Abstract
Rejection sensitivity is invovled in a defensive mechanism that triggers an individual's flight or fight response to social exclusion. Inspired by the roles of testosterone in defensive behaviors, we explored the relationships between three indicators of testosterone and rejection sensitivity through two studies. In Study 1, we recruited a cohort (
Keywords
Introduction
The pursuit of acceptance and avoidance of rejection stand as foundational motivations with social contexts. Despite this, many people often anticipate rejection when expressing their needs to significant others. This tendency is a personality characterized by oversensitivity to social rejection (Downey & Feldman, 1996). Rejection sensitivity significantly influences how people respond to negative stimuli and interpersonal challenges, contributing to the development of psychopathological symptoms (Beeson et al., 2020; Downey & Feldman, 1996; Minihan et al., 2023; Romero-Canyas et al., 2010). In recent years, the scientific community, particularly in biology, has recognized the critical role of biological factors in rejection sensitivity (Auer et al., 2015; Way et al., 2009; Woods et al., 2018). Nonetheless, our understanding of the biological underpinnings of rejection sensitivity remains in its infancy.
Rejection sensitivity is a key aspect of defensive motivation, influencing individuals to adopt fight or flight response (Downey et al., 2004; Gao et al., 2021). It encompasses specific cognitive-affective dimensions, including the anticipation of rejection and an enhanced perception of such rejection (Downey & Feldman, 1996). At the heart of rejection sensitivity, the expectation of rejection prompts individuals to be excessively vigilant towards any hints of disapproval (Downey & Feldman, 1996). This anticipatory mechanism often intertwines with defensive emotions, such as anxiety and anger, further affecting an individual's response to perceived social cues. The concept of readily perception, a successive cognitive process of rejection expectation, involves the beliefs in the likelihood of being rejected by others. Individuals with a heightened sense of perception are more prone to interpret others’ actions as having negative intentions (Downey & Feldman, 1996). This skewed perception, when combined with a strong anticipation of rejection, can lead to either withdrawal or aggressive behavior as a means of self-defense. Given the link between rejection sensitivity and defensive behaviors (Downey et al., 2004; Gao et al., 2021), we investigated whether the biological underpinnings of defensive behaviors influence rejection sensitivity.
Testosterone, a crucial hormone in humans, influences defensive behaviors in contexts of social rejection or provocation. In females, it has been found that those with higher levels of testosterone are more inclined to reject unfair offers (Probst et al., 2018). Similarly, among males, higher levels of testosterone are not only linked to the rejection of lower offers (Burnham, 2007), but also to an increased propensity to decline unfair proposals from members of out-groups (Reimers et al., 2019). These findings collectively suggest that testosterone may contribute to defensive responses in scenarios involving social rejection. However, more research is needed to further understand the relationship between testosterone levels and rejection sensitivity as a personality trait.
Testosterone influences the brain and behavior through different mechanisms. Current testosterone levels have an activation effect, influencing behaviors that are responsive to fluctuations in testosterone levels (Ostatnikova et al., 2020). In contrast, prenatal testosterone levels exert an organizational effect, shaping behaviors dependent on testosterone throughout an individual's lifespan. An indirect measure of prenatal testosterone exposure is the 2D:4D ratio, which is the relative lengths of the second to fourth fingers. This ratio is hypothesized to be inversely correlated to prenatal testosterone levels (Teatero & Netley, 2013; Ventura et al., 2013). Furthermore, testosterone levels are regulated by specific genes, including the steroid 5 alpha-reductase 2 (
This research aimed to explore the relationships between testosterone functions and rejection sensitivity through two distinct studies. Given that testosterone has been found to relate differently to social behaviors in males and females (Rehbein et al., 2021; van Wingen et al., 2010), we investigated whether the associations between these testosterone indicators and rejection sensitivity are differed by gender in sex-mixed samples, which would enrich the theory that testosterone's impact on social cognition varies by gender. In Study 1, we investigated the relationships between rejection sensitivity and two measures: salivary testosterone levels and the 2D:4D ratio. Drawing on evidence that links higher testosterone levels with increased rejection in response to unfair offers (Burnham, 2007; Probst et al., 2018; Reimers et al., 2019), as well as the relationship between lower prenatal testosterone levels and a higher 2D:4D ratio (Teatero & Netley, 2013; Ventura et al., 2013), we hypothesized that individuals with high salivary testosterone levels or a low 2D:4D ratio would exhibit greater sensitivity to rejection. This effect might be particularly pronounced in females, as testosterone more directly influences neural circuits involved in emotional regulation and social cognition in females (Rehbein et al., 2021; van Wingen et al., 2010). In Study 2, we examined the associations between testosterone-related genetic polymorphisms and rejection sensitivity. Due to the scarcity of existing research, we did not formulate specific hypotheses regarding the links between the genotypes and rejection sensitivity.
Study 1: The relationships between rejection sensitivity and salivary testosterone levels and the 2D:4D
Methods
Participants
One hundred eighty-three participants (88 females, mean age = 22.40 ± 2.34 years) were recruited through campus posters. Each one received ¥ 50 for participation. All the participants self-reported good health and confirmed that they were not taking any oral contraceptives. Female participants were specifically asked to avoid participation during their menstrual period. Additionally, all participants were instructed to refrain from eating, drinking, brushing their teeth, or engaging in any physical exercise for one hour prior to the collection of saliva samples. This study was approved by the ethics committee of our institution.
Rejection sensitivity assessment
Rejection sensitivity was assessed using the Chinese version of the Rejection Sensitivity Questionnaire (RSQ; Downey & Feldman, 1996). This scale consists of 18 hypothetical interpersonal interactions involving the possibility of rejection by a significant other. For each interaction, participants rated their level of concern or anxiety regarding the significant other's response to an important request (refers to anxious expectations of rejection) and their perception of the likelihood that the other person would accept the request (referred to as readily perceive). Ratings were made on a 7-piont scale (anxious expectation: “1” = not worried about at all to “7” = very worry; readily perceive: “1” = completely impossible to “7” = completely possible). To calculate RSQ scores, the expected likelihood of rejection for each scenario was weighted by the corresponding degree of anxiety reported by the participant. These weighted scores were then averaged across all situations to obtain the total RSQ score. In this sample, the internal consistencies (Cronbach's α) for anxious expectations, readily perceive, and total scale were 0.92, 0.86, and 0.89, respectively.
Salivary testosterone levels assessment
Saliva samples were collected between 13:30 and 14:00 p.m. (Dabbs, 1990; Touitou & Haus, 2000). To account for random fluctuations in testosterone levels, saliva samples were collected at two time points. The first sample was collected after a 10-min rest upon participants arrived at laboratory. The second sample was collected after a 20-min interval, during which participants signed the informed consent form. For each sample collection, following a rinse with water 15 min prior to collection, saliva was obtained by participants lowering their heads for 1 min and spiting into sterile polypropylene tubes. The samples were then stored at −80 °C. Salivary testosterone levels were quantified using a commercial enzyme-linked immunosorbent assay (ELISA) kit (Cloud-Clone Corp., China). The assay had a concentration range of 12.35 to1000 pg/mL and a sensitivity of more than 4.65 pg/mL. To perform the measurement, thawed saliva samples underwent centrifugation at 2000 g and 4 °C for 20 min. The resulting supernatant (50 μL) was extracted and added into sample wells of an ELISA plate. The ELISA plate included blank and standard wells, with reference standards added in concentration gradients (12.35, 37.04, 111.11, 333.33, and 1000 pg/mL). A detection reagent was then added to all wells, followed by incubation and washing steps. Subsequently, a chromogenic reaction occurred with the catalytic effect of HRP, and the reaction was terminated with a stop solution. The optical densities of all wells were measured at a wavelength of 450 nm using a microplate reader. Each plate included two sets of reference samples, and the mean optical density of these reference samples was calculated. A standard curve was created using the four-parameter logistic (4PL) method with ELISA Calc software. The intra-assay coefficient of variability was found to be less than 10%, indicating minimal variability within the same plate. The inter-assay coefficient of variability was less than 12%, demonstrating a high consistency across plates.
Digit ratio assessment
The measurement on the 2D:4D ratio followed the method proposed by Kemper's suggestion (Kemper & Schwerdtfeger, 2009). The lengths of index finger (2D) and ring finger (4D) digits were determined as the distance from the tip to ventral proximal crease. To calculate the digit ratio, the length of 2D was divided by the length of 4D of the right hand. The details for obtaining the lengths of 2D and 4D were available in a previous study (Zhang et al., 2020). The ratio of 2D:4D was obtained by two raters. The intra-class correlation coefficient for the ratio of 2D:4D between the two raters reached to 0.501.
Statistical analysis
The analysis of gender differences in the 2D:4D ratio was performed using an independent-sample
Results
The relationship between salivary testosterone level and rejection sensitivity
In this sample, the females exhibited higher level of rejection sensitivity than males (Mean ± Se = 7.69 ± 0.37 vs. 7.42 ± 0.37), although the difference was not significant (
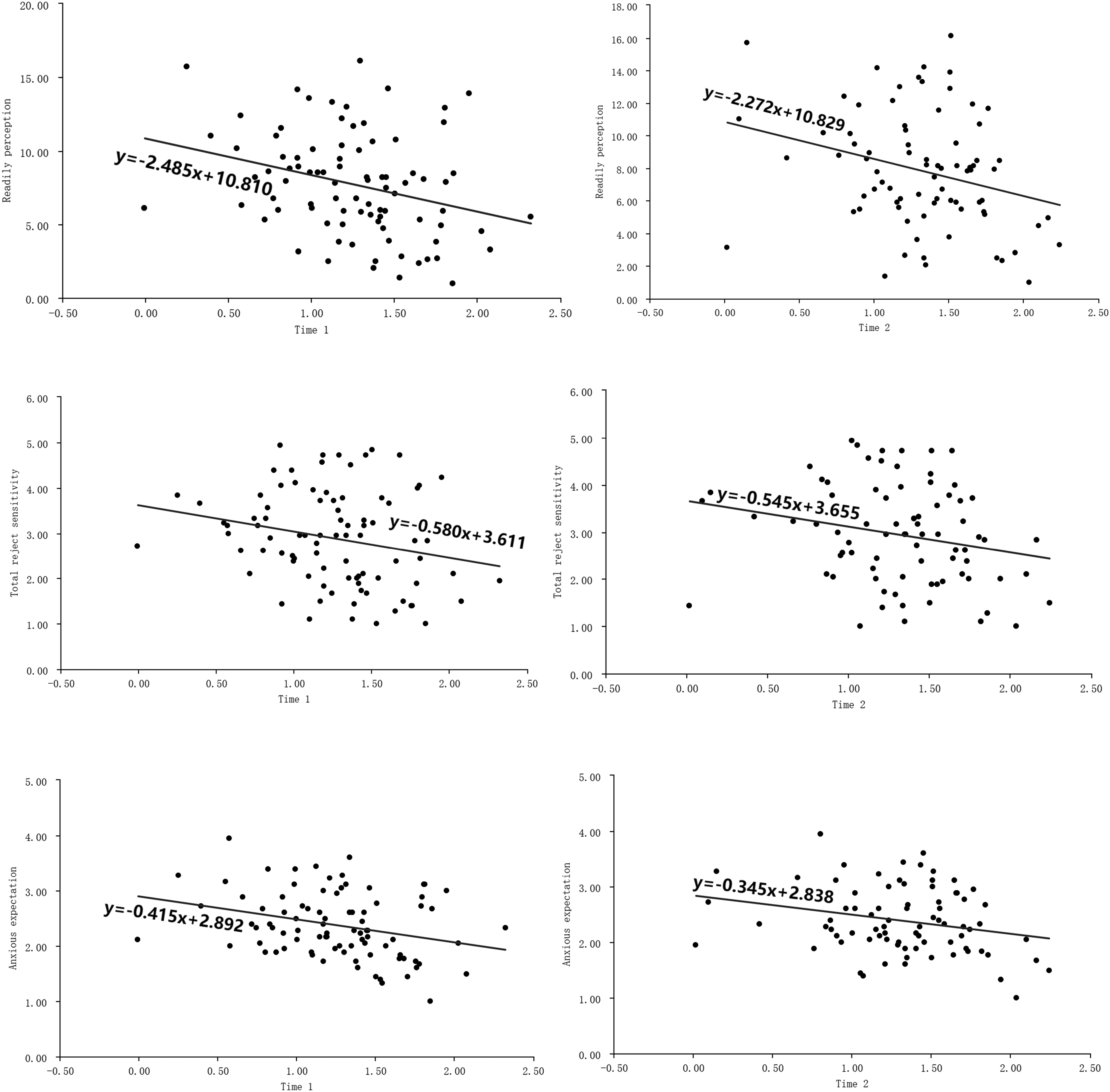
Testosterone levels at both time points are significantly related to anxious expectation, readily perceive, and total reject sensitivity. Females with higher testosterone levels at the both time points exhibit lower anxious expectation, readily perceive, and total reject sensitivity.
Regressions on the effects of testosterone levels and the 2D:4D on rejection sensitivity.
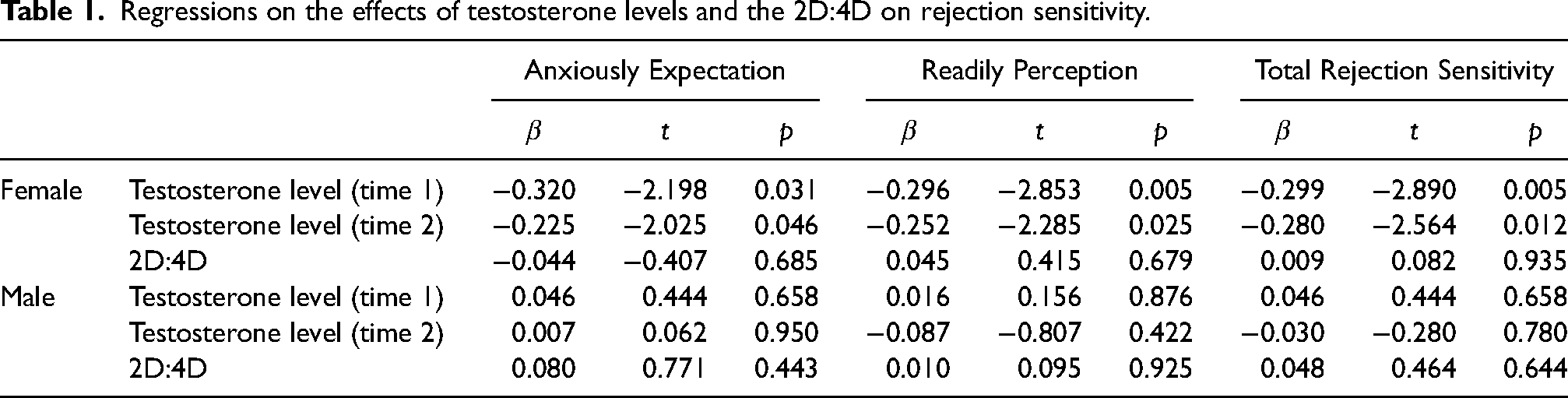
The relationship between the 2D:4D and rejection sensitivity
The ratio of 2D:4D of males was significantly smaller than that of the females (Mean ± SD: males vs. females = 0.953 ± 0.028 vs. 0.977 ± 0.058;
Interim discussion
The study demonstrates that testosterone levels are associated with rejection sensitivity in females. Specifically, individuals with higher testosterone levels tend to exhibit lower levels of anxious expectation, readily perceive, and overall rejection sensitivity. These findings provide new insights into the roles of testosterone in emotional responses in the contexts of rejection. By contrast, this study indicates significant sexual differences in the 2D:4D ratio, with a smaller ratio in males than females. However, duo to the sexual difference, the study did not observed a significant relationship between the 2D:4D ratio and rejection sensitivity. This implicates that the 2D:4D ratio is not an available indicator for predicting individual differences in rejection sensitivity.
Study 2: The relationships between testosterone-related genetic polymorphisms and rejection sensitivity
Methods
Participants
A power analysis was conducted using G*Power 3.1 to ensure adequate statistical power. With a conservative effect size of 0.01 (Border & Keller, 2017; Duncan & Keller, 2011), a two-tailed alpha level of 0.05, and a desired power of 0.85, the analysis indicated that a sample size of 900 participants would be required. To account for the uncertainty in the estimated effect size, the sample size was increased to approximately 120% of the initial estimation, resulting in a target sample size of 1,080 participants. Finally, 1,112 college students (677 females, age = 20.89 ± 2.05 years) were recruited through advertisements posted on university website, and each participant received ¥ 5 for participation. This study was approved by the ethics committee of our institution.
Rejection sensitivity assessment
Rejection sensitivity this sample was also assessed by using the Chinese version of the RSQ, with administering in pencil-and-paper format to a group of 15–20 participants. In this sample, the Cronbach's α for anxious expectation, readily perceive, and total scale were 0.89, 0.87, and 0.92, respectively.
Genotyping
We collected 3–5 hair follicle cells from each participant to extract genomic DNA. The
Statistical analysis
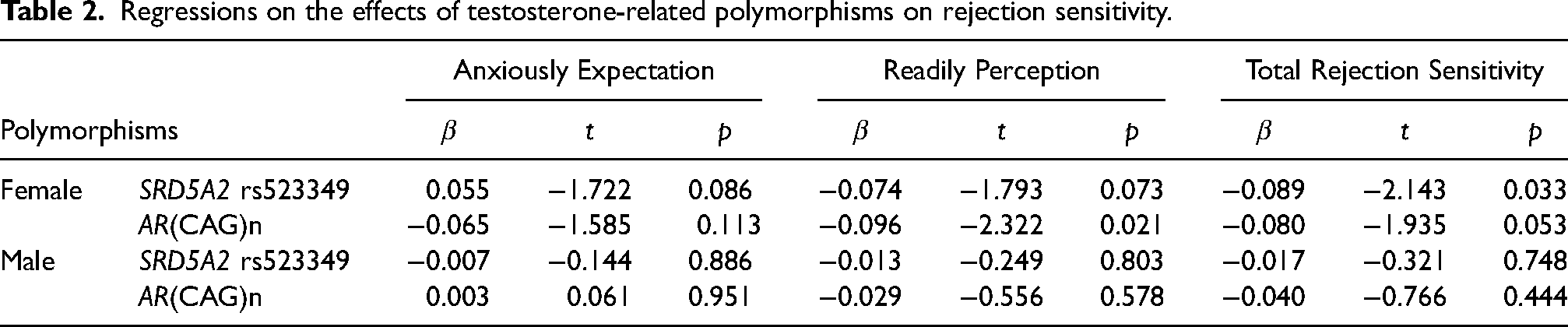
A power analysis was conducted using G*Power 3.1, software, targeting an effect size of 0.01, a significance level (alpha) of 0.05 with a two-tailed approach, and aiming for a statistical power of 0.80. The associations between genotypes and rejection sensitivity was examined with linear regression analysis on SPSS 18.0 software (SPSS Inc., Chicago, IL, USA). In analysis, genotypes were categorized based on their roles in testosterone sensitivity and metabolism. Specifically, the
Results
In this sample, females exhibited higher levels of rejection sensitivity compared to males (Mean ± Se: 8.00 ± 0.21 vs. 7.34 ± 0.14;
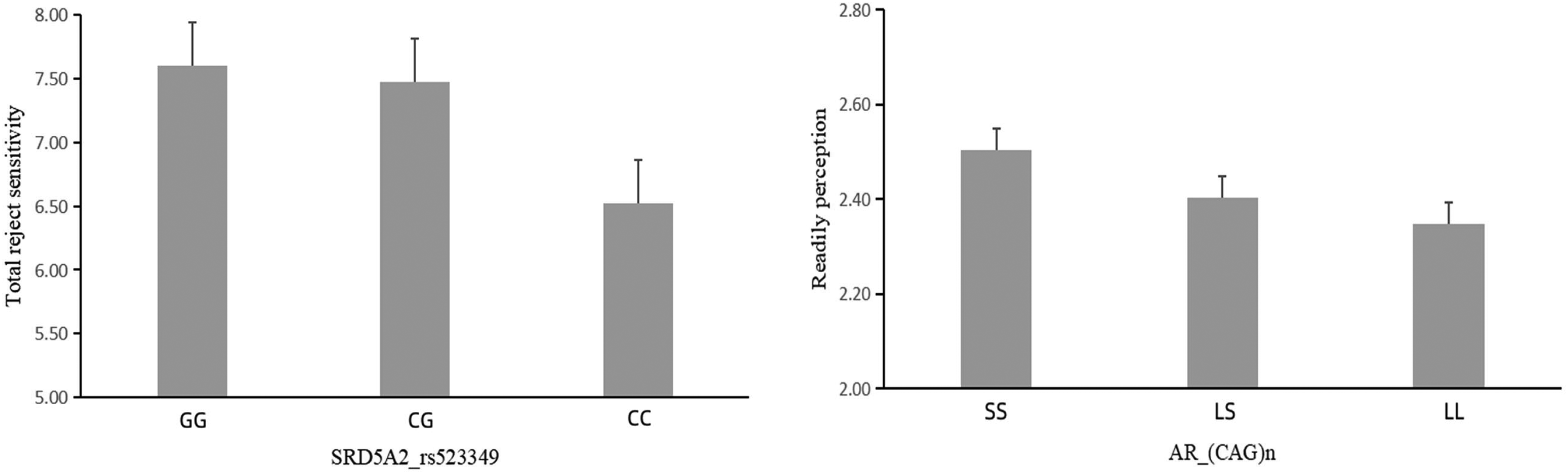
Females with the CC genotype exhibited lower reject sensitivity than those with the CG or GG genotype (Mean ± Se = CC vs. CG vs. GG = 6.521 ± 0.32 vs. 7.471 ± 0.201 vs. 7.602 ± 0.246; Figure 2a). Individuals with the LL genotype showed lower readily perceive than those with the SS genotype (LL vs. LS vs. SS = 2.348 ± 0.041 vs. 2.402 ± 0.047 vs. 2.503 ± 0.053; Figure 2b).
Regressions on the effects of testosterone-related polymorphisms on rejection sensitivity.
Interim discussion
The study indicates the relationships between testosterone-related polymorphisms, and rejection sensitivity, particularly in females. Specifically, females with the CC genotype of
General discussion
This research examines the relationships between three indicators of testosterone and rejection sensitivity. The findings demonstrate that testosterone levels are associated with dispositional rejection sensitivity in females. Additionally, this research demonstrates significant relationships between testosterone-related polymorphisms and rejection sensitivity in females. These findings highlight the significant influences of testosterone functions on emotional and cognitive processes of rejection sensitivity.
In contrast to previous findings that individuals with higher levels of testosterone are more likely to reject unfair offers and to exhibit reactive aggression (Burnham, 2007; Probst et al., 2018; Reimers et al., 2019), we observe that females with higher levels of testosterone tend to exhibit lower levels of rejection sensitivity. This discrepancy could be attributable to the differences in the types of social rejection assessed across studies. Anxious expectation of rejection is associated with social anxiety and withdrawal, while angry expectation of rejection is linked to increased aggression (Gao et al., 2021). Prior studies primarily focused on reactions to unfair financial propositions in the ultimatum game, where rejection is often a defensive response driven by anger towards unfair treatment. This form of rejection aligns with the angry expectation of rejection, which is correlated with aggression. In contrast, our study utilizes the RSQ to examine reactions to interpersonal situations, which may evoke anxiety over the anticipated response to significant requests. This anxious expectation of rejection is linked to social anxiety and withdrawal, rather than aggression (Gao et al., 2021). Such discrepancies underscoring the relationship between testosterone levels and responses to social rejection is context-dependent. This highlights the importance of considering the specific social context and type of rejection when examining the role of testosterone in social behaviors.
This study highlights that females with relatively higher levels of testosterone tend to exhibit lower rejection sensitivity. The mechanisms underlying this association are not fully elucidated. However, several plausible explanations exist. Firstly, testosterone has been shown to suppress acute fear responses and foster a reactive dominance posture (Enter et al., 2014; Radke et al., 2015; Terburg & van Honk, 2013). Females with higher testosterone levels might prioritize social dominance over avoiding rejection, leading to a stronger focus on pursuing personal goals rather than being overly concerned about potential rejection. Secondly, testosterone is linked to increased assertiveness and self-confidence (Chichinadze et al., 2012; Metzger & Boettger, 2019). This could result in women with elevated testosterone levels presenting with greater confidence and self-assurance, rendering them less vulnerable to cues of implicit subordination or threats (Terburg et al., 2016; van Peer et al., 2017). As a consequence, these individuals might experience lower levels of anxious anticipation concerning rejection (Hutschemaekers et al., 2020). These findings pave the way for understanding the roles of testosterone in modulating sensitivity to rejection.
Our findings indicate that the CC genotype of
The theory of defensive motivation offers a comprehensive framework for understanding the intricate relationships among testosterone, rejection sensitivity, and the defensive system. According to this theory, individuals instinctively activate their defensive mechanisms when faced with social threats, such as rejection and social exclusion (Downey et al., 2004). Testosterone, a hormone known for its association with dominance and social aggression (Enter et al., 2014; Radke et al., 2015; Terburg & van Honk, 2013), influences the activation of defensive system. Research indicates that higher testosterone levels are linked to increased dominance, competitiveness, and a diminished sensitivity to social threats (Eisenegger et al., 2017; Grant & France, 2001; van Honk & Schutter, 2007; Welling et al., 2016), leading to lower rejection sensitivity. This means that individuals with elevated testosterone levels may be less attuned to cues of rejection or may display a reduced emotional response to such cues. Within the context of this theory, testosterone may modulate the functioning of the defensive system and influence rejection sensitivity through multiple pathways. For instance, testosterone's effects on dominance and the pursuit of status can shift an individual's focus towards achieving social dominance and pursuing goals, rather than dwelling on the potential for rejection (Enter et al., 2014; Radke et al., 2015; Terburg & van Honk, 2013). This reorientation of priorities is likely to result in reduced sensitivity to rejection. By exploring these mechanisms, the theory of defensive motivation provides valuable insights into how testosterone modulates the defensive system and the perception of social threats.
This research suggests that rejection sensitivity may be influenced by testosterone levels and genetic polymorphisms, but not by the 2D:4D ratio. A prior study, utilizing replicated analysis and meta-analysis, found no significant correlation between current testosterone levels,
The current study reveals that testosterone levels and related genetic polymorphisms are specifically associated with rejection sensitivity in females. These results broaden our understanding of testosterone's gender-specific roles in social sensitivity. Firstly, females, having typically lower baseline testosterone levels than males, may experience a significant impact on the sensitivity to social cues from even slight increases within their physiological range (Harteveld et al., 2019; Im et al., 2018). This highlights the influence of baseline testosterone differences on rejection sensitivity in females. Secondly, testosterone may differentially affect areas of the brain involved in emotion regulation between genders. In females, testosterone could particularly alter neural circuits tied to social cue processing and emotional responses (Rehbein et al., 2021; van Wingen et al., 2010), potentially intensifying emotional reactions to rejection or heightening awareness of potential rejection. Thirdly, socialization process significantly shapes how social rejection is perceived and its consequences for females (Guadagno & Cialdini, 2007; London et al., 2012). Behaviors influenced by testosterone that diverge from socially conditioned roles might heighten rejection sensitivity, pointing to a clash between testosterone-driven behaviors and societal expectations for females. Together, this research not only enhances our understanding of the gender-specific effects of testosterone on social rejection, but also offers a multidimensional perspective on the biological, psychological, and social determinants of rejection sensitivity.
The research greatly benefits from adopting an interdisciplinary approach that integrates insights from psychology, endocrinology, and genetics. This strategy would offer a more thorough understanding of the biological foundations underlying rejection sensitivity. However, it is important to acknowledge several limitations of our current study. Firstly, the method of measuring testosterone levels and rejection sensitivity presents challenges. Testosterone levels are subject to fluctuation due to a variety of factors, such as the time of day and levels of stress (Rehbein et al., 2021). Our decision to measure testosterone at only two time points within a single day may not provide an accurate representation of baseline levels. Similarly, our assessment of rejection sensitivity relied on a self-report questionnaire, which is susceptible to response biases (Adams et al., 1999). Secondly, our study's focus on specific genetic polymorphisms may not adequately address the intricate interactions among multiple genes. Consequently, our approach might fail to capture the entire genetic landscape associated with this condition. Lastly, the pathways through which testosterone and genetic polymorphisms affect rejection sensitivity are likely complex and involve multiple facets. This study falls short of fully elucidating these mechanisms and the biological processes they entail.
Conclusion
This research demonstrates that both testosterone levels and genetic polymorphisms underpin the individual differences in rejection sensitivity, which highlight the influences of testosterone functions on emotional processes related to social rejection.
Supplemental Material
sj-docx-1-pac-10.1177_18344909241254499 - Supplemental material for The influence of testosterone on rejection sensitivity: Insights from salivary testosterone levels and genetic polymorphisms
Supplemental material, sj-docx-1-pac-10.1177_18344909241254499 for The influence of testosterone on rejection sensitivity: Insights from salivary testosterone levels and genetic polymorphisms by Pingyuan Gong, Xiaohan Li, Yuting Yang, Yafang Yang, Mengying Xue and Mengfei Zhang in Journal of Pacific Rim Psychology
Supplemental Material
sj-docx-2-pac-10.1177_18344909241254499 - Supplemental material for The influence of testosterone on rejection sensitivity: Insights from salivary testosterone levels and genetic polymorphisms
Supplemental material, sj-docx-2-pac-10.1177_18344909241254499 for The influence of testosterone on rejection sensitivity: Insights from salivary testosterone levels and genetic polymorphisms by Pingyuan Gong, Xiaohan Li, Yuting Yang, Yafang Yang, Mengying Xue and Mengfei Zhang in Journal of Pacific Rim Psychology
Footnotes
Acknowledgments
We thank Mr. Guomin Shen for his assistance in data collection. This study was supported by the National Social Science Foundation of China (20BSH165) to Pingyuan Gong.
Authors contributions
Performed the experiment: Xiaohan Li, Yuting Yang, Yafang Yang, Mengying Xue,Mengfei Zheng, Pingyuan Gong. Analyzed the data: Pingyuan Gong and Xiaohan Li. Wrote the paper: Pingyuan Gong. Designed the study: Pingyuan Gong.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Participants signed informed consent before taking part. The study was carried out according to the Declaration of Helsinki and was approved by the ethics committee of Life Science College, Northwest University, China.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the the National Social Science Foundation of China (grant number 20BSH165).
Open research data
We have uploaded our data on the Figshare (10.6084/m9.figshare.24269215).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
