors experienced distinct sales impacts. 20 For instance, while Californian companies faced an average sales loss of 17% in the second quarter of 2020 compared to 2019, the e-tailing industry witnessed a remarkable 180% growth. 2 Moreover, small businesses, due to the liability of newness, were disproportionately affected. A survey 21 of over 5800 small businesses indicated that the majority struggled with cash shortages, with less than 1 month of cash on hand at the onset of the pandemic.
Surprisingly, there has been a dearth of research efforts to aggregate the firm-level impacts of COVID-19 on the pharmaceutical industry, a sector at the forefront of combating the adverse effects of the pandemic on society. The impact of COVID-19 on pharmaceutical supply chains has been among the most studied. 9 For example, Tirivangani et al. 9 interviewed 21 informants in the public and private pharmaceutical sectors of Namibia and summarized several supply chain issues, such as access and availability of essential medicines, long lead time, bureaucratic bottlenecks and lack of emergency readiness of the medicine’s logistics supply chain. Latonen et al. 22 studied the impact of COVID-19 on the pharmaceutical supply chain and how stakeholders joined forces to combat the crisis. Combining qualitative and survey data, Aivazi et al. 23 investigated vulnerabilities that predispose a pharmaceutical firm supply chain to disruption and the capabilities that empower a firm to sustain its supply chain functionalities during COVID-19. Additionally, some scholars have highlighted the significant disruption of clinical trial activities by COVID-19, 4 which has also spurred opportunities for innovative clinical trial practices, including distributed large-scale trials, 5 the use of observational data over randomized trials, and the promotion of collaborative structures. 24
To date, the existing literature has focused on a specific area of COVID-19 impact. A systematic understanding of the major impact areas of the COVID-19 outbreak and their variations across pharmaceutical companies is lacking. The research methodologies are primarily case or survey studies and there is an insufficient use of 10-K reports as a source of data to understand the impact. 10-K reports are widely accepted as an authoritative data source to study firm performance and behavior. 10-K reports during COVID-19 contain valuable, objective information on how COVID-19 impacted various aspects of pharmaceutical firms.25,26 This research aims to provide an in-depth and comprehensive understanding of the impact of the COVID-19 outbreak on pharmaceutical firms.
Part I study: A topic modeling analysis
This research employs a topic modeling approach to identify the principal impact areas of COVID-19 as outlined in pharmaceutical companies’ annual reports. Figure 1 provides an overview of the key steps in data collection and analysis, including data collection, pre-processing, visualization, and result analysis. Data processing flow.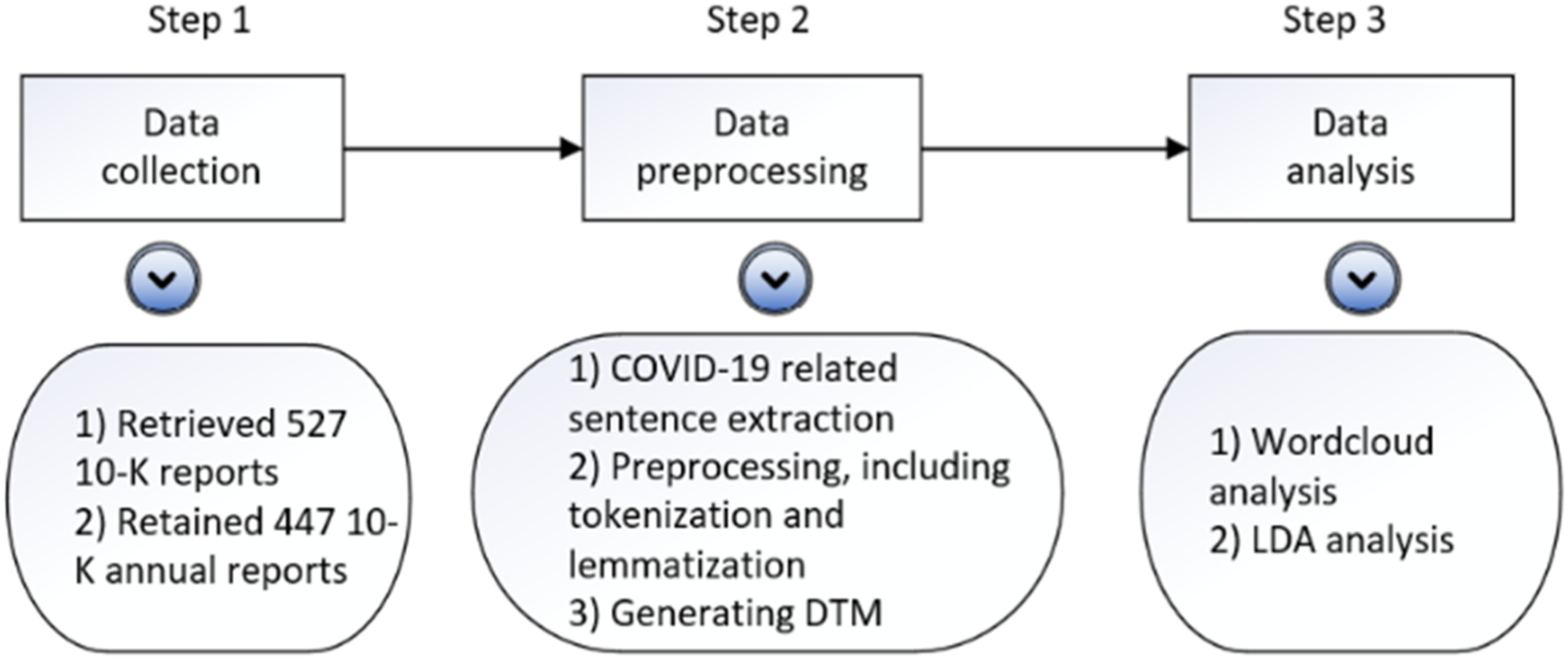
Data collection
We collected Form 10-K annual reports of U.S.-listed pharmaceutical companies from April 2020 to March 2021, a period significantly impacted by the COVID-19 pandemic. The Form 10-K is mandated by the U.S. Securities and Exchange Commission for public companies to disclose comprehensive financial performance and operations details. Widely recognized as a rich source of corporate operating information, 27 the Form 10-K provides more extensive insights compared to other types of company annual reports.28–30 Prior research has extensively used annual reports to investigate topics such as investor trading, 31 company reputation, 32 financial forecasting, 28 and business performance.30,33 Although annual reports have been valuable in understanding various aspects of business, their potential for exploring the business impact of COVID-19 remains untapped.
Our focus narrowed down to companies with SIC Code 2834, designated as Pharmaceutical Preparations [https://www.osha.gov/sic-manual/2834], indicating pharmaceutical companies. SIC, or Standard Industrial Classification, is a classification framework established by the U.S. government to define an organization’s primary business scope [https://siccode.com/]. A total of 527 U.S. pharmaceutical companies’ annual reports were initially retrieved. However, some reports lacked discussions on COVID-19, leading to their exclusion from the analysis. Consequently, our final sample comprised 447 public pharmaceutical companies.
Data preprocessing
Data preprocessing involves cleaning and transforming textual data into machine-friendly formats using natural language processing techniques. 34 To ensure high-quality results, machine learning methods typically necessitate processes such as tokenization, lemmatization, and labeling of textual data. 35 In this study, we utilized NLTK [https://www.nltk.org], a natural language processing toolkit in Python, for data cleaning and transformation.
Initially, for each report, we extracted sentences containing the keyword “COVID-19” through string matching. Subsequently, the text collection, comprising these filtered sentences, underwent several preprocessing operations, including: (1) converting all words to lowercase letters; (2) removing punctuation marks and common stop words; (3) eliminating meaningless words of certain lexical nature, such as quantifiers, prepositions, and conjunctions; (4) standardizing common equivalents and extracting roots for word form reduction to mitigate the sparsity of the document-term matrix (DTM) caused by repeated words with the same meaning (e.g., reducing plural forms to singular, transforming other tense participles of verbs to the general present tense); (5) constructing a collection of two-gram words to retain two-gram words with multiple occurrences (e.g., the common term “clinical trial” in medicine). The data cleaning and transformation result in a DTM for the subsequent topic modeling analysis.
LDA model
We applied the Latent Dirichlet Allocation (LDA) model to analyze sentences containing “COVID-19” in the selected annual reports. Based on Bayesian probability theory, LDA utilizes the probability of co-occurring words in the document corpus to identify topic distribution and its associated word distribution. It is highly effective in analyzing large document collections, organizing them based on identified topics, 36 and has found widespread application in various research domains such as literature reviews,37,38 user-generated content,39–41 and online reviews. 42
LDA assumes that the entire corpus of document precursors comprises a certain number of topics, and these documents exhibit a mixture of these topics. Each topic is conceptualized as “an infinite mixture over an underlying set of topic probabilities” 43 (p.993). By specifying the number of topics, LDA generates information about each topic in the entire corpus, including words closely associated with each topic and the probability distribution of topics for each document. LDA posits that words have different probabilities in different topics, generating the probability of word occurrence under each topic. This helps identify words closely related to each topic. In addition to word probabilities, the assigned topic probabilities for each document provide a clear representation of a document, aiding in understanding how each document is represented across different topics.
Perplexity and coherence scores serve as metrics to determine the optimal number of topics.43,44 Generally, the preferred number of topics (K) is the one with the smallest perplexity value and the highest coherence score among various LDA models with different Ks. Perplexity indicates the uncertainty of the topic to which a document belongs, with a smaller value suggesting higher prediction accuracy. 43 Topic coherence measures the interpretability of potential topics. A topic is considered easily interpretable when the top words in that topic co-occur more frequently in documents of the corresponding corpus, and a larger coherence score indicates a better model. 45
Data analysis results
Intensity of impacts
The frequency of COVID-19 keywords can be indicative of the intensity of the pandemic’s impact on pharmaceutical companies. Among the 447 10-K annual reports in the pharmaceutical preparation industry category, 84.8% contain the keyword “covid”. Figure 2 depicts the histogram of the keyword “covid,” revealing that over 80% of the reports mention it more than 20 times. This high frequency of “covid” mentions in annual reports suggests the substantial impacts of the pandemic on pharmaceutical companies. Histogram of the keyword “covid” mentioning.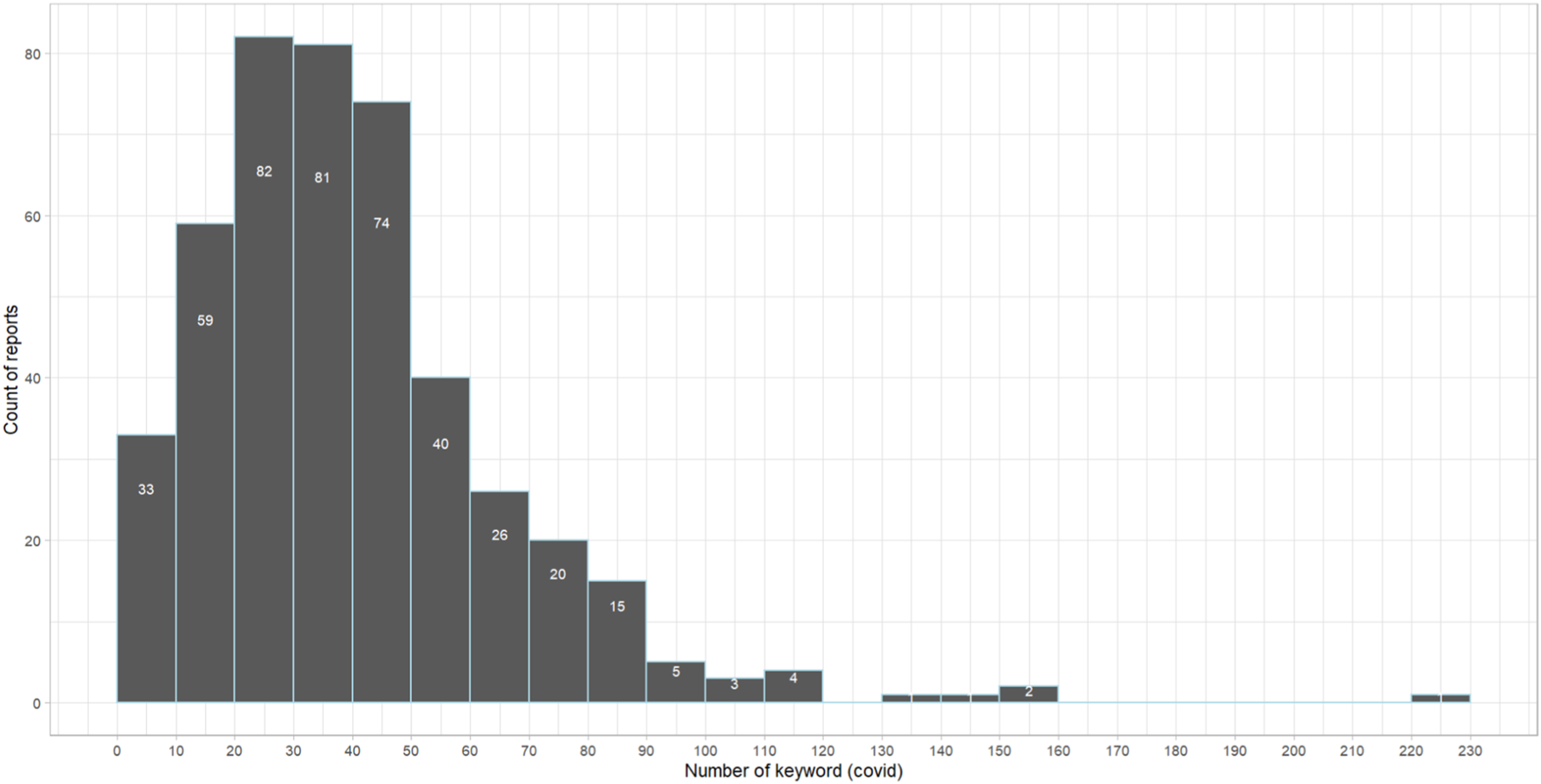
Impact of COVID-19: Overview through high-frequency keywords
To identify key topics related to COVID-19, we visualize the frequently appearing words in the annual reports of the sampled companies through a word cloud diagram, as shown in Figure 3. The two most prominent words in the diagram are “product candidate” and “clinical trial”, followed by “clinical development”, “trial site”, and “clinical study”, which are commonly associated with product development activities of pharmaceutical companies. This suggests that the drug development process is likely the most affected by the COVID-19 pandemic. Additionally, negative terms such as “adversely impact”, “adversely affect”, and “highly uncertain” are frequently appearing, indicating that pharmaceutical companies are primarily concerned with the negative impacts of the COVID-19 pandemic. The word cloud of 10-K reports.
LDA analysis results
LDA analysis enables us to identify the impact areas caused by COVID-19. The initial step is to determine the optimal number of topics (K). Figures 4(a) and (b) display the number of topics and the corresponding model perplexity and coherence scores, respectively. Both suggest that the most suitable LDA model has 4 topics, as it boasts the lowest perplexity score and a relatively high coherence score, with minimal differences in scores beyond that point. Therefore, we select the LDA model with 4 topics. Perplexity and coherence scores of the candidate models. (a) Perplexity scores for the candidate models. (b) Coherence scores for the candidate models.
Topics identified from 10k reports.

aNumber of companies whose topic probability of a specific topic (i.e., topic n) is the highest among all four topic probabilities; also called the topic n companies hereafter (n is the topic number under consideration).
bThe average topic n probability of the topic n companies.
cThe ratio of the number of negative words over that of positive words (neutral words are excluded from the calculation) for the topic n companies.
Topic 1: New product development process
This topic contains keywords related to new product development, a pivotal aspect of pharmaceutical companies’ operations. Particularly, a large portion of companies have expressed concerns about the disruption and delay of clinical trials for new products caused by the COVID-19 outbreak. Johnson & Johnson, in its annual report, highlighted that “due to the ongoing impacts of the COVID-19 pandemic, certain trials have been rescheduled or delayed.” Similarly, Biomarin Pharmaceutical Inc. acknowledged that “the continued spread of COVID-19 has adversely affected and will likely continue to adversely impact our product development programs, including preclinical study and clinical trial operations.” The primary reasons cited for the delayed or disrupted clinical trials include challenges in patient identification and recruitment, uncertainties related to product commercialization, and delays in obtaining materials from third-party suppliers. For instance, Aprea Therapeutics, Inc. noted difficulties in locating and enrolling patients due to the pandemic, interference with patient trials caused by the COVID-19 virus, slowed FDA review of trial applications, and the inability of third-party suppliers to manufacture drugs for trials.
This impact area is widely discussed, with 323 out of the total 447 companies in our sample mentioning it. Its average topic probability is 0.842, indicating a high concentration of the topic discussion among all topics for these companies. The impact is notably adverse, with a negative sentiment ratio of 0.752, the second-highest among all sentiment ratios.
Topic 2: Sales and operations
This topic focuses on keywords related to the sales and business operations of pharmaceutical companies, such as “sales”, “impact”, “product”, “revenue”, “demand” and “operations”. Business operations, such as sales, manufacturing, supply chain and collaboration, are adversely affected. Sales can come down to an abnormally low level due to the demand and economic change. For example, China Pharma Holdings Inc. which is traded on Nasdaq, noted negative impacts on assay sales attributed to the suspension of commercial activities, staff quarantine, and a decline in primary hospital visits during COVID-19. Prestige Consumer Healthcare Inc. reported decreased revenues from pharmaceutical product sales due to reduced general consumer illness and exercise following social quarantine measures.
However, exceptions were observed, particularly for segments producing products addressing COVID-19 symptoms, leading to increased revenue or reduced operational costs in specific cases. Johnson & Johnson’s Tylenol, for instance, experienced elevated sales revenue during the pandemic due to increased demand and shortages of competitor products. Additionally, Pieris Pharmaceuticals, Inc. reduced spending by $2 million on the respiratory system program, as increased patient enrollment in clinical trials resulted in lower costs for related chemical component manufacturing and control products.
Out of the 447 companies in our sample, a total of 93 discussed the impact on sales and operations as their top concern. These companies exhibited an average topic probability of 0.744, indicating a concentrated focus on this topic over others. Moreover, the negative sentiment ratio of 0.833 was the highest among all four groups of companies, suggesting a significant adverse impact.
Topic 3: COVID-19 treatment and prevention drug clinical trials
This topic revolves around keywords related to clinical trial activities specific to COVID-19 treatment and prevention, such as “respiratory”, “vaccine”, “ARDS” (“Acute respiratory distress syndrome), and “APL-9”. Enanta Pharmaceuticals Inc., due to its expertise in developing respiratory virus treatment, mentioned that “we announced our newest discovery program for the treatment of COVID-19, we have been leveraging our expertise in direct-acting antiviral mechanisms to discover new compounds to treat COVID-19, using a combination of screening and targeted drug design”. Additionally, the combined words, such as “patient” and “phase”, often appear, indicating that the COVID-19 treatments for the companies in this group have progressed to the clinical trials stage involving human subjects rather than animals (i.e., Phase 3 trial).
In our sample of 447 companies, only 11 discussed COVID-19 treatment and prevention drug clinical trials as their most prominent impact area. The negative sentiment ratio for these discussions is the lowest among all four groups (0.580), indicating the least adverse impacts of the COVID-19 pandemic on these companies, possibly due to their profit prospects of engaging in COVID-19 treatment and prevention medications. The average topic probability of this topic for the 11 companies is the lowest among the four categories (i.e., 0.532), suggesting that these companies are also impacted by COVID-19 in other areas.
Topic 4: COVID-19 treatment and prevention drug early development
This topic centers around keywords related to Coronavirus disease drug development, such as “test”, “develop”, “antibody”, “treatment” and “SARS-CoV-2”. Some keywords overlap with those in the topic of COVID-19 treatment and prevention drug clinical trials, but compared to those, this topic reflects a more focused view on drug development in early, pre-clinical stages rather than clinical trials (Phase 1 and 2 trials), and the drugs seem to target patients with less severe syndromes as “COVID-19 has three distinct phases: early infection, pulmonary/inflammatory and hyperinflammatory.” Companies leverage their expertise to develop such drugs. For example, Humanigen, Inc mentioned that their COVID-19 treatment drug targeting GM-CSF brought uncertainty to the company since “the scientific rationale behind the hypothesis that GM-CSF is a cause of the cytokine storm that leads to adverse results in COVID-19 patients is still being tested and may not prove accurate”. Another example is Immunic Inc, which is also “investigating IMU-838 as a potential treatment option for coronavirus disease 2019 (COVID-19)” and is in “Phase 2 Trial of IMU-838 in Moderate COVID-19”.
In our sample, 20 companies discussed this topic as their top impact area. However, they are also likely to be impacted in other areas, as reflected by their low average topic probability of 0.527. The sentiment of these companies is modest (0.655), indicating a more positive prospect in engaging with respiratory disease drug development during the pandemic.
In sum, the topic modeling analysis of the annual reports indicates that the COVID-19 pandemic brought forth both challenges and opportunities for pharmaceutical companies. The majority of companies experienced adverse effects, particularly in the areas of new product development or sales and operations. However, a small portion of companies engaged in the sale or development of COVID-19 and respiratory disease treatment and prevention drugs tended to report more positive impacts.
Part II study: The association of firm characteristics and COVID-19 impacts
Pharmaceutical companies are affected by COVID-19 disruption differently. Consequently, we conduct the statistical regression analysis to investigate the associations between firm characteristics and the varied impacts experienced by pharmaceutical companies. Particularly, we seek to identify the associations of specific company characteristics with: (1) the likelihood of a pharmaceutical company experiencing and reporting an impact area; (2) the diversity of the topic distribution; and (3) negative sentiment of firms in reporting their impact.
Five categories of company characteristics are evaluated: company size, performance, risks, future development opportunities, and financial market features such as liquidity. By answering these questions, the study aims to discern the reasons behind the heterogeneity of COVID-19 impacts on pharmaceutical companies and identify the types of firms that may be less resilient to these impacts.
Methodology
Variables and measurements
For research question 1, i.e., the associations of firm characteristics with the likelihood of a pharmaceutical company experiencing an impact area, the dependent variables are PROB_TOPICs 1–4 in Table 1. These variables represent the likelihood of a pharmaceutical company being impacted in a specific area, as measured by topic probabilities derived from the LDA analysis.
For research question 2, i.e., the associations of firm characteristics with the diversity of the topic distribution, the dependent variable is measured by VARIETY. VARIETY is calculated as the number of topics with a probability higher than zero. To ensure the robustness of the results, an alternative measurement of variety, Blau’s index, is also used. Blau’s index is a common metric for diversity and heterogeneity. Its formula is given by:
Independent variables.

Size
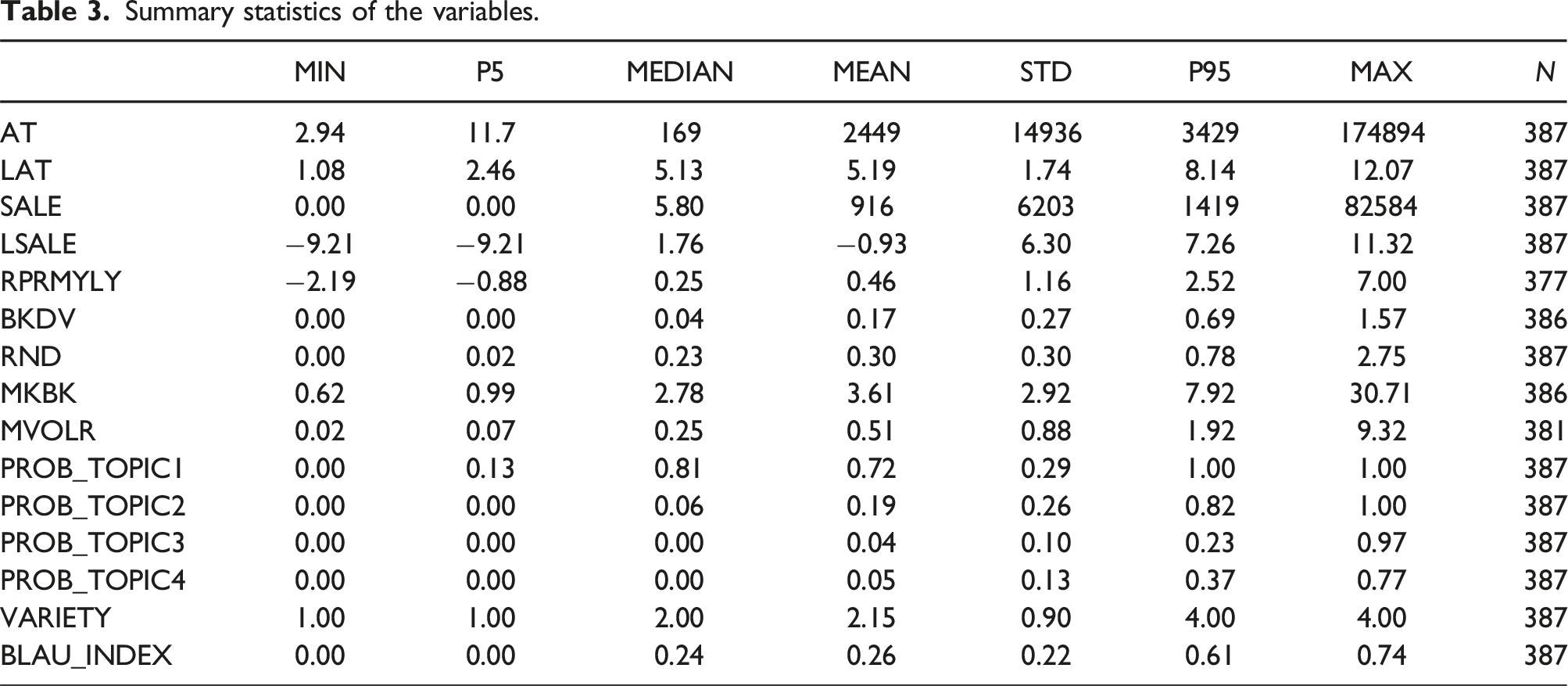
Size is one of the most important characteristics of a company. Larger companies often have the advantage of generating profits from a diverse portfolio of business activities, while smaller companies tend to be more focused on a limited set of activities. Larger companies generally demonstrate greater resilience to economic shocks, such as COVID-19, owing to redundant resources and a diversified business portfolio that can help mitigate or absorb risks. In this study, we use two measures of company size: total assets (AT) and sales (SALE). Due to the huge contrast of values between the topic probabilities and AT and SALE, the logarithms of the assets (LAT) and sales (LSALE) are used due to the significant contrast in values between the company size measures and the dependent variables.
Overall performance
A company with high overall performance typically has a better capability to manage and absorb market risks while maintaining high economic profit, thus can be more resilient to shocks caused by events like COVID-19. In this study, we use risk-adjusted return per month, year to last year (RPRMYLY) as a measure of a company’s market performance. RPRMYLY is the annualized risk-adjusted return (RET), obtained by compounding the monthly risk premium into an annualized return. A high RPRMYLY demonstrates superior market performance, indicating a firm’s capability to effectively manage risks without compromising overall performance.
Financial risk
A firm’s financial risk is closely tied to its ability to mobilize monetary resources when confronting disruptive events. Companies with high financial risks often exhibit lower resilience to external shocks, primarily due to the constraints imposed by financial limitations. These constraints hinder the company’s capacity to allocate financial resources effectively in response to disruptive events. The measure of financial risk employed in this study is the book value debt over book value asset (BKDV) ratio, calculated as total liabilities over total assets. This ratio provides insights into the proportion of a company’s assets financed by debt, serving as a crucial indicator of its financial risk profile.
Future development opportunity
A company’s future growth opportunities may negatively influence its resilience to external shocks, as its potential for monetization is yet to be fully realized. This study utilizes two metrics to assess a firm’s future development opportunities: the research and development (R&D) ratio and the market-to-book value ratio (MKBK). The R&D ratio is measured as the ratio of a firm’s R&D expenses to its total assets. A higher R&D ratio indicates a greater allocation of resources to R&D, suggesting a heightened potential for future development opportunities. However, companies heavily reliant on R&D for growth may be more vulnerable to external shocks if disruptions occur in R&D activities. Additionally, MKBK compares a company’s market value (market capitalization) to its book value (the value of its assets as recorded on the balance sheet). A high market-to-book value ratio suggests that the market values the company’s potential for future growth, while a lower ratio may indicate a more conservative outlook.
Financial market liquidity
High liquidity in the financial market indicates the attention that a firm attracts and its potential ability to acquire funds. Such a firm may have an advantage in mobilizing financial resources to mitigate the adverse effects of disruptive events. We employ the monthly share turnover of stock, also called MVOLR (monthly trading volume ratio), to measure a firm’s financial market liquidity. It is calculated by dividing the monthly trading volume (VOL) by the average monthly shares outstanding of a firm’s stock. The higher the share turnover, the more liquid the firm’s shares are.
Control variables
We include subindustries as a control variable to account for their fixed effect. The subindustry for each company is obtained using S&P Global Industry Classification Standard (GSECTOR) code. GSECTOR is a six-digit industry classification that is more granular than SIC CODE. It provides codes for multiple subindustries within the SIC 2834 industry (Pharmaceutical Preparations). Our sample includes three subindustries: Biotechnology (352010), Pharmaceuticals (352020), and others. There are 110 firms in 352020, 259 firms in 352010, and 18 other firms.
Sample
The data for company operations were obtained from two major databases: the Center for Research in Security Prices (CRSP) and COMPUSTAT North America (CSTT). CRSP provides monthly stock market data, including variables such as RET and trading volume (VOL). CSTT provides annual company data, such as total assets (AT), sales (SALE), net income (NI), and others. These data are either directly used in regression analysis or indirectly used in the calculation of the independent variables.
Matching companies present in both CRSP and CSTT in the fiscal year 2020 (April 2020 to March 2021), with historical and current SIC CODEs equal to the sample used for the LDA analysis, resulted in a sample of 387 unique firm-year observations. Some samples were lost due to: (1) status changes of companies (e.g., delisting or merging), or lack of data in the database; (2) the mismatching of time periods in the data obtained from different sources.
Summary statistics of the variables.
Correlations of the variables.

Model specification
Topic probability and sentiment
We used Tobit regression to investigate the influence of company characteristics on the probability of a firm being assigned to a specific topic and the sentiment expressed in its reporting (i.e., research questions 1 and 3). Tobit regression, also known as a censored regression model, is a statistical tool that can be useful for analyzing data with censored dependent variables.
46
Given that the dependent variables—namely, the probability of the four topics—are constrained between 0 and 1, we employed a Tobit regression with the following model: • Yi represents PROB_TOPIC1 to PROB_TOPIC4 and NegSentiRatio, • X1i denotes the size measure, either LAT or LSALE, • X2i is the performance measure, RPRMYLY, • X3i is the financial risk measure, BKDV, • X4i represents the future development opportunities measure, either RND or MKBK, • X5i is the market liquidity measure, MVOLR, • 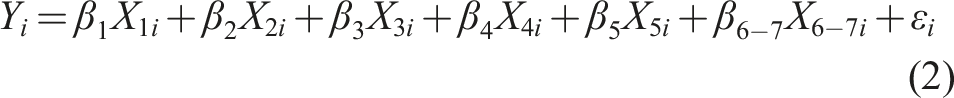
Diversity of topic distribution
We use a logistic model to examine the impact of firm characteristics on the diversity of impact areas during the pandemic. Specifically, we run nominal logistic regression of VARIETY on the aforementioned explanatory variables to identify which firm characteristic can predict a firm’s diversity of impact areas. In this context, a value of 1 for VARIETY indicates that the firm only experiences one impact area, while a value of 4 indicates that the firm reports all four impact areas. We selected VARIETY = 1 as the base category in the nominal logistic regression, and the subsequent three logistic regressions explored the change in likelihood from the base category (VARIETY = 1) to the other three categories of VARIETY: • • X
j
includes each of the five independent variables discussed above.
A significant regression parameter
To ensure the robustness of the results associated with the diversity of topic distribution, we re-run the above analysis using model (2) with Blau’s index as the dependent variable.
Data analysis results
Topic probability
Tobit regression of topic probabilities on firm-specific characteristics.

Topic 1: New product development process
The results for the probability of Topic 1, i.e., new product development processes, reveal several key insights. Notably, the risk-adjusted return (RPRMLY) and future development opportunities, measured by R&D expenditure (RND), demonstrate positive and significant effects on the probability of firms reporting issues related to new product development. On the other hand, firm size (both LAT and LSALE), financial risk (BKDV), and financial market liquidity (MVOLR) exhibit negative and significant effects. For instance, as illustrated in Model 1 of Table 4, a 1% increase in the R&D ratio is associated with a 0.17% higher probability of a firm reporting challenges in new product development. Conversely, a 1% higher trading turnover (MVOLR) correlates with a 0.11% lower probability of experiencing similar issues. Interestingly, the effect of MKBK is deemed insignificant in this context.
These findings suggest that firms of larger size, lower financial constraints and risks, and higher liquidity in the stock market, are less likely to report COVID-19 impacts on new product development. This is probably because such companies are established and possess diverse product portfolios, making the development of a specific new product less critical for their survival. On the other hand, firms with higher risk-adjusted returns and a focus on R&D are more likely to report COVID-19 impacts on new product development. These companies are often driven by new drug development, where the successful creation of multiple drugs is crucial for their survival.
Topic 2: Sales and operations
Models 5–8 in Table 5 present the TOBIT regression results with PROB_TOPIC2 as the dependent variable. The findings suggest that financial risk (BKDV), size (both LAT and LSALE), and financial market liquidity (MVOLR) have significant positive associations with the probability of firms discussing COVID-19 impacts on sales and operation. This implies that larger firms, those with high financial risk and constraints but high leverage, and those with high liquidity in the stock market are more likely to be impacted in sales and operations. Meanwhile, performance (RPRMYLY) and R&D ratio (RND) show negative associations with Topic 2 probability, indicating that firms with high market performance and future development opportunities are less likely to report Topic 2 impact areas.
In summary, larger firms that are mature and well-established, where sales and operations are crucial to their survival, are more concerned about disruptions in these areas due to COVID-19. Conversely, firms with higher risk-adjusted returns or higher R&D ratios may prioritize new product development, and sales and operations are of lesser concern for them.
Topics 3 and 4: COVID-19 treatment and prevention drug early development and clinical trial
Models 9–12 in Table 5 present regression results for the probabilities of Topics 3 and 4. The results show that none of the variables are consistently and significantly related to topic 3 and 4 probabilities. Business engagement with COVID-19 treatment and prevention-related drug development does not appear to be particularly tied to firm characteristics of pharmaceutical companies in our study.
Diversity of topic distribution
Logistic regression results for VARIETY.

Sentiments of reporting
Tobit regression results for NegSentiRatio.

Discussions
This research studies the major business impacts experienced and reported by public pharmaceutical companies in North America. Analyzing textual data extracted from 447 companies’ annual reports, this study identifies four major areas of COVID-19 impacts on pharmaceutical companies: new product development processes, sales and operations, COVID-19 treatment and prevention drug clinical trials, and early development. The findings affirm the observation from the medical field that clinical trials and new drug development processes are the main areas demanding attention from managers. 5 Moreover, it identifies additional impact areas. Some companies expressed concerns about decreased product demand and sales revenue, while COVID-19-related treatment and prevention drug development bring companies opportunities.
Notably, this study underscores that the effects of COVID-19 vary among firms. Mature companies with more financial leverage, more assets and higher sales scale are more likely to be affected by and therefore report sales and operations issues, while firms with high risk-adjusted returns and more spending on R&D are more likely to suffer from the problem of disrupted new product development processes. In addition, mature firms with high financial leverage and larger size are more likely to report multiple impact areas, and those with high assets are more likely to report impact areas positively relative to other firms.
Contributions to the literature
This research makes several significant contributions to the existing literature on the impact of the COVID-19 pandemic on the pharmaceutical industry. Firstly, it provides a systematic and comprehensive analysis of the pandemic’s effects on pharmaceutical companies, filling a notable gap in firm-level insights. Unlike previous studies that primarily focus on market-level impacts such as infoveillance of twits and public emotions,6–8 or relying on anecdotal evidence to derive firm-level insights, 9 this research leverages empirical data from 447 public pharmaceutical companies’ 10-K annual reports, offering a robust and data-driven perspective.
Secondly, the study offers a comprehensive understanding of the impact areas of COVID-19 on pharmaceutical firms by employing advanced topic modeling techniques. This methodological approach not only enhances the comprehensiveness of the analysis but also ensures that the findings are grounded in a rigorous analytical framework. The identification of four common themes—new product development, sales and operations, COVID-19 treatment and prevention drug early development, and clinical trials—provides a nuanced understanding of how different aspects of the pharmaceutical value chain were affected. Such an unsupervised learning approach does generate unexpected findings that extend the existing literature. For example, while prior research mainly focuses on supply chain impacts,9,14 this study highlights the major impact areas that extend beyond supply chain management, including new product development processes, sales and operations, and COVID-19 treatment and prevention drug early development and clinical trials.
Thirdly, the research contributes to the understanding of differential impacts across firms of varying sizes and focuses, adding to a more nuanced understanding of the COVID-19 impact on firms and firm resilience to the COVID-19 crisis. By conducting statistical analyses such as Tobit and logistic regressions, the study reveals that larger firms experienced lower negative effects, while companies with a primary focus on research and development were more likely to report impacts related to new product processes. Furthermore, firms with more assets, higher sales scale, and financial leverage are likely to report more areas of impact, but only those with high assets exhibit strong resilience to COVID-19 disruption, as judged from their reporting sentiment. This insight also adds to the organization resilience literature that larger firms and firms with more resources are more resilient to external shocks.48,49
Practical implications
The findings of this research offer substantial practical implications for pharmaceutical business managers. By providing a detailed analysis of firm annual reports and financial data, this study delivers actionable insights that can help managers strategically navigate potential risks and opportunities in future business disruptions. Understanding the specific areas of impact and the factors influencing these impacts enables managers to better prepare for and mitigate the adverse effects of similar crises in the future.
Firstly, the identified impact areas—new product development, sales and operations, COVID-19 treatment and prevention drug early development, and clinical trials—serve as a comprehensive guide for managers to re-examine and reflect on the impacts their companies experienced during the COVID-19 pandemic. This re-examination is crucial for identifying vulnerabilities and strengths within their operations and value chains. By understanding the areas most affected by the pandemic, managers can implement targeted strategies to safeguard these areas against future disruptions. This proactive approach includes diversifying supply chains, investing in digital transformation to support remote operations, and enhancing the flexibility of clinical trial processes.
Secondly, the research highlights the importance of firm characteristics in determining resilience to disruptions. Larger firms experienced fewer negative effects, while those focused on research and development were more negatively affected in the area of new product development processes. Managers can use these insights to tailor their strategies based on their firm’s size and focus, ensuring a more resilient and adaptive approach to future disruptions.
Thirdly, the study’s methodological approach, which includes advanced topic modeling and statistical analyses, provides a robust framework for managers to analyze their own data. By adopting similar analytical techniques, managers can gain deeper insights into their firm’s specific challenges and opportunities, leading to more informed decision-making.
Lastly, the research underscores the value of continuous monitoring and assessment of business environments. Managers are encouraged to establish systems for ongoing evaluation of external and internal factors that could impact their operations. This vigilance will enable them to quickly adapt to changing circumstances and maintain business continuity.
Research limitation
The research is limited in several aspects, which suggests future research opportunities. First, we only focus on limited firm characteristics, including firm size, performance, financial risk, future growth opportunities and market liquidity. Future research can study more characteristics such as collaborative networks of firms and IT resources, which may predict the extent to which a firm suffers from the COVID-19 disruption. Second, this research is retrospective, and the main purpose is to study the impact of COVID-19. Future research can study how firms recover from COVID-19 and the factors that contribute to such recoveries. Finally, this research reveals two distinct groups of companies in terms of the impact of COVID-19: topic 1 companies and topic 2 companies. Topic 2 companies are companies that have multiple business focuses and reported their impact most negatively among all companies, while topic 2 companies are those that have a focus on RND. Future research can also compare their patterns of recovery due to the distinct resources these two types of companies own.
Conclusions
The COVID-19 outbreak has significantly changed how organizations operate, and such impacts vary with industries. Our research focusing on the pharmaceutical industry suggests that the new product development process, sales and operations, COVID-19 treatment and prevention drug early development and clinic trials are major impact areas that managers need to pay high attention to. The impact of COVID-19 is heterogeneous among firms, depending on their characteristics such as firm size and performance. Future research can continue to monitor the after-pandemic changes and understand the transformation meaningful for the pharmaceutical sector.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work was supported by Social Sciences and Humanities Research Council of Canada [grant numbers: 892-2021-0031 and 435-2021-0941].

