Abstract
Background:
This study shares insights from clinical trialists who have conducted investigator-initiated trials that have linked trial data to administrative data, focusing on the challenges and facilitators of this approach.
Objective:
To provide recommendations for evaluating the feasibility and suitability of using administrative data in clinical trials.
Method:
A convergent parallel mixed-methods study was conducted, surveying Australian clinical trialists and operations staff. Participants could opt-in to in-depth interviews. Survey data were analysed using descriptive statistics, while thematic analysis was applied to interview data, with findings integrated during interpretation.
Results:
Four main themes and 10 sub-themes were identified as critical when evaluating the suitability of administrative data for clinical trials: (i) “trial management considerations” covers operational factors like budgeting, timelines and staffing; (ii) “assessing burdens vs. gains” encourages weighing up the research benefits with the additional operational and consent considerations; (iii) “data preparation and analysis” addresses the processes involved in preparing and analysing data for linkage between trial and administrative datasets; and (iv) “training and support” emphasises the need for researcher support when using linked data.
Conclusion:
Researchers should carefully evaluate the feasibility of using administrative data, considering costs, required skills, timelines and data accuracy. They must also be prepared for delays due to data request processes, participant consent requirements and the mandated use of data access platforms. Early planning can mitigate later complexities.
Implications for health information management practice:
This study highlights the value of health information managers in clinical research, particularly in managing electronic health records and clinical coding. Their expertise in these areas, as well as in data governance and system architecture, can support clinical trials that link to administrative data.
Keywords
Introduction
Linking clinical trial data with administrative data, or routinely collected health data, has great potential worldwide for answering research questions. Administrative data refer to information collected by government departments, businesses and organisations for reasons other than research, such as registrations, billing and record keeping (Australian Bureau of Statistics, 2021), and in this paper, we use “linkage” to describe the process of linking clinical trial data to administrative data. The drive to conduct trials more efficiently, reduce costs and lessen the burden on trial sites is a common goal for trial units. Administrative data are often viewed as a cost-effective alternative for clinical trial data collection, and it can provide data, such as health service utilisation and medication adherence, which are often challenging to obtain through traditional methods.
A scoping review of administrative data use in Australian randomised controlled trials (Fahridin et al., 2024) reported a steady increase in publications that linked routinely collected health data to clinical trial data. While the reviewed articles identified several barriers and facilitators to administrative data use, these were limited to the information available in the included studies and did not address the practical use of the datasets or how challenges were overcome. The review found that there were many more studies planning to access administrative data than there were publications, but reasons for this discrepancy were not explored. Guidance around the use of linked administrative data for research purposes is available through state and federal data custodians, or the national data linkage unit network (Flack et al., 2019); however, a knowledge gap remains as to how to best work within the known limitations of administrative data.
In this study, we aimed to share learnings from clinical trialists who have conducted investigator-initiated trials that linked clinical trial data to administrative data, and to understand the challenges and facilitators to its use, while providing suggestions for evaluating its feasibility and suitability for a clinical trial.
Method
Study design and sample selection
A convergent parallel mixed-methods design was used (Katz-Buonincontro, 2024), integrating responses from a survey with interview data from a subsection of the original sample to achieve a comprehensive understanding of administrative data use. The target population were individuals who worked in academic clinical research and who had experience linking clinical trial data with administrative data at any stage of the linkage process. Purposive sampling was utilised to recruit potential participants, through an invitation to complete the anonymous online survey, which was advertised within relevant trial, network and association newsletters, as well as snowball sampling (Braun and Clarke, 2013), where respondents were encouraged to share the survey within their networks. The survey was open for 10 weeks, between March and May 2024. Survey respondents provided consent before completing the survey. (For a copy of the survey, see Figure S3, online Supplemental File). At the conclusion of the survey, all respondents were invited to participate in a single in-depth interview. Those who agreed were asked to provide an email address on a separate form to ensure the anonymity of their survey responses.
Survey data collection
For the survey, a REDCap (Research Electronic Data Capture; Harris et al., 2009, 2019) questionnaire (Supplemental File 3) hosted at the University of Sydney, Australia, was designed to collect responses by mobile phone/tablet or desktop browser. Survey questions collected information via a combination of structured response options (such as yes/no or multiple choice) and open-ended free-text responses regarding participants’ responsibility in the data linkage processes, available support, required skills, storage/accessibility considerations, cost and barriers/enablers to linkage. Participant details such as occupation, employer and experience with data linkage were also collected.
Interview data collection
An information sheet was emailed to the subsample of respondents who had indicated that they were willing to participate in an interview, together with a link to the interview consent form for completion and so they could nominate the times they were available to be interviewed. The interviewer arranged for interviews to take place within 1–3 weeks of the survey completion date. Semi-structured, one-on-one, in-depth interviews were then conducted with this subsample of interview participants. The interviews took place via video call from their usual workplace (university, health service, or home) and lasted between 20 and 64 minutes (average of 45 minutes).
Interviews were based on an interview guide (see Figure S1, online Supplemental File 1), which had been piloted previously with three colleagues who were experienced in clinical trials that linked to administrative data. These pilot interviews were not included in the analysis. With the consent of participants, interviews were audio-recorded using Zoom (2024) and were conducted by one researcher (SF). The interviewer was a health information management graduate and PhD candidate at the time of the study, who also held a leadership role in overseeing data management operations at a large academic clinical trial coordinating centre. This professional context informed the motivation for the research, which was disclosed to the participants at the beginning of each interview. The interviewer had also undergone postgraduate qualitative methods training prior to conducting interviews and was known to some interview participants through existing working relationships. Interview participants were advised that any identifying information in their responses would be anonymised and their responses would not impact their relationship with the interviewer or the organisation at which she (SF) was employed. The interviews focused on the experiences of the participants in requesting, obtaining and using administrative data, specifically, barriers and facilitators to these processes, as well as on the purpose of using the data, support received and overall attitudes about linking trial data to administrative data. The semi-structured interview format allowed participants to speak freely.
Transcripts were produced directly from the audio recordings and were manually checked for accuracy by the researcher (SF). All comments and responses were anonymised during the process of transcription. Transcriptions were emailed to participants to confirm accuracy. As with the survey data, anonymised transcripts of the interviews were stored in the University of Sydney’s Research Data Management System and accessible only to the study team. Data saturation was discussed over two coding sessions by four researchers (SF, KB, CKL and RLM) at which point it was decided that no new themes had been identified and further interviews were not required.
Data processing and analysis
Quantitative survey data were coded and analysed using descriptive statistics. Interview data and qualitative survey data were analysed using the six-phase approach for thematic analyses proposed by Braun and Clarke (2013): (i) familiarising yourself with your data, (ii) generating initial codes, (iii) searching for themes, (iv) reviewing themes, (v) defining and naming themes and (vi) producing the report. The interview transcripts and qualitative survey data from the open-ended, free-text responses were imported into NVivo (Version 14), qualitative analysis software (Lumivero, 2023). Four researchers (SF, KB, CKL and RLM) independently conducted preliminary coding on 20% of the qualitative data and derived themes or patterns by systematically identifying and labelling relevant data characteristics. The coded elements were discussed to identify similarities and differences, and organised into potential themes and sub-themes, and further refined through a review process to generate cohesive and meaningful themes, until consensus was reached. The remaining transcripts were coded and labelled by one researcher (SF). Following the initial thematic analysis, interview participants were emailed a summary of the themes and invited to provide feedback. Their suggestions were considered and incorporated where appropriate to enhance the credibility and accuracy of the findings.
Ethics approval
This research was approved by the University of Sydney Human Research Ethics Committee (Project no. 2023/793). The study has been reported in accordance with the Standards for Reporting Qualitative Research (O’Brien et al., 2014) and Consolidated Criteria for Reporting Qualitative Studies (Tong et al., 2007) (Supplemental File 2).
Results
Sample characteristics
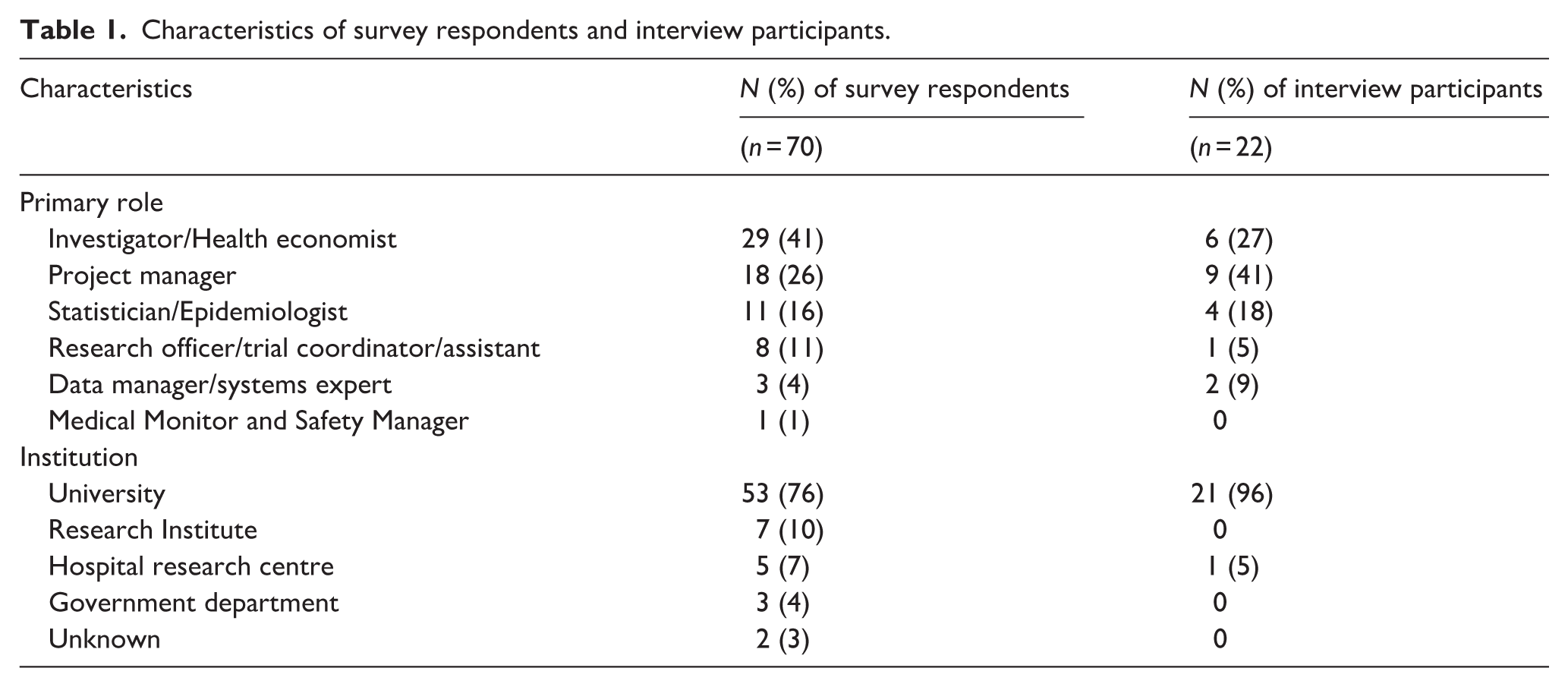
Survey respondents
Seventy survey responses were received, with respondents spread across 18 different Australian institutions, with three-quarters working in an Australian university (n = 53; 76%). Ten universities across five Australian states were represented among these respondents. The majority were investigators, including health economists (n = 29; 41%), project managers (n = 18; 26%), or statisticians/epidemiologists (n = 11; 16%) (Table 1). There was a broad range of data linkage experience, with 66% (n = 46) having worked on between one to four trials involving data linkage, and 29% (n = 20) having worked on at least five trials involving data linkage (Figure 1). Survey respondents were involved in a broad range of duties in the data linkage process. However, of the 10 pre-defined tasks, the majority of respondents played a role in leading and/or assisting processes related to receiving the data (n = 60; 86%), requesting the data (n = 59; 84%) and seeking ethics approval (n = 57; 81%) (Figure 2).
Characteristics of survey respondents and interview participants.
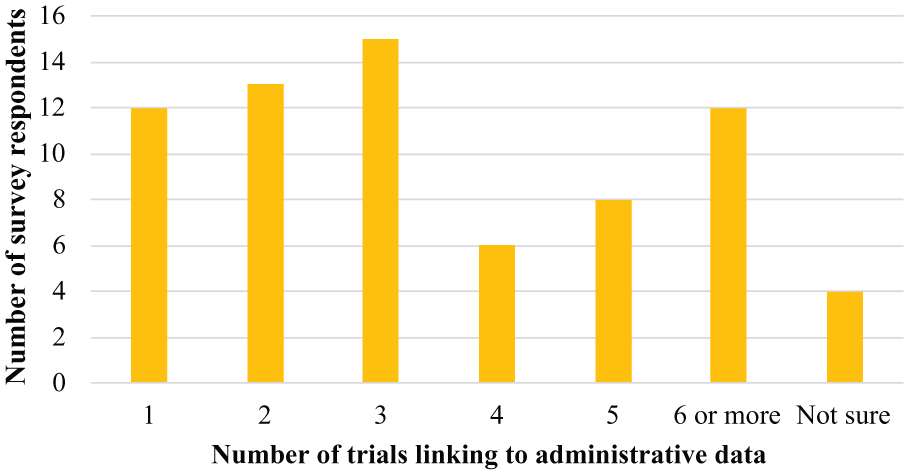
Number of trials with linked administrative data per survey respondent (n = 70).
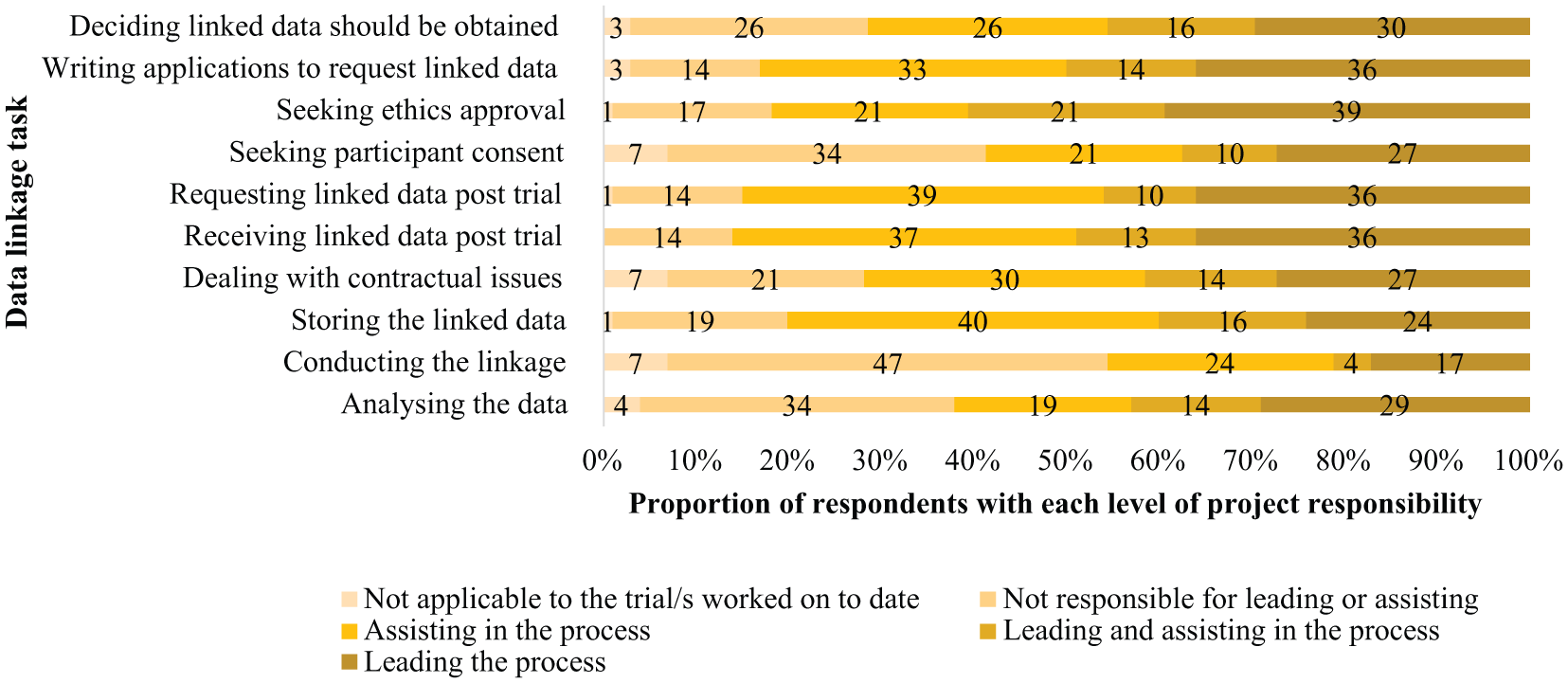
Survey respondents’ level of project responsibility in data linkage project/s (n = 70).
Information was collected about 201 trials from the 70 survey respondents. For each trial, respondents could select between one and six different purposes for using administrative data. The most common uses were measuring economic outcomes (n = 111 trials; 55%), tracking health service use (n = 107 trials; 53%), measuring disease or health outcomes (n = 99 trials; 49%) and tracking participant survival status (n = 84 trials; 42%). Fewer trials used administrative data to measure drug adherence (n = 22; 11%) (see Figure 3).
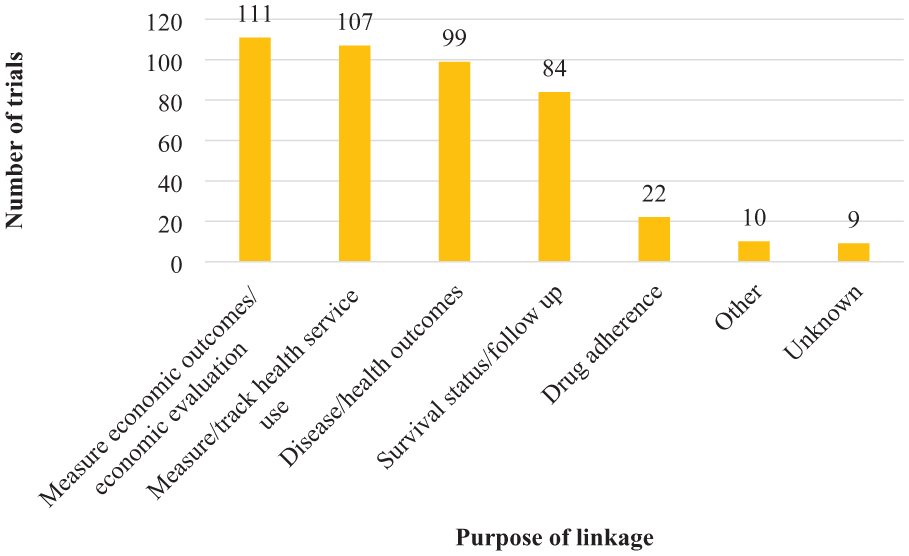
Purpose of administrative data linkage among survey respondents’ trials (n = 201).*
Interview participants
Twenty-two (31%) of the survey respondents agreed to participate in an in-depth interview and all but one of the interview participants worked at an Australian university (n = 21, 96%).
Themes
The thematic analysis of interview and survey data produced four overarching themes and 10 sub-themes (Table 2): “Trial management considerations” covers operational factors like budgeting, staffing, and timelines; “Assessing burdens vs. gains” evaluates whether the research benefits of administrative data outweigh the additional operational and consent considerations; “Data preparation and analysis” addresses the processes involved in preparing and analysing data for linkage between trial and administrative datasets, while “Training and support” emphasises the need for researcher support in using linked data. The quotes associated with these themes and subthemes are listed in Tables 3–6.
Outline of themes and sub-themes.

Quotations to illustrate Theme 1 – Trial management considerations.

Quotations to illustrate Theme 2 – Assessing burdens versus gains.

Quotations to illustrate Theme 3 – Data preparation and analysis.

Quotations to illustrate Theme 4 – Training and support.

Theme 1 – Trial management considerations
The use of administrative data involves key operational considerations that researchers must navigate. Of the 70 survey participants, almost half (n = 31; 44%) felt that they would do something differently the next time they are involved in a data linkage process, including better planning or having clearer processes (n = 12), engaging data custodians earlier (n = 7) or allowing for more money and time (n = 5). Some participants said they would reconsider using administrative datasets altogether (n = 8) due to the added complexities. These findings highlight the operational complexities that can significantly impact the feasibility and success of administrative data projects.
Good documentation and communication practices
Participants commented that maintaining detailed records of correspondence and decisions was a valuable practice as the duration between initial discussions and requesting of data can be years. This may include discussions with data custodians and internal decisions, such as when and why datasets were required, who was responsible for each process, such as preparing applications, managing consent forms and receiving and storing the data. Good communication was seen as particularly important where trials involved multiple parties, especially if they were from different institutions. Participants agreed that connecting early and regularly with the full research team would help to agree on plans for the data; however, some project managers mentioned the challenge of balancing investigator expectations and project demands to keep the trial on time and under budget, as changes to project scope or expectations typically result in increased time and cost. Health economists, in particular, expressed frustration that they were consulted too late in the planning or were not kept updated on the progress of the trial.
Cost of obtaining data
Interview participants stressed the importance of evaluating the benefits of obtaining the data in relation to the fees charged and noted the substantial differences in data access fees among various data custodians. The cost of obtaining data influenced study design, such as whether an additional jurisdiction would be included in the participant sample, or whether an endpoint could be assessed. There were varying opinions on whether the cost of obtaining data was considered reasonable, with interview participants acknowledging that research is not the custodians’ core business, and some citing considerable fee increases over time. However, participants generally agreed that the most important factor was ensuring a quote is obtained from the data custodians and factored into grant applications. Challenges raised by the participants were the unpredictability of the final fee for the data, as often quotes are received years before data are required and are subject to change when the dataset is finally generated, suggesting a buffer should be added to all quoted costs when developing a budget. Further, some were unaware they would need to add the cost of the data storage and curation platform, which is increasingly becoming a mandatory requirement when working with administrative data (see subtheme “Mandated data access platforms”).
Setting realistic timelines
A recurring theme was the significant time required to prepare data linkage applications, delays in receiving approval for data requests and delays in receiving data once the final request is submitted to data custodians. Participants observed that it was common for less experienced researchers to underestimate the work required to prepare data linkage applications, and this was further exacerbated by the fact that linkage requirements are becoming increasingly complicated. Some said it was worthwhile developing a strategy for applying for access to administrative data, especially in multi-jurisdictional trials, where the order of events seemed to impact the outcome. Participants employed different strategies to deal with this. Some decided to approach the custodians who had very specific requirements first, whereas others would approach these custodians last to ensure that they were satisfied with all the changes made following applications to prior custodians. It was clear that applying to multiple custodians increased the workload significantly, as applications were not streamlined. Reusing responses from prior approved applications was one way to avoid unnecessary rework; however, custodian requirements could change between applications. Appreciating custodians have limited resources to deal with research requests; delays of up to 2 years were not uncommon and depended on the custodian and the complexity of the linkage, in particular, the number of jurisdictions and datasets involved, as these added layers of complexity in terms of multiple ethics submissions and approvals. Often the frustration stemmed from a lack of or unreliable alternatives, such as hospital episodes of care for a health economic analysis, where the alternative to the administrative data is patient recall.
Skilled staff availability
Many participants, regardless of their position, emphasised that prior experience in data linkage or access to mentoring is crucial, as the skills required are unique. Of the 70 survey participants, 60 (86%) agreed that specific skills are required. Given trials linking to administrative data are already prone to delays, having less experienced staff can lead to further setbacks. Participants felt that resourcing additional time for trial operations staff was important, particularly at the beginning of the trial, to account for increased administrative requirements, and this should increase accordingly with each additional custodian data set request. Several participants also emphasised the importance of having access to staff with strong data manipulation and programming skills to prepare the data, whether it be a statistical programmer, biostatistician or data manager (see subtheme “Data preparation”).
Theme 2 – Assessing burdens versus gains
The burden of additional operational processes and resources, as well as the administrative burden of added consent processes, means that researchers should weigh up the value added by linking to administrative data with the burden of including it in the trial design.
Assessing value added by administrative data
Participants emphasised the trial design, such as the trial population, planned length of follow-up or the number of trial participants, can help decide whether administrative data are preferable over traditional case report form (CRF) data collection. In general, participants agreed that while there are known limitations to administrative data, such as missing data that cannot be followed up through sites, there are instances where it is a preferred data source, such as where the alternative is patient recall, or where researchers seek straightforward data, such as the death status. Some interview participants felt that administrative data can hold great potential for including priority populations who may otherwise not take part in trials due to barriers such as their regional/remote geographical location, language and socioeconomic status. Trial participants who are less likely to attend follow-up appointments are also better suited to be followed up through administrative data.
From a trial coordination perspective, an advantage for sites can be reduced data entry into trial databases for data which will be obtained administratively, with one participant commenting that CRF data collection has its own challenges, such as obtaining timely and complete data. Another interview participant suggested that administrative data should supplement, not replace, CRF data, as some data sources are more difficult to obtain, such as educational and general practice data. Researchers are also influenced by the data custodians’ level of experience in working with researchers, which impacts on the choice of data source. Establishing which data are available through linkage is vital, with more than half of participants mentioning direct discussions with data custodians were the most effective way to gather this information.
Additional participant consent requirements
Although trials involving data linkage often add complexities to the participant consent process, several factors can contribute to a more streamlined consent process. Establishing a consent process for data linkage at the beginning of the trial was viewed as essential to the success of administrative data related outcomes. Challenges included the struggle to have consent forms approved before commencing recruitment, particularly where there are competing interests and a pressure to meet trial milestones. There were instances where trial participant consent to access administrative data was collected as a contingency, despite the uncertainty of funding, particularly where the data would not be required until years later, such as a health economic analysis or long-term follow-up. Overall, it was felt that it was better to wait for the consent form to be approved and finalised as the reconsenting process created a high administrative burden on sites and coordinating staff, as well as confusion for trial participants. There was some frustration with specific or lengthy mandated text in consent forms, as well as poorly designed forms resulting in the need for additional site monitoring to ensure that the consent form information was correct. Programming the consent form into the clinical trial database for site data entry seemed to help with reducing potential errors. Training site staff as well as monitors in how to administer the consent forms, as well as the coordinating centre project team on what to expect from sites, was viewed as crucial as missing information may hinder linkage. Reviewing consent forms early to identify potential errors or low response rates could help to identify sites requiring additional training or processes. While there is increasing uptake of e-consent in trials, acceptability of e-consent amongst data custodians for administrative data access remained unclear. Without e-consent, it was difficult to obtain consent if trial visits were performed remotely.
Theme 3 – Data preparation and analysis
Participants highlighted the technical complexities involved in preparing and analysing administrative datasets, as well as accessing them where it had to be done through a curated data access platform.
Data preparation
Participants responsible for preparing administrative datasets for analysis said it was a skill mostly learnt through trial and error, as datasets are unique, and contained certain intricacies only learnt over time. While data dictionaries supplied by custodians are essential to understanding the datasets, they can vary considerably in terms of quality and detail between custodians. Deciphering classification and coding systems, such as the International Classification of Diseases and Diagnostic Related Groups, were particularly challenging for researchers, as knowledge in classification and coding is a niche skill and researchers did not always have access to an experienced clinical coder, skilled in understanding historical changes to the coding of diagnoses and procedures when analysing longitudinal data. The large size of the datasets and lengthy computer processing times for analyses was something those working with the data needed to be prepared for, even with the help of a high-performance computer and the highest speeds available through virtual trusted research environments.
Mandated data access platforms
Participants’ experiences with receiving data from custodians varied considerably depending on the custodian and their requirements. Some custodians permitted data sharing via a secure download, while others only allowed access through a curated data platform. Curated platforms enable secure sharing and analysis of sensitive data and are managed independently by universities or research institutes rather than the custodians themselves; therefore, researchers are required to enter into separate agreements with the platform provider. Besides the cost (see subtheme “Cost of obtaining data”), issues around using curated data platforms ranged from challenges in setting it up, the speed of running a program and the difficulty in retrieving results. Participants who were aware of plans of having a single national linked data system, with all data stored centrally, said they hoped it would make the process more seamless.
Theme 4 – Training and support
Those involved in operationalising a trial with linked administrative data, such as project managers and data managers, wanted to be better equipped to respond to investigator requests for data linkage in their trials and felt that they would have benefited from better training and support.
Adequate internal training and procedures
Of the 70 survey participants, 51 (73%) reported following some form of written guidance; however, 39 (77%) of these also faced challenges or felt that processes could be improved. Of the 19 (27%) participants who did not follow written guidance, 15 (79%) felt that guidance would have been helpful. Of the 51 survey participants who followed written guidance, 40 (78%) used procedures or guidance directly provided by the data custodian and 17 (33%) used their own institutions procedures. Resources such as standard operating procedures, templates and checklists were seen as ideal tools to have access to and could be broadly written to account for the variability across custodians and data sets. Participants felt that understanding the terminology around data linkage would be particularly helpful, and some participants developed their own guidelines which they shared with colleagues when requested. Interview responses varied when asked whether they understood the purpose of data linkage, and unsurprisingly, it was those who were involved in the operations of the trial who felt that they had the most limited understanding, despite being responsible for overseeing the process to ensure it functioned well.
Access to support
Participants, particularly in larger research centres, felt that resourcing a local subject matter expert would be beneficial as they could be consulted during concept and protocol development and could provide a framework for how to apply data linkage processes within their institutions. Some participants had access to support networks within their institutions, of whom questions could be asked and resources shared. A fixed contact within the custodian unit was seen as helpful for obtaining timely information about the data request process, with 45 of the 70 survey participants (64%) receiving some form of support from a data custodian.
Discussion
This study contributes to enhancing the use of administrative data in trials by offering insights into the experiences of clinical trialists and trial operations staff across diverse roles, expertise and therapeutic areas. The issues identified in the Australian context broadly align with those reported in previously published studies from international jurisdictions (Toader et al., 2024; Williams et al., 2023). Some challenges are specific to Australia. A large proportion of health data are collected by individual Australian states, and there is limited uniformity between state collections. Further, Australia does not have a centralised health record, and the movement towards this (Australian Digital Health Agency, 2024) has been met with mixed reactions from Australians (Knaus, 2019), which has been further impacted by data breaches across the country (Department of Home Affairs, 2024). Internationally, there are initiatives pushing for more standardised planning for trials using linked administrative data, including the SPIRIT-ROUTINE extension for protocols of trials conducted using administrative data (McCarthy et al., 2021), CONSORT-ROUTINE for the reporting of randomised controlled trials using cohorts and administrative data (Kwakkenbos et al., 2021), and RECORD for reporting of observational studies using administrative data (Benchimol et al., 2015). These guidelines aim to assist the transparency and quality of trial reporting.
Progress has been made within Australia in recent years to streamline access to administrative data for research purposes. The National Health Data Hub (Australian Institute of Health and Welfare, 2024) and the National One Stop Shop (Australian Commission on Safety and Quality in Health Care, 2024) both aim to eliminate redundant processes through centralisation of datasets, and the introduction of The Data Availability and Transparency (DAT) Act 2022 (Australian Government, 2023) aimed to create and support transparency and data sharing in Australia and provides a searchable government data catalogue for policy makers and researchers. This Act also introduced the role of the National Data Commissioner. The formation of a network of researchers with the stated goal of offering a coordinated approach for researchers to engage with the National Data Commissioner and contribute to shaping the national data sharing and release framework (University of New South Wales, 2018) represents another initiative that has empowered researchers by involving them in addressing a shared challenge, and funding was recently awarded to create better services for clinical trials using administrative data (University of Western Australia, 2023). Although these initiatives were viewed as positive steps forward by some interview participants, they noted that they were yet to observe significant changes in practice. The Australian Research Data Commons (ARDC, 2025), who were involved in discussions with the Office of the National Data Commissioner to implement the DAT Scheme, recently commented that data access continues to be a challenge, and that the Act has not been successful in its goal of increasing access for researchers.
The themes identified in this study were consistent across survey and interview participants, despite the broad range of researcher roles and experiences. Overall, participants agreed the process of obtaining data, while cumbersome, was worth repeating where the data are crucial to analysing trial outcomes; however, they expressed a strong desire for a more streamlined and accessible process.
Strengths and limitations
A key strength of this study was the in-depth one-on-one interviews, which enabled researchers to speak candidly and allowed the interviewer to delve deeper into responses. Further, surveying researchers allowed the capture of a larger number of participant views.
A limitation of this study was that clinical trials were the focus; however, many of the themes could be applicable to other study designs that utilise administrative data, such as prospective cohorts or implementation trials. An additional limitation may be the generalisability of study findings, as it is possible that those surveyed and interviewed represented individuals who faced more difficulties and were keen to share their experiences, compared with researchers who found the process straightforward. While the interviewer’s background provided valuable insight into the topic, during interviews it may have introduced a potential bias toward promoting efficient and positive data management practices. The use of snowball sampling excluded the possibility of assessing response rates.
Implications for practice and future research
Researchers can facilitate a streamlined outcome by establishing a well-planned process for submitting applications, which should involve regular and early communication with data custodians, while maintaining a detailed record of decisions and conversations, given the potential delays in between decision making and obtaining the data. Additionally, institutions are well-equipped to manage trials linking to administrative data if they have access to local expertise and provide comprehensive training and procedures.
Conclusion
This study presents candid experiences of researchers in administrative data use, and practical information for Australian researchers when considering applicability of administrative data to their own clinical trials. Primarily, researchers should assess whether obtaining administrative data is worthwhile, considering the fees and charges, availability of skills, time to obtain and analyse data and whether the data are more accurately captured through administrative data or through traditional data collection. Further, researchers should be aware of the additional processes required to access administrative data, including lengthy application processes, extra requirements for participant consent and the mandated use of data access platforms. Consideration of these factors will enable researchers to make pragmatic decisions around the inclusion of jurisdictions and datasets in their trial. Considerations are best made during the planning stages of the trial, so that complexities, such as reconsenting trial participants and overspending on trial budgets, are avoided.
Supplemental Material
sj-docx-1-him-10.1177_18333583251413652 – Supplemental material for Using linked administrative data: Insights and tips from academic clinical trialists
Supplemental material, sj-docx-1-him-10.1177_18333583251413652 for Using linked administrative data: Insights and tips from academic clinical trialists by Salma Fahridin, Karen Bracken, Chi Kin Law and Rachael L Morton in Health Information Management Journal
Supplemental Material
sj-docx-2-him-10.1177_18333583251413652 – Supplemental material for Using linked administrative data: Insights and tips from academic clinical trialists
Supplemental material, sj-docx-2-him-10.1177_18333583251413652 for Using linked administrative data: Insights and tips from academic clinical trialists by Salma Fahridin, Karen Bracken, Chi Kin Law and Rachael L Morton in Health Information Management Journal
Supplemental Material
sj-docx-3-him-10.1177_18333583251413652 – Supplemental material for Using linked administrative data: Insights and tips from academic clinical trialists
Supplemental material, sj-docx-3-him-10.1177_18333583251413652 for Using linked administrative data: Insights and tips from academic clinical trialists by Salma Fahridin, Karen Bracken, Chi Kin Law and Rachael L Morton in Health Information Management Journal
Footnotes
Acknowledgements
The authors thank all of the survey and interview participants for their time and thoughtful responses and contribution to the article.
Accepted for publication December 20, 2025.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
