Abstract
Background:
Vasovagal responses (VVR) are common transient autonomic reactions to invasive procedures such as injections or needling, characterized by bradycardia, hypotension, and transient loss of consciousness; however, their occurrence and underlying autonomic mechanisms during dry needling (DN) for myofascial pain syndrome (MPS) remain poorly understood.
Objectives:
This study aimed to determine the frequency of VVR during DN in patients with MPS and to compare autonomic parameters—heart rate variability (HRV), pupil diameter, and skin perfusion—between those with and without VVR.
Design:
Prospective observational study.
Methods:
In total, 100 adults with acute cervical MPS underwent a standardized DN procedure. Outcomes included pressure pain threshold (PPT), heart rate (HR), blood pressure (BP), HRV indices [average NN interval (AVNN), standard deviation of NN intervals (SDNN), low-frequency (LF)/high-frequency (HF) ratio], pupil diameter, and skin perfusion, assessed at baseline, 5 min, and 30 min post-intervention. Participants were classified post hoc as VVR positive (VVR+) or VVR negative (VVR−) based on clinical signs of vasovagal reaction.
Results:
No significant changes in skin perfusion or BP were found over time or between groups (p > 0.05). Significant main effects of time and Time × Group interactions were observed for PPT (p < 0.001), HR (p < 0.001), AVNN (p < 0.001), SDNN (p < 0.001), LF/HF ratio (p < 0.001), and pupil diameter (p < 0.001). Post hoc analyses revealed that only the VVR+ group showed significant increases in PPT (p < 0.001), AVNN (p < 0.001), SDNN (p < 0.001), and pupil diameter (p < 0.001), alongside decreases in HR (p < 0.001) and LF/HF ratio (p < 0.001) from baseline to post-intervention. The VVR− group showed no significant changes.
Conclusion:
Patients experiencing VVR during DN demonstrated distinct autonomic modulation with parasympathetic predominance, pupil dilation, and increased pain threshold. These findings suggest individual variability in autonomic reactivity during DN and suggest the need for awareness and monitoring of susceptible patients.
Trial registration:
The clinical registration number is ISRCTN16484644, date: January 28, 2025.
Plain language summary
Vasovagal responses (VVR) are common transient autonomic reactions to invasive procedures such as injections or needling, characterized by bradycardia, hypotension, and transient loss of consciousness; however, their occurrence and underlying autonomic mechanisms during dry needling (DN) for myofascial pain syndrome remain poorly understood. This study aimed to determine the frequency of VVR during DN in patients with myofascial pain syndrome and to compare autonomic parameters “heart rate variability (HRV), pupil diameter, and skin perfusion” between those with and without VVR. Design: prospective observational study. The findings demonstrate that individuals who experience a VVR exhibit distinct autonomic patterns (marked by parasympathetic dominance) and sensory modulation, as evidenced by changes in HRV, pupillometry, and pressure pain threshold. These responses were not observed in patients without VVR, highlighting the importance of recognizing and monitoring this subset of individuals in clinical practice. Although further research is needed to confirm these findings and explore long-term effects, the current results underscore the need for individualized assessment and vigilance during DN procedures, particularly in patients with higher stress levels or other predisposing factors.
Keywords
Introduction
Myofascial pain syndrome (MPS) is a highly prevalent musculoskeletal condition characterized by myofascial trigger points (MTrPs)—hyperirritable spots within taut bands of skeletal muscle—that cause referred pain, motor dysfunction, and localized tenderness.1,2 It is estimated to affect up to 85% of individuals during their lifetime and accounts for 30%—93% of cases seen in pain clinics.2,3 MPS has a substantial negative impact on physical function and quality of life, with clinical outcomes often worse than in healthy individuals and, in some domains, comparable to those observed in fibromyalgia patients.4,5 The maxillofacial region is particularly vulnerable, contributing significantly to head and neck pain complaints. 6 Diagnosis is primarily clinical and relies on the identification of trigger points and their characteristic pain referral patterns through palpation. 2 Management typically involves a multidisciplinary approach that prioritizes conservative treatments—such as physical therapy and exercise—while more invasive interventions, including needling techniques, are reserved for refractory cases.7,8 Despite its high prevalence and clinical burden, MPS remains frequently underdiagnosed, underscoring the need for increased clinical awareness and a more comprehensive approach to care. 6
Dry needling (DN) has become an increasingly popular therapeutic approach for managing MPS.9,10 It is a minimally invasive technique that involves inserting fine, solid filament needles into active trigger points to elicit a local twitch response (LTR), deactivate the point, reduce muscle tension, restore local perfusion, and relieve pain.7,11 Clinical studies have shown that DN is moderately to highly effective in reducing pain, improving range of motion, and enhancing patients’ quality of life.12,13 Despite its benefits, DN is a percutaneous and neurologically active intervention that may provoke unintended physiological reactions, particularly autonomic responses such as vasovagal episodes.14,15 These reactions highlight the importance of better understanding the underlying mechanisms and ensuring appropriate patient monitoring during and after treatment.
Although DN is an effective therapeutic technique widely employed by physical therapists, it carries potential risks, including vasovagal responses (VVRs)—transient autonomic reactions that may compromise patient safety.16,17 VVRs are characterized by a sudden drop in blood pressure (BP) and heart rate (HR), often accompanied by dizziness, nausea, pallor, diaphoresis, or transient syncope.18,19 These episodes are relatively uncommon, with incidence during acupuncture and DN ranging from 0.02% to 7%, and most are mild and self-limiting.16,19 The physiological basis of VVR involves increased vagal tone, typically triggered by nociceptive input, emotional distress, or needle penetration, and mediated by afferent vagal pathways and central autonomic centers such as the nucleus tractus solitarii. 20
Despite the growing use of DN and increasing awareness of VVRs as a potential adverse effect, the physiological mechanisms underlying these reactions remain poorly understood. Most existing studies emphasize the efficacy and general safety of DN, 9 while few have investigated objective autonomic markers, such as heart rate variability (HRV), pupillary responses, or skin perfusion, that could signal dysregulation associated with VVRs. Furthermore, data on incidence rates, predisposing factors, and predictive biomarkers for VVR in DN-treated populations are scarce and inconsistent. The absence of controlled observational studies integrating both clinical outcomes and autonomic indicators hampers efforts to identify vulnerable individuals and to establish evidence-based prevention strategies. This gap is especially concerning given the potentially distressing nature of VVRs and their impact on patient safety, treatment adherence, and clinician preparedness. Further research is therefore essential to elucidate the neurophysiological correlates of VVR and to develop predictive models based on demographic, psychological, and physiological variables.
Despite increasing DN utilization, objective physiological correlates of VVR during DN remain insufficiently characterized, and integrated multimodal monitoring (HRV, pupillometry, and perfusion) has rarely been applied to identify autonomic signatures of VVR in DN-treated MPS populations. Clarifying whether autonomic changes occur broadly after DN or selectively in those who experience VVR has direct safety relevance for outpatient practice. Although VVR is often self-limited, it can lead to treatment interruption, patient distress, and (in seated procedures) risk of fall-related injury if not recognized early.17,19 Therefore, identifying objective autonomic signatures that distinguish VVR-prone patients has practical value for monitoring, positioning, and immediate management in outpatient physiotherapy settings. Therefore, the present study aimed to determine the frequency of VVRs during DN treatment in patients with MPS and to compare physiological changes—specifically HRV, pupil diameter, and skin perfusion—between those with and without a vasovagal reaction. Furthermore, this study sought to identify potential predictors of VVR, including sex, age, body mass index (BMI), stress level, and pressure pain threshold (PPT). Primary hypothesis (H1): participants who develop VVR positive (VVR+) will show an acute shift toward parasympathetic predominance (decreased HR and low-frequency/high-frequency (LF/HF) ratio; increased AVNN and SDNN) and pupil dilation at 5 min post-intervention compared with participants without VVR negative (VVR−).21,22 Secondary hypothesis (H2): VVR+ participants will show a greater increase in PPT than VVR− participants.13,23 Exploratory hypothesis (H3): baseline clinical/demographic factors (sex, age, BMI, baseline PPT, and baseline stress level) will be associated with the odds of VVR occurrence. 24
Methods
Study design
This study was conducted as a prospective, controlled observational investigation (Figure 1) aimed at evaluating the frequency of VVRs following DN in individuals diagnosed with acute cervical MPS, defined as the presence of symptoms for no longer than 1 month before enrollment. A secondary objective was to assess the physiological responses associated with these reactions and to identify potential predictive factors for their occurrence.

Study design.
All participants underwent the same intervention (DN), and classification into groups was determined post-intervention based on the presence or absence of a VVR (VVR+ or VVR−). This design allowed for comparison of physiological and clinical variables between those who exhibited a vasovagal reaction and those who did not, without manipulating exposure or using random allocation.
No randomization was applied because the study’s primary aim was not to test the efficacy of an intervention, but rather to observe natural physiological and clinical variations in response to a standardized procedure. Since the occurrence of VVRs cannot be predicted or induced ethically, group assignment was necessarily determined after the intervention, based on the presence of predefined clinical signs of VVR.
To minimize the potential influence of confounding variables, all procedures were conducted under standardized environmental and procedural conditions. Assessments were carried out in a seated position in a quiet, temperature-controlled room (21°C–22°C) with constant humidity. All participants were evaluated at the same time of day to reduce circadian variability. They were also instructed to avoid caffeine, alcohol, and strenuous physical activity for at least 24 h prior to testing. The use of a single trained physiotherapist for all procedures further reduced inter-rater variability.
Each participant underwent assessments at three time points: T0 (baseline, pre-intervention), T1 (5 min post-intervention), and T2 (30 min post-intervention). At each time point, physiological measurements included HRV, pupil diameter (using infrared pupillometry), skin perfusion (via laser Doppler flowmetry, LDF), systolic and diastolic BP, and HR. Additional baseline data were collected, including age, sex, height, weight, BMI, self-reported stress level (visual analog scale, VAS), and PPT (PPT in N/cm2), to explore their potential as predictors of VVR.
VVRs were identified based on clinical signs such as dizziness, pallor, nausea, sweating, bradycardia, hypotension (defined as a drop of 20 mmHg or more in systolic pressure), and syncope. This criterion was consistent with syncope consensus statements and vasovagal pathophysiology descriptions.19,25 These events were recorded by the treating physiotherapist using a standardized observation form. Participants who exhibited two or more defined signs were classified as VVR+, while those who showed none were classified as VVR−.
A convenience sample of 100 participants was established based on clinical feasibility and existing data regarding the estimated incidence of VVRs in needling procedures. Although no formal power calculation was performed, this sample size was considered sufficient to detect medium effect sizes in between-group differences of autonomic outcomes (HRV, pupillometry, skin perfusion) and to explore potential predictors through logistic regression analyses in an exploratory framework.
The study was carried out between December 2024 and March 2025 under standardized and environmentally controlled conditions. The study was registered with ethical approval by the Polish Society of Physiotherapy (registration number: 2/03/2025, registration date: 12/03/2025), and the clinical registration number is https://doi.org/10.1186/ISRCTN16484644, and conducted per the Declaration of Helsinki. All participants gave written informed consent before inclusion in the study.
Participants
A total of 100 adult participants (Figure 2) with clinically diagnosed acute cervical MPS, with a duration of symptoms not exceeding 1 month, were included in the study. Participants (Table 1) were recruited during physiotherapy consultations at the Provita Medical Center in żory between December 2024 and March 2025.
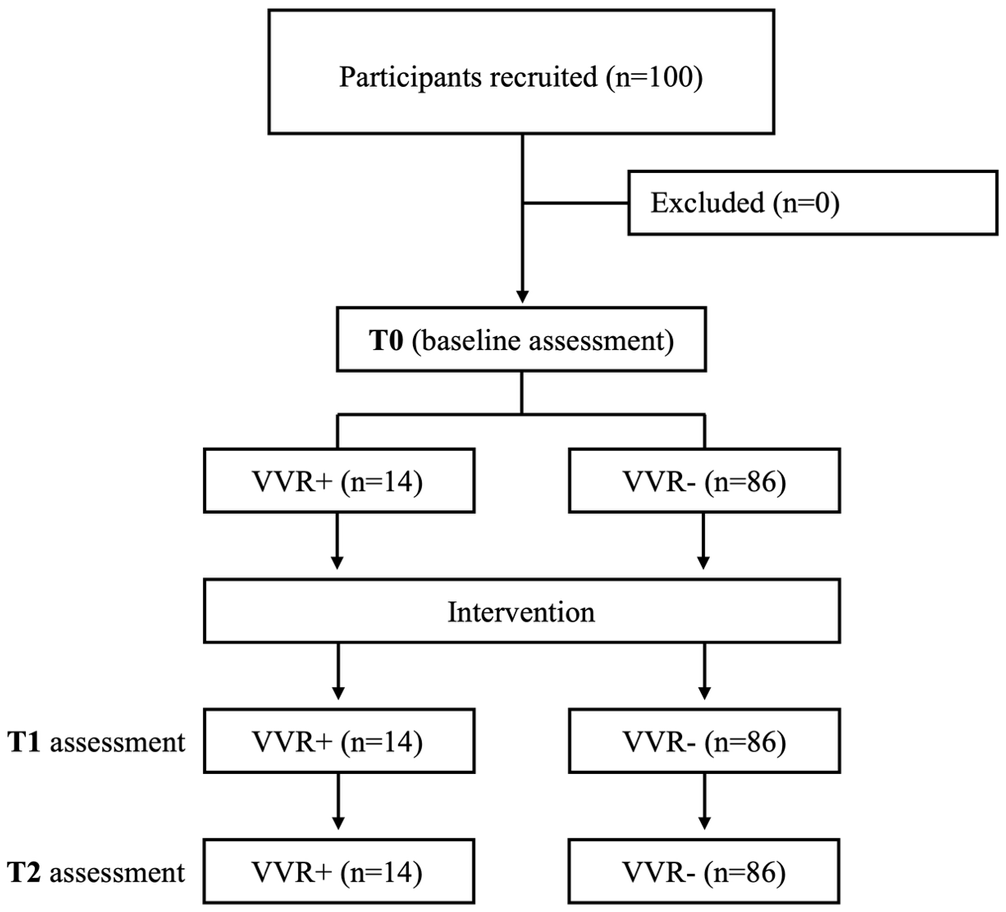
Participant flow and study timeline. Number assessed for eligibility, excluded (with reasons), included (N = 100), completed T0 baseline measures, received DN, completed T1 (5 min) and T2 (30 min) measures, and post hoc classification into VVR+ (n = 14) and VVR− (n = 86) based on predefined clinical criteria.
Demographic and clinical characteristics compared between positive and negative vasovagal patients (VVR+ vs VVR−).

BMI, body mass index; SD, standard deviation; VVR, vasovagal responses.
The inclusion criteria were as follows: (i) age between 18 and 65 years, (ii) presence of at least one active trigger point in the cervical musculature (confirmed by palpation and reduced PPT), (iii) absence of prior DN or other invasive techniques within the last 3 months, and (iv) no other significant health conditions. Participants were not on any regular medication. Exclusion criteria included the following: (i) hypertension (BP ⩾140/95 mmHg), (ii) use of corticosteroids or hormonal contraceptives, nicotine dependence, medications affecting systemic hemodynamics (e.g., β-blockers, calcium antagonists, or renin–angiotensin system inhibitors), (iii) diagnosed cardiovascular or neurological disorders, and (iv) use of drugs influencing autonomic function, pregnancy, and history of non-vasovagal syncope.
Before the intervention, each participant completed a standardized clinical questionnaire covering demographic data (age, sex), previous experience with DN, history of syncope, and perceived stress level using a 0–10 VAS. A single familiarization session was conducted 14 days prior to data collection, during which DN was applied to the gastrocnemius muscle to standardize procedural exposure. All participants gave written informed consent before participating in the study and were informed of their right to withdraw at any time without providing a reason.
Intervention
A single experienced physiotherapist, with over 10 years of specialized clinical practice in DN techniques, performed all procedures. The intervention protocol began with the identification of MTrPs in the middle portion of the trapezius muscle, following standardized and evidence-based criteria. All procedures adhered to established safety and hygiene protocols for DN. A sterile SOMA needle (0.30 × 30 mm) was used for the intervention. The technique involved the insertion of a single needle into the identified MTrP, with manual manipulation of the needle angle and depth to elicit LTRs, following established protocols.26,27 Figure 2 illustrates the DN procedure combined with simultaneous monitoring of HRV and pupillometry.
The treatment was immediately discontinued if the participant reported a burning sensation or any other adverse event. The number of insertions depended on the number of active trigger points detected; however, for all physiological measurements and safety assessments, the most sensitive trigger point (lowest PPT value) was consistently selected. In addition to monitoring for vasovagal reflexes, other adverse events such as hematoma, post-needling soreness, or increased pain were also recorded.
Outcomes
Primary outcome was the occurrence of a VVR (yes/no) during or immediately after DN, defined by prespecified clinical criteria. The secondary outcomes were the acute autonomic change from T0 to T1 and T2 in HR, AVNN, SDNN, and LF/HF, and pupil diameter. Additional secondary outcomes were PPT, systolic/diastolic BP, and skin perfusion (LDF). Exploratory outcomes were baseline predictors of VVR (sex, age, BMI, baseline stress, baseline PPT).
Characteristics of measurements
Measurements were conducted using the same sequence, technical procedures, and environment over the time points. Standardization of HRV acquisition/analysis followed published short-term HRV standards and artifact-handling recommendations. 28 Pupillometry procedures followed published recommendations regarding ambient light and adaptation. 29 LDF measurement followed published guidance on reproducibility and methodological control. 30
Tissue perfusion
Skin microcirculatory perfusion was assessed using the PeriFlux System 5000 (Perimed AB, Järfälla, Sweden), equipped with a PF 457 laser Doppler probe, based on LDF technology. The probe was positioned directly over the most active MTrPs in the middle portion of the trapezius muscle, identified by palpation as the site with the lowest PPT. Participants remained seated with back support during the assessment. The chin was stabilized using the pupillometry device mount, and the upper limbs rested naturally on the thighs. The LDF probe was fixed in a perpendicular and stable position using an adjustable flexible arm to ensure measurement reproducibility. Recordings lasted 2 min under resting conditions, in a temperature-controlled room (21°C–22°C) with stable humidity, following a 10-min silent adaptation period. Data were analyzed using PeriSoft (Perimed AB, Järfälla, Sweden) for Windows (version 2.50). LDF is considered a reliable and reproducible method for static perfusion assessment, with reported intraclass correlation coefficients ranging from 0.80 to 0.92 under standardized conditions.30,31
Ppt
PPT was measured using the FPIX algometer (Wagner Instruments, Greenwich, CT, USA; 2013), a well-established method known for its high reliability and reproducibility. 32 A 4 mm diameter probe applied progressive compressive force to a marked area over the identified trigger point. Participants were instructed to indicate when the pressure became unpleasant but not painful. The force value, displayed in Newtons per square centimeter (N/cm2), was calculated as the average of three consecutive measurements. 33
Hrv
RR intervals (i.e., the time between two consecutive R peaks – heartbeats) were recorded using a validated chest-strap system (Polar H10; Polar Electro Oy, Kempele, Finland) with 1-ms resolution (1000 Hz). At each time point (T0 baseline, T1 5 min post-DN, T2 30 min post-DN), participants remained seated and breathing spontaneously. HRV was derived from 5-min stable segments consistent with short-term HRV standards. Raw RR series were processed in Kubios HRV Scientific (Kubios Oy, Kuopio, Finland). Artifacts were detected and corrected using Kubios’ validated automatic beat correction algorithm. 34 Recordings were excluded if the proportion of corrected/removed beats exceeded 5%. Time-domain indices included AVNN and SDNN. Frequency-domain power was estimated using an FFT (Welch periodogram) on the detrended RR series after cubic-spline interpolation and resampling (4 Hz); LF (0.04–0.15 Hz) and HF (0.15–0.40 Hz) bands were computed, and LF/HF was reported descriptively as an index of sympathovagal balance. AVNN reflects the mean NN interval, and the SDNN reflects the overall HRV. Another indicator is the LF/HF ratio, which represents the relationship between LF and HF power and is used as an index of sympathovagal balance. LF/HF values below 1 indicate parasympathetic predominance, associated with relaxation and recovery states; values between 1 and 3 suggest a balance between the sympathetic and parasympathetic systems; while values above 3 reflect sympathetic predominance, typical of stress or activation states. 35
Pupillometry
Autonomic responses during DN therapy were assessed using pupillometry, a technique considered innovative in both scientific research and advanced clinical diagnostics. For this purpose, a Carl Zeiss Meditec corneal tomography device (Germany, 2012) was employed (Figure 3). Although originally designed for anterior segment evaluation, its technical specifications allow for precise measurement of pupillary responses, which are closely linked to autonomic nervous system activity. The device utilizes 22 rings (18 of which are full rings), capturing a total of 3425 measurement points. It provides topographic mapping of the corneal surface within a measurement diameter ranging from 0.75 to 9.4 mm at 42.125 diopters (D). It covers a diopter range from 25 to 65 D, corresponding to a corneal radius between 13.5 and 5.2 mm. The system ensures high precision and reliability, with a reported measurement accuracy of ±0.05 D (±0.01 mm) and repeatability of ±0.10 D (±0.02 mm), as supported by previous validation studies.35,36 These parameters support its suitability for evaluating subtle autonomic fluctuations in response to clinical stimuli such as DN.

Dry needling procedure combined with simultaneous monitoring of heart rate variability and pupillometry.
Sample size calculation
This study was designed as an observational cohort 37 with post hoc classification into VVR+ and VVR− based on predefined clinical criteria. Because VVR incidence in DN/acupuncture is typically low and group sizes cannot be predetermined, a conventional two-group power calculation assuming balanced allocation is not appropriate. Therefore, the sample size was justified primarily to (i) estimate VVR incidence with reasonable precision and (ii) allow exploratory between-group comparisons of autonomic outcomes. With N = 100, an observed VVR proportion in the range reported for needling procedures (0.02%—7%) 19 yields a 95% confidence interval with acceptable precision for feasibility/observational inference. Predictor modeling was considered exploratory and constrained by the number of VVR events.
Given the post hoc formation and imbalance of VVR+ and VVR− groups, all between-group inferential analyses are interpreted as observational and exploratory. For the exploratory logistic regression, the number of candidate predictors was limited a priori to minimize overfitting, consistent with event-limited modeling practice, and results are presented with odds ratios and confidence intervals as hypothesis-generating. 38
Statistical procedures
Normality of continuous variables was assessed using the Shapiro–Wilk test, and homogeneity of variances was evaluated with Levene’s test. Descriptive statistics were used to summarize the sample characteristics. Continuous variables are reported as mean ± standard deviation (SD), and categorical variables as frequencies and percentages. To compare baseline characteristics (e.g., age, sex, BMI, perceived stress, and PPT) between participants who experienced a VVR (VVR+) and those who did not (VVR−), independent-samples t tests were used for normally distributed variables. A mixed-design repeated measures ANOVA was conducted to assess changes in physiological parameters (HRV, pupil diameter, and skin perfusion) across three time points (T0—baseline, T1—5 min post-intervention, and T2—30 min post-intervention), with group (VVR+ vs VVR−) as the between-subjects factor and time as the within-subjects factor. When Mauchly’s test indicated a violation of sphericity, the Greenhouse–Geisser correction was applied. Bonferroni-adjusted post hoc comparisons were used to further explore significant effects. Statistical significance was set at p < 0.05. All analyses were conducted using SPSS version 29.0 (IBM Corp., Armonk, NY, USA).
Results
Table 2 shows the descriptive statistics for physiological variables by group and time point. Supplementary material 1 shows the mean and standard deviation for all the outcomes. For tissue perfusion (TP), Mauchly’s test indicated a violation of sphericity (χ2(2) = 7.605, p = 0.022), so Greenhouse–Geisser correction (ϵ = 0.930) was applied. No significant main effect of Time was found (F(1.86, 182.26) = 1.509, p = 0.225,
Descriptive statistics (mean ± SD) for physiological variables by group and time point.

BP, blood pressure; PU, perfusion units; SD, standard deviation; VVR, vasovagal responses.
Regarding PPT, the sphericity was violated (χ2(2) = 12.912, p = 0.002), and Greenhouse–Geisser correction (ϵ = 0.889) was applied. A significant main effect of Time was observed (F(1.78, 174.28) = 11.149, p < 0.001,
In BP systolic, the sphericity was met (χ2(2) = 1.598, p = 0.450). There was no significant main effect of Time (F(2, 196) = 0.424, p = 0.655,
Considering BP diastolic, the sphericity was met (χ2(2) = 1.195, p = 0.550). No significant main effect of Time was found (F(2, 196) = 0.424, p = 0.655,
Sphericity was violated for HR (χ2(2) = 12.912, p = 0.002), and Greenhouse–Geisser correction (ϵ = 0.889) was applied. A significant main effect of Time was observed (F(1.38, 135.04) = 111.679, p < 0.001,
In AVNN, the sphericity was met (χ2(2) = 2.519, p = 0.284). A significant main effect of Time was observed (F(2, 196) = 67.616, p < 0.001,
Considering SDNN, the sphericity was met (χ2(2) = 0.478, p = 0.787). A significant main effect of Time was observed (F(2,196) = 13.692, p < 0.001,
With sphericity met in the LF/HF ratio (χ2(2) = 0.445, p = 0.800), a significant main effect of Time was observed (F(2,196) = 78.098, p < 0.001,
Finally, in pupillometry, sphericity was violated (χ2(2) = 26.436, p < 0.001), and Greenhouse–Geisser correction (ϵ = 0.807) was applied. A significant main effect of Time was observed (F(1.62, 158.25) = 26.118, p < .001,
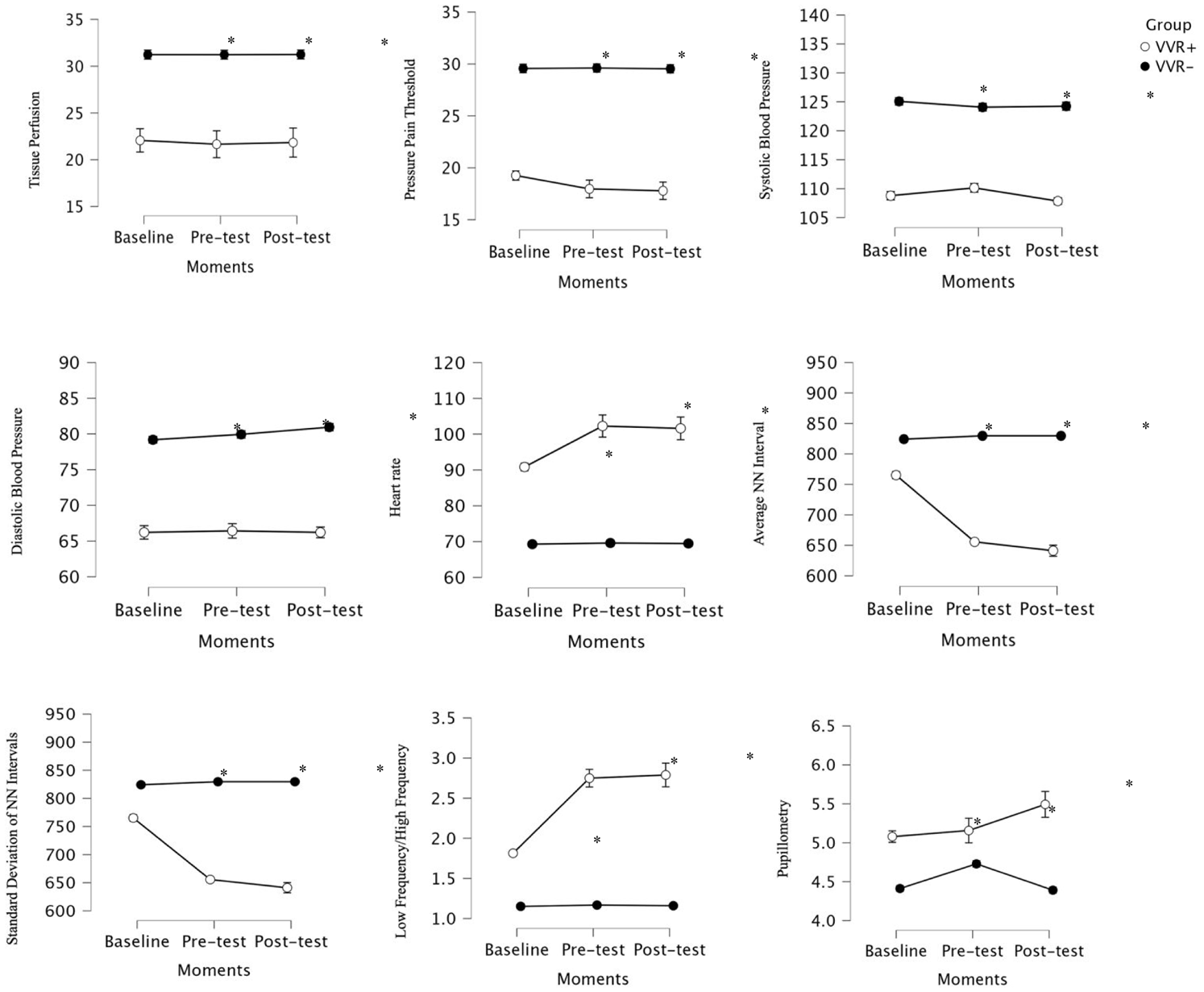
Comparison between the groups in each time point observed in tissue perfusion, pressure pain threshold, systolic BP, diastolic BP, heart rate, average NN interval, standard deviation of NN intervals, low frequency/high frequency, and pupillometry.
Discussion
The primary aim of this study was to determine the frequency of VVR during DN in patients with MPS and to compare physiological changes—specifically HRV, pupil diameter, and skin perfusion—between those who did and did not experience a VVR. In addition, the study sought to identify potential predictors of VVR, including sex, age, BMI, stress level, and PPT. The results revealed that patients in the VVR+ group exhibited significant autonomic alterations compared to the VVR− group, including a marked reduction in HR, increased AVNN and SDNN values, and a significant decrease in the LF/HF ratio, reflecting heightened parasympathetic activity. Furthermore, pupillometry showed contrasting trends: pupil diameter significantly increased in the VVR+ group and decreased in the VVR− group. Only the VVR+ group demonstrated a significant increase in PPT over time, suggesting a differential sensory modulation. Collectively, these findings support the hypothesis that individuals who experience a VVR during DN present distinct autonomic and sensory profiles compared to those who do not.
The VVR− group exhibited minimal or no autonomic and sensory modulation across time points, suggesting that the observed autonomic signature is not a generic response to DN per se but is primarily expressed in individuals who develop VVR. One interpretation is that needle-related autonomic modulation is contingent on the vasovagal reflex cascade, which can be triggered by pain, anxiety, or contextual factors (e.g., seated posture), rather than by DN as a uniform physiological stimulus. This interpretation is consistent with broader literature17,19,39 indicating that vasovagal reactions (while typically benign) may meaningfully influence procedure tolerance and continuation, and are strongly context-dependent.
In-depth analysis of the autonomic parameters revealed marked differences between the VVR+ and VVR− groups, particularly in HR and HRV indices. The VVR+ group demonstrated a marked parasympathetic shift, as evidenced by reductions in HR and LF/HF ratio and increases in AVNN and SDNN—patterns consistent with vasovagal episodes.40–42 Conversely, the VVR− group showed no significant changes in autonomic markers, suggesting a more stable physiological response. These differences may reflect an exaggerated vagal reflex triggered by nociceptive or emotional stimuli during DN.39,43 Similar profiles are observed in tilt-test responses among vasovagal-prone individuals, 40 and psychological factors such as perceived control may modulate VVR expression. 44 Overall, these results highlight the importance of HRV as a marker of autonomic regulation during DN.
Pupillometric analysis further confirmed divergent autonomic responses: dilation in the VVR+ group likely reflects increased sympathetic arousal or vagal activation preceding the VVR,45,46 while constriction in the VVR− group may indicate habituation or reduced sympathetic tone. 47 These pupil dynamics, influenced by both subcortical autonomic circuits and cognitive processing, reinforce the value of pupillometry in assessing real-time autonomic regulation during invasive procedures.48,49 No significant changes were observed in skin perfusion between groups or across time. This suggests that cutaneous blood flow, as measured by LDF, may not be a sensitive marker for detecting acute autonomic changes in this context.50,51 Limitations of LDF (such as motion artifacts, probe pressure, circadian variability, and high interindividual variability) may obscure transient perfusion changes.52,53 Moreover, compensatory vascular mechanisms may buffer expected shifts. 54
The PPT increase in VVR+ participants may reflect reflex-linked analgesia accompanying vagal predominance, rather than a direct therapeutic analgesic effect attributable to DN exposure alone. Experimental work shows that vagal afferent activation can modulate nociceptive transmission and engage descending inhibitory circuits, which could plausibly elevate PPTs in the acute post-event window. 55 This distinction is important since a transient increase in PPT associated with VVR physiology should not be over-interpreted as evidence of superior DN efficacy, but rather as a marker of autonomic reflex engagement in susceptible individuals. 56 By contrast, the absence of significant changes in PPT in the VVR− group reinforces the notion that autonomic activation—particularly vagal dominance—may play a central role in mediating the immediate analgesic effects of DN in this context. Supporting this, prior research has shown that DN can activate the autonomic nervous system and produce short-term analgesic effects through both opioid and non-opioid pathways, although the extent of central modulation remains under investigation.57,58
No significant changes were found in systolic or diastolic BP across time or between groups, except for a small, isolated drop in diastolic BP in the VVR− group, which lacked support in the ANOVA and is likely not clinically relevant. BP regulation is highly complex and sensitive to multiple factors and may not reliably capture transient autonomic shifts.59,60 Despite HRV changes in the VVR+ group, these did not translate into sustained BP variations, reinforcing the limited utility of BP as a marker for acute vasovagal detection. 61 Indeed, although decreases in BP are commonly reported at the onset of prodromal symptoms during vasovagal episodes, such changes may be transient or occur too late to serve as early indicators in experimental settings. 62 These findings underscore the importance of considering additional autonomic markers, such as HRV or pupillometry, when evaluating acute VVRs in clinical or research contexts.
From a clinical workflow perspective, the value of multimodal monitoring in this study is not to “over-medicalize” DN, but to delineate whether measurable autonomic shifts occur broadly or are concentrated in patients who develop VVR. If changes are largely confined to VVR+ individuals, clinicians may prioritize risk screening and targeted precautions (supine positioning, anxiety mitigation, readiness protocols) rather than assuming DN routinely modulates autonomic physiology. Clinically, identifying patients prone to VVR is important given the distinct autonomic and sensory responses observed. VVR+ individuals were more frequently female and reported higher stress, aligning with predictors reported in prior studies.52,63 Given the potentially abrupt nature of VVR, individualized monitoring is recommended (especially for at-risk patients), including screening for stress or anxiety, observing autonomic signs, and ensuring clinical readiness for adverse events. 17 Addressing these factors can improve both safety and patient adherence. 64
This study presents several methodological limitations that should be acknowledged when interpreting the findings. First, the uneven group sizes may have affected statistical power. Second, the intervention was limited to a single DN session, with no follow-up to determine whether effects persisted over time. Another limitation is the absence of psychological variables such as anxiety, pain catastrophizing, or trait vagal tone, which could provide further information on individual susceptibility to vasovagal reactions. The VVR frequency in the present cohort (14%) exceeds many published rates 19 for acupuncture/needling procedures. This discrepancy may reflect differences in posture (seated testing) and the VVR case definition that includes presyncope, which can inflate incidence compared with studies counting only frank syncope. Importantly, vasovagal rates vary by procedure context; for example, musculoskeletal injection settings report VVR in the ~2%—3% range, 52 with higher risk in females and younger patients, emphasizing that positioning and context materially affect observed incidence. All procedures were performed by a single experienced physiotherapist at one clinical site, which improves procedural consistency but may limit external validity. Future multicenter studies, including multiple clinicians with predefined competency criteria, should evaluate reproducibility across settings. Future studies should aim to include larger and more balanced samples, adopt longitudinal designs with repeated DN sessions, and incorporate psychophysiological profiling to better understand the interplay between emotional, autonomic, and pain responses. Moreover, exploring preventive strategies—such as patient education, stress regulation techniques, or modified needling protocols—may help reduce the incidence or intensity of VVRs in clinical settings.
Conclusion
In conclusion, this study provides novel information on the physiological and clinical characteristics associated with VVR during DN in patients with MPS. The findings demonstrate that individuals who experience a VVR exhibit distinct autonomic patterns (marked by parasympathetic dominance) and sensory modulation, as evidenced by changes in HRV, pupillometry, and PPT. These responses were not observed in patients without VVR, highlighting the importance of recognizing and monitoring this subset of individuals in clinical practice. Although further research is needed to confirm these findings and explore long-term effects, the current results underscore the need for individualized assessment and vigilance during DN procedures, particularly in patients with higher stress levels or other predisposing factors.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X261422368 – Supplemental material for Physiological correlates and predictors of vasovagal responses following dry needling in myofascial pain syndrome: a controlled observational study
Supplemental material, sj-docx-1-tab-10.1177_1759720X261422368 for Physiological correlates and predictors of vasovagal responses following dry needling in myofascial pain syndrome: a controlled observational study by Robert Trybulski, Adrian Kużdżał, Gracjan Olaniszyn, Bartosz Kapustka, Wiesław Marcol and Katarzyna Walicka-Cupryś in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-docx-2-tab-10.1177_1759720X261422368 – Supplemental material for Physiological correlates and predictors of vasovagal responses following dry needling in myofascial pain syndrome: a controlled observational study
Supplemental material, sj-docx-2-tab-10.1177_1759720X261422368 for Physiological correlates and predictors of vasovagal responses following dry needling in myofascial pain syndrome: a controlled observational study by Robert Trybulski, Adrian Kużdżał, Gracjan Olaniszyn, Bartosz Kapustka, Wiesław Marcol and Katarzyna Walicka-Cupryś in Therapeutic Advances in Musculoskeletal Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
