Abstract
Background:
Osteoarthritis (OA) is the leading cause of disability among US adults and most commonly affects the knee. Guidelines for knee OA treatment include behavioral, nonpharmacological, pharmacological, and surgical interventions. While emerging knee OA treatments show promise for pain control, data gaps remain regarding the efficacy, safety, comparative effectiveness, and real-world value of treatments.
Objectives:
The Innovations in Genicular Outcomes Registry (IGOR) is prospectively collecting real-world data to assess clinical effectiveness, safety, health-related quality of life, and healthcare resource utilization of knee OA treatments.
Design:
The IGOR is a prospective, observational, longitudinal, multicenter registry (NCT05495334) examining knee OA pain treatment outcomes at intervals up to 18 months after treatment.
Methods and analysis:
All clinical management decisions are made via shared decision-making involving both the physician and the patient. Index joint-directed treatments may include various intra-articular injections, oral opioid and nonopioid medications (including nonsteroidal anti-inflammatory drugs), cryo nerve blocks, radiofrequency ablations, novel treatment modalities, other physical therapy modalities (including muscle strengthening), and total knee arthroplasties. Patient-reported assessments along with physician-provided medical record data are recorded. Regular data quality assessments are conducted for each site, and an outside monitor ensures data quality and integrity. A steering committee ensures transparency and oversees administrative, legal, ethical, and scientific decisions. Treatment outcomes within and between therapies are compared.
Ethics:
Ethical approval was granted by Advarra, Inc. (protocol number, Pro00050981).
Discussion:
Data from the IGOR registry study will further elucidate the effectiveness, safety, and real-world value of knee OA treatments individually or in combination. Characterization of real-world treatment patterns will help better understand the impact of specific treatments.
Trial registration:
Clinicaltrials.gov, NCT05495334.

Keywords
Introduction
Osteoarthritis (OA) is a chronic joint disease that results in joint pain, stiffness, mobility impairment, and functional limitation, with the highest prevalence (9961 per 100,000) being in the United States.1,2 The knee is the most commonly affected joint. In 2019, knee OA accounted for approximately 61% of total OA cases, making it the leading cause of disability among US adults.2,3 Most OA treatments are primarily focused on decreasing pain and improving function.1,4,5 However, treatments are not always effective, which may lead to suboptimal outcomes or abandoning a given method in favor of others. In one study examining real-world OA treatment patterns, 80% of patients who failed multiple interventions changed because a modality was ineffective. 6 The current armamentarium of knee OA treatments may not meet patient’s needs; however, emerging knee OA treatments may offer improved efficacy. The Food and Drug Administration (FDA)-approved nonsurgical knee OA pain treatments that are moderately recommended in guidelines include denervation-based therapies (e.g., cryo nerve block, radio frequency ablation techniques) and intra-articular injections (corticosteroids, hyaluronic acid compounds, extended-release injectable corticosteroid formulations). 1 These treatment alternatives may improve function and help manage knee OA pain, but the evidence regarding their benefits, risks, and impacts on healthcare resource utilization outcomes is sparse, inconsistent, and potentially conflicted.7 –9 Therefore, larger studies are needed to further characterize their therapeutic profile.
As new treatments for OA are developed and implemented, reliable assessments of their efficacy and safety are paramount. Randomized controlled trials (RCTs) are important for evaluating the efficacy and safety of novel knee OA treatments. However, RCT study designs typically assess single interventions in controlled settings and cannot capture the heterogeneous real-world treatment experience of patients undergoing chronic OA treatment, which often involves combinations of multiple treatments on individualized schedules. 10 Furthermore, RCTs typically have strict and narrow inclusion and exclusion criteria and have at most one or two comparative arms, limiting generalizability. Alternatively, real-world, retrospective studies often use claims records of patients who have undergone knee OA treatments to better reflect meaningful treatment patterns. However, these studies often lack patient-reported outcome (PRO) measures that allow examination of treatment impacts from the patient’s perspective as well as clinical contexts (including phenotypic granularity), measures of disease severity, rates of adherence by intervention, and whether treatments are provided to individual patients in lieu of total knee arthroplasty (TKA; i.e., surgical candidates) or as palliative care among those not considered candidates for surgical intervention.
Unlike administrative- or claims-based studies, real-world registries can identify additional factors not captured in clinical trials (e.g., patient and physician preferences) and can capture more abundant and standardized data for emerging therapies that better reflect typical knee OA treatment in real-world clinical practice. Such registries can help determine the impact of treatments and their values to patients and healthcare systems. 11 The Innovations in Genicular Outcomes Registry (IGOR) combines a prospective, observational study design with standardized real-world evidence collections of clinical data, PRO assessments, reimbursements, and healthcare resource utilization data across the spectrum of available clinician-selected treatment approaches. This research registry is designed to capture information related to treatments used in real-world scenarios to identify best practices and outcomes for knee OA, including clinical response to treatment, safety, health-related quality of life (HRQOL), and healthcare resource utilization data.
Methods and analysis
Overall design and study population
The prospective, observational, multicenter registry IGOR (registered at Clinicaltrials.gov, NCT05495334) examines clinical- and health-related outcomes after knee OA pain interventions at intervals for up to 18 months after an administered treatment. This registry is designed for a non-randomized and observational study, with all clinical management decisions made by patients and their physicians together in a shared decision-making manner as part of routine care independent of participation in the registry. Physicians are free to modify each patient’s treatment at their discretion during the registry period without withdrawing the patient from the registry. To maintain fidelity with real-world practice, the selection of treatments and the schedule of clinic visits are expected to be unaltered by registry participation. The registry enrolls patients who have any knee OA treatment(s) for the management of pain, which may include, but are not restricted to, various denervation therapies (e.g., cryoneurolysis, radiofrequency ablation), various intra-articular injections (e.g., corticosteroids, viscosupplementations, platelet-rich plasma injections, stromal cell products, amniotic fluid products), analgesic medications (opioids and nonopioids), or surgeries (e.g., arthroplasty). All treatments are captured prospectively, from those applied initially through any new or repeated treatments subsequently pursued.
Patients who plan to receive treatment for OA pain within 60 days are screened and, if eligible, offered enrollment in the registry. Registry inclusion and exclusion criteria are described in Table 1. The “treatment day” is defined as the day the treatment is administered, and the “target knee” is the affected knee undergoing OA treatment. If a patient is scheduled for bilateral knee treatment, a care provider selects the most symptomatic knee as the target knee, which remains the index knee for all assessments. The planned data analyses will separate patients who have unilateral versus bilateral knee symptoms. For patients screened on the treatment day, the screening assessments are administered before treatment administration. The follow-up period includes the time from the day after the treatment until the earliest of the following dates: end of the registry follow-up (i.e., 18 months following treatment) or death. A follow-up period of 18 months will be used for each treatment that patients receive for knee OA management. If a patient receives the next line of treatment (e.g., another intra-articular injection) during this follow-up period, their follow-up assessment will be reset from the beginning for a new 18-month follow-up cycle. Patients are compensated for their participation in research activities.
Inclusion and exclusion criteria.
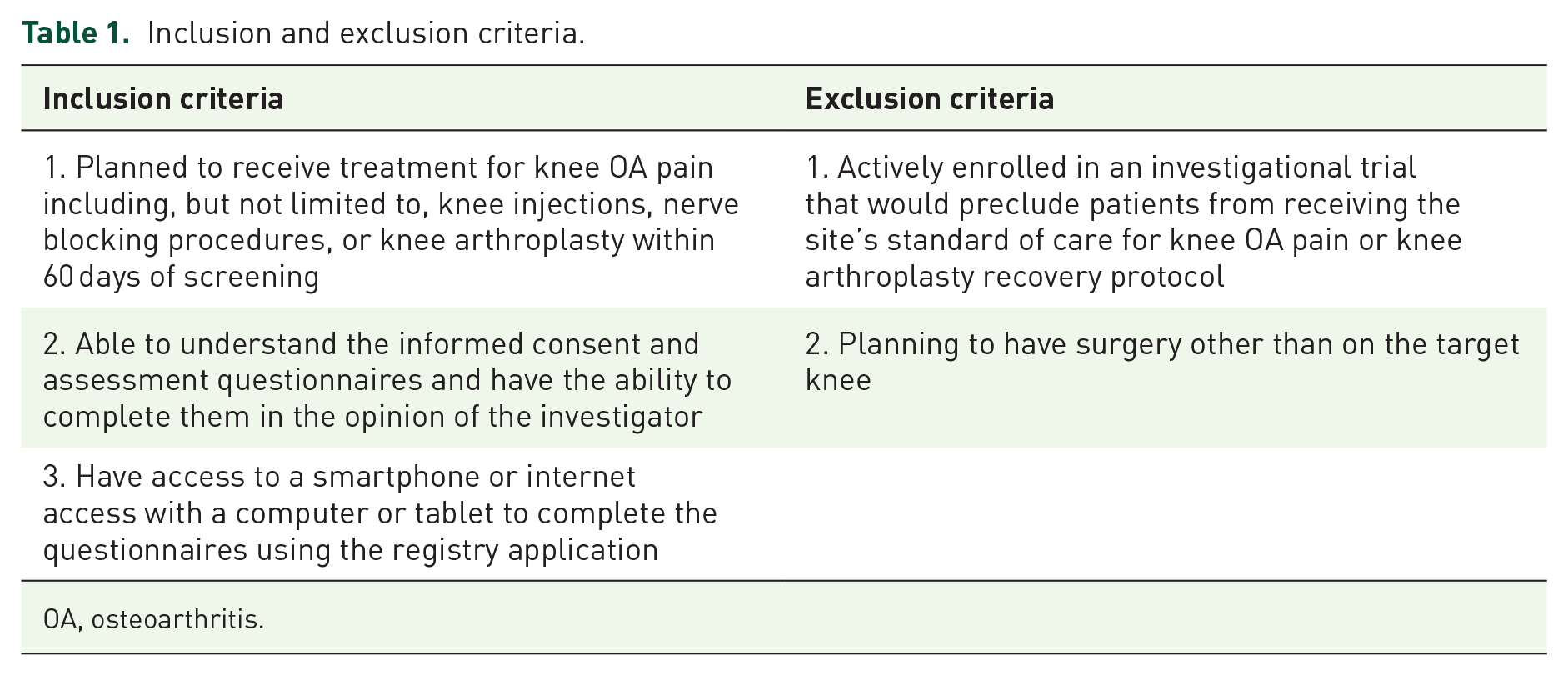
OA, osteoarthritis.
A total of up to 14 sites are planned across the United States in outpatient clinics and teaching hospitals; these sites include principal investigators within multiple specialties such as orthopedic surgery, pain management, and rheumatology (Supplemental Table 1). Once enrolled, patients gain access to, and are trained to respond to, the web-based Health Insurance Portability and Accountability Act (HIPAA)-compliant PRO instruments from a mobile phone to capture numerous clinical-, and health-related, outcomes at baseline through follow-up; these assessments are described further below and in Supplemental Table 2 and Supplemental File. Registry site staff enter provider-reported data into a web-based, electronic data capture system; these data include any knee OA-directed nonsurgical treatment information, surgical information (preoperative and intraoperative), discharge information, and other relevant data from patients’ medical records. Registry data from each patient are collected for up to 18 months from the initial treatment day (Figure 1(a)). For any patients undergoing a subsequent course of knee OA-direct treatment, data capture will restart, and that patient will continue in the registry for up to 18 months after the final treatment. Each episode of treatment includes follow-up until the beginning of the next episode of treatment (Figure 1(b)).

Registry period and individual assessment time periods at screening and baseline and during follow-up. Registry data from each patient are collected for up to 18 months from the initial treatment day. Patients who undergo two or more treatments continue in the registry for up to 18 months after the final treatment, and each episode of treatment includes follow-up until the beginning of the next episode of treatment. (a) Patients with one treatment; (b) patients with two or more treatments.
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 12
Assessments and outcomes
The schedule of assessments during screening, on treatment day, and during the follow-up period is detailed in Table 2. Baseline characteristics collected during screening include patient demographics, socioeconomic, medical, surgical, OA treatment, and prior analgesic medication histories. The Pain Catastrophizing Scale (PCS), a self-reported measure of catastrophic thinking associated with pain, is administered during screening through the web-based ePRO website. 13 Patients enrolled in the registry are grouped as high (PCS > 16) or low (PCS ⩽ 16) catastrophizers (all PROs and their descriptions are included in Supplemental Table 2).
Schedule of assessments.
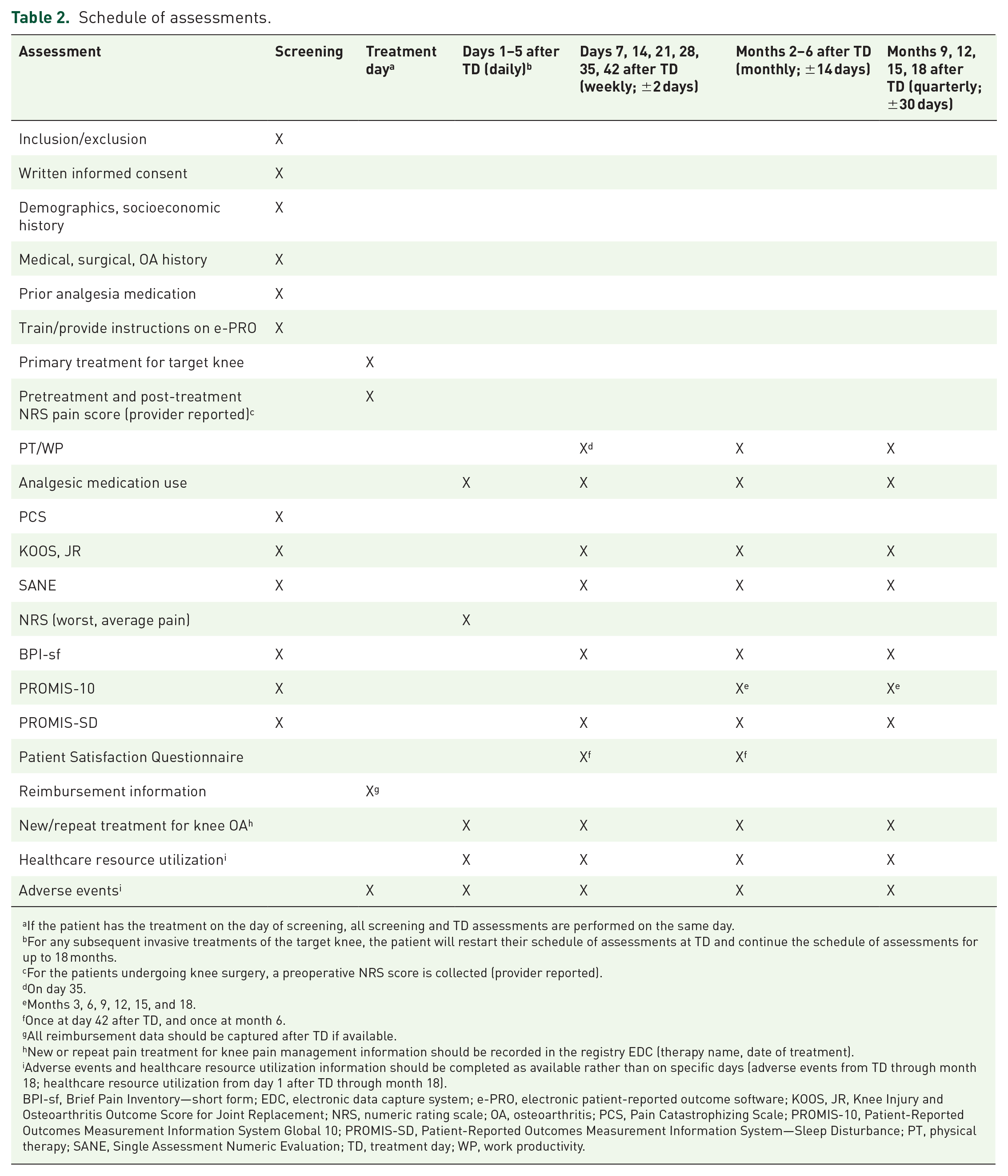
If the patient has the treatment on the day of screening, all screening and TD assessments are performed on the same day.
For any subsequent invasive treatments of the target knee, the patient will restart their schedule of assessments at TD and continue the schedule of assessments for up to 18 months.
For the patients undergoing knee surgery, a preoperative NRS score is collected (provider reported).
On day 35.
Months 3, 6, 9, 12, 15, and 18.
Once at day 42 after TD, and once at month 6.
All reimbursement data should be captured after TD if available.
New or repeat pain treatment for knee pain management information should be recorded in the registry EDC (therapy name, date of treatment).
Adverse events and healthcare resource utilization information should be completed as available rather than on specific days (adverse events from TD through month 18; healthcare resource utilization from day 1 after TD through month 18).
BPI-sf, Brief Pain Inventory—short form; EDC, electronic data capture system; e-PRO, electronic patient-reported outcome software; KOOS, JR, Knee Injury and Osteoarthritis Outcome Score for Joint Replacement; NRS, numeric rating scale; OA, osteoarthritis; PCS, Pain Catastrophizing Scale; PROMIS-10, Patient-Reported Outcomes Measurement Information System Global 10; PROMIS-SD, Patient-Reported Outcomes Measurement Information System—Sleep Disturbance; PT, physical therapy; SANE, Single Assessment Numeric Evaluation; TD, treatment day; WP, work productivity.
Clinical effectiveness and safety outcomes
Pain control assessment measures
Pain control is assessed using a numeric rating scale (NRS), portions of the Brief Pain Index—short form (BPI-sf), the pain subscale of the Knee Injury and Osteoarthritis Outcome Score for Joint Replacement (KOOS, JR), and a patient-reported analgesic medication use log (including opioid and nonopioid medications used to treat pain). Postoperative opioid consumption is captured for patients undergoing surgery.14,15 Moreover, analgesic medication use is assessed following each non-operative treatment. The status of opioid use for knee OA pain (yes, no) will be captured monthly and quarterly in all patients during follow-up. In addition, discharge prescriptions of opioids will be assessed in patients who received knee replacement surgery. Each of the pain-related measures is completed by patients at baseline and on a weekly, monthly, and quarterly basis (except the NRS, which is measured daily in the first 5 days after treatment day only). Patient-reported NRS scores are recorded by the healthcare provider immediately before and after the pain-directed intervention. Patients record both the average and worst pain intensity in the web-based electronic patient-reported outcome (e-PRO) application at multiple scheduled times in the follow-up period.
Functional assessments
Functional status is assessed using the KOOS, JR and the Single Assessment Numeric Evaluation at baseline as well as weekly, monthly, and quarterly in the follow-up period.15,16
Sleep quality evaluations
Sleep quality in the past 7 days is assessed via the Patient-Reported Outcomes Measurement Information System–Sleep Disturbance (PROMIS-SD). 17
Adverse events
Adverse events (AEs) and adverse device effects (ADEs) related to the OA treatment or device are reported directly to the manufacturer of the knee OA therapy immediately per standard reporting and also recorded in the electronic data capture system throughout the follow-up period by the registry site provider or staff (Table 2). The distribution of AEs and ADEs is assessed in the overall cohort and according to treatments.
HRQOL outcomes
Responses to the Patient-Reported Outcomes Measurement Information System 10 (PROMIS-10) are collected via e-PRO at baseline and quarterly during the follow-up period. 18 The raw PROMIS-10 scores are converted to standard T scores and EuroQol 5 Dimension (EQ-5D) index scores.
Patient satisfaction assessments
Patient satisfaction with pain management is assessed on a scale of 1–5 (1 = very dissatisfied, 2 = dissatisfied, 3 = neutral, 4 = satisfied, and 5 = very satisfied) at week 6 and month 6 after treatment.
Healthcare resource utilization outcomes
Nonsurgical treatment reimbursement
Treatment coverage, reimbursement codes (Current Procedural Terminology, Healthcare Common Procedure Coding System, International Classification of Diseases-10–Procedure Coding System), and diagnostic codes (International Classification of Diseases-10) are recorded by the study sites. During the follow-up period, patients may receive repeat or new treatments, which are also recorded. The coding systems enable the specification of the laterality of the treated joint (Table 2).
Surgical treatments
Postoperative healthcare resource utilization outcomes for patients treated with surgery include length of stay and discharge disposition. Discharge dispositions encompass the post-anesthesia care unit and the hospital.
Inpatient, emergency department, and outpatient visits during follow-up
Emergency department usage, hospital admissions, unscheduled phone calls, unscheduled office visits, and reimbursement data related to the therapy are collected from medical records (as available), during and after the follow-up period (up to 18 months), and entered into the electronic data capture system (Table 2).
Physical therapy and work productivity
Physical therapy (PT) utilization is assessed by the patient-reported PT/work productivity questionnaire at multiple time periods after treatment (daily for 5 days, weekly for 6 weeks, monthly from months 2 to 6, and quarterly at months 9, 12, 15, and 18).
Data analyses
While this registry is not hypothesis-driven, descriptive analyses of registry data summarize outcomes of changes from baseline in pain control, functional status, therapy-related AEs, sleep quality, HRQOL, and healthcare resource utilization. The response rate for IGOR participation will be reported as the ratio of total patients who have knee OA who are approached prior to enrollment to those who agree and are eligible to participate in the registry. In the case of patients undergoing multiple episodes of treatment, outcomes are analyzed for each treatment episode and grouped together by treatment type. Descriptive statistics are generated to summarize baseline characteristics as well as any measurements collected during follow-up.
Multivariable generalized linear mixed-effect modeling accounting for within-patient relatedness is used to assess changes between pre-therapy and post-therapy (and post-surgery) outcomes, assuming normal distributions with identified link function (e.g., for NRS; BPI pain severity; KOOS, JR interval score; and SD-T score), negative-binomial distribution with log link function (for PT use, healthcare resource utilization frequency), or binomial distribution with logit link function (for opioid use and opioid refill status). The analytical models are adjusted for confounders assessed at baseline, including patient demographics, medical histories, lifestyle factors, prior knee OA treatments, and baseline PRO scores.
Comparisons of changes in patient outcomes between therapies over the course of follow-up are conducted using multivariate generalized linear mixed-effect modeling. Multivariable generalized linear modeling may be used when comparing the impact of therapies on non-repeated outcomes (e.g., length of facility stay in TKA patients). All analytical models are adjusted for confounders. To determine whether the pain therapy is perceived as beneficial by patients, minimal clinically important difference values and pain intensity scores (area under the curve) for response to each pain therapy are calculated for each PRO measure.
All analyses are performed separately by pathway cohort using Statistical Analysis Software, Version 9.4 (SAS Institute, Cary, NC, USA). All p values are from two-sided tests.
Sample size estimations
Power and sample size calculations were performed using the proc power command in SAS. Assuming a mean difference of 0.5 in NRS average pain scores before and after pain therapy, a two-sided alpha of 0.05, and a standard deviation of 1.2, 63 patients will be required for individual pain therapy to reach 90% power. To compare changes in pre- and post-NRS average pain scores between therapy groups, a total of 123 patients (62 in either pain therapy group) will be required to detect a mean difference of 0.5 in NRS average pain scores between the two therapy groups at 90% power.
Data quality control
Data management staff oversee the completeness of the data and inform site coordinators of any concerns with data. Regular assessments of data quality (missingness, distribution) are conducted for each participating site to establish the completeness of the data being entered into the registry database. Regular overall and site-specific reports are generated and circulated to all participating sites.
At each study site, a data monitor is enlisted to ensure data quality and integrity and review data entry for 2–3 patients for consistency with site and subject source records. Data monitor review frequency can be increased as needed. Additional data monitoring by an experienced third party (Advent Advisory Group) is planned to further investigate the integrity and consistency of data collection and security.
The web-based PRO instruments are designed to safeguard against missing data by requiring responses in mandatory fields. In addition, site coordinators remind patients to respond to questionnaires in a timely manner. Patients who miss ⩾50% of the questionnaires may be excluded from the final analyses. No imputation of data is planned. The proportion of missing data will be reported in any subsequent publications. The quality of the outcome data from the PRO assessments is reviewed using internal reliability methods (e.g., intraclass correlation coefficients or Cohen’s alpha methods) and external construct validity measures (e.g., Spearman’s correlations across multiple assessments).
Ethics and dissemination
Patient consent and protection
Each site obtains approval from an Institutional Review Board (IRB; local and/or central) that complies with the International Conference on Harmonisation (ICH), Good Clinical Practice (GCP), and the US FDA Title 21 Code of Federal Regulations Part 56. Attention is directed to the basic elements that are required for incorporation into the informed consent form under Title 21 Code of Federal Regulations Part 50.25 and ICH GCP. Central IRB approval for the registry protocol was obtained from Advarra, Inc. (protocol number, Pro00050981).
The principal provider at each site conducts the registry in accordance with this protocol, obtains written informed consent, and reports unexpected problems in accordance with the individual site’s IRB policies and procedures as well as all applicable federal requirements. Patients are informed that they are free to withdraw from the study at any time. Accurate data collection, recording, and reporting are performed in a manner that ensures the privacy, health, and welfare of patients during and after registry participation. Each registry site has a principal provider and site-supporting staff who have access to patient medical records and the registry database. Patients receive a registry-generated unique identifier, and their deidentified information is transferred to the IGOR Registry Data Coordinating Center, which monitors data quality using a secure electronic data capture system. Access to the electronic data capture system is password-protected and is given to authorized IGOR Registry Data Coordinating Center personnel.
Governance
A steering committee for IGOR was established from a multispecialty group of US providers. The objective of the steering committee is to ensure transparency in operations, decision-making, and reporting of results. This committee is responsible for study design, data monitoring, data review, plans for data analyses, and publication planning. The steering committee is also responsible for oversight of administrative, legal, ethical, and scientific decisions. However, as stated above, the treating physicians make all treatment decisions without direction from the steering committee or funding source. Funding for the IGOR registry and compensation for the steering committee members is provided by Pacira BioSciences, Inc., Tampa, Florida. Data analyses with statistical analysis plans are also facilitated by Pacira BioSciences, Inc., and reviewed and approved by the steering committee. Results from these analyses will be submitted for publication in peer-reviewed journals. To maintain objectivity and minimize conflict of interest, an experienced third-party consultant will review and document the overall procedures involved in the registry.
Discussion
Results derived from IGOR may inform optimal real-world treatment approaches by stratifying the relative ability of various clinical interventions for knee OA to impact symptoms, healthcare resource utilization, and health-related quality of life outcomes. Analyses of registry data may also provide evidence for use in future treatment guidelines that distinguish which OA pain treatments are preferable among subgroups of patients who have knee OA and inform the design of future study protocols.
The IGOR is designed to capture real-world clinical and health-related quality-of-life outcomes for all knee OA treatments, which allows for comparison across treatments and patient subgroups. Because physician providers in conjunction with patients participating in IGOR make all treatment decisions, the registry is treatment agnostic, and the steering committee and registry funding source will not direct or influence treatment choices. The phenotype of real-world knee OA patient populations is likely to differ considerably from clinical trial populations that tend to be homogeneous. 10 The IGOR intentionally captures a diverse patient population across different regions and clinical settings in the United States, as geographic regions, economic backgrounds, practice environments, and varying healthcare costs or insurance types can affect treatment patterns for knee OA. 19 The robust inclusiveness of the treatments captured by IGOR but not typically included in claims/record-based studies (e.g., over-the-counter medication use and treatment sequence) will help chronicle the journey of patients who have OA, allowing for a more complete picture of the available OA treatments in the United States and their usage patterns and associated outcomes. Capturing validated, objective clinical measures and PROs should help to determine whether a given treatment is perceived as beneficial relative to the other available interventions. In particular, PROs are clinically meaningful for understanding the patient experience of treatment, and several PROs included in IGOR (PROMIS-SD; PROMIS-10; and KOOS, JR) have been previously validated.15,18,20 Furthermore, the objective endpoints and analyses in IGOR may help generate hypotheses that stimulate future studies to validate the findings. Ultimately, the results derived from IGOR may more accurately inform patient expectations for treatment effectiveness among patients undergoing interventions for knee OA in real-world environments.
The IGOR may offer advantages over other registries in which data are retrospectively retrieved for quality improvement or accreditation purposes or where data gaps occur given a lack of follow-up. Large-quality registries may have inconsistencies in reported data and a lack of standardized research protocols, which may hinder proper follow-up of patients. 21 For example, the American Academy of Orthopaedic Surgeons’ American Joint Replacement Registry captures a large number of patients but may encounter high amounts of missing data for certain variables (e.g., PROs) as patient consent is not required for quality improvement studies.22,23 Patient consent is required for participation in the Function and Outcomes Research for Comparative Effectiveness in Total Joint Replacement (FORCE-TJR) registry, and researchers and clinicians have made efforts to validate patient self-reporting. While FORCE-TJR has gathered PROs from 20,000 patients, gaps in data capture remain given a lack of early and consistent patient follow-up and/or patients not reporting care received from an institution other than the one that performed the total joint arthroplasty.24,25 Missing data compromise longer-term outcomes and the ability to follow-up, which is important for understanding chronic conditions like knee OA. The IGOR is best classified as a research-oriented registry and overcomes these challenges by leveraging rigorous, long-term, in-depth data capture encompassing PROs, analgesic medication, AEs, and healthcare resource utilization outcomes from patients (via direct patient data capture) and study sites. Follow-up for IGOR extends up to 18 months, with some patients followed for longer time frames in the case of repeat treatments. This long-term, prospective data collection from patients in IGOR that specifically addresses OA outcomes meets the FDA standard for a patient registry that generates real-world data. 26 Direct patient engagement and participation with IGOR is enhanced by patient compensation, incentivizing patient follow-up. In addition, electronic capture of PROs can reduce the number of visits after treatment, focusing patient time and effort on reducing and preventing gaps in data capture. The design of IGOR also helps ensure uniform data collection across multiple study sites, allowing for more robust comparisons of patient outcomes within and between visits; this approach also allows for greater temporal granularity in response assessment than typically afforded by other approaches (e.g., electronic medical record-based assessments that are limited to in-person, visit-based data capture). To our knowledge, this is the only industry-sponsored registry that tracks all OA treatment options rather than only those products associated with the sponsor. Ultimately, future collaboration with quality registries will allow for more robust assessment and may validate the relatively smaller IGOR database while providing insights from complete, prospective patient data to the quality registries.
Despite the value of collecting real-world evidence in this manner, there are some inherent potential limitations in designing a registry. Obtaining patient consent requires time and effort, which may result in fewer participants. In addition, because there is no mandated treatment protocol for registry participants, patients enrolled in IGOR are not randomized or stratified into treatment groups, and sample size variations between different treatments may be created by the variability of treatment approaches applied at different sites and among specialties. The resulting variation in sample size precludes the preplanning of analytic comparisons between specific treatments. Certain biases, whether conscious or unconscious, can also be a limitation and may affect the selection of one treatment over another by different providers and for a given patient or set of patients; however, the number of patients available for analysis is a summation of the ecosystem forces that drive shared decision-making for the selected treatments, resulting in a combination of desirable attributes from clinical trials (e.g., preplanned and rigorous data collection) and retrospective claims analyses (e.g., organically determined treatment choices that support representative rates of real-world use). Efforts are made to assess for such bias in the analysis by comparing data across sites/clinicians by order and frequency of application to assess clinician preferences and by comparing treatment applied by phenotype to determine treatment preferences by patient types. Furthermore, the use of standardized outcome measures may offset the impact of unconscious bias by objectifying treatment decisions as discrete inputs to clinical judgment. The determination of the threshold levels of change in specific outcome measures that result in repeated treatments versus new treatments may assist in addressing potential bias in the future. Another potential limitation is that patient knowledge of being observed may impart bias (i.e., the Hawthorne effect 27 ) that is difficult to assess or control for, particularly in the absence of a control group. In addition, operational challenges in implementation are primarily managed by the individual participating sites, which are a mixture of clinically focused academic centers and clinical practices without formal Contract Research Organization involvement. While efforts were made to ensure a diverse set of study sites, some selection bias may be inherent given the availability of study staff and clinic space for data collection. Similarly, although data capture using technology allows for wider participation, not all populations have access to technology or technology education, which could bias the study population toward those with higher socioeconomic status and educational attainment.
Conclusion
The IGOR registry generates information regarding real-world knee OA treatment patterns and outcomes in response to established and more innovative knee OA treatments. Collectively, these analyses are designed to further characterize the relative clinical and health economic benefits of treatments for subgroups of patients who have knee OA. Results from registry analyses provide an additional perspective to that available from clinical trials and claims analyses as to the individual and comparative effectiveness, safety, and economic impact to inform treatment decisions for optimal and precise patient care and future research.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X241304193 – Supplemental material for Innovations in Genicular Outcomes Registry (IGOR): protocol for a real-world registry study of treatments for knee osteoarthritis
Supplemental material, sj-docx-1-tab-10.1177_1759720X241304193 for Innovations in Genicular Outcomes Registry (IGOR): protocol for a real-world registry study of treatments for knee osteoarthritis by Vinod Dasa, William Mihalko, Adam Rivadeneyra, Joshua Urban, Andrew Wickline, David Rogenmoser, Andrew Concoff, Andrew Spitzer and Michael A. Mont in Therapeutic Advances in Musculoskeletal Disease
Footnotes
Appendix
Acknowledgements
Writing and editorial assistance was provided under the direction of the authors by Emma Hinkle, PhD, of MedThink SciCom, and was funded by Pacira BioSciences, Inc. The data management and analysis team included Anne Yeakel at White Star Consulting and Swapnabir Kakoty, Yanfen Zhou, Sandy Shi, and Masato Nakazawa at Pacira BioSciences, Inc. Data monitoring assistance was provided by Kensington Meister, Kate Fasel, and Kathryn Eten at Pacira BioSciences, Inc. Medical consultation was provided by Stan Dysart and Partha Saha at Pacira BioSciences, Inc. Mary DiGiorgi, Kathryn Eten, Jennifer H. Lin, and Partha Saha of Pacira BioSciences, Inc., provided critical review of the manuscript.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
