Abstract
Background:
Older age is a risk factor for serious infection (SI) associated with biologic or targeted synthetic DMARDs (b/tsDMARDs) use in rheumatoid arthritis (RA). Among older adults with RA, one-third are diagnosed at ⩾60 years (late-onset RA, LORA), while others are diagnosed earlier (young-onset RA, YORA). LORA is characterized by a more acute onset and heightened inflammatory burden due to age-related immune dysregulation. Whether RA onset age independently affects infection risk with b/tsDMARDs remains unclear.
Objective:
Evaluate SI risk in older adults with RA initiating b/tsDMARDs, stratified by RA onset age: LORA versus YORA.
Design:
Retrospective cohort study using prospectively collected registry data.
Methods:
From FORWARD, the National Databank for Rheumatic Diseases (2001–2019), we identified RA patients aged ⩾60 years who initiated (1) anti–TNF then (2) subsequent non-TNF b/tsDMARDs. Patients were categorized as LORA versus YORA and matched using kernel-based propensity scores. SI was defined as an infection requiring hospitalization, intravenous antibiotics, or death. Multivariable Cox models estimated the risk of SI in LORA versus YORA.
Results:
Among 1379 LORA and 2727 weighted YORA patients initiating anti-TNF therapy, the crude incidence of SI was 28.2 and 20.5 per 1000 person-years. In adjusted models, LORA was not associated with increased anti-TNF-related SI risk compared to YORA (HR 0.82, 95% CI 0.63–1.02). Among 198 LORA and 675 weighted YORA patients subsequently initiating non-TNF b/tsDMARDs, the crude incidence of SI was 17.3 versus 19.5 per 1000 person-years. In adjusted models, LORA was not associated with increased non-TNF related SI risk (aHR 0.81, 95% CI 0.31–2.12). Across cohorts, older age was independently associated with increased SI risk. For anti-TNF, prior SI and recent long-term glucocorticoid use, and for non-TNF, HAQ disability were also associated with SI.
Conclusion:
Age at RA onset was not independently associated with SI risk following b/tsDMARD initiation. Risk stratification should prioritize functional status and treatment history.
Plain language summary
Older adults with rheumatoid arthritis (RA) are known to have a higher risk of serious infections when treated with biologic or targeted synthetic medications. However, it is unclear whether this risk is influenced by the age at which RA first develops. Some people develop RA later in life, while others are diagnosed earlier but continue treatment into older age.
In this study, we examined adults aged 60 years and older with RA who started biologic or targeted therapies. We compared people whose RA began at age 60 or later (late-onset RA) with those who were diagnosed earlier in life (young-onset RA). We evaluated the risk of serious infections, defined as infections requiring hospitalization, intravenous antibiotics, or resulting in death.
We found that the risk of serious infection did not differ meaningfully based on whether RA began earlier or later in life, both among patients starting anti-TNF therapies and among those starting other biologic or targeted treatments after prior anti-TNF use. Instead, infection risk was more strongly related to other factors, including older age, a prior history of serious infection, worse physical function, and recent long-term use of glucocorticoids (steroids).
These findings suggest that, in older adults with RA, infection risk is influenced less by the age at which RA began and more by functional status and treatment-related factors. Efforts to reduce long-term steroid use, optimize disease-modifying therapy, and support physical function may be important strategies to lower infection risk in this population.
Keywords
Introduction
As global life expectancy increases, the number of older adults living with rheumatoid arthritis (RA) continues to grow. 1 In this population, RA may reflect long-standing disease that began earlier in life, referred to as young-onset RA (YORA), or new disease developing at older ages, typically at or after age 60 or 65, referred to as late-onset RA (LORA). LORA, first formally described in 1994, 2 is characterized by a more abrupt symptom onset, constitutional symptoms, and predominant large joint involvement.3–5 Among Medicare beneficiaries, the average age at LORA diagnosis is in the mid-70s, underscoring its clinical importance in the aging RA population. 6
Older adults, particularly those with LORA, are less likely to receive standard-of-care treatment with disease-modifying anti-rheumatic drugs (DMARDs), especially biologic and targeted synthetic agents (b/tsDMARDs).6–8 This is despite evidence demonstrating the effectiveness and tolerability of b/tsDMARDs in older adults. Data from international registries have shown comparable rates of remission, drug retention, and treatment discontinuation due to adverse events among older adults receiving b/tsDMARDs, and between LORA and YORA patients.9–12
Serious infection (SI) is a major concern when prescribing b/tsDMARDs.13–15 While SI risk has been broadly characterized in rheumatoid arthritis (RA), older adults are often underrepresented in both clinical trials and large observational cohorts due to exclusion criteria based on age or comorbidities. 16 As a result, the applicability of existing evidence to this growing patient population remains limited. Furthermore, few studies have specifically evaluated whether age at RA onset (i.e., LORA vs YORA) modifies the risk of SI in older adults receiving b/tsDMARDs.
In clinical practice, RA treatment with b/tsDMARDs typically follows a stepwise approach, beginning with anti–tumor necrosis factor (anti-TNF) agents and advancing to non-TNF b/tsDMARDs in cases of inadequate response or adverse events. This treatment pattern aligns with clinical guidelines 15 and real-world prescribing practices across age groups. However, infection risk may vary over the course of treatment due to cumulative immunosuppressive exposure, including glucocorticoids (GCs), and prior history of SI. 17 Therefore, evaluating SI risk at multiple treatment stages is essential for a more comprehensive and clinically meaningful understanding of infection risk in older adults with RA.
In this study, we conducted a two-part observational analysis using data from FORWARD, a U.S.-based longitudinal cohort. First, we evaluated the risk of SI following initiation of anti-TNF therapy in older adults with RA, comparing outcomes between those with LORA and YORA. Second, we assessed SI risk following the subsequent initiation of non-TNF b/tsDMARDs among individuals with prior anti-TNF exposure.
Methods
We conducted an observational cohort study to evaluate the risk of SI in older adults with RA, stratified by age at disease onset. The study included two parts: (1) evaluation of SI risk following initiation of anti-TNF therapy and (2) evaluation of SI risk following subsequent initiation of non-TNF b/tsDMARDs.
Data were drawn from FORWARD, the National Databank for Rheumatic Diseases, a U.S.-based, prospective, longitudinal observational registry. 18 FORWARD collects biannual self-reported data from patients with physician-confirmed rheumatic diagnoses. Available data include sociodemographic, comorbidities, medication use, healthcare utilization, and patient-reported outcomes. Diagnoses are confirmed at enrollment, and outcomes are validated using medical records and linkage to national mortality databases.
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 19
Study cohort
We identified adults aged ⩾60 years with physician-confirmed RA who initiated a b/tsDMARD between January 1, 2001 and December 31, 2019. The study period was restricted to the end of 2019 to avoid bias introduced by the COVID-19 pandemic, which substantially altered infection risk and immunosuppressive prescribing practices. Participants were required to have completed at least two semiannual FORWARD questionnaires to ensure adequate follow-up.
Patients were classified into two groups based on age at RA diagnosis: LORA, defined as diagnosis at age 60 or older, and YORA, defined as diagnosis before the age of 60. The age threshold used to define LORA varies across studies, with cutoffs of either 60 or 65 years commonly reported, often depending on the data source.20,21 For example, studies using claims data typically use age 65 to reflect Medicare eligibility. In this study, we defined LORA as RA diagnosis at age 60 or older, consistent with prior registry-based studies, and because FORWARD includes patients regardless of insurance status.
Individuals with baseline diagnoses of psoriatic arthritis, ankylosing spondylitis, systemic lupus erythematosus, inflammatory bowel disease, cancer, or HIV were excluded.
Exposure definition: RA treatment
Anti-TNF initiation
For the first part of the study, we identified individuals initiating anti-TNF therapy (etanercept, adalimumab, infliximab, certolizumab, or golimumab) at age ⩾60, with or without concurrent conventional synthetic DMARDs (csDMARDs). New use was defined as no reported anti-TNF use at enrollment and first reported use at a subsequent follow-up. All anti-TNF agents were grouped into a single therapeutic class; switching among anti-TNFs was treated as continuous exposure.
Subsequent non-TNF b/tsDMARD initiation (after anti-TNF exposure)
In the second part of the study, we identified patients who initiated a non-TNF b/tsDMARD at age ⩾60 after anti-TNF therapy, regardless of prior SI history. The non-TNF b/tsDMARDs of interest were abatacept, tocilizumab, Janus kinase inhibitors (JAKi; tofacitinib, upadacitinib, or baricitinib), anakinra, and rituximab. This nested subgroup analysis evaluated the incremental risk of SI after switching therapies.
For both parts, the index date was defined as the first questionnaire at which the relevant treatment initiation was reported: (1) anti-TNF initiation or (2) non-TNF b/tsDMARD initiation was reported.
Outcome and follow-up
The primary outcome was the first occurrence of an SI after the index date, defined as any infection requiring hospitalization, intravenous antibiotics, or resulting in death. Events were identified based on patient self-report and underwent a structured validation process using medical record review, physician report, and a linkage to the US National Death Index when available. Validation status was classified as strong (hospital records, other medical records, physician-reported) or moderate (patient-reported physician diagnosis or self-diagnosis); events with pending validation (<1%) were excluded from analysis.
Follow-up continued from the index date until the earliest of the following: discontinuation of the treatment (defined as ⩾90 days without use) plus 3 months (to allow for SI risk window, except rituximab where the risk window was set at 12 months), switch to a different therapeutic class, death, loss to follow-up, occurrence of an SI, or the end of the study period on December 31, 2019.
Covariates
Covariates were assessed at enrollment and at the treatment index date. Sociodemographic variables included age at treatment initiation, sex or gender, race and ethnicity, educational attainment, annual household income, rural versus urban residence, and insurance type (categorized as Medicare vs other). Clinical variables included RA disease duration, prior history of SI, smoking status, functional status, and comorbidity burden, measured using the Rheumatic Disease Comorbidity Index (RDCI).22,23 Disease activity was assessed using the Patient Activity Scale (PAS), a composite index incorporating the HAQ score, patient global assessment, and pain score. In a previous FORWARD study, a PAS score of 3.7 or lower was defined as remission or low disease activity, and a score greater than 3.7 was defined as moderate or high disease activity.14,24
Medication-related covariates included concurrent methotrexate use and GC exposure. GC use was categorized using several binary indicators, including current and long-term use. Long-term GC use was defined as receipt of a prednisone-equivalent dose of at least 5 mg/day for at least 90 consecutive days.25,26 For the primary multivariable time-to-event analyses, GC use was modeled as a time-varying covariate using a recent long-term GC definition to capture sustained higher-dose exposure proximal to the outcome (i.e., SI). Recent long-term use was defined as receipt of ⩾5 mg/day prednisone equivalent for at least 90 consecutive days during the 90-day window immediately preceding the SI event or censoring.
Statistical analysis
To address potential confounding from baseline differences between patients with LORA and YORA, we applied kernel-based propensity score matching (PSM). Propensity scores were estimated separately for each treatment cohort using logistic regression models that included: age at treatment initiation, sex or gender, race and ethnicity, Medicare coverage, RDCI score, and HAQ disability score. Matching was conducted twice: once for patients initiating anti-TNF therapy and again for those initiating non-TNF b/tsDMARDs after prior anti-TNF use. Kernel matching with an Epanechnikov kernel function (bandwidth 0.06, Stata default specification) was used to match each LORA patient to a weighted average of YORA comparators, optimizing statistical efficiency and reducing bias relative to nearest-neighbor methods. 27 Covariate balance before and after matching was evaluated using standardized mean differences.
After matching, Cox proportional hazards models were used to estimate hazard ratios (HRs) for SI in LORA versus YORA patients with robust standard errors to account for the matched setting. Separate models were developed for (1) anti-TNF initiation and (2) subsequent non-TNF b/tsDMARD initiation. The proportional hazards assumption was assessed using Schoenfeld residuals and log–log survival plots. Where proportionality was violated, we employed time-dependent covariates or stratified Cox models as appropriate.
Missing data were addressed using targeted imputation strategies. Hot-deck imputation was performed for demographic variables, including ethnicity, educational level, marital status, smoking status, and total income. Sex was imputed when missing using pregnancy- and menstrual-related variables. HAQ was completed using HAQ-II when available and otherwise imputed using hot-deck methods informed by other patient-reported outcomes. Comorbidity burden (RDCI score) was imputed using hot-deck methods based on individual comorbidities, total joint replacement status, body mass index, number of painful joints, and total income.
Time-varying treatment variables were handled using last observation carried forward when missing intervals were flanked by observation periods with the same treatment status. Overall, methotrexate and glucocorticoid use had less than 3% missing observations.
Sensitivity analyses were performed to assess the robustness of findings, including alternative definitions of GC use (e.g., recent long-term use vs current use). To assess potential survivor bias among patients with YORA who initiated biologic therapy at older ages, we conducted a sensitivity analysis restricting the sample to patients with shorter RA duration (⩽5 years) at treatment initiation, using the same model specifications as in the primary analyses. In addition, competing risk sensitivity analyses were performed using Fine–Gray subdistribution hazard models, treating all-cause mortality as a competing event for SIs, given the potential influence of death on SI risk estimates in older populations.
All statistical analyses were conducted using Stata MP 17.0, and two-sided
Results
We present findings separately for the two parts of the study: (1) risk of SI following anti-TNF initiation and (2) risk of SI following subsequent non-TNF b/tsDMARD initiation among those previously treated with anti-TNF agents.
Most SI events (90.5%) met strong validation criteria, and 8.8% met moderate validation criteria. A small proportion (0.73%) of reported SI events had pending validation and were excluded from the analysis. Hospitalization was the most frequent source of validation, accounting for 88.1% of SIs following anti-TNF initiation and 86.5% following non-TNF initiation. Other sources of validation included death (8.1% and 13.1%, respectively), intravenous antibiotic administration (2.5% and 0%), and outpatient-treated events (1.3% and 0%).
Risk of SI with anti-TNF initiation
Among 1379 individuals with LORA and 2727 kernel-weighted comparators with YORA, all of whom initiated anti-TNF therapy at age ⩾60 (Figure 1), baseline characteristics were well balanced between groups (Supplemental Table 1). There were no significant differences in age at initiation, sex, race and ethnicity, Medicare coverage, comorbidity burden, or HAQ disability scores. However, LORA patients were more likely to report concurrent methotrexate use and current smoking (Table 1).

Cohort selection.
Baseline characteristics of propensity-score-matched older adults with anti-TNF initiation by age at RA diagnosis.

Each LORA patient was matched to YORA patients based on age at anti-TNF initiation, sex, race/ethnicity, Medicare coverage, comorbidity burden, and HAQ disability.
The values are presented as mean (SD) unless indicated otherwise.
SMD obtained for the variables used in the propensity score matching (after matching).
anti-TNF, anti-tumor necrosis factor; HAQ, Health Assessment Questionnaire; LORA, late-onset rheumatoid arthritis; PAS, Patient Activity Scale; RDCI, Rheumatic Disease Comorbidity Index; SMD, standard mean difference; VAS, Visual Analog Scale; YORA, young-onset rheumatoid arthritis.
With anti-TNF use, LORA patients experienced 149 SIs over 5290.7 patient-years, while YORA patients experienced 381 SIs over 18,622.3 patient-years, corresponding to crude SI incidence rates (95% CI) of 28.2 (24.0–33.1) and 20.5 (18.5–22.6) per 1000 patient-years, respectively (Supplemental Table 2).
In adjusted Cox regression models, LORA was not associated with increased risk of anti-TNF-related SI compared to YORA (adjusted HR (aHR) 0.82, 95% CI 0.63–1.02). Older age at anti-TNF initiation was independently associated with higher infection risk: aHR 1.71 (95% CI 1.27–2.29) for individuals aged 70–80, and aHR 1.63 (95% CI 1.08–2.48) for those aged ⩾80, with age 60–70 as the reference. Additional independent predictors included prior SI (aHR 1.95, 95% CI 1.13–3.40) and recent long-term GC use (aHR 2.17, 95% CI 1.68–2.81). Concurrent methotrexate use and current smoking were not significantly associated with SI risk (Table 2).
Adjusted hazard ratios for risk of serious infection related to anti-TNF use in older adults with LORA versus YORA.

LORA and YORA Kernel propensity score matched on age at treatment initiation, sex or gender, race and ethnicity, Medicare coverage, RDCI score, and HAQ disability score.
Recent long-term glucocorticoid use defined as at least 5 mg/day prednisone equivalent for 90 consecutive days prior to serious infection.
Bold values indicate adjusted hazard ratios with 95% confidence intervals that exclude 1.
anti-TNF, anti-tumor necrosis factor; CI, confidence interval; HR, hazard ratio; LORA, late-onset rheumatoid arthritis; YORA, young-onset rheumatoid arthritis.
We performed several sensitivity analyses to assess the robustness of our findings. Sensitivity analyses using the current GC use definition yielded similar results. Analyses addressing potential survival bias, restricted to patients with RA duration ⩽5 years at anti-TNF initiation, yielded similar inferences (Supplemental Tables 4 and 5). Finally, all-cause mortality was considered as a competing risk. The proportion of patients experiencing a competing event (all-cause mortality) was 4.7% among those who initiated an anti-TNF. The subdistribution HR for LORA versus YORA was 0.80 (95% CI) 0.64–1.00), confirming that LORA was not associated with an increased risk of SI when accounting for death as a competing risk (Supplemental Table 6). In addition, the direction of associations for all covariates was comparable to those observed in the cause-specific Cox models. Comparison of the Kaplan–Meier-based failure function with the cumulative incidence function accounting for death as a competing risk showed highly similar curves, with the complement of the Kaplan–Meier curve slightly overestimating the true failure probability (Supplemental Figure 1).
Risk of SI with non-TNF b/tsDMARD initiation after anti-TNF exposure
We identified 198 LORA patients and 675 kernel-weighted YORA comparators who initiated a non-TNF b/tsDMARD at age ⩾60 following prior anti-TNF therapy. The groups were similar in age at treatment initiation, sex, and comorbidity burden (Supplemental Table 3). Compared to those with YORA, LORA patients had shorter RA disease duration and lower educational attainment (Table 3).
Baseline characteristics of propensity-score matched older adults with non-TNF b/tsDMARD initiation following anti-TNF therapy use by age of RA diagnosis.
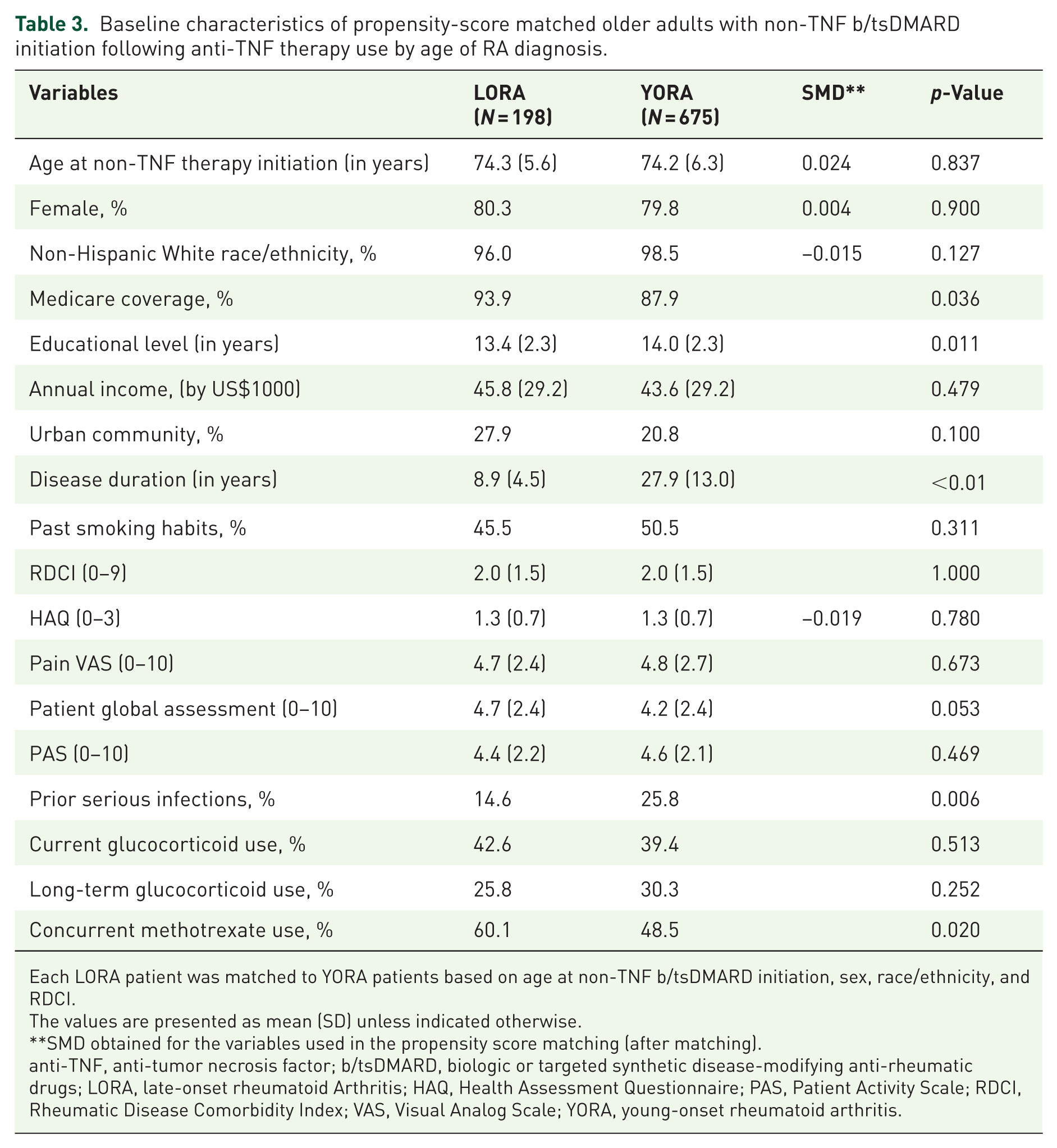
Each LORA patient was matched to YORA patients based on age at non-TNF b/tsDMARD initiation, sex, race/ethnicity, and RDCI.
The values are presented as mean (SD) unless indicated otherwise.
SMD obtained for the variables used in the propensity score matching (after matching).
anti-TNF, anti-tumor necrosis factor; b/tsDMARD, biologic or targeted synthetic disease-modifying anti-rheumatic drugs; LORA, late-onset rheumatoid Arthritis; HAQ, Health Assessment Questionnaire; PAS, Patient Activity Scale; RDCI, Rheumatic Disease Comorbidity Index; VAS, Visual Analog Scale; YORA, young-onset rheumatoid arthritis.
With non-TNF use, LORA patients experienced 7 SIs over 404.2 patient-years, and YORA patients experienced 30 SIs over 1534.6 patient-years, corresponding to crude SI incidence rates (95% CI) of 17.3 (8.3–36.3) and 19.5 (13.7–27.8) per 1000 patient-years, respectively (Supplemental Table 2).
In adjusted models, SI risk increased with older age at non-TNF initiation, with aHR of 2.85 (95% CI 1.20–6.76) among those aged 70–80 years compared with 60 to 70 years. Higher HAQ disability scores were also strongly associated with SI risk (aHR 2.86, 95% CI 1.45–5.64). No significant associations were observed by RA onset age (LORA vs YORA; aHR 0.81, 95% CI 0.31–2.12), prior infection (aHR 1.82, 95% CI 0.74–4.56), recent long-term GC use (aHR 1.84, 95% CI 0.79–4.31), or methotrexate use (aHR 0.76, 95% CI 0.32–2.83; Table 4).
Adjusted Hazard Ratio for risk of serious infection related to subsequent non-TNF b/tsDMARD use after exposure to anti-TNF in older adults with LORA versus YORA.

LORA and YORA Kernel propensity score matched on age at treatment initiation, sex or gender, race and ethnicity, Medicare coverage, and RDCI.
Recent long-term glucocorticoid use defined as at least 5 mg/day prednisone equivalent for 90 consecutive days prior to serious infection.
Bold values indicate adjusted hazard ratios with 95% confidence intervals that exclude 1.
anti-TNF, anti-tumor necrosis factor; b/tsDMARD, biologic or targeted synthetic disease-modifying anti-rheumatic drugs; CI, confidence interval; LORA, late-onset rheumatoid arthritis; HAQ, Health Assessment Questionnaire; HR, hazard ratio; YORA, young-onset rheumatoid arthritis.
Sensitivity analysis addressing potential survival bias could not be performed for non-TNF initiation due to the low sample size of patients with short RA disease duration. In a competing risk sensitivity analysis, analogous to the anti-TNF analysis, the subdistribution HR for LORA was 0.83 (95% CI 0.32–2.19) and did not indicate an increased risk of SI, even when death was accounted for as a competing risk (Figure 1 and Supplemental Table 6).
Discussion
In this large U.S. registry-based study, SI risk did not differ between older adults with LORA and YORA after initiation of either anti-TNF or subsequent non-TNF b/tsDMARD therapy. Across treatment classes, chronological age at treatment initiation was associated with SI risk. Among anti-TNF initiators, SI risk was also associated with prior serious infection and long-term glucocorticoid use, whereas among non-TNF initiators, SI risk was associated with higher HAQ disability, a potentially modifiable marker of functional status.
The lack of association between risk of SI and age of RA onset is consistent with findings from the Kansai Consortium for Well-being of Rheumatic Disease Patients (ANSWER) cohort, a large multicenter registry in Japan. 11 Among 989 RA patients initiating bDMARDs, the ANSWER study found similar rates of treatment discontinuation due to adverse events, including infections, between LORA and YORA groups (7.4% vs 8.2%; HR 0.78, 95% CI 0.38–1.18), with no significant differences in infection-related events.
Our study provides additional insights by focusing solely on adults aged 60 and older at treatment initiation, ensuring that both LORA and YORA groups were within a high-risk age range. By contrast, the ANSWER study included adults aged 18 and older, meaning their YORA group included many younger individuals with inherently lower infection risk. Moreover, ANSWER grouped all bDMARDs and JAKi together, whereas we explicitly evaluated treatment sequences, separately analyzing SI risk following anti-TNF initiation and after subsequent non-TNF b/tsDMARD use. This approach provides greater clinical granularity and reflects real-world prescribing patterns in older adults with RA.
Chronological age at treatment initiation was associated with SI risk across both treatment phases, independent of RA onset age. This finding aligns with prior studies demonstrating higher infection risk with advancing age in RA that is likely multifactorial and reflects the combined effects of immune dysregulation, multimorbidity, and cumulative immunosuppression. 28 Importantly, observed age-related associations varied by treatment phase and were characterized by greater uncertainty at the oldest ages, suggesting that chronological age may function primarily as a marker of underlying vulnerability rather than as a uniform or linear risk factor. Immunosenescence, characterized by age-related alterations in both the innate and adaptive immune systems, contributes to “inflammaging,” a chronic pro-inflammatory state that increases susceptibility to serious infections and impairs immune responses to protective strategies such as vaccination. 29 In older adults with RA, these age-related immune changes are often accelerated and compounded by chronic systemic inflammation, higher comorbidity burden, and cumulative immunosuppressive treatment exposures, thereby amplifying immune dysregulation and further increasing susceptibility to serious infection. 30
GC use was a predictor of SI risk among anti-TNF initiators, aligning with prior large cohort studies.25,31,32 Even at low doses of prednisone ⩽5 mg/day, GCs substantially increase the risk of infection, especially in older adults. 25 In addition to dose, the timing and duration of GC use substantially increased SI risk. 32 Specifically, continuous use of GC 5 mg/day over 3 years doubled the SI risk, comparable to that of high short-term doses. Importantly, infection risk declined after GC discontinuation, underscoring the value of deprescribing when feasible. Supporting these findings, a Swedish inception cohort study confirmed a strong dose–response relationship between GC use and SI risk, even after accounting for time-varying confounding. 31 In this study, recent use carried the greatest risk, with one additional SI for every 83 individuals treated with low-dose GCs for 6 months and every 125 individuals treated with high-dose GCs for 3 months, compared with non-users. Overall, these studies emphasize that GC-related infection risk is influenced by dose, duration, and recency of use. Our findings extend this evidence to older adults regardless of RA onset age, underscoring the importance of minimizing GC exposure and regularly reassessing its necessity.33,34
Prior SI also predicted subsequent SI with anti-TNF use, in line with previous evidence. 35 The German RABBIT registry showed that one prior SI more than doubled future SI risk, while two or more prior infections quadrupled risk, independent of treatment type. 36 These findings highlight prior SI as a readily identifiable and clinically actionable factor that should be routinely considered in infection risk assessments and preventive strategies, including vaccination.
Functional disability, as measured by HAQ, also emerged as a key modifiable risk factor. This association was observed after non-TNF b/tsDMARD initiation, but not after anti-TNF therapy, likely because HAQ was included in the propensity score matching for the latter analysis, limiting its ability to emerge as an independent predictor. This finding aligns with prior research linking higher physical disability and frailty to adverse outcomes in RA. Frailty, defined as diminished physiologic reserve and increased vulnerability to stressors such as infection, disability, and hospitalization, is highly relevant in this context. In a prospective RA cohort, baseline frailty predicted worsening physical function over time, with frail individuals experiencing significant increases in HAQ scores over 2 years, independent of RA disease activity (β: 0.4, 95% CI 0.1–0.8;
Frailty has also been directly associated with increased SI risk. A large claims-based study of adults with RA under age 65 initiating b/tsDMARDs found that frailty was associated with a 50% higher risk of SI among patients initiating anti-TNF therapies (aHR for SI 1.5, 95% CI 1.2–1.9). 38 These findings suggest that frailty captures infection risk beyond traditional demographic or clinical variables. Together, these results support the use of HAQ as a practical proxy for frailty and physiologic reserve, making it a valuable marker for infection risk stratification in older adults with RA.
This study has several notable strengths. It draws on a large, longitudinal, U.S.-based cohort with validated rheumatologic diagnoses and detailed patient-reported outcomes. The two-part study design enables evaluation of infection risk across treatment sequences, reflecting real-world prescribing patterns. By focusing exclusively on older adults initiating b/tsDMARDs, the study addresses an important evidence gap in a population frequently underrepresented in RA studies. Moreover, including both late-onset and young-onset RA patients, each initiating advanced therapy at age 60 or older, allows for comparisons within a clinically relevant, high-risk age group. Lastly, the use of kernel-based propensity score matching enhanced covariate balance and statistical efficiency, strengthening internal validity.
However, several important limitations warrant consideration. First, although we used propensity score methods to reduce confounding, residual confounding by unmeasured factors may persist. Variables such as RA disease activity, frailty, or social determinants of health were not fully captured in the dataset and could influence both treatment decisions and infection risk. Second, channeling bias may have affected treatment selection, particularly in the non-TNF cohort. Clinicians may have preferentially prescribed certain agents based on comorbidities or a history of prior SI, which could confound observed associations despite covariate adjustment. However, the relatively small number of SIs in the non-TNF cohort precluded reliable estimation of class-specific effects, and non-TNF agents were therefore analyzed as a pooled group. Third, survivor bias is a potential concern in the YORA group. Sensitivity analyses restricting to shorter RA duration did not reveal systematic differences; however, individuals with YORA who survive into older age may represent a healthier subgroup of patients with long-standing disease. Although our matching approach balanced age, comorbidity burden, functional status, and patient-reported disease activity, survivor effects may still influence the observed results. Fourth, SI was identified through patient self-report, and although validated when possible, outcome misclassification is a possibility. Moreover, our definition of SI may not fully reflect current clinical practice. Many infections historically considered serious are now managed in outpatient settings with oral antibiotics, potentially leading to undercounting. Fifth, while RDCI is a validated tool for capturing overall comorbidity burden, its use as a summary measure may obscure the impact of specific conditions, such as pulmonary disease, that differentially affect infection risk or treatment selection. Finally, the FORWARD cohort may represent a healthier, more engaged group of individuals with RA. As a result, the findings may not fully generalize to frailer, socioeconomically disadvantaged, or medically underserved populations.
Conclusion
In conclusion, among older adults with RA initiating b/tsDMARD therapy, SI risk was not influenced by the age of RA onset but was more strongly associated with modifiable factors, including functional disability, GC use, and prior history of SI. This finding is especially important given the low use of DMARDs, particularly b/tsDMARDs, in older adults with newly diagnosed LORA, while long-term GC use remains common, 6 raising concerns about avoidable infection risk in this population. Clinically, these findings suggest that age at RA onset alone should not drive treatment decisions in older adults. Instead, risk stratification should incorporate functional status and GC exposure, with management efforts directed toward mitigating functional impairment, minimizing chronic GC use, and reinforcing infection prevention measures, including vaccination.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X261433254 – Supplemental material for Serious infection risk in older adults with RA receiving biologic or targeted synthetic DMARDs: the role of age at disease onset
Supplemental material, sj-docx-1-tab-10.1177_1759720X261433254 for Serious infection risk in older adults with RA receiving biologic or targeted synthetic DMARDs: the role of age at disease onset by Jiha Lee, Sofia Pedro, Sara H. Bares, Gulsen Ozen, Ted R. Mikuls and Kaleb Michaud in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-docx-2-tab-10.1177_1759720X261433254 – Supplemental material for Serious infection risk in older adults with RA receiving biologic or targeted synthetic DMARDs: the role of age at disease onset
Supplemental material, sj-docx-2-tab-10.1177_1759720X261433254 for Serious infection risk in older adults with RA receiving biologic or targeted synthetic DMARDs: the role of age at disease onset by Jiha Lee, Sofia Pedro, Sara H. Bares, Gulsen Ozen, Ted R. Mikuls and Kaleb Michaud in Therapeutic Advances in Musculoskeletal Disease
Footnotes
Acknowledgements
The authors thank the staff of the FORWARD, the National Databank for Rheumatic Diseases, and the participants whose contributions made this research possible.
Declarations
Supplemental material
Supplemental material for this article is available online.
