Abstract
Background:
Giant cell arteritis (GCA) is a chronic inflammatory vasculopathy associated with an increased risk of cardiovascular and thromboembolic complications, including acute coronary syndrome (ACS). Early identification of high-risk subgroups is essential for targeted prevention. This scoping review summarizes the demographic and clinical characteristics of patients with GCA who are at elevated risk for ischemic cardiac events.
Design:
Scoping review.
Methods:
We systematically searched PubMed, Cochrane Central Register of Controlled Trials in the Cochrane Library (CENTRAL), and EMBASE (Ovid interface) for studies evaluating the association between GCA and ACS, applying predefined eligibility criteria. Eligible studies included adults aged ⩾50 years with confirmed GCA and reported ACS outcomes. Data were synthesized qualitatively, and key quantitative variables were extracted.
Results:
Nine studies met inclusion criteria, encompassing 14,484 patients with GCA. Of these, 71.5% were women, 77% had a new GCA diagnosis, and 9.8% experienced an ischemic cardiac event during follow-up. Consistently reported predictors of ACS included the early post diagnosis period, male sex, and older age at diagnosis. Other potential risk factors such as hypertension, corticosteroid use, and concomitant polymyalgia rheumatica were inconsistently reported or lacked statistical significance. Substantial heterogeneity in study design, definitions, and follow-up duration precluded pooled analysis.
Conclusion:
Older age, male sex, and the period shortly after GCA diagnosis appear to confer the highest risk for ACS, though current evidence is limited by methodological variability and incomplete reporting. Prospective studies with standardized definitions, detailed treatment data, and longitudinal follow up are needed to refine cardiovascular risk stratification in GCA. Meanwhile, clinicians should maintain heightened vigilance for cardiac events in high-risk patients, particularly early in the disease course.
Keywords
Introduction
Giant cell arteritis (GCA) is a chronic granulomatous disease through which medium- and large-caliber vessels are inflamed. The aorta and its branches are affected, with preferential involvement of cranial arteries. It is the most common type of systemic vasculitis in adults over 50, and it is more common in women. 1 The classic clinical presentation of the condition is characterized by a new-onset headache, scalp tenderness, jaw claudication, visual changes, and constitutional symptoms. 2 The objective of treating GCA with high-dose glucocorticoids is to prevent severe ischemic complications. 3 If left untreated, GCA can result in irreversible vision loss, cerebrovascular accidents, aortic aneurysms or dissections, and myocardial infarction (MI). 4
Previous studies on the risk of cardiovascular disease (CVD), particularly coronary artery disease (CAD), in patients with GCA have yielded conflicting results. An earlier meta-analysis found no statistically significant association, likely due to substantial heterogeneity in event definitions. 5 In contrast, a more recent meta-analysis reported an increased risk of MI in GCA patients compared to matched controls, although moderate heterogeneity was still present. 6 These findings highlight ongoing uncertainty and variability in the evidence regarding cardiovascular risk in GCA.
The biological plausibility of increased cardiovascular risk in GCA is supported by the systemic inflammation that characterizes the disease, along with its direct and indirect effect on vascular health. One mechanism involves direct inflammation of the vessel wall, leading to luminal narrowing and downstream ischemia. 7 This process is mediated by CD4+ T cells (specifically Th1 and Th17 subsets) which, once activated by cytokines, recruit macrophages, CD8+ T lymphocytes, and growth factors such as VEGF and PDGF.8,9 The resulting cascade promotes vascular damage, intimal hyperplasia, and progressive occlusion.7,10 A second mechanism is the potential acceleration of atherosclerosis, a phenomenon well documented in inflammatory diseases such as rheumatoid arthritis, systemic lupus erythematosus, and other vasculitides including Takayasu’s arteritis. 11 While this has not been objectively demonstrated in GCA, systemic inflammation is known to have proatherogenic effects by inducing dyslipidemia, insulin resistance, hypercoagulability, endothelial dysfunction, and oxidative stress. 12 A third contributing factor is glucocorticoid therapy, the cornerstone of GCA treatment, which involves high initial doses followed by a prolonged taper. 13 Corticosteroids have been shown in vitro to exert procoagulant effects by increasing the synthesis of von Willebrand factor (VWF) and plasminogen activator inhibitor-1, thereby promoting thrombosis through enhanced coagulation and impaired fibrinolysis. However, in the context of inflammation, they may instead exert anticoagulant effects by decreasing fibrinogen and VWF while increasing antithrombin III, protein C, and protein S. 14 Despite these mixed effects on coagulation, chronic glucocorticoid use is clearly associated with increased atherosclerotic disease. 15
Given the increased risk of MI in patients with GCA, cardiologists, rheumatologists, and other involved clinicians should incorporate this risk into early management decisions and longitudinal follow-up. Greater awareness of this association may support timely cardiovascular risk stratification and prevention efforts, ultimately reducing morbidity and mortality in this vulnerable population. Importantly, a subset of patients with GCA may be at particularly high risk for ischemic complications, and identifying clinical characteristics associated with this group could enable a more personalized approach to care.
Accordingly, this scoping review aims to characterize the frequency, timing, and risk factors of acute coronary syndrome (ACS), including unstable angina (UA) and MI, among patients with GCA.
Methods
The reporting of this scoping review adheres to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses extension for Scoping Reviews (PRISMA-ScR) guidelines. 16 We chose a scoping review methodology to map and synthesize the breadth of heterogeneous evidence on the association between GCA and ACS and to identify key knowledge gaps that preclude systematic review or meta-analysis. The primary research question was formulated using the PECO (Population, Exposure, Comparator, Outcome) framework: What risk factors are associated with the development of ACS, including UA and MI, in patients diagnosed with GCA?
Eligibility criteria
We included articles examining the association between GCA and ACS, including in a broad sense any terms referring to ischemic cardiac events. Studies were eligible for inclusion if they involved adult patients aged 50 years and older with a confirmed diagnosis of GCA and reported the occurrence of ACS. The included studies had to be randomized controlled trials, cohort studies, case–control studies, or case series with at least five patients. GCA was defined as per each study’s criteria, requiring evidence consistent with GCA based on histological confirmation (e.g., temporal artery biopsy showing vasculitis) or imaging findings (e.g., vascular wall thickening or halo sign on ultrasound, or any large vessel imaging compatible with GCA). ACS was defined as the occurrence of UA, non-ST-segment elevation MI, or ST-segment elevation MI, as reported by study authors. Diagnosis was based on clinical presentation, cardiac biomarkers, electrocardiographic changes, or imaging consistent with ischemia, according to the definitions provided in each included study.
Studies were excluded if they were not written in English or French, if the full text was unavailable despite multiple attempts, or if they were case reports. Studies were also excluded if the reported cardiovascular outcomes did not include ACS, or if the characteristics of the group experiencing ACS were not described.
Information source and search strategy
Using Medical Subject Headings and text words related to GCA and ACS, we searched the following electronic bibliographic databases: PubMed, Cochrane Central Register of Controlled Trials in the Cochrane Library (CENTRAL), and EMBASE (Ovid interface). The search strategy included terms for GCA (“giant cell arteritis” OR “temporal arteritis”) and for ischemic heart disease/ACS, including “acute coronary syndrome,” “myocardial infarction,” “Non-ST Elevated Myocardial Infarction,” “ST Elevation Myocardial Infarction,” and “unstable angina.”
To align with the introduction of the first classification criteria for GCA, a publication date filter was applied to include only articles published from August 1990 onward. The search was performed on July 2, 2025, and updated on December 19, 2025. Retrieved records were then screened for relevance and assessed according to the predefined eligibility criteria for this review. To identify additional eligible studies, we conducted a manual review of the reference lists from the included studies as well as other relevant literature.
Selection process
Two independent reviewers screened the titles and abstracts for eligibility. Full texts of potentially relevant articles were subsequently assessed for inclusion. Any uncertainties were resolved through discussion with a third reviewer. The study selection process was documented in accordance with the PRISMA flow diagram.
Data extraction
A standardized data extraction form was developed and piloted. One reviewer performed data extraction for each study. Several details were retrieved, including the first author’s name, year of publication, country of origin, study design, mean patient age, percentage of female patients, characteristics of patients with GCA, reported risk factors for ACS, occurrence of ACS, and outcomes and effect estimates such as relative risks, odds ratios, and hazard ratios (HRs).
Data synthesis
A qualitative synthesis of the included studies was conducted. Due to the anticipated heterogeneity in study design, populations, outcome definitions, and follow-up durations, no quantitative analysis or pooled effect estimate was prespecified. As such, findings were narratively summarized and interpreted. We reported descriptive summary of findings for each study. Quantitative variables were reported as mean with standard deviation or median with interquartile range, as appropriate. Continuous and categorical variables were summarized as counts and percentages.
Ethics
This scoping review was based exclusively on publicly available data and previously published literature; therefore, institutional ethics approval was not required.
Results
Study selection and population characteristics
Out of 3174 records screened, 9 studies met the eligibility criteria and were included in this review (Figure 1).17–25 These studies reported on a total of 14,484 patients with GCA, of whom 1418 (9.8%) experienced ischemic heart disease during follow-up. Among studies that reported baseline characteristics, 10,361 patients (71.5%) were women, and 11,155 had newly diagnosed GCA (Table 1). Only one study directly compared the characteristics of GCA patients who developed ACS to those who did not. The remaining studies assessed the association between GCA and cardiovascular events by comparing patients with GCA to matched control populations.
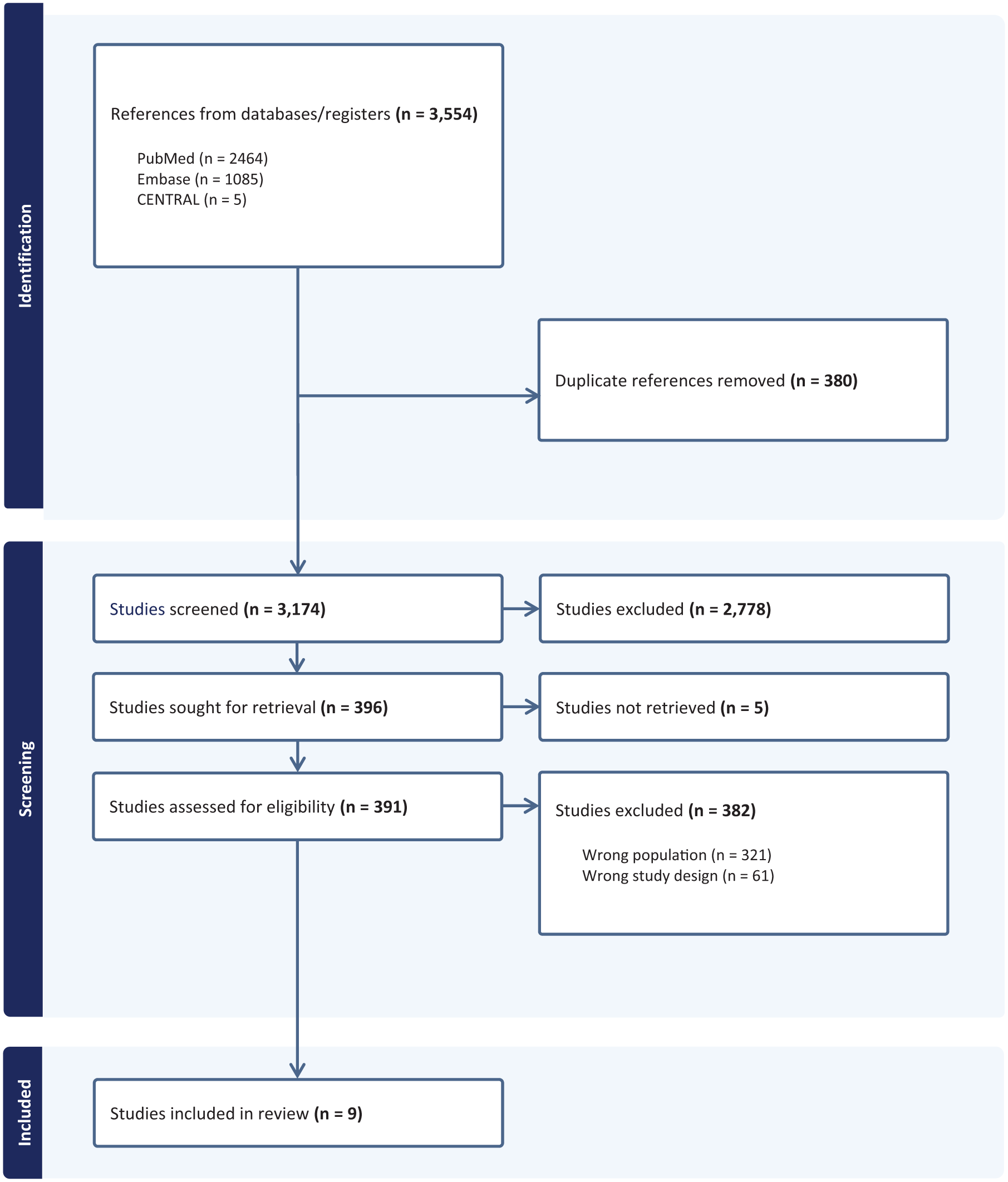
PRISMA flow diagram.
Characteristics of studies and patients included.
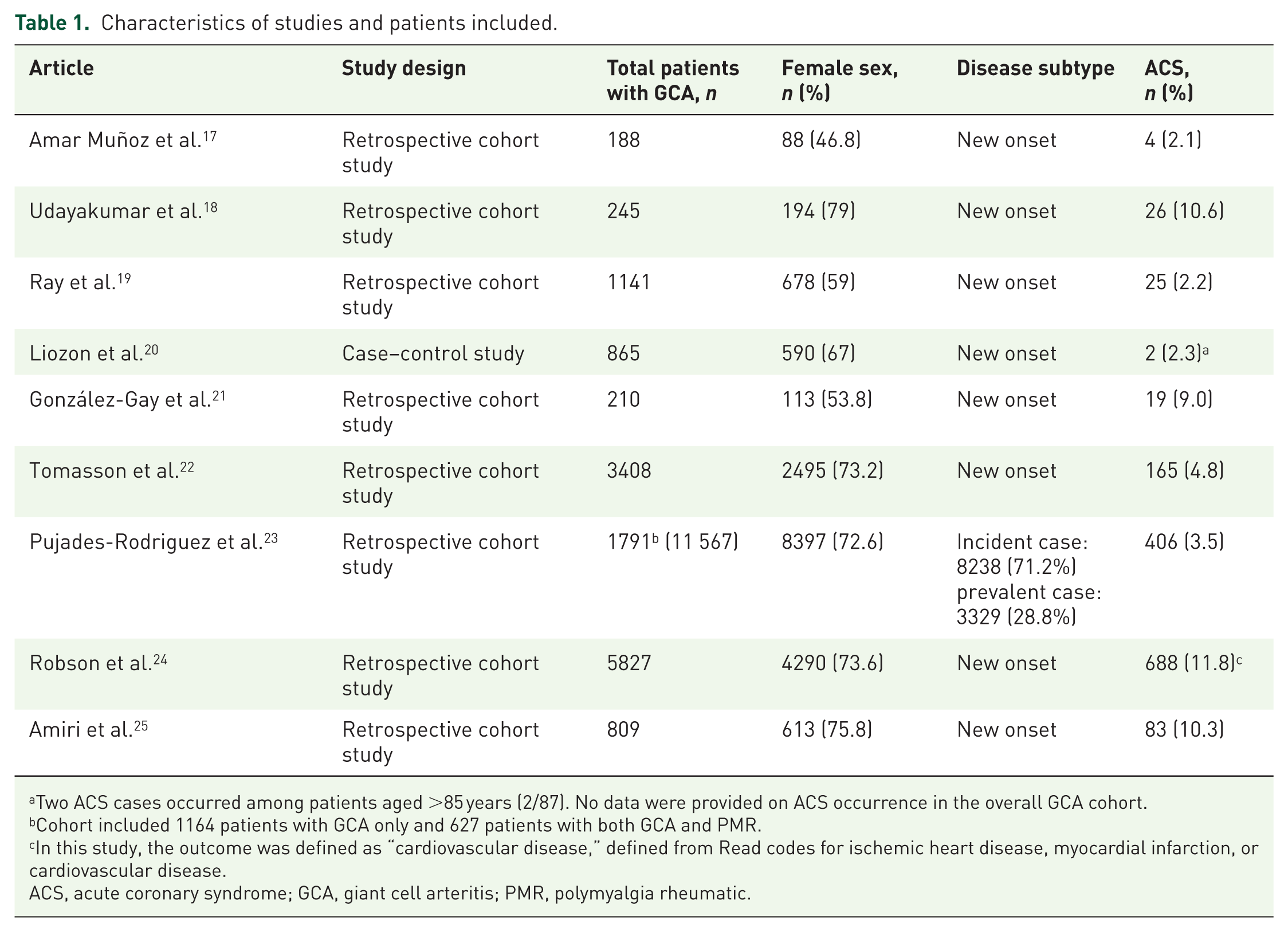
Two ACS cases occurred among patients aged >85 years (2/87). No data were provided on ACS occurrence in the overall GCA cohort.
Cohort included 1164 patients with GCA only and 627 patients with both GCA and PMR.
In this study, the outcome was defined as “cardiovascular disease,” defined from Read codes for ischemic heart disease, myocardial infarction, or cardiovascular disease.
ACS, acute coronary syndrome; GCA, giant cell arteritis; PMR, polymyalgia rheumatic.
Predictors of ACS within the GCA population
The only study that examined individual-level predictors of ACS among patients with GCA was a 2005 retrospective cohort study by González-Gay et al., 21 which included 210 patients with biopsy-confirmed disease. Nineteen patients (9%) experienced a cardiac ischemic event. Those who developed ischemic heart disease were significantly older, with a mean age of 78 years (vs 74 years in those without ischemic heart disease) at the time of GCA diagnosis (HR 1.15, 95% confidence interval (CI): 1.04–1.26, p = 0.004). Hypertension was more prevalent among patients with ischemic events (58% vs 31%, p = 0.017), although this association did not reach statistical significance in adjusted analysis (HR 2.51, 95% CI: 0.87–7.25, p = 0.089). No other demographic, clinical, or biochemical characteristics were significantly different between patients with and without cardiac ischemia.
In a large UK cohort study, Robson et al. 24 examined 5827 patients with GCA and 37,090 matched controls. Among GCA patients, predictors of cardiovascular events included male sex (standardized hazard ratio (SHR) 1.32, 95% CI: 1.12–1.56), hypertension (SHR 1.53, 95% CI: 1.14–2.05), and age over 65 years (SHR 1.34, 95% CI: 1.04–1.73). No other variables significantly differentiated patients with and without cardiovascular complications within the GCA group.
In a 2019 retrospective case–control study, Liozon et al. 20 compared outcomes in 865 patients with GCA, stratified by age. Among the 87 patients aged 85 years and older, ischemic complications were significantly more frequent compared to younger patients (40.2% vs 19.0%, p < 0.0001). However, specific data on ACS were limited, with only two cases of MI reported among the older group. Key predictors of ACS in GCA are summarized in Figure 2.

Risk factors for ACS in GCA.
Effect of concomitant polymyalgia rheumatica on cardiovascular risk
A 2016 study by Pujades-Rodríguez et al. 23 evaluated the relationship between polymyalgia rheumatica (PMR), GCA, and cardiovascular outcomes using the CALIBER database. The study included 11,567 patients with PMR and/or GCA, along with 105,504 matched controls. The incidence rate ratio (IRR) for MI was highest among patients with isolated GCA (IRR 3.00, 95% CI: 1.25–7.18), while the IRR was lower among patients with both GCA and PMR (IRR 1.23, 95% CI: 0.58–2.62). This suggests that the co-presence of PMR may attenuate cardiovascular risk in GCA, although the results were not statistically significant in most subgroups.
Temporal trends in ACS risk after GCA diagnosis
Several studies identified the early postdiagnosis period as a time of heightened cardiovascular risk. Tomasson et al., 22 using data from the UK Health Improvement Network, reported that the incidence rate of MI was 9.97 per 1000 person-years in the GCA group compared to 4.91 per 1000 person-years in controls (IRR 2.03, 95% CI: 1.70–2.43). The HR for MI was highest shortly after diagnosis, with values of 11.89 at 1 month (95% CI: 2.40–59.00), 4.13 at 3 months (95% CI: 2.05–8.35), and 2.06 by the end of follow-up (95% CI: 1.72–2.46). Adjustment for traditional cardiovascular risk factors did not attenuate these associations.
Consistent findings were reported in a Canadian study by Amiri et al., 25 which included 809 GCA patients and 8577 matched controls. The adjusted HR for MI in the GCA cohort was 4.76 during the first year after diagnosis (95% CI: 3.29–6.88), declining to 3.11 within 5 years (95% CI: 2.43–3.99), and to 2.75 beyond 5 years (95% CI: 2.16–3.50). These results support a temporal pattern in which the risk of ischemic events is highest in the early period following diagnosis.
Influence of corticosteroid therapy
Two studies reported post hoc analyses suggesting a potential protective effect of corticosteroid therapy. Ray et al. 19 evaluated older adults from administrative datasets in Ontario (Canada) and found that the association between GCA and cardiovascular events was attenuated and no longer statistically significant among those who received glucocorticoid therapy (adjusted HR 1.6, 95% CI: 0.9–2.8, vs 2.1 in the overall cohort). Similarly, Amiri et al. 25 found that the association between GCA and MI was no longer statistically significant in a sensitivity analysis limited to patients who received at least five glucocorticoid prescriptions (HR 1.35, 95% CI: 0.93–1.95).
Role of GCA imaging findings
A recent study by Amar Muñoz et al. 17 investigated whether vascular ultrasound findings could predict ischemic outcomes in GCA. Among 188 patients referred to rapid access GCA clinics, 43 (22.9%) experienced an ischemic complication within 3 months of diagnosis, including four cases of ACS. While cranial artery involvement was more common in patients with anterior ischemic optic neuropathy (AION), extracranial involvement was equally frequent in those with non-AION ischemic events such as ACS. The distribution of vascular involvement did not differentiate patients who developed ACS from those who did not.
Comparison with matched control groups
Several studies evaluated cardiovascular event rates in GCA compared to matched controls. Robson et al. 24 found a significantly higher rate of cardiovascular events in GCA patients compared to controls (11.8% vs 7.8%, SHR 1.49, 95% CI: 1.37–1.62). Tomasson et al. 22 and Amiri et al. 25 also reported increased rates of MI in GCA patients relative to matched non-GCA populations. In contrast, a smaller study by Udayakumar et al. 18 involving 245 patients with GCA and 245 controls found no significant difference in ACS incidence between groups (HR 0.74, 95% CI: 0.44–1.26). Furthermore, within the GCA group, none of the evaluated variables, including clinical symptoms, comorbidities, or medications, were associated with increased ACS risk.
Role of low-dose aspirin to prevent ACS in GCA
None of the included studies explicitly reported on the use of aspirin, either as a preventive therapy or as part of clinical management in GCA. Aspirin use was not captured in baseline characteristics, treatment variables, subgroup analyses, or outcome models. Even studies that utilized large-scale administrative or electronic health record datasets with prescription linkage did not report on aspirin exposure or its association with cardiovascular outcomes.
Discussion
This scoping review summarizes the current data on characteristics of patients with GCA that increase their risk of ACS. Most recent articles on the subject have focused on evaluating the strength of the association between cardiovascular events and GCA. Recent data confirm that GCA increases the occurrence of ischemic cardiac events, so it is important for clinicians to identify patients with GCA at the highest risk for serious cardiac complications. 6
A plausible causal model is that active arterial inflammation in GCA promotes endothelial dysfunction, intimal hyperplasia, and platelet activation, leading to a transient prothrombotic state that is most pronounced early after diagnosis and may explain the clustering of ACS events in the first months of disease. 26 Although glucocorticoids rapidly suppress vascular inflammation, they may also independently increase cardiovascular risk through metabolic effects such as hyperglycemia, dyslipidemia, and hypertension, as well as fluid retention and potential procoagulant changes, thereby modifying risk during the early treatment phase.8,15 These inflammation and treatment effects likely interact with baseline cardiovascular risk factors, including older age, male sex, and preexisting atherosclerosis, suggesting that ACS risk in GCA reflects the convergence of inflammation-driven vascular injury and treatment-related cardiometabolic stress in a vulnerable population. 10 Clinically, these findings support early cardiovascular risk assessment at diagnosis, aggressive optimization of modifiable risk factors, and heightened surveillance during the initial treatment period, while reinforcing the importance of steroid-sparing strategies when appropriate to reduce cumulative glucocorticoid exposure. This review shows a clear increase in ACS risk after GCA diagnosis, peaking in the first month and remaining elevated in the first year, with relative risks as high as 11. The risk then declines gradually but persists over time. Older age at diagnosis increases ischemic risk, especially in those 65 and older.
Some studies suggest potential predictive factors for ACS in GCA, though findings are inconsistent and often not statistically significant. Hypertension at diagnosis was associated with over a 50% increased risk in two cohorts, but not in another that found no elevated ACS risk in GCA overall.18,21,24 Glucocorticoid use shows mixed effects: in two studies, restricting analyses to those with prescriptions reduced the strength of association with ACS, suggesting a possible protective role.19,25 In contrast, another study found a stronger association when limiting to those with more than 10 prescriptions. 18 Variability in definitions, dosing, and timing may explain these differences. A concurrent diagnosis of PMR showed a nonsignificant trend toward reduced ACS risk in three studies.21,23,24 Data on clinical presentation or imaging phenotype showed no significant associations with ACS risk.17,18 Importantly, although extracranial GCA may plausibly confer a different (potentially higher) risk of ACS than cranial disease, the included studies did not report subtype (cranial/extracranial/mixed) data in a sufficiently consistent manner to allow subtype-specific analyses, and whether ACS risk varies across GCA phenotypes therefore remains unclear.
Comparing factors associated with stroke versus ACS in GCA is highly relevant, since these ischemic outcomes may reflect overlapping but distinct vascular mechanisms; notably, our recent review on GCA presenting with ischemic stroke highlights characteristic neurovascular patterns and diagnostic clues that differ from typical atherosclerotic presentations, underscoring the need for future studies directly contrasting risk factors and phenotypes associated with stroke versus ACS in GCA. 27
The absence of data on aspirin use in the included studies represents an important knowledge gap, particularly given the established cardioprotective benefits of low-dose aspirin in other high-risk inflammatory settings. However, the lack of observational or randomized evidence precludes conclusions regarding the effectiveness and safety of aspirin in this context, underscoring the need for well-designed prospective studies, including interventional trials that also carefully evaluate bleeding risk. 26
Our review has several strengths. It systematically compiles and analyzes data on the prevalence and predictors of ACS in GCA, drawing from large cohort studies to provide a comprehensive overview of current evidence and knowledge gaps. However, important limitations remain. Because eligibility was restricted to English- and French-language publications, relevant studies published in other languages may have been missed, potentially introducing language bias. Definitions of GCA vary widely with some studies rely solely on biopsy, while others include patients with no biopsy or require glucocorticoid use for case inclusion. In one study, fewer than half of GCA patients received glucocorticoids. Definitions of cardiac ischemia also differ and are sometimes imprecise, limiting clinical applicability and comparability. Most studies have follow-up periods under 10 years, restricting the ability to assess late-onset ACS, which may be influenced by long-term corticosteroid use.
Conclusion
This review highlights important demographic and clinical factors such as older age, male sex, and recent GCA diagnosis that may help stratify cardiovascular risk in this population. However, the current literature is limited by heterogeneity in study design, inconsistent reporting of outcomes, and a lack of data on relevant interventions such as aspirin. To address these gaps, prospective studies with standardized definitions, detailed treatment data, and longitudinal follow-up are urgently needed. Until such evidence emerges, clinicians should maintain a high index of suspicion for cardiac events in patients with GCA, particularly during the early phases of disease, and consider individualized cardiovascular risk mitigation strategies.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X261425813 – Supplemental material for Risk factors for acute coronary syndrome in patients with giant cell arteritis: a scoping review
Supplemental material, sj-docx-1-tab-10.1177_1759720X261425813 for Risk factors for acute coronary syndrome in patients with giant cell arteritis: a scoping review by Eugénie Pelletier, Caitlyn Kanters, Stéphanie Ducharme-Bénard and Jean-Paul Makhzoum in Therapeutic Advances in Musculoskeletal Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
