Abstract
Background:
The recent availability of infliximab subcutaneous biosimilar in Europe represents a significant advancement in therapeutic delivery. Real-world data regarding the new formulation are still limited.
Objectives:
To describe subcutaneous infliximab (scIFX) use in immune-mediated inflammatory diseases (IMIDs), evaluating clinical effectiveness, treatment persistence, and factors influencing drug retention rates (DRRs).
Methods:
Adult patients with IMIDs treated with scIFX either de novo or after switching from intravenous infliximab (ivIFX) were included. Disease activity was assessed using standard indices, and persistence was evaluated through 18-month DRRs.
Results:
In total, 201 patients were included (66.7% women): 68 with psoriatic arthritis (33.8%), 66 axial spondyloarthritis (32.8%), 18 rheumatoid arthritis (9.0%), 18 Behçet’s disease (9.0%), 16 juvenile idiopathic arthritis (8.0%), 10 Takayasu arteritis (5.0%), and 5 other IMIDs (2.5%). Median treatment duration was 14 months (interquartile range 13.0). Ninety-five patients (47.3%) switched from ivIFX, while 106 initiated scIFX de novo, of whom 74 (78.3%) received induction. Median disease activity grades improved from baseline through 24 months (p < 0.001). Fifty-five subjects (27.4%) discontinued scIFX. DRRs were 88.5%, 76.9%, and 68.3% at 6, 12, and 18 months, respectively. DRRs were higher in switchers versus de novo initiators (p = 0.038), also controlling for baseline activity and loading protocol (hazard ratio = 0.25 [95% confidence interval: 0.075–0.848], p = 0.026). De novo patients without induction had lower DRRs (p = 0.033) than those receiving induction, whereas line of biologic therapy (p = 0.066) and body mass index (BMI; p = 0.445) had no effect on scIFX DRRs. Adverse events occurred in 21 patients (10.4%).
Conclusion:
ScIFX appears effective across IMIDs, with sustained retention over time. Prior ivIFX exposure and use of induction protocols are associated with improved persistence, while treatment line and BMI appear not to influence outcomes.
Plain language summary
This study looked at how well a medicine called subcutaneous infliximab (scIFX) works for people with several inflammatory diseases, such as arthritis, Behçet’s disease, and Takayasu arteritis. These diseases cause the immune system to attack the body and lead to pain and inflammation. Infliximab is usually given through a drip (an IV infusion), but the newer form can be injected under the skin, which is quicker and easier. Doctors collected information from 201 adults who received scIFX. About half had already been treated with infliximab through IV infusion and then switched to the injection. The other half started directly with scIFX. Some people starting treatment received extra early doses (induction) to help the medicine work faster. After more than a year of treatment, most people’s symptoms improved and stayed better over time. About 7 out of 10 people were still using scIFX after 18 months. People who had previously used the IV version or received the extra starting doses were more likely to continue treatment. Side effects happened in about 1 in 10 people, but no unexpected safety problems were found. Overall, scIFX seems to be an effective and well-tolerated treatment that many patients can continue long-term. Having used IV infliximab before, or starting with extra doses, may help people stay on the treatment longer.
Introduction
While intravenous infliximab (ivIFX) remains a cornerstone in the treatment of various immune-mediated inflammatory diseases (IMIDs),1,2 the recent approval of its subcutaneous biosimilar formulation (scCT-P13) by the European Medicines Agency 3 represents a significant advancement in therapeutic delivery. Although scCT-P13 has been authorized for the same indications as the IV formulation, its approval was primarily based on clinical trials conducted in patients with rheumatoid arthritis (RA) and inflammatory bowel disease (IBD),4,5 leaving a paucity of data regarding its use across the broader spectrum of rheumatologic disorders. In alignment with a recent Delphi consensus, 6 scCT-P13 has been increasingly recognized not merely as a biosimilar but as a potential biobetter, offering enhanced pharmacokinetics, lower immunogenicity, and greater convenience for patients. This distinction is particularly meaningful considering emerging evidence suggesting that patients weigh the route of administration as heavily as the risk of adverse events (AEs),7,8 with subcutaneous delivery being strongly preferred, especially at treatment initiation or following multiple biologic failures. 9 Against this backdrop, the present multicenter Italian study provides a comprehensive real-world assessment of scCT-P13 across a wide range of rheumatologic conditions, with the aim of evaluating its clinical effectiveness, treatment persistence, and safety profile in a diverse patient population.
Methods
A multicentric observational study was conducted at six Italian rheumatology centers. The general scope of the study was to provide a snapshot of scCT-P13 uses in IMIDs, including both licensed and off-label rheumatological indications. The study aims were (1) to describe cross-sectionally the population at the start of scCT-P13 treatment regarding demographic and clinical characteristics as well as previous therapeutical history; (2) to assess the global impact of scCT-P13 treatment on disease activity over time taking into account the diverse disease diagnoses; (3) to estimate the drug retention rates (DRRs) of scCT-P13 over 18 months in the entire population; (4) to evaluate potential differences in the DRRs based on critical stratifications of the cohort, including sex, nosological groups, body mass index (BMI) classes, baseline disease activity groups, and different therapeutical strategies (de novo scCT-P13 or switch from ivIFX, use of loading dose, combination with conventional Disease-Modifying Anti-Rheumatic Diseases (DMARDs) or monotherapy, line of biologic treatment); (5) to provide safety information on scCT-P13 in patients with immune-mediated diseases.
This study consecutively enrolled all adult patients (⩾18 years) with IMIDs treated with scCT-P13 at six rheumatology units in Italy between 2021 and 2025. Subcutaneous CT-P13 could be prescribed either as first-line IFX therapy or following a switch from ivIFX, according to routine clinical practice. Patients who initiated scCT-P13 before the age of 18 were excluded from the analysis. Data were collected retrospectively in a standardized database. Demographics, clinical, and therapeutic data were recorded at baseline (T0, start of scCT-P13 treatment) and every 6 months thereafter, extending through a 24-month observational period on a routine clinical basis. Regarding axial spondyloarthritis (axSpA), both the radiographic and non-radiographic forms were included, without distinguishing them as separate groups.
Disease activity was assessed with standard activity indexes according to diagnosis: disease activity scores (DAS)28 for RA, disease activity in psoriatic arthritis (DAPSA) for psoriatic arthritis (PsA), ankylosing spondylitis disease activity score (ASDAS)-C-reactive protein (CRP) for axSpA, and clinician’s evaluation (grade 0 = remission, grade 1 = low disease activity, grade 2 = moderate disease activity, and grade 3 = high disease activity) for the other conditions. The following ranges were applied to define disease activity groups based on DAS28: <2.6 (grade 0 = remission), ⩾2.6–⩽3.2 (grade 1 = low disease activity), >3.2–⩽5.1 (grade 2 = moderate disease activity), and >5.1 (grade 3 = high disease activity). The following ranges were applied to define disease activity groups based on DAPSA: ⩽4 (grade 0 = remission), >4–⩽14 (grade 1 = low disease activity), >14–⩽28 (grade 2 = moderate disease activity), and >28 (grade 3 = high disease activity). The following ranges were applied to define disease activity groups based on ASDAS-CRP: <1.3 (grade 0 = remission), ⩾1.3–<2.1 (grade 1 = low disease activity), ⩾2.1–⩽3.5 (grade 2 = disease activity), and >3.5 (grade 3 = high disease activity). When stratifying the cohort by BMI classes, the following ranges were applied: <18.5 (underweight), 18.5–24.9 (normal weight), 25.0–29.9 (overweight), ⩾30 (obese).
Reasons for treatment discontinuation were classified into efficacy-related causes (lack or loss of efficacy) and non-efficacy–related causes, including AEs and patient preference or tolerability concerns. When reporting efficacy-related causes, standard terminology was adopted. The term “lack of efficacy” was used to indicate primary non-response, defined as the absence of a clinically meaningful response within the first 6 months of treatment, whereas “loss of efficacy” referred to secondary non-response, defined as the loss of treatment effectiveness over time after an initial clinical response.
Subcutaneous CT-P13 was administered according to the approved therapeutic regimens for RA, axSpA, and PsA, that is, 120 mg every other week. When a loading protocol was used, patients received 120 mg subcutaneously at baseline followed by injections at weeks 1, 2, 3, and 4, or two intravenous infusions of 3–5 mg/kg 2 weeks apart, followed by initiation of subcutaneous maintenance therapy 4 weeks after the second infusion. In case of switching from intravenous maintenance therapy to scCT-P13, patients initiated the subcutaneous formulation at the time of the next scheduled intravenous dose. For all patients, the choice of loading protocol (no loading doses, intravenous, or subcutaneous loading doses), as well as the therapeutic regimen in cases of off-label use, was determined by the clinician’s judgment, considering patient characteristics and preferences, local clinical practice, and administrative considerations.
All analyses were performed using JASP software (version 0.19.3.0; University of Amsterdam, Amsterdam, The Netherlands). Descriptive statistics (means, medians, standard deviations, interquartile ranges, and ranges, as appropriate) were computed to characterize the study cohort. The Shapiro–Wilk test was used to evaluate the normality of continuous variable distributions. Group differences in categorical variables were assessed using the Chi-square test. For comparisons of continuous variables across more than two independent groups, the Kruskal–Wallis test was employed due to the non-normal distribution of data. Longitudinal comparisons of paired ordinal data were analyzed using the Friedman test (non-parametric repeated measures ANOVA). Time-to-event data (i.e., time to drug discontinuation for reasons other than remission) were analyzed using Kaplan–Meier survival curves, and between-group differences in survival distributions were tested using the log-rank test. Cox proportional hazards models were employed to evaluate predictors of drug discontinuation and to calculate hazard ratios (HRs) and 95% confidence intervals (CIs), accounting for potential confounders. Missing data were addressed via complete-case analysis (listwise deletion). All statistical tests were two-sided, and a p-value < 0.05 was considered indicative of statistical significance.
This study was conducted and reported in accordance with the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) guidelines for cohort studies.
Results
A total of 201 patients were included, 134 women (66.7%) and 67 men (33.3%). Mean ± SD age at the time of enrollment was 49.9 ± 13.8 years (range 18.0–82.0). Sixty-six (32.8%) patients had a diagnosis of axSpA, 68 (33.8%) of PsA, 18 (9.0%) RA, 18 (9.0%) of Behçet’s disease (BD), 10 (5.0%) of Takayasu arteritis (TA), 16 (8.0%) of Juvenile Idiopathic Arthritis (JIA, non-systemic types), and 5 (2.5%) of other IMIDs.
Patients were treated with scCT-P13 for a median duration of 14.0 months (interquartile range (IQR) 13.0; range 2.0–40.0). Among them, 71 (35.3%) were treated with IFX as first-line biologic, 75 (37.3%) as second-line biologic, and 55 subjects were treated as third or more lines of treatment. Ninety-five (47.3%) patients received ivIFX before switching to scCT-P13. Among the 106 patients who started scCT-P13 as a new molecule, an induction protocol was applied in 74 cases (78.3%; intravenously in 45, subcutaneously in 29), while 32 subjects started scCT-P13 every other week without a loading dose. Table 1 provides the characteristics of the population at baseline, that is, at the start of scCT-P13. Table 2 provides a breakdown of the treatments administered before scCT-P13.
Baseline characteristics of the population stratified by diagnosis groups.

The number of available observations is reported in brackets when missing data were present.
BMI, body mass index; IFX, infliximab; IV, intravenous; N., number; SD, standard deviation; SpA, spondyloarthritis.
Breakdown of the treatments administered before scCT-P13 in the population.

DMARD, disease-modifying anti-rheumatic drug.
Disease activity status variations (from grade 0 = remission to grade 3 = high disease activity) measured by the disease activity indexes specific to each diagnosis at scCT-P13 start and at the 6-, 12-, and 18-month timepoints are represented in Figure 1. The median disease activity grade was 2.0 (IQR 1.0, range 0.0–3.0) at the baseline (N = 200), 1.0 (IQR 1.0, range 0.0–3.0) at 6-month follow-up (N = 183), 0.0 (IQR 1.0, range 0.0–3.0) at 12-month follow-up (N = 136), 0.0 (IQR 1.0, range 0.0–2.0) at 18-month follow-up (N = 92), and 0.0 (IQR 1.0, range 0.0–2.0) at 24-month follow-up (N = 61; p < 0.001). The statistical significance was maintained when analyzing separately patients starting scCT-P13 as a new treatment (p < 0.001) and those who switched from ivIFX (p < 0.001). To account for heterogeneity in disease activity assessment between diagnoses evaluated using validated indices (RA, PsA, and axSpA) and those assessed by physician clinical judgment (BD, TA, and JIA), a sensitivity analysis of disease activity variations was restricted to patients with RA, PsA, and axSpA. In this subgroup, the median disease activity grade significantly decreased over time, from 2.0 (IQR 1.0, range 0.0–3.0) at baseline to 1.0 (IQR 1.0, range 0.0–3.0) at 6 months, 0.0 (IQR 1.0, range 0.0–2.0) at 12 months, 0.0 (IQR 1.0, range 0.0–2.0) at 18 months, and 0.0 (IQR 1.0, range 0.0–2.0) at 24 months (p < 0.001).
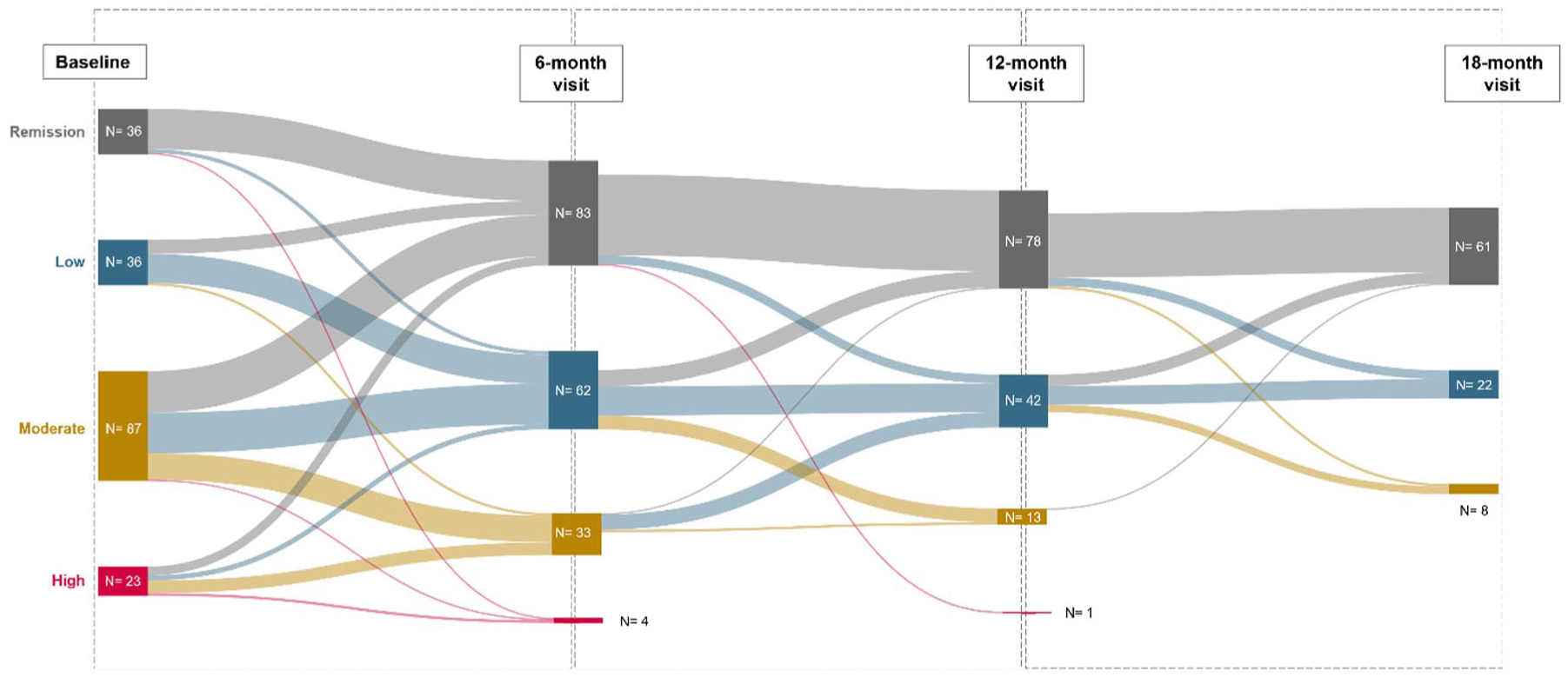
Disease activity status variations measured by the disease activity indexes specific to each diagnosis at scCT-P13 start and at the 6-, 12-, and 18-month timepoints.
The DRRs of scCT-P13 were 88.5% at 6 months (95% CI: 84.1–0.93), 76.9% at 12 months (95% CI: 70.7–83.5), and 68.3% at 18 months (95% CI: 61.0–76.5; Figure 2(a)). They were significantly higher in patients switching from ivIFX than starting scCT-P13 as a new molecule (p = 0.038). Patients starting scCT-P13 without a loading dose had significantly lower DRRs (p = 0.033) than those who received a loading dose (Figure 2(b)). Patients starting scCT-P13 as a new molecule had similar DRRs regardless of the line of biologic treatment (p = 0.066). Similarly, in the whole cohort, there were no statistically significant differences in scCT-P13 DRRs according to BMI class (p = 0.445; Figure 2(c)), sex group (p = 0.18), diagnosis group (p = 0.871), baseline disease activity grade (p = 0.064), and mono- or combination therapy (p = 0.057). In a Cox Proportional Hazards analysis including the use of a loading protocol, biologic treatment line (0 = first-line scCT-P13; 1 = switch from first-line ivIFX; 2 = second-line scCT-P13; 3 = third or more line scCT-P13), and baseline disease activity (0–4 grades) as factors, switch from first-line ivIFX resulted a protective factor from discontinuation compared to first-line scCT-P13 (HR = 0.25 (95% CI: 0.075–0.848) p = 0.026), the use of a loading scheme was borderline protective (HR = 0.55 (95% CI: 0.298–1.016) p = 0.056), while disease activity was not statistically significant in the model. The DRRs of scCT-P13 at 6-, 12-, and 18-month follow-up in patients affected by different diseases are reported in Table 3.

Survival curve of scCT-P13 in the entire cohort (a), split by loading protocol and previous exposition to ivIFX (b), and split by body mass index classes (c).
DRRs of scCT-P13 at 6-, 12-, and 18-month follow-up in patients affected by different diseases.
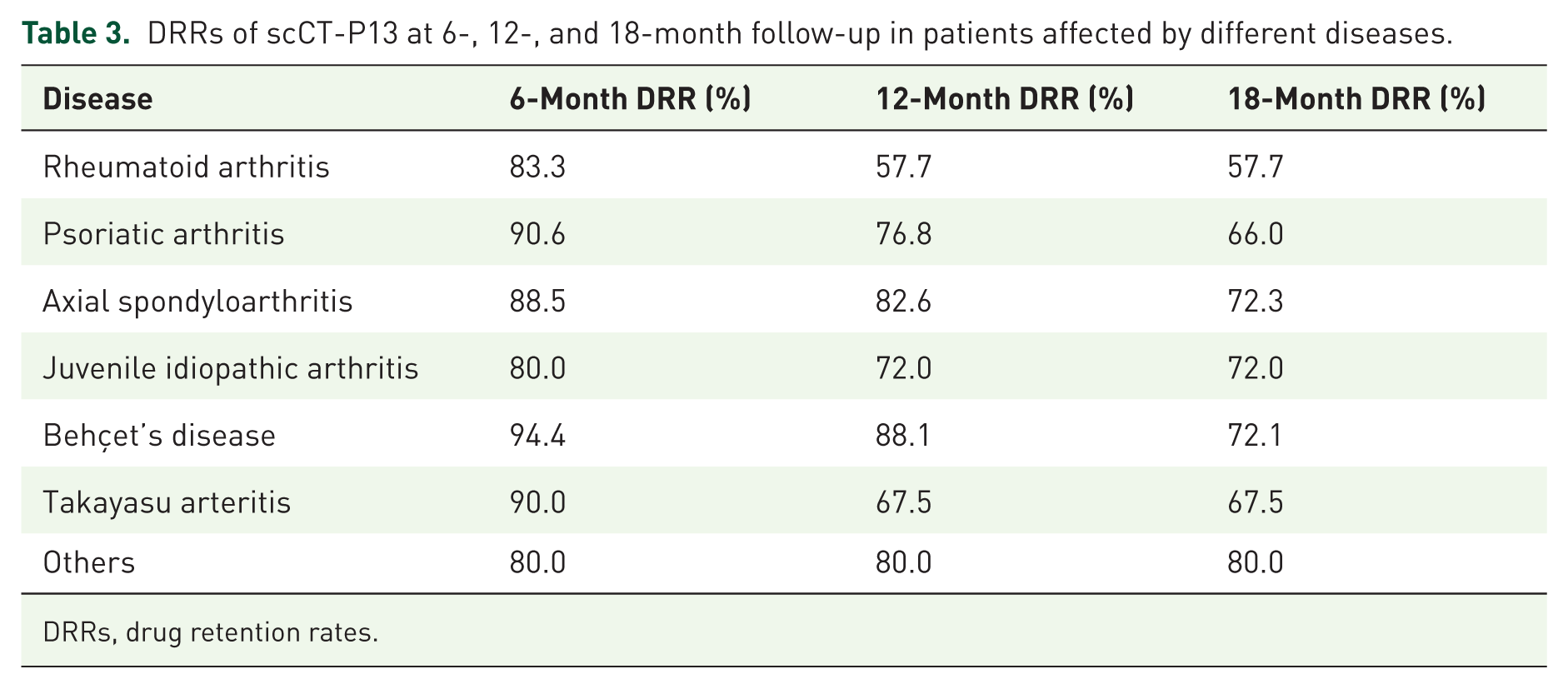
DRRs, drug retention rates.
Fifty-five subjects (27.4%) discontinued scCT-P13 during the observation time: 10 for lack of efficacy (5.0%), 22 for loss of efficacy (10.9%), 13 for AEs (6.5%); no patients discontinued the drug because of personal preference or tolerability concerns, while reasons were not available for 10 patients (5.0%). Discontinuation occurred within the first 6 months of treatment in 25 cases (45.5%), within the first 12 months in 41 cases (74.5%), and after the first 12 months in 14 cases (25.5%).
AEs were recorded in 21 patients (10.4%), of whom 5 experienced serious AEs (2.5%). A detailed breakdown of AEs is reported in Table 4. The overall incidence rate of AEs was 10.0 events per 100 patient-years.
Adverse events reported in the cohort.

Discussion
The present work provides a comprehensive real-world evaluation of scCT-P13 across a wide range of rheumatologic conditions in an Italian multicenter cohort.
We included 201 patients with a median follow-up of 14 months, encompassing a wide range of rheumatologic diseases, including axSpA, PsA, RA, BD, TA, JIA, and other conditions. Notably, this is the first report evaluating scCT-P13 across various treatment strategies, including first- and later-line use, transition from ivIFX to SC maintenance, SC induction followed by maintenance, and initiation directly with the subcutaneous maintenance dose.
In RA, previous phase I/III studies have demonstrated the non-inferiority of scCT-P13 compared to ivIFX when started after intravenous induction, with post hoc analyses suggesting even superior efficacy outcomes compared to continued intravenous administration. 10 A recent systematic review and meta-analysis confirmed the favorable performance of scCT-P13 over IV IFX, adalimumab, and etanercept in patients with moderate-to-severe RA, reporting higher efficacy at weeks 30 and 54. 11 Evidence for axSpA and PsA remains limited to small case series. In one such study, 11 axSpA patients switched from ivIFX to scCT-P13: 5 (45.5%) maintained SC therapy at 14.7 months, while 6 (54.5%) reverted to the intravenous formulation due to perceived loss of efficacy, although CRP levels remained stable. 12 Among seven PsA patients, 57.1% continued subcutaneous treatment at follow-up, with some reporting clinical benefit; the remainder switched back due to a similar perception of reduced efficacy. 13 A recent study by Viazis et al. examined the outcomes of patients with IMIDs following the switch from intravenous to subcutaneous formulation. In this study, all patients transitioning to scCT-P13 were in clinical remission or, in the case of those with RA, axSpA, and PsA, at least in a state of low disease activity. The cohort included 344 patients (CD = 136, UC = 62, axSpA = 52, PsA = 38, RA = 7, PsO = 44, co-existence of more than one disease = 5). After a mean ± SD follow-up period of 8 ± 4 months, 12 patients (3.5%) discontinued treatment with scCT-P13. Among these, one had PsA, one had RA, two had axSpA, and three had SpA associated with an IBD. The authors concluded that switching from ivIFX to scCT-P13 was a valid and safe strategy for maintaining long-term remission in patients with various IMIDs. 14
Data on TA, BD, and JIA are particularly scarce despite the well-established role of anti-TNF therapy in these conditions,15–19 as neither ivIFX nor scCT-P13 is currently approved for these indications. Although no published studies to date have evaluated scCT-P13 in these diseases, ivIFX has shown promising results. A prospective study in 12 TA patients demonstrated complete or partial remission in 9 cases (81.8%) at week 30 and a reduction in inflammation detected by positron emission tomography (PET). 20 Similarly, the TAKASIM study reported a 90.4% treatment persistence rate at 52 weeks in 23 refractory patients, with significant glucocorticoid-sparing effects and PET-confirmed remission in all patients at the end of follow-up. 21 In BD, while ivIFX is well-documented across various organ involvements,16,22–25 only one study has explored the use of scCT-P13: 7 patients with refractory uveitis showed reduced relapse rates and a 71.4% remission rate at 6 months, with only 1 AE reported. 26 In both BD and TA, infliximab is often administered at higher doses than those typically used in the treatment of RA, PsA, and axSpA, to achieve greater efficacy across various organ manifestations.27,28 In this regard, the higher and more stable plasma concentrations of infliximab achieved through subcutaneous administration of scCT-P13 11 may offer a valuable therapeutic option for patients with these complex and refractory conditions. Additionally, a retrospective study of 26 pediatric patients with non-infectious uveitis, including 16 with JIA and 3 with BD, treated with ivIFX, demonstrated high remission rates (95.4% of affected eyes), fewer flares, and an AE rate of 26.9% over 14 months. 29
Patients in our cohort were predominantly female (66.7%) with a mean age of 50 years. This female predominance should be interpreted in light of the heterogeneous diagnostic composition of the study population. The included diseases are characterized by distinct and well-established sex distributions, with a marked female predominance in RA, JIA, and TA, a more balanced distribution in PsA, and a relative male predominance in axSpA and BD. Therefore, the overall male–female ratio likely reflects the relative representation of each diagnosis rather than a potential selection bias. For the same reason, sex-based comparisons at the level of the entire cohort should be approached carefully.
In relation to the composition of our cohort, the preferential use of infliximab among patients with axSpA and PsA is unsurprising. This trend likely reflects the more limited range of therapeutic options available with distinct mechanisms of action in these conditions, particularly when compared to RA. Furthermore, the increased prevalence of associated comorbidities—such as IBDs, psoriasis, and anterior uveitis—may also contribute to the observed prescribing patterns. Most patients received scCT-P13 as first- or second-line therapy, often following prior treatment with anti-TNF, anti-IL17, or anti-IL6 agents. Approximately half switched from ivIFX, and most underwent subcutaneous induction before transitioning to maintenance dosing. In over half the cases, scCT-P13 was administered in combination with conventional synthetic (cs) DMARDs, primarily methotrexate.
Disease activity analysis showed that the proportion of patients in remission increased from 17.9% at baseline to 30.3% at 18 months, with corresponding reductions in the proportions with moderate and high disease activity. Importantly, improvements were observed in both patients starting scCT-P13 de novo and those switching from intravenous to subcutaneous formulation.
At 18 months, the overall DRR was 68.3%, consistent with retention rates reported in real-world studies of other biologic and targeted therapies across immune-mediated rheumatic diseases. When stratified by disease, the highest DRRs were observed in patients with axSpA, BD, and JIA, all exceeding 70%, whereas lower values were noted in PsA and especially in RA, with DRRs of approximately 66% and 58% at 18 months, respectively. These differences should be interpreted with caution. The sub-cohort of enrolled patients with RA was smaller in size and characterized by a higher proportion of individuals receiving advanced lines of therapy and combination regimens, thereby representing a difficult-to-treat patient subgroup. Notably, none of the patients with RA were in remission at the time of scCT-P13 initiation. However, when considering the whole cohort, DRRs did not significantly differ according to whether scCT-P13 was used as first-, second-, or later-line biologic therapy. It should also be considered that patients with axSpA, BD, and JIA may be more likely to persist with treatment, partly due to the more limited availability of alternative advanced therapeutic options compared with RA and PsA.
Regarding treatment strategies, DRRs were significantly higher in patients who switched from ivIFX to scCT-P13 compared to those who started scCT-P13 de novo with subcutaneous induction, and even lower in those initiating scCT-P13 de novo without the recommended induction phase. Although multivariate analysis underscored the greater impact of prior IFX exposure compared with the loading protocol, our findings are consistent with pharmacokinetic data from preliminary studies demonstrating higher trough levels following intravenous induction.4,5 It must be acknowledged that the decision to administer or omit a loading dose was not standardized across centers and may have reflected local clinical practice or administrative constraints. This therapeutic heterogeneity could have influenced drug retention and outcomes, particularly during the first 6 months of therapy. However, after 6 months, median disease activity had significantly decreased, suggesting a rapid onset of efficacy regardless of the initial strategy. Most discontinuations (74.5%) occurred within the first 12 months, more frequently because of secondary loss of efficacy (10.9%) than primary failure (5.0%). Given the clinical relevance of these observations, further prospective studies are needed to define the optimal induction strategy with scCT-P13.
On the other hand, the high DRR observed among patients switching from stable ivIFX to scCT-P13 may have implications for clinical decision-making. It’s important to acknowledge that switchers do not represent a randomized group and are likely enriched for patients with prior treatment success (good response, tolerability, established adherence, and clinical stability), factors that may inherently predict persistence. However, our data support considering a switch in clinically stable patients given the expected benefits, such as reduced need for infusion visits, potential optimization of healthcare resources, and alignment with patient preference.
No significant effect of BMI classes was observed on DRRs in our cohort, consistent with the stable pharmacokinetics demonstrated by scCT-P13. 30 This finding is of considerable interest, as it is well documented that the efficacy of TNF inhibitors tends to decline with increasing BMI. 31 ivIFX, administered at a weight-adjusted dose, may offer a partial solution to this limitation, and one could hypothesize that switching to a fixed-dose subcutaneous formulation might be less advantageous in this patient population. However, data from our cohort suggest the contrary, possibly due to the higher and more stable plasma drug concentrations achieved with the subcutaneous formulation.30,32 Nonetheless, further studies are warranted to elucidate this clinically relevant issue.
Interestingly, RA patients showed similar DRRs regardless of concomitant conventional DMARDs use, a finding that may reflect reduced immunogenicity of scCT-P13 compared to ivIFX, potentially minimizing the need for combined immunosuppressive treatment. 4
Safety data on scCT-P13 use in the literature are limited but globally favorable. In our cohort, a total of 21 patients (10.4%) experienced AEs, with an incidence of 10 events per 100 patient-years. The most common AEs were urticarial reactions and upper respiratory tract infections. Notably, no serious infections or tuberculosis reactivations were observed. Severe AEs occurred in 2.5% of participants: these included isolated cases of interstitial lung disease, lupus-like syndrome, and supraventricular tachycardia, as well as one relapse of colon cancer and one death due to myocardial infarction in a patient with high cardiovascular risk. Among the severe AEs, the lupus-like syndrome was considered potentially related to the study treatment, whereas the remaining events were deemed unlikely to be treatment-related. Treatment was discontinued in 13 patients (6.5%) due to AEs. These findings support a favorable safety profile, comparable to that reported in the literature for both ivIFX and scCT-P13.3,4,10,11,30
Strengths and limitations of the study
This study fills a gap in the existing literature by providing real-world evidence on scCT-P13 treatment in inflammatory rheumatic diseases. Its major strengths are the inclusion of low-prevalence diseases and the evaluation of multiple treatment strategies. Limitations relate to its retrospective observational design and the relatively small sample size in some disease subgroups. Also, the generalizability of our findings might be limited by the heterogeneity of underlying diseases and the potential for confounding by indication, including patient selection for switching versus de novo initiation and the choice of induction regimen.
An additional limitation relates to heterogeneity in disease activity assessment across diagnoses, as validated composite indices were available for RA, axSpA, and PsA, whereas disease activity in other conditions relied on physician clinical judgment, potentially introducing inter-observer variability. However, a sensitivity analysis restricted to diseases with standardized activity measures yielded results consistent with the primary analysis, supporting the robustness of our findings.
Longitudinal disease activity data were increasingly missing at later follow-up time points, largely due to treatment discontinuation or censoring, which may limit the interpretability of long-term effectiveness outcomes. In addition, baseline covariates (notably BMI class and concomitant combination therapy) were missing in a subset of patients; although a conservative complete-case analysis suggested no relevant impact on drug retention, some residual bias and reduced generalizability cannot be excluded.
Nonetheless, this study provides the first real-life assessment of scCT-P13 across a broad range of rheumatologic conditions, filling an important gap in current literature and highlighting the prominent role of this treatment in both approved and off-label indications.
Conclusion
In conclusion, this multicenter real-world study provides novel and supportive data regarding the utility of scCT-P13 across a broad spectrum of rheumatologic conditions, including low-prevalence diseases. The observed clinical efficacy, sustained treatment persistence, and favorable safety profile across multiple indications underscore the value of scCT-P13 as a therapeutic option beyond RA and IBDs. Subcutaneous administration offers additional potential benefits in terms of patient autonomy, satisfaction, and treatment adherence, factors increasingly recognized as critical in the long-term management of chronic disease. In particular, the administration of subcutaneous IFX following an induction phase appears to be associated with lower discontinuation risk. Further prospective studies are warranted to validate these findings and clarify the role of scCT-P13 within individualized treatment strategies for rheumatologic diseases.
