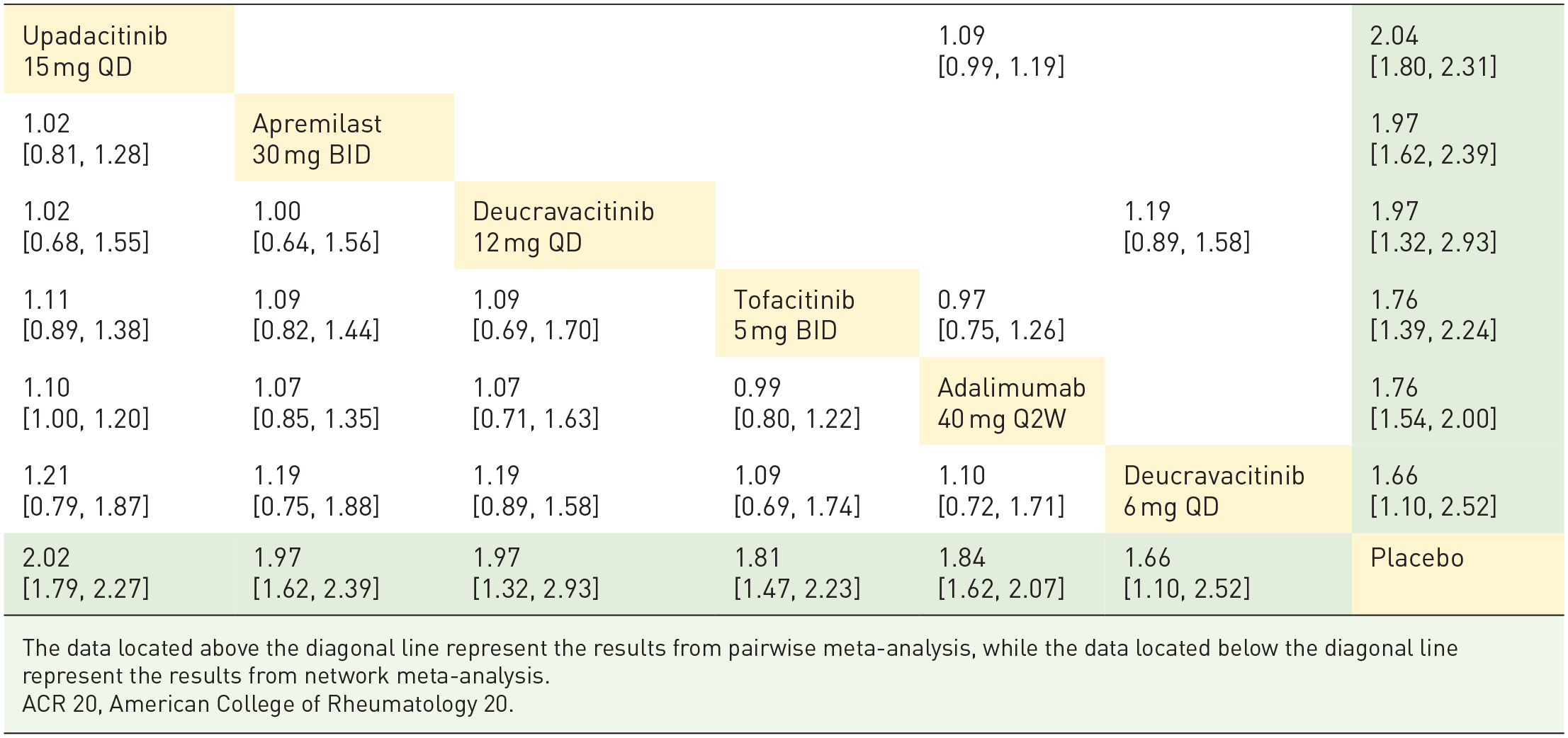Abstract
Background:
Although there have been network meta-analyses (NMAs) regarding the efficacy and safety of targeted immunotherapy for psoriatic arthritis, most of them primarily focus on biologics. However, small molecules and biologics have many differences in properties and onset time. This NMA put emphasis on small molecule drugs and incorporates medications including upadacitinib and deucravacitinib, which have been less compared previously.
Objective:
To compare the efficacy and safety of small molecule drugs (apremilast, deucravacitinib, tofacitinib, and upadacitinib) in active psoriatic arthritis (PsA) using NMA.
Design:
This study was conducted and reported following the guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-analyses Extension Statement for Network Meta-Analyses (PRISMA-NMA), with relevant articles identified through searches across electronic databases.
Data sources and methods:
Databases including PubMed, Cochrane Library, and ClinicalTrial.gov were searched. Randomized controlled trials (RCTs) of targeted synthetic small molecules for PsA controlled by either placebo or active comparators within weeks 12–16 were eligible. A frequentist framework and a random-effect model were employed for the analysis. The revised Cochrane risk-of-bias tool for RCTs was used to assess the quality of the enrolled studies.
Results:
A total of 9 RCTs involving 3699 patients were analyzed. Apremilast 30 mg bid, deucravacitinib 6 mg and 12 mg qd, tofacitinib 5 mg bid, and upadacitinib 15 mg qd all demonstrated superior efficacy over placebo in achieving American College of Rheumatology (ACR) 20/50/70, Psoriasis Area and Severity Index (PASI) 75, and Health Assessment Questionnaire-Disability Index (HAQ-DI) scores at weeks 12–16. When compared to adalimumab, upadacitinib achieved borderline superiority in PASI 75 at week 12 (risk ratio (RR) = 1.20, 95% confidence interval (CI): 1.02–1.40). Among the four medications studied, only deucravacitinib showed significantly better HAQ-DI scores compared to apremilast (RR = −0.16, 95% CI: −0.29 to −0.02), with no statistically significant differences observed in other parameters.
Conclusion:
All four small molecule oral medications exhibited superior efficacy to placebo and comparable safety profiles. Across different assessment criteria, including ACR 50, ACR 70, PASI 75, or HAQ-DI, deucravacitinib 12 mg and upadacitinib consistently ranked in the top two positions.
Keywords
Introduction
Psoriatic arthritis (PsA) is a chronic inflammatory disease that affects both peripheral and axial joints. 1 Manifestations include psoriasis skin plaques, joint swelling, stiffness, and limited mobility. The arthritis is mainly categorized into five types: asymmetric oligoarticular type, symmetric polyarticular type, distal interphalangeal predominant, arthritis mutilans, and spondylitis type.1–3 Both bone erosion and pathologic bone formation cause significant impacts on quality of life.3,4
Treatment goals encompass controlling inflammation, preventing joint deformities, minimizing skin lesions, normalizing functional activities, facilitating reintegration into society, and ultimately enhancing overall quality of life.5,6 Therefore, this analysis incorporates the results of the American College of Rheumatology (ACR) 20/50/70, Psoriasis Area and Severity Index (PASI) 75, Health Assessment Questionnaire-Disability Index (HAQ-DI), and the incidence of serious adverse events (SAE).5–9
Treatment has revolutionized from conventional disease-modifying antirheumatic drugs (csDMARDs) such as cyclosporine, sulfasalazine, methotrexate, and leflunomide to targeted immunotherapy, which encompasses biologic agents and oral small molecule drugs.10,11 The FDA-approved biologics for PsA including TNFα inhibitors (adalimumab, certolizumab, etanercept, golimumab, and infliximab), IL-12/23 inhibitors (ustekinumab), IL-17 inhibitors (secukinumab, ixekizumab, and brodalumab), and IL-23 inhibitors (guselkumab and risankizumab). However, there are still some remaining gaps in treatment. In recent years, several small molecule drugs have become available as alternatives. These encompass a phosphodiesterase-4 (PDE4i) inhibitor (apremilast), Janus kinase (JAK) inhibitors (tofacitinib and upadacitinib), and a tyrosine kinase 2 (Tyk2) inhibitor (deucravacitinib).5,6,10–12
Numerous randomized controlled trials (RCTs) have assessed the efficacy and safety of treatments for PsA. However, there is a limited number of direct comparisons between different medications. Therefore, network meta-analysis (NMA) becomes necessary to integrate both direct and indirect comparisons, providing evidence-based decision-making for clinical applications. While there have been several NMA articles discussing the therapeutic effects of targeted immunotherapies on PsA, most of them have primarily focused on biologic drugs,13–17 lacking a comprehensive report on small molecule medications. Some include coverage of tofacitinib and apremilast,14–17 but there is limited inclusion of upadacitinib18,19 and deucravacitinib. 20
The reason for separately discussing small molecules and biologics lies in their distinct characteristics. Small molecules possess unique properties that significantly differ from those of biologics, as outlined below: (1) small molecules generally exhibit a faster onset, with primary endpoints often assessed within 12–16 weeks, whereas biologics are evaluated at the 24-week mark; (2) the impact on quality of life differs between oral and injectable formulations; and (3) small molecules tend to have broader specificity, interacting with multiple targets due to their smaller size and structural features. By contrast, biologics typically exhibit high specificity, selectively binding to their intended targets with high affinity. This could potentially lead to an increased likelihood of immune imbalance between anti-Th1/Th2/Th17 with the use of biologics. (4) Small molecules have a lower likelihood of inducing an anti-drug immune response, while biologics have a higher potential for immunogenicity since they are derived from non-self sources (e.g., foreign proteins).21,22 Taking into account the factors mentioned above, this analysis exclusively concentrates on the selection of oral small molecule inhibitors for the treatment of PsA. The objective of this article is to provide recommendations for selecting small molecule drugs for PsA through the use of NMA, allowing for the simultaneous comparison of multiple treatment options.
Methods
Protocol
The NMA was conducted and reported following the guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-analyses Extension Statement for Network Meta-Analyses (PRISMA-NMA). 23 The systematic review has been registered in the PROSPERO database with the registration number CRD42024517595.
Search strategy
We performed a search of electronic databases, including PubMed, Cochrane Library, and ClinicalTrials.gov to identify relevant RCTs from their respective inception dates up to January 19, 2024. In addition, we manually searched the references of eligible studies to find any additional pertinent articles. The keywords used for the search encompassed terms such as “psoriatic arthritis,” “apremilast,” “deucravacitinib,” “tofacitinib,” “upadacitinib,” and “small molecule.”
Eligibility criteria and study selection
The NMA applies the PICO model (Population, Intervention, Comparison, Outcome), comprising the following criteria: (1) P: individuals diagnosed with moderate-to-severe psoriatic arthritis; (2) I: administration of oral small molecule drugs; (3) C: comparison against either placebo or active comparators; and (4) O: assessment of changes in ACR response.
The inclusion criteria were defined as follows: (1) Enrollment of PsA patients in studies controlled by either placebo or active comparators. (2) Trials designed to assess the efficacy and safety of small molecule drugs. (3) RCTs reporting the following outcomes: ACR 20, ACR 50, ACR 70, PASI 75, HAQ-DI scores at weeks 12–16, and SAEs at weeks 12–24. However, clinical trials involving patients who were naïve to both csDMARDs and biologics were excluded. Reports concerning open-label phases or post hoc analyses were also not considered. Y.-C. Tsai and C.-Y. Hung independently reviewed all titles and abstracts after removing duplicates, and thoroughly examined full texts to identify potential trials. Any discrepancies were resolved through discussion with a third author, T.-F. Tsai.
Data extraction and risk of bias evaluation
The extracted data of interest included baseline demographic information (age, gender, and body weight), treatment regimens, participant numbers, and outcome assessments. Our primary outcome measure was ACR 20, while secondary outcomes comprised ACR 50, ACR 70, PASI 75, HAQ-DI, and SAEs. The quality assessment of the enrolled studies was conducted by two investigators (Y.-C. Tsai and C.-Y. Hung) using the revised Cochrane risk-of-bias tool for RCTs. 24 Methodological evaluation determined the overall risk of bias to be categorized as low, high, or of some concern. In the event of discordance, the third author (T.-F. Tsai) was engaged to arbitrate.
Data synthesis and statistical analysis
This study encompassed pharmacological interventions involving all oral small molecule agents for PsA, including apremilast, deucravacitinib, tofacitinib, and upadacitinib. The analysis was executed utilizing MetaInsight (version 5.1.2; Complex Reviews Support Unit, National Institute for Health Research, London, UK) with a frequentist framework. 25 A random-effect model was employed for the NMA. 26 MetaInsight serves as a web-based platform designed for NMA, utilizing the netmeta package in R software to conduct frequentist statistical calculations. 25
Binary outcomes such as ACR 20/50/70 and PASI 75 were reported as risk ratios (RR) with corresponding 95% confidence intervals (CI). Continuous outcomes, like HAQ-DI, were depicted as mean differences (MD) with their respective 95% CI. SAEs were illustrated as risk differences with 95% CI. The treatments were systematically ranked, and numerical data for both indirect and direct comparisons were organized in league tables. In addition, inconsistency tests were conducted to scrutinize any potential disparities within the dataset. Statistical significance was determined by a two-tailed p-value criterion of less than 0.05.
Sensitivity analyses were undertaken using a one-study removal method. Specifically, eliminating one study at a time from the analysis in a sequential manner was done to ascertain that the results were not disproportionately influenced by any single study. Through this process, we could evaluate whether the conclusions and rankings remained consistent regardless of which study was excluded.
Results
Study identification and network model formation
The PRISMA flowchart outlining the literature search process is illustrated in Figure S1.
When initially searching the PubMed and the Cochrane Library databases, a total of 1256 articles were identified. Following the removal of duplicate articles and screening of titles and abstracts, the remaining articles underwent a full-text assessment. Additional findings from ClinicalTrial.gov were integrated. Ultimately, nine RCTs met the eligibility criteria for qualitative and quantitative analyses.27–35
The network diagram for the interventions is presented in Figure 1. In this NMA, the sample size includes a total of 3699 patients across 9 RCTs,27–35 encompassing 4 small molecule drugs (apremilast, deucravacitinib, tofacitinib, and upadacitinib), 1 biologic agent (adalimumab), along with the placebo group.

Network plot of small molecule drugs for PsA.
Patient characteristics
Among the 3699 participants included in our analysis, female patients accounted for 47%–60% of the total, with an average age ranging from 47 to 54 years and an average weight ranging from 82 to 92 kg. Regarding medication history, five studies included patients who previously had inadequate response (IR) to csDMARDs or bDMARDs,31–35 one study required prior use of at least one csDMARD or bDMARD, 30 and the other three studies did not impose any restrictions on whether participants had previously used DMARDs.27–29 For detailed information on the trial phases, the patient numbers in each intervention arm, and the inclusion criteria of individual trials, please refer to Table 1.
Summary of included trials.
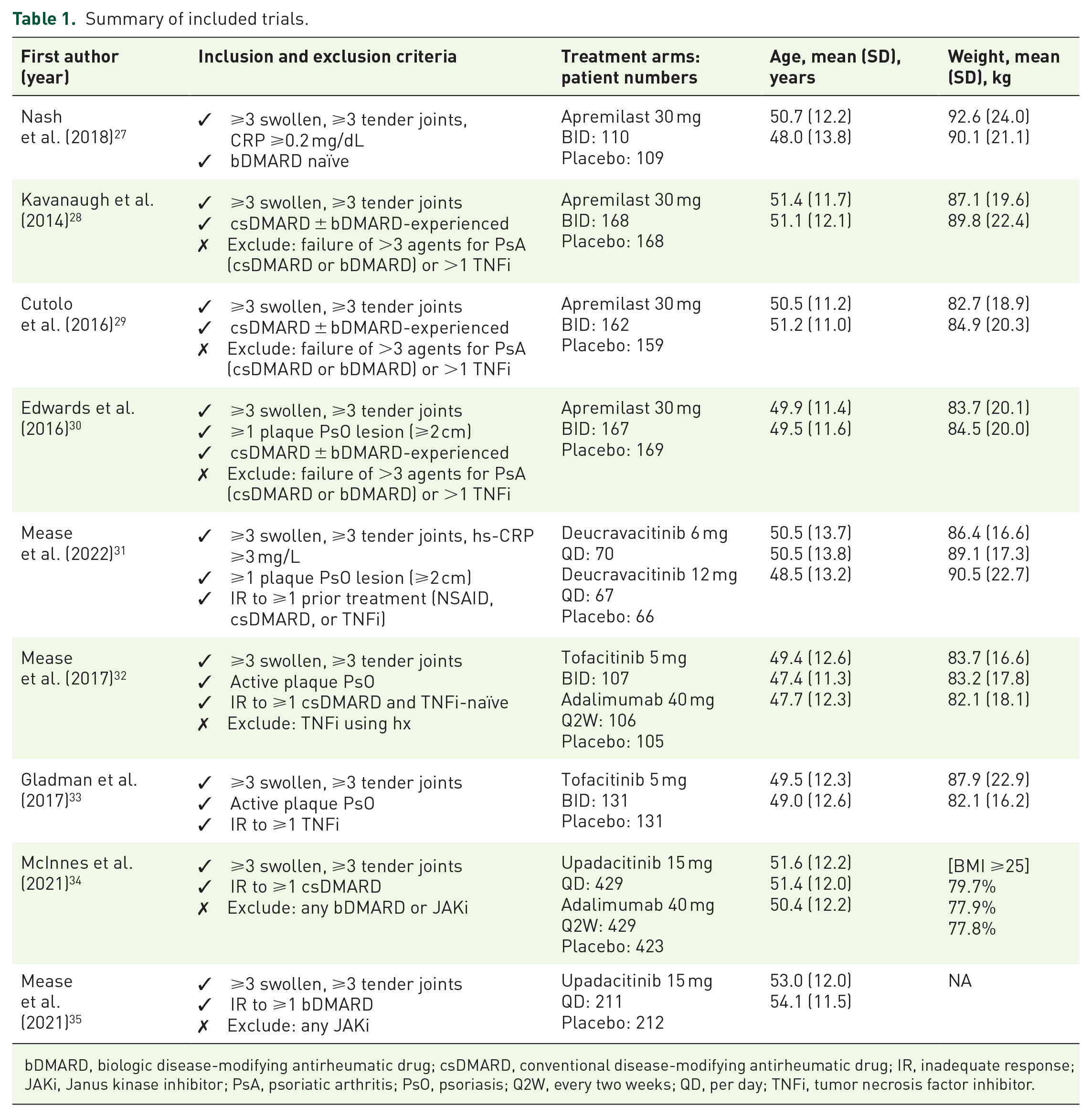
bDMARD, biologic disease-modifying antirheumatic drug; csDMARD, conventional disease-modifying antirheumatic drug; IR, inadequate response; JAKi, Janus kinase inhibitor; PsA, psoriatic arthritis; PsO, psoriasis; Q2W, every two weeks; QD, per day; TNFi, tumor necrosis factor inhibitor.
Methodological quality of the included studies
Concerning the overall methodological quality of the studies, it was observed that 77.8% (7/9) of the studies had a low risk of bias, while 22.2% (2/9) had some risk of bias (refer to Figure S2). The studies with some risk of bias displayed differences in their protocols and in the selection of reported results, which could potentially impact the outcomes of the interventions. The details of the risk of bias assessment are provided in Figure S3.
Primary endpoint: ACR 20
American College of Rheumatology 20
At weeks 12–16, the NMA indicates that all administered drugs surpassed placebo in terms of ACR 20 improvement, but there was no significant difference among all the other drugs. In comparison to the biological agent adalimumab, there was also no statistically significant discrepancy between the oral small molecule agents and adalimumab (Table 2).
League table of ACR 20 at weeks 12–16.

The data located above the diagonal line represent the results from pairwise meta-analysis, while the data located below the diagonal line represent the results from network meta-analysis.
ACR 20, American College of Rheumatology 20.
Secondary endpoint: ACR 50, ACR 70, PASI 75, HAQ-DI, and safety
ACR 50 and ACR 70
Similar results were noted in the outcomes of ACR 50 and ACR 70. All administered oral small molecule drugs demonstrated superior efficacy compared to placebo. However, these oral targeted drugs did not significantly differ from adalimumab in terms of ACR 50 and ACR 70 improvements (Figure 2). Also, there were no statistically significant differences in efficacy observed between the various small molecule inhibitors (Tables S1 and S2).
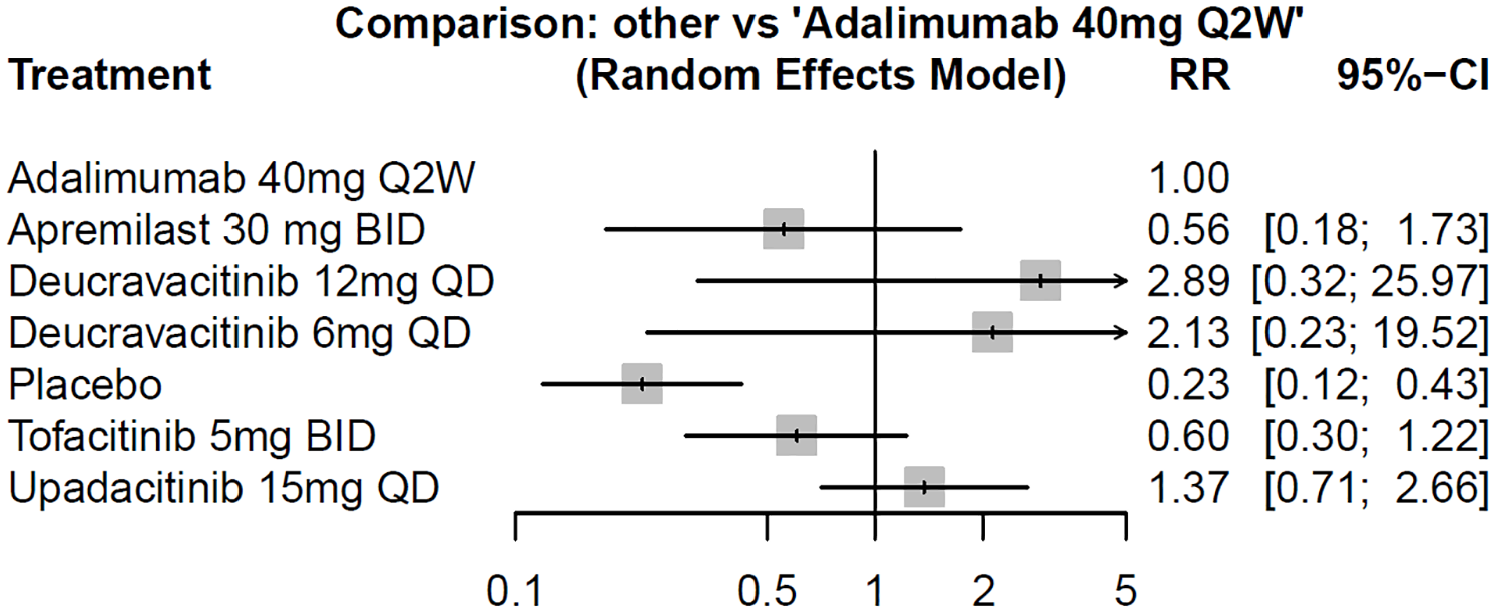
Comparison of ACR 70 between adalimumab and small molecule drugs at weeks 12–16.
Psoriasis Area and Severity Index 75
Regarding the PASI 75 response in patients with PsA, all medications demonstrated better efficacy than placebo (Table S3). Notably, upadacitinib showed a borderline effect in surpassing adalimumab (RR = 1.20, 95% CI: 1.02–1.40) for cutaneous manifestations when using PASI 75 as the assessment tool. Besides, deucravacitinib 12 mg tended greater efficacy than deucravacitinib 6 mg, although the difference was not statistically significant (RR = 1.39, 95% CI: 1.00–1.95).
Health Assessment Questionnaire-Disability Index
In the context of using HAQ-DI as a clinical measure, all administered drugs demonstrated superior performance compared to the placebo. In addition, both deucravacitinib doses of 12 and 6 mg exhibited statistically significant improvements over apremilast. Furthermore, deucravacitinib doses of 12 and 6 mg, as well as upadacitinib, trended toward better outcomes compared to adalimumab, although the differences did not reach statistical significance (Table 3).
League table of HAQ-DI at weeks 12–16.

HAQ-DI, Health Assessment Questionnaire-Disability Index.
Safety
The analysis of the risk difference for SAE occurring within 24 weeks revealed no statistically significant difference between the group treated with each small molecule drug and the placebo group. This result is presented in the league table and the forest plot detailing SAE, both of which can be referenced in Table S4.
Inconsistency test and sensitivity analyses
The inconsistency between indirect and direct comparisons for the outcomes was assessed. All available comparisons (ACR 20/50/70, PASI 75, HAQ, and SAE) yielded p values greater than 0.05, which indicated no evidence of inconsistency between indirect and direct comparisons.
With regard to the sensitivity analysis, the findings of the one-study removal analysis revealed constant rankings and clinical significance across all treatments. Specifically, eliminating one study at a time from the analysis in a sequential manner was done to ascertain that the results were not disproportionately influenced by any single study. This indicates the outcomes of our study remain consistent and are not affected by the inclusion or exclusion of individual studies.
Discussion
Main research findings and clinical implications
The findings of this study indicated that all four small molecule oral drugs exhibited superior efficacy compared to the
In terms of
In addition, the ranking results of HAQ-DI and ACR 70 were quite similar, with both showing deucravacitinib 12 mg > deucravacitinib 6 mg > upadacitinib > tofacitinib > apremilast. This might infer an ACR 70 response, rather than ACR 20 or 50, which may better reflect improvements in impaired daily functioning. Furthermore, it also highlighted the correspondence between objective evaluations by physicians and subjective patient-reported outcomes.
Significance of the findings in comparison to existing literature
Different from previous NMAs for PsA, we specifically focus on oral small molecule drugs. Small molecules tend to have a quicker onset, leading to primary endpoints typically falling within the 12–16-week range. By contrast, biologics in phase III trials commonly use 24 weeks as the main assessment time point. 15 Therefore, our analysis centers on an earlier time frame (12–16 weeks) as the primary endpoint. In addition, our article is distinctive in that it is the only NMA to include an analysis of deucravacitinib. Furthermore, we only included approved doses for comparison (except for deucravacitinib, where the approved dose for PsA is yet to be determined), omitting other dosage variations. This approach aims to better align with clinical requirements and provide a more practical analysis.
The NMA conducted by Ruyssen-Witrand et al.
13
primarily focuses on biological agents but includes two small molecule drugs,
In the NMA study conducted by Mease et al.,
15
with guselkumab as the primary focal drug, the ACR 20 data show that
In the analysis of
Recently, Nash et al.
14
reported an NMA focusing on the
Limitations
The limitations are as follows. First, the inclusion of
Second, due to the limited number of studies,
Lastly, apremilast differs slightly in its
Conclusion
The efficacy of all four small molecule drugs in treating PsA was superior to placebo over 12–16 weeks, and their effectiveness was comparable to adalimumab. In addition, the risk difference for SAEs within 24 weeks showed no significant difference between the small molecule drugs, placebo, and adalimumab. When considering ACR 50/70, PASI 75, and HAQ-DI simultaneously, upadacitinib 15 mg QD and deucravacitinib 12 mg QD consistently ranked in the top two.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X241305459 – Supplemental material for Efficacy and safety of small molecule oral medications for psoriatic arthritis: a network meta-analysis of randomized controlled trials
Supplemental material, sj-docx-1-tab-10.1177_1759720X241305459 for Efficacy and safety of small molecule oral medications for psoriatic arthritis: a network meta-analysis of randomized controlled trials by Ya-Chu Tsai, Chen-Yiu Hung and Tsen-Fang Tsai in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-docx-2-tab-10.1177_1759720X241305459 – Supplemental material for Efficacy and safety of small molecule oral medications for psoriatic arthritis: a network meta-analysis of randomized controlled trials
Supplemental material, sj-docx-2-tab-10.1177_1759720X241305459 for Efficacy and safety of small molecule oral medications for psoriatic arthritis: a network meta-analysis of randomized controlled trials by Ya-Chu Tsai, Chen-Yiu Hung and Tsen-Fang Tsai in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-docx-3-tab-10.1177_1759720X241305459 – Supplemental material for Efficacy and safety of small molecule oral medications for psoriatic arthritis: a network meta-analysis of randomized controlled trials
Supplemental material, sj-docx-3-tab-10.1177_1759720X241305459 for Efficacy and safety of small molecule oral medications for psoriatic arthritis: a network meta-analysis of randomized controlled trials by Ya-Chu Tsai, Chen-Yiu Hung and Tsen-Fang Tsai in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-docx-4-tab-10.1177_1759720X241305459 – Supplemental material for Efficacy and safety of small molecule oral medications for psoriatic arthritis: a network meta-analysis of randomized controlled trials
Supplemental material, sj-docx-4-tab-10.1177_1759720X241305459 for Efficacy and safety of small molecule oral medications for psoriatic arthritis: a network meta-analysis of randomized controlled trials by Ya-Chu Tsai, Chen-Yiu Hung and Tsen-Fang Tsai in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-docx-5-tab-10.1177_1759720X241305459 – Supplemental material for Efficacy and safety of small molecule oral medications for psoriatic arthritis: a network meta-analysis of randomized controlled trials
Supplemental material, sj-docx-5-tab-10.1177_1759720X241305459 for Efficacy and safety of small molecule oral medications for psoriatic arthritis: a network meta-analysis of randomized controlled trials by Ya-Chu Tsai, Chen-Yiu Hung and Tsen-Fang Tsai in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-docx-6-tab-10.1177_1759720X241305459 – Supplemental material for Efficacy and safety of small molecule oral medications for psoriatic arthritis: a network meta-analysis of randomized controlled trials
Supplemental material, sj-docx-6-tab-10.1177_1759720X241305459 for Efficacy and safety of small molecule oral medications for psoriatic arthritis: a network meta-analysis of randomized controlled trials by Ya-Chu Tsai, Chen-Yiu Hung and Tsen-Fang Tsai in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-jpg-7-tab-10.1177_1759720X241305459 – Supplemental material for Efficacy and safety of small molecule oral medications for psoriatic arthritis: a network meta-analysis of randomized controlled trials
Supplemental material, sj-jpg-7-tab-10.1177_1759720X241305459 for Efficacy and safety of small molecule oral medications for psoriatic arthritis: a network meta-analysis of randomized controlled trials by Ya-Chu Tsai, Chen-Yiu Hung and Tsen-Fang Tsai in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-tiff-8-tab-10.1177_1759720X241305459 – Supplemental material for Efficacy and safety of small molecule oral medications for psoriatic arthritis: a network meta-analysis of randomized controlled trials
Supplemental material, sj-tiff-8-tab-10.1177_1759720X241305459 for Efficacy and safety of small molecule oral medications for psoriatic arthritis: a network meta-analysis of randomized controlled trials by Ya-Chu Tsai, Chen-Yiu Hung and Tsen-Fang Tsai in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-tiff-9-tab-10.1177_1759720X241305459 – Supplemental material for Efficacy and safety of small molecule oral medications for psoriatic arthritis: a network meta-analysis of randomized controlled trials
Supplemental material, sj-tiff-9-tab-10.1177_1759720X241305459 for Efficacy and safety of small molecule oral medications for psoriatic arthritis: a network meta-analysis of randomized controlled trials by Ya-Chu Tsai, Chen-Yiu Hung and Tsen-Fang Tsai in Therapeutic Advances in Musculoskeletal Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.