Abstract
Background:
Treating rheumatic musculoskeletal diseases (RMDs) during pregnancy and breastfeeding presents significant complexities, mainly due to inconsistencies between the clinical guidance documents and the reference safety information, including the summary of product characteristics (SmPC) and the patient information leaflets (PIL).
Objectives:
To assess healthcare professionals’ (HCPs) prescribing behaviors, comfort levels, and challenges when advising patients, focusing on discrepancies between clinical guidance documents and SmPC/PIL.
Design:
Online survey entitled PRAISE (Perception of healthcare providers Regarding Antirheumatics in pregnancy and breastfeeding: advice, Information and patient perSpEctives) and disseminated through HCPs groups and social media.
Methods:
A cross-sectional survey was conducted among 414 HCPs globally. Respondents were divided into prescribers (n = 336) and non-prescribers (n = 78) based on their self-reported role in prescribing antirheumatic medications to pregnant or breastfeeding patients with RMDs. The survey covered demographics, clinical experience, confidence in prescribing, use of clinical guidelines, and experiences managing conflicting information between guidelines and SmPC/PIL.
Results:
Prescribers were more likely than non-prescribers to feel comfortable discussing medication safety during pregnancy. Most prescribers found clinical guidance documents useful, with 48% rating them as “very useful” and 38% as “extremely useful.” In case of conflicting information between clinical guidance documents and SmPC/PIL, 58% of HCPs reported that it caused confusion and tension in patient–doctor relationships, and almost 20% of them are “likely” or “very likely” to discontinue ongoing treatment. Clear communication and shared decision-making were the most common strategies used to address patient concerns.
Conclusion:
HCPs often face significant challenges when advising patients with RMDs on the use of medications during pregnancy and breastfeeding. Conflicting information between clinical guidance documents and SmPC/PIL can disrupt patient–doctor relationship and lead to treatment discontinuation, with potential consequences on maternal disease control. Improved alignment between clinical guidance documents and the SmPC/PIL could enhance patient care and prevent confusion among HCPs and patients.
Plain language summary
Confidence in prescribing: HCPs who prescribe medications were more comfortable discussing medication safety during pregnancy than those who do not prescribe. Usefulness of guidelines: Most prescribers found clinical guidelines very useful in managing patient care. Conflicting information: Over half of the HCPs reported feeling confused or tense when dealing with conflicting information between guidelines and medication safety documents. This confusion can lead to discontinuing treatments, which might negatively impact disease control. Communication strategies: HCPs often use clear communication and shared decision-making to address patient concerns.
In conclusion, PRAISE highlights the need for better alignment between clinical guidelines and medication safety information to improve patient care and reduce confusion. This could help ensure that pregnant and breastfeeding women with RMDs receive the best possible.
Keywords
Introduction
Treating rheumatic musculoskeletal diseases (RMDs) during pregnancy is essential to minimize the risk of adverse maternal and perinatal outcomes such as miscarriage, fetal growth restriction, preeclampsia, and preterm birth, all of which can be exacerbated by active maternal disease. 1 Discontinuation of established medical treatment during the preconception period or in early pregnancy can result in disease flares and suboptimal pregnancy outcomes.1 –3 Therefore, the benefit–risk assessment of medication use during pregnancy should be addressed during preconception counseling, with a treatment plan established through shared decision-making.4,5
Clinical guidance documents from leading rheumatology societies, including the European Alliance of Associations for Rheumatology (EULAR), the British Society for Rheumatology (BSR), and the American College of Rheumatology (ACR), generally advocate for the use of compatible antirheumatic drugs during the periconception period, pregnancy, and breastfeeding.6 –10
In 2023, the European Medicines Agency (EMA) updated the reference safety information for hydroxychloroquine (HCQ), citing a single study that indicated a slight increase in congenital malformations, especially at higher doses. 11 Prior evidence from observational studies and meta-analyses, which unanimously supported HCQ’s safety at standard doses for RMDs, was removed in this update. The French National Agency for Medicines Safety (ANSM) promptly issued a public warning about using HCQ during pregnancy, which led to great confusion among patients and physicians, who promptly liaised with ANSM to clarify the issue. 12 This scenario soon ignited an international debate among healthcare professionals (HCPs) and patients. 13
This series of events prompted worldwide discussion among HCPs who treat patients with RMDs during their family planning journey. The conflicting message between regulatory guidance in the summary of product characteristics (SmPC) and patient information leaflet (PIL) and established clinical guidance documents has been brought to attention, alongside its possible implications on the management of patients. 13
To gain further insight into this matter, the PRAISE (Perception of health care providers Regarding Antirheumatics in pregnancy and breastfeeding: advice, Information and patient perSpEctives) survey was conducted to explore how HCPs perceive and approach the advice on antirheumatic medications during pregnancy and breastfeeding. The survey also examined the impact of conflicting information from clinical guidance documents and SmPC/PIL on shared decision-making, focusing on how it affects the patient–HCPs relationship and therapeutic decisions during pregnancy.
Methods
From June to December 2023, we conducted a multinational, electronic survey to capture the perspectives of HCPs regarding the use of antirheumatic drugs in pregnancy and breastfeeding. The survey, designed as an open survey using SurveyMonkey, was disseminated to a convenience sample HCPs, including doctors, nurses, and midwives worldwide. The survey was promoted by the EULAR Study Group on Reproductive Health and Family Planning (ReHFaP), which targeted networks of HCPs specializing in the management of RMDs and pregnancy (e.g., the Study Group on Gender Medicine of the Italian Society for Rheumatology). The survey was launched at the EULAR annual congress in Milan in June 2023 and was circulated on several social media platforms by the members of the ReHFaP Study Group.
The 24-question survey was designed to examine how HCPs perceive and approach the prescription of antirheumatic medications during pregnancy and breastfeeding. The survey delved into the usage of clinical guidance documents and their influence on HCPs’ clinical practice. Furthermore, it sought to understand how HCPs manage conflicting information between clinical guidance documents and reference safety information, including the SmPC/PIL.
The questionnaire was developed to gather both quantitative and qualitative data. It included a mix of closed-ended questions with predefined response options (e.g., multiple-choice, Likert scales, and Visual Analog Scales (VAS)) and open-ended responses to allow for more detailed feedback.
The survey was divided into four sections: demographic information, professional background, clinical practice regarding pregnancy-related prescribing, and perceptions of guideline use and patient communication. The initial questions focused on the respondents’ profession, country of practice, specialty, and years of experience, to contextualize their answers and allow for subgroup analysis based on these variables.
Subsequent questions aimed to assess the respondents’ confidence in prescribing antirheumatic medications during pregnancy and breastfeeding, their familiarity with clinical guidance documents (e.g., EULAR, ACR, BSR), and their experiences managing the conflicting information between guideline recommendations and the SmPC/PIL. Several questions used Likert scales to assess the utility of guidelines, comfort with decision-making, and potential impacts on the patient–doctor relationship. VAS were employed to quantify subjective responses, particularly regarding comfort in discussing complex risk–benefit scenarios with patients. The full survey is shown in Supplemental Material.
Before circulation, the survey was face-validated. Face validity was assessed using a panel of three experts in the field of rheumatology, and minor modifications were made to the wording of items to improve clarity. The survey design adhered to the CHERRIES (Checklist for Reporting Results of Internet E-Surveys) framework, 14 ensuring precise question phrasing, logic validation, and respondent anonymity to enhance reliability and reduce response bias. A completeness check was performed after submitting the questionnaire by highlighting missing items (all items were mandatory). Where applicable, items provided a non-response option such as “not applicable/prefer not to say.” No ethical approval was required for this survey.
Data were automatically downloaded into a spreadsheet. According to the response to the question, “Do you prescribe anti-rheumatic medications in patients who are planning pregnancy, are pregnant, or lactating?,” HCPs were divided into two groups: prescribers and non-prescribers.
Data were reported as numbers and proportions for categorical variables and as median values (interquartile range (IQR)) for continuous variables. Proportions were calculated according to the appropriate denominator, considering prescribers and non-prescribers.
Results
A total of 414 HCPs from 42 countries (Supplemental Figure 1) from various specialties completed the PRAISE survey (85% of 468 individuals who attempted the survey). Responses were checked for duplicates to ensure that the same respondent did not complete the survey twice. Most respondents (n = 336, 81%) were prescribers of antirheumatic medications, while 78 (19%) were non-prescribers (Supplemental Figure 2). The characteristics of respondents are shown in Table 1.
Characteristics of respondents to the PRAISE survey (N = 414), subdivided according to the prescription of antirheumatic medications to people who are planning for a pregnancy or are pregnant/lactating.
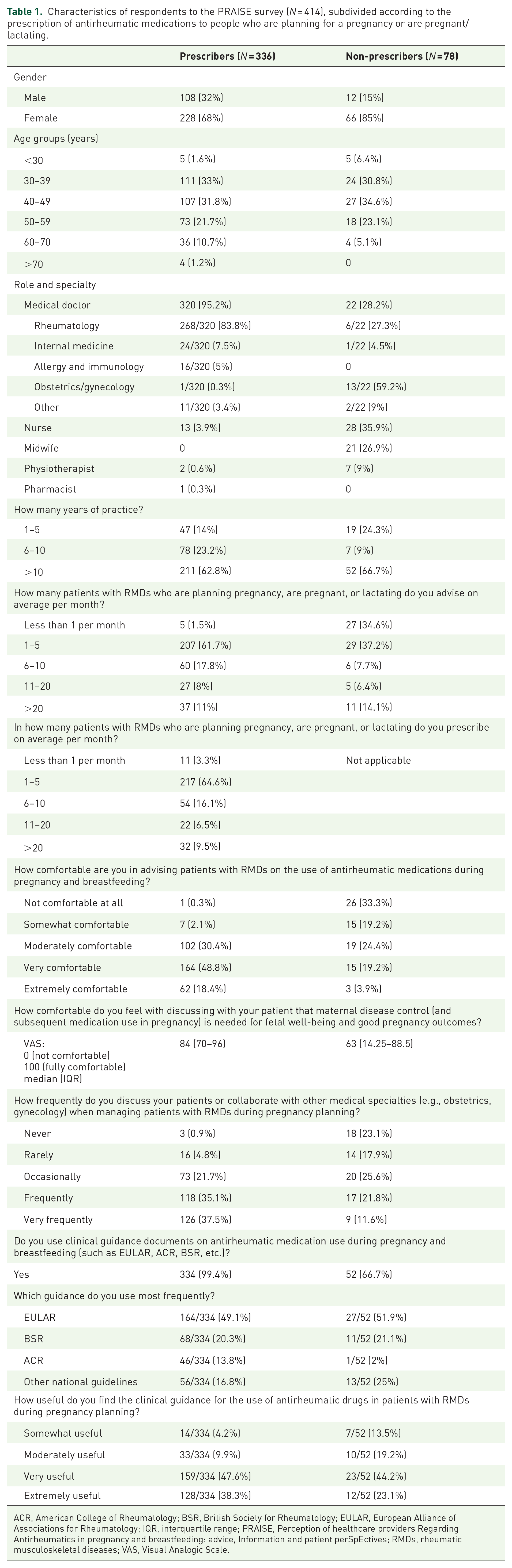
ACR, American College of Rheumatology; BSR, British Society for Rheumatology; EULAR, European Alliance of Associations for Rheumatology; IQR, interquartile range; PRAISE, Perception of healthcare providers Regarding Antirheumatics in pregnancy and breastfeeding: advice, Information and patient perSpEctives; RMDs, rheumatic musculoskeletal diseases; VAS, Visual Analogic Scale.
Among prescribers, 228 (68%) were female, compared to 66 (85%) among non-prescribers. The age group ranging 30–49 years comprised 218 (65%) and 51 (66%) respondents among prescribers and non-prescribers, respectively.
Prescribers were mostly medical doctors (n = 320, 95%), with the majority specializing in rheumatology (n = 268/320, 84%). Other specialties included internal medicine (n = 24/320, 7%), allergy and immunology (n = 16/320, 5%), and obstetrics/gynecology (n = 1/320, 0.3%). Non-prescribers were more evenly distributed across roles, with 22 (28%) being medical doctors—predominantly in obstetrics/gynecology (n = 13/22, 59%)—in addition to nurses (n = 28/78, 36%) and midwives (n = 21/78, 27%). Most prescribers (n = 211, 63%) had more than 10 years of practice, while 78 (23%) had 6–10 years, and 47 (14%) had 1–5 years of experience. By contrast, 52 (67%) of non-prescribers had over 10 years of practice, with fewer in the 1–5 years (n = 19, 24%) and 6–10 years (n = 7, 9%) categories. Nearly all prescribers (n = 334, 99%) reported using clinical guidance documents such as those endorsed by EULAR, ACR, or BSR. EULAR documents were the most frequently used (49%). Among non-prescribers, 52 (67%) used guidance documents, with the same preference for EULAR (52%).
Addressing conflicting information
On a scale from 0 to 100, where 0 means “not comfortable” and 100 means “fully comfortable,” the median comfort level of respondents in discussing the lack of data as a reason for pregnancy warnings was 74 (IQR 51–90). Prescribers demonstrated greater comfort with a median comfort level of 77 (IQR 60–91), compared to 52 (IQR 15–80) for non-prescribers (Question 1/Figure 1). Prescribers were more likely to continue treatment when clinical guidance documents supported drug safety in pregnancy, even if product information suggested otherwise, as compared to non-prescribers (63% of prescribers vs 31% of non-prescribers; Question 2/Figure 2). However, the survey also revealed that almost 20% of HCPs are likely or very likely to discontinue ongoing treatment when confronted with conflicting information between clinical guidance documents and SmPC/PIL (Question 2/Figure 2). The probability that a patient would stop taking a medication if the SmPC or PIL states that the drug may be associated with “a small increased risk of major malformations and should not be used during pregnancy unless your doctor considers the benefits outweigh the risk” was overall rated as a median value of 63 (IQR 43–80) on a scale where 0 corresponded to “absolutely unlikely” and 100 to “absolutely likely” (Questions 3/Figure 3).

Question 1—Ease of discussion of a lack of data for the use of medications during pregnancy. The scores were subdivided into three categories: <30, 30–70, and >70. Percentages within the bars were calculated with reference to prescribers and non-prescribers as denominators.
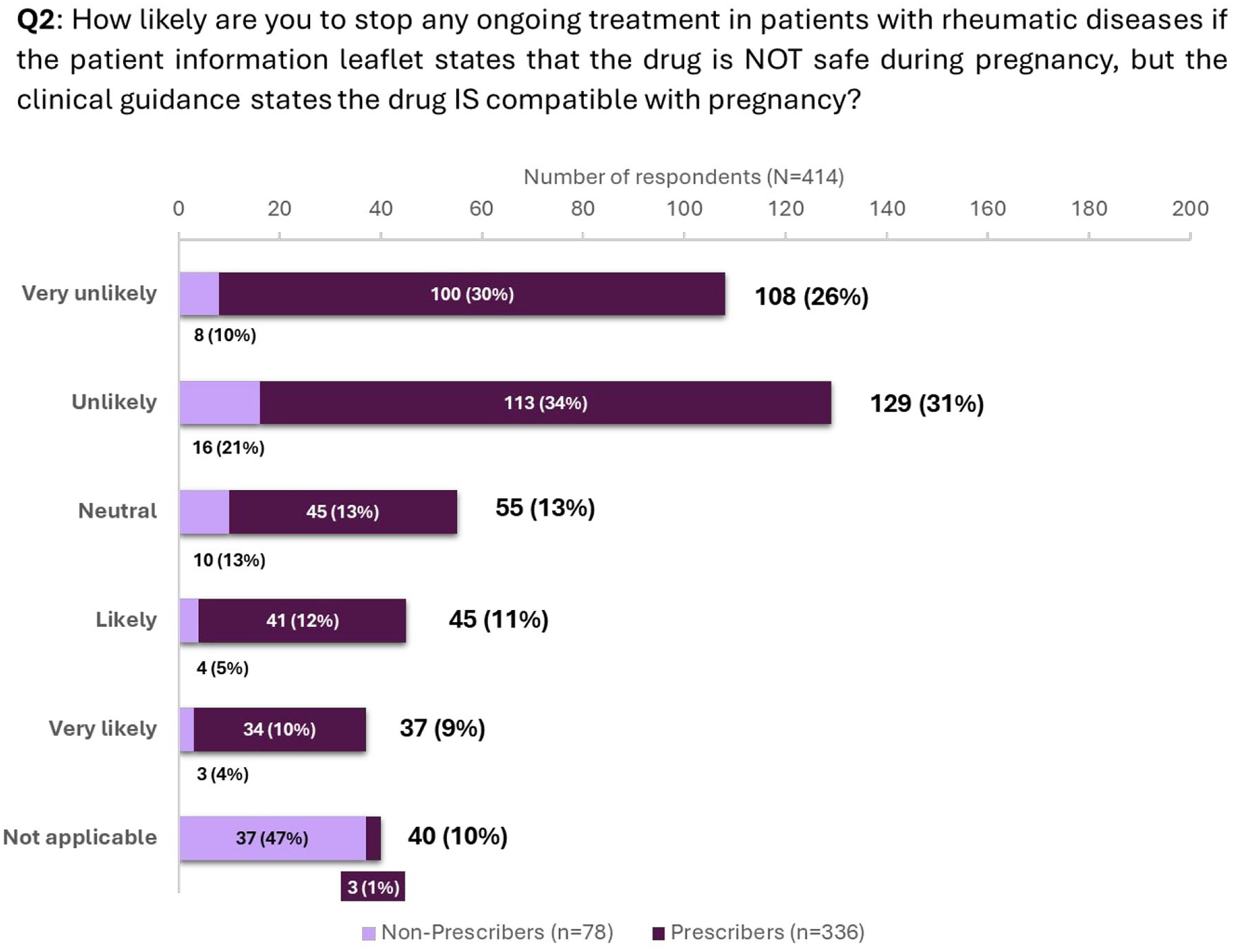
Question 2—Likelihood of healthcare professionals discontinuing medications in the case of conflicting information. Percentages within the bars were calculated with reference to prescribers and non-prescribers.

Question 3—Likelihood of patients discontinuing medications in the case of conflicting information. The scores were subdivided into three categories: <30, 30–70, and >70. Percentages within the bars were calculated with reference to prescribers and non-prescribers as denominators.
Patient concerns and doctor–patient relationship
A total of 249 (60%) HCPs reported that patients frequently or very frequently expressed concerns or worries about discrepancies between PIL and their medical advice (Question 4/Figure 4). In 58% of cases, prescribers indicated that conflicting information caused confusion and tension in the patient–doctor relationship (Question 5/Figure 5), and 52% engaged in shared decision-making with patients to address their concerns (Question 6/Figure 6). Assessing the impact of conflicting guidance between specialties, when encountering conflicting information between HCP specialties, 69% of prescribers and 73% of non-prescribers reported that it caused tension in the patient–doctor relationship (Question 7/Figure 7).

Question 4—Likelihood of patients expressing concerns when informed about conflicting information. Percentages within the bars were calculated with reference to prescribers and non-prescribers.

Question 5—Impact of conflicting information on patient–physician relationship. Percentages within the bars were calculated with reference to prescribers and non-prescribers.
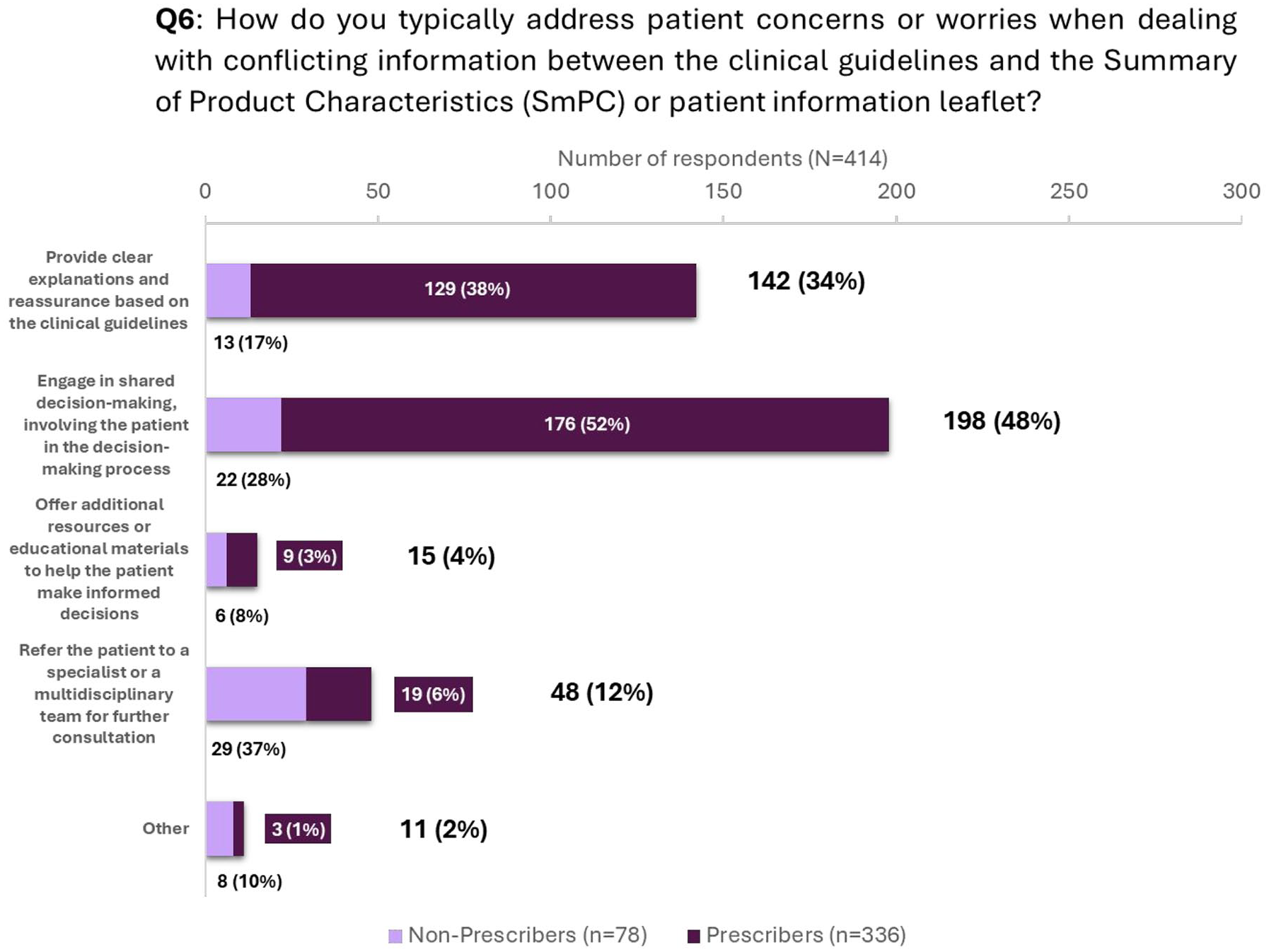
Question 6—Strategies to address patient concerns in the case of conflicting information. Percentages within the bars were calculated with reference to prescribers and non-prescribers.

Question 7—Impact of conflicting advice between different healthcare professionals on patient–physician relationship. Percentages within the bars were calculated with reference to prescribers and non-prescribers.
The respondents were grouped upon demographic variables such as sex, age (< or ⩾40 years of age), profession (doctors vs non-doctors), experience (⩽10 years of experience or >10 years of experience), and geographic areas of practice (Geographic Europe vs other continents); there were no significant differences in the answers of the respondents while comparing the two groups identified upon each variable (Supplemental Tables for Questions 1–7).
Discussion
To the best of our knowledge, the PRAISE survey is the first study to highlight critical challenges that HCPs face in a real-world setting when prescribing antirheumatic medications to patients with RMDs who are planning pregnancy, are pregnant, or breastfeeding.
Prescribers, particularly rheumatologists with more than 10 years of experience, responded that they were generally comfortable with advising patients on antirheumatic drug use during pregnancy. By contrast, non-prescribers, often less experienced with clinical guidance documents, showed lower confidence levels, reflecting their reliance on SmPC and PIL over clinical judgment.
One crucial observation is that respondents comprised mostly experienced HCPs (with >10 years of experience). Still, one out of five HCPs reported that they are “likely” or “very likely” to stop ongoing medication if the SmPC states that the drug should not be taken during pregnancy. This highlights a vital strength of the study: It captures real-world prescribing behavior and a strong clinician reliance on advice in the SmPC, even in the face of clinical guidance documents indicating that the drug is compatible with pregnancy and breastfeeding. In real-world practice, where HCPs may be less specialized in this area, the number of HCPs stopping medications based solely on the SmPC and PIL might be significantly higher.
The PRAISE study also showed that conflicting information between clinical guidance documents and SmPC/PIL often leads to confusion and strain in the patient–doctor relationship, with over half of prescribers reporting tension in these situations. Shared decision-making emerged as an important strategy for resolving patient concerns, with more than half of prescribers involving patients in treatment decisions. However, conflicting information between specialties, such as rheumatology and obstetrics, further complicates these discussions, highlighting the need for coordination between different disciplines.
For the first time, we investigated the impact of misalignment in the information from clinical guidance documents and SmPC/PIL on medication use during pregnancy and breastfeeding on the relationship between HCPs and patients and their shared decision-making process. The study’s major strengths include its relatively large sample size, distribution across several countries, and demographic homogeneity. Its focus on real-world clinician experiences provides valuable insights into the management of RMDs in pregnancy.
Our study has some limitations. An important limitation is that the study heavily represents prescribers from high-income countries, with some countries being overrepresented, potentially overlooking variations in prescribing practices in lower-income settings. To keep the survey as concise as possible, we missed out on variables that would have informed us about regional and healthcare setting differences, such as rural versus urban setting, and academic versus community practice. Dissemination through social media and congress attendees is likely to have generated a selection bias among respondents. Particularly, the data sampling may be skewed toward experts in the field, as those with a strong interest in family planning for patients with RMDs may be more likely to respond to the survey. This bias could lead to an overrepresentation of HCPs with advanced knowledge and experience, potentially influencing the results toward a more expert-driven perspective. Consequently, the findings may only partially reflect the practices or decision-making processes of the broader healthcare provider population, who might have less familiarity with the topic. From an HCP perspective, the skewing toward experts is essential because it may overestimate the confidence and knowledge in managing medications during pregnancy; this might potentially not reflect the uncertainty that less experienced providers may encounter in clinical practice. For patients, this implies that the results may not mirror the typical advice they receive, as non-expert providers may be more conservative in their approach, possibly leading to higher rates of discontinuation of medications based on safety information leaflets rather than clinical guidelines.
Tying the discussion back to the safety update of HCQ in 2023, the PRAISE survey highlights a critical gap that reflects the misalignment of SmPC/PIL and clinical guidance documents. The controversy surrounding HCQ may have led to the potentially unnecessary cessation of established long-term treatment in patients requiring immune regulation during pregnancy. HCQ is a cornerstone drug in pregnant patients requiring immune modulation. In patients with systemic lupus erythematosus (SLE) in particular, risk factors for developing a flare during pregnancy include hypocomplementemia, active disease in the 6 months before pregnancy, and discontinuation of HCQ therapy.15 –18 Data from other cohort studies suggest that continuing HCQ during pregnancy decreases pregnancy loss, preterm birth, and SLE flare rates.19,20 Infra-therapeutic blood HCQ levels that can be explained by sub-optimal adherence (possibly due to safety concerns in the context of pregnancy) are also associated with severe flares. 21 Maintaining HCQ treatment in SLE patients is crucial to improve the chances of a successful pregnancy. 22
The safety update of HCQ may have resulted in conservative decisions on other antirheumatic medications that display conflicting information. If non-expert HCPs or patients prioritize outdated or overly cautious safety warnings in SmPC/PIL, this could lead to the unnecessary discontinuation of vital medications, which, in turn, may harm both mothers and their babies by failing to control maternal disease effectively during pregnancy.
Putting the study findings in context and determining action plans, there is an urgent unmet need for harmonization between clinical guidance documents and SmPC/PIL. Collaborative efforts between regulatory bodies, clinical societies, and other stakeholders are crucial to ensure that safety information is up-to-date, complete, and reflective of the best available evidence, particularly when new data emerge from pregnancy registries and other observational studies. 23 In addition, the involvement of multidisciplinary teams in a decision process can provide a more comprehensive approach to managing these often complex cases, ensuring that the risks and benefits of treatment options are thoroughly considered and communicated to the patient. 24
For harmonization between clinical guidance documents and SmPC/PIL, it is worth looking into how regulatory bodies other than EMA provide safety information for medication use during the periconception period, pregnancy, and breastfeeding. For example, the US Food and Drug Administration (FDA) updated the Pregnancy and Lactation Labeling Rule in 2015, eliminating the previous A, B, C, D, and X grading system for medication use in pregnancy. Although physicians and patients had appreciated this system’s relative ease of use, it was often misconstrued. It was not regularly updated to include new data suggesting enhanced compatibility of medications with pregnancy. 3 The new label was designed to include more clinically relevant data, incorporating data from human studies and links to existing pregnancy registries while relying less on animal data. 25 A vital goal of the new label was to assist physicians and patients in weighing the risks and benefits of medications against the risks during pregnancy in women with chronic, untreated illnesses. 3 The FDA approach may highlight one crucial aspect, in that a label change alone may not be sufficient to ensure patient safety, and that cross-sectoral stakeholder involvement is essential in the implementation of new label formats.26 –32 The imminent revision of the EMA guideline on risk assessment of medicinal products on human reproduction and lactation presents a timely opportunity to align the perspectives of regulators and HCPs. 33
Conclusion
The PRAISE survey underscores the significant negative impact that conflicting information between clinical guidance documents and SmPC/PIL has on the management of RMDs during the reproductive journey. Discrepancies between clinical guidelines and regulatory information can lead to confusion and suboptimal decisions. Therefore, complete labeling information is essential for optimizing care. The periodic label structure update for pregnancy and breastfeeding information represents a unique window of opportunity to enhance the ecosystem that supports regulatory decisions, to foster a positive experience of shared decision-making for both patients and HCPs.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X251350087 – Supplemental material for Impact of conflicting information on the use of antirheumatic drugs in pregnancy and breastfeeding: perspectives of healthcare providers from the global PRAISE survey
Supplemental material, sj-docx-1-tab-10.1177_1759720X251350087 for Impact of conflicting information on the use of antirheumatic drugs in pregnancy and breastfeeding: perspectives of healthcare providers from the global PRAISE survey by Karen Schreiber, Christine Graversgaard, Ioannis Parodis, Nancy Agmon-Levin, Silvia Aguilera, Aleksandra Antovic, George K. Bertsias, Ilaria Bini, Anca Bobirca, Susana Capela, Ricard Cervera, Nathalie Costedoat-Chalumeau, Radboud Dolhain, Oseme Etomi, Julia Flint, Joao Eurico Fonseca, Ruth Fritsch-Stork, Frauke Förger, Ian Giles, Bethan Goulden, Carina Götestam Skorpen, Iva Gunnarsson, Latika Gupta, Hege Svean Koksvik, Louise Linde, Jacob Lykke, Yvette Meissner, Anna Molto, Louise Moore, Marta Mosca, Catherine Nelson-Piercy, Luis Fernando Perez-Garcia, Luigi Raio, Ane Lilleoere Rom, Amihai Rottenstreich, Muna Saleh, Savino Sciascia, Anja Strangfeld, Elisabet Svenungsson, Maria G. Tektonidou, Angela Tincani, Anne Troldborg, Jelena Vojinovic, Anne Voss, Marianne Wallenius, Nuria Zuniga-Serrano and Laura Andreoli in Therapeutic Advances in Musculoskeletal Disease
Footnotes
Acknowledgements
The Study Group on Reproductive Health and Family Planning (ReHFaP) endorsed by the European Alliance of Associations for Rheumatology (EULAR) is kindly acknowledged for its participation in designing and disseminating the survey and for critically discussing the results. We thank all the networks who helped to disseminate the survey, and Kirsten Frøhlich from the Centre of Expertise in Rheumatology (CeViG) for her unwavering logistic support.
Declarations
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
