Abstract
Background:
Osteoarthritis (OA) is a common degenerative joint disease that poses a significant global healthcare challenge due to its complexity and limited treatment options. Advances in metabolomics have provided insights into OA by identifying dysregulated metabolites and their connection to altered signaling pathways. However, a comprehensive understanding of these biomarkers in OA is still required.
Objectives:
This systematic review aims to identify metabolomics biomarkers associated with dysregulated signaling pathways in OA, using data from various biological samples, including in vitro models, animal studies, and human research.
Design:
A systematic review was conducted following Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines.
Data sources and methods:
Data were gathered from literature published between August 2017 and May 2024, using databases such as “PubMed,” “Scopus,” “Web of Science,” and “Google Scholar.” Studies were selected based on keywords like “metabolomics,” “osteoarthritis,” “amino acids,” “molecular markers,” “biomarkers,” “diagnostic markers,” “inflammatory cytokines,” “molecular signaling,” and “signal transduction.” The review focused on identifying key metabolites and their roles in OA-related pathways. Limitations include the potential exclusion of studies due to keyword selection and strict inclusion criteria.
Results:
The meta-analysis identified dysregulated metabolites and associated pathways, highlighting a distinct set of related metabolites consistently altered across the studies analyzed. The dysregulated metabolites, including amino acids, lipids, and carbohydrates, were found to play critical roles in inflammation, oxidative stress, and energy metabolism in OA. Metabolites such as alanine, lysine, and proline were frequently linked to pathways involved in inflammation, cartilage degradation, and apoptosis. Key pathways, including nuclear factor kappa B, mitogen-activated protein kinase, Wnt/β-catenin, and mammalian target of rapamycin, were associated with changes in metabolite levels, particularly in proinflammatory lipids and energy-related compounds.
Conclusion:
This review reveals a complex interplay between dysregulated metabolites and signaling pathways in OA, offering potential biomarkers and therapeutic targets. Further research is needed to explore the molecular mechanisms driving these changes and their implications for OA treatment.
Plain language summary
Graphical abstract

Introduction
Osteoarthritis (OA) is a complex and multifactorial disease characterized by the progressive degradation of joint cartilage. 1 Metabolic components have also been involved in disease development. Subchondral bone ischemia, articular cartilage destruction, and dysfunction of the synovial endothelium are the three symptoms of metabolic syndrome, which is characterized by the triad of hypertension, dyslipidemia, and diabetes. 2 Joint stiffness and discomfort are two OA symptoms brought on by the growth of osteophytes, ligamentous laxity, and weakened muscles. 3 Metabolites are the intermediates and end products of cellular biochemical processes. Their steady-state levels can be regarded as the ultimate response of biological systems to genotype, phenotype, and environment.
Metabolomics can play a key role in unraveling the mechanistic aspects of OA pathogenesis by identifying the metabolic pathways and dysregulated signaling associated with OA pathogenesis. A total of around 140 metabolites have been specifically identified as being associated with OA, and ongoing research continues to explore their potential as biomarkers for the disease. 4 However, despite the identification of numerous metabolites, there is still no single consensus biomarker or panel of biomarkers universally accepted for clinical use in diagnosing or differentiating OA from healthy individuals. 5
Metabolomics offers several advantages over other omics approaches, such as genomics and proteomics, particularly its ability to provide real-time insights into physiological states. First, they directly reflect the current physiological state of an organism, as metabolites are the end products of gene expression and protein activity. While genomics provides information on potential predispositions based on genetic sequences, and proteomics captures protein expression, metabolomics offers a snapshot of real-time biological activity, making it more relevant for understanding disease states and treatment responses. 6 Second, metabolites are directly involved in biochemical pathways, so changes in their levels provide immediate insights into functional disruptions. This makes metabolomics especially useful for understanding metabolic pathways in disease progression, drug effects, and environmental exposures. By contrast, genomic data offer more predictive or potential insights than real-time functional outcomes. 4 Signaling pathways are critical in the development and progression of OA. Several signaling pathways have been implicated in OA pathogenesis, including inflammatory, catabolic, and anabolic pathways. Metabolomics can provide valuable insights into the metabolic changes associated with these pathways and identify potential biomarkers for OA.
Several common signaling pathways have been implicated in the development and progression of the disease. Activation of nuclear factor kappa light chain enhancer of activated B cells (nuclear factor kappa B, NFκB) leads to the production of inflammatory mediators, including cytokines, chemokines, and matrix metalloproteinases (MMPs), which contribute to cartilage degradation of extracellular matrix (ECM) components and joint inflammation. 7 In OA, the mitogen-activated protein kinase (MAPK) pathway is activated by mechanical stress and proinflammatory cytokines, which regulate the production of inflammatory mediators, catabolic enzymes (such as MMPs and aggrecanases), and apoptotic signals, leading to cartilage degradation and joint inflammation. 8 The transforming growth factor-beta (TGF-β) signaling pathway is another example that dysregulation will lead to an imbalance between anabolic and catabolic processes in cartilage. 9 Reduced TGF-β signaling impairs chondrocyte proliferation and differentiation, inhibits the production of ECM components, and promotes the expression of MMPs, thereby contributing to cartilage degradation. Previous literature reported that increased Wnt signaling leads to the stabilization and nuclear translocation of β-catenin, which activates target genes’ expression in cartilage degradation, inflammation, and osteophyte formation. 10
Metabolic syndrome, characterized by abdominal obesity, high blood pressure, elevated blood sugar levels, high triglycerides, and low HDL cholesterol, is a cluster of conditions that increase the risk of cardiovascular diseases and diabetes. In the context of OA, metabolic syndrome has been identified as a significant risk factor, contributing to OA progression through mechanisms such as low-grade systemic inflammation and disrupted lipid and glucose metabolism. Metabolomics, which analyzes metabolic changes, helps uncover these pathways by identifying alterations in metabolites linked to metabolic syndrome, shedding light on how these metabolic disruptions contribute to inflammation and cartilage degradation in OA. 11
This review aims to build on existing information by addressing the current state of dysregulated metabolomics as biomarkers for OA. By performing a systematic review of individual metabolites associated with OA and the signaling pathways disrupted in OA pathogenesis, we seek to summarize insights provided by metabolomics and identify candidate biomarkers for future studies. Our investigation was conducted following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines to ensure a rigorous and transparent process. We carried out an extensive search across multiple databases, including PubMed, Scopus, Web of Science, and Google Scholar, for articles published between August 2017 and May 2024. Using specific search strategies, we retrieved relevant literature and applied explicit inclusion and exclusion criteria to focus on original research related to metabolomics markers and disrupted signaling pathways in OA. The article selection involved two stages: initial screening of abstracts followed by a full-text review, with final selections independently validated by three co-authors. Data were extracted using a standardized Comma-separated values (CSV) file format from the National Centre for Biotechnology Information (NCBI) database. This structured methodology ensures a comprehensive assessment of the literature, enhancing the reliability and depth of our findings on the role of metabolites and signaling pathways in OA while addressing potential limitations such as keyword specificity and inclusion criteria.
Methods
Information sources and search strategy
The systemic review was conducted in English and by the PRISMA guidelines. 12 PubMed, Scopus, and Web of Science articles were initially searched. However, due to the limited availability of relevant articles from established databases, we expanded our search to include Google Scholar to increase the chances of identifying pertinent articles. The PRISMA guidelines conducted the systematic literature review. 12 The PRISMA guidelines were utilized to ensure a rigorous and transparent review process. PRISMA provides a comprehensive checklist that standardizes reporting practices, enhancing the quality and consistency of systematic reviews. The guidelines were instrumental in defining explicit eligibility criteria, which ensured that only high-quality, relevant studies were included. The PRISMA checklist guided the process of data extraction, ensuring that key information on metabolites and disrupted signaling pathways was systematically collected and analyzed. Adhering to PRISMA also facilitated clear and transparent reporting, allowing readers to understand the review process and assess the reliability of the findings. By following these guidelines, the review benefited from a structured and unbiased approach, improving the credibility and depth of the assessment of dysregulated metabolites and signaling pathways in OA. This systematic approach involved developing a detailed search strategy, specifying databases such as PubMed, Scopus, Web of Science, and Google Scholar, and setting a clear time frame from August 2017 and May 2024 using the search strategy and standard terms in Table 1. The 7-year timeframe was chosen to ensure that the review remains current, relevant, and manageable. It balances the need for recent data with practical considerations related to the scope and resources available for the review.
Search strategy for a systematic review process.

The search strategy used for this systematic review is detailed in Table 1, which lists the specific keywords and Boolean operators entered into each database, including Scopus, Web of Science, PubMed, and Google Scholar. The key terms selected were designed to capture relevant articles on metabolomics and signaling pathways related to OA. For example, combinations such as “metabolomics* OR metabolic profiling” along with “osteoarthritis*” and “signal* OR cellular signaling OR molecular signaling” ensured that the search was comprehensive, targeting both metabolomic data and relevant signaling pathways. In addition, the inclusion of terms such as “biomarkers* OR molecular markers OR diagnostic markers” helped narrow the search to articles related to markers of interest in OA.
We also considered that several metabolites might be mentioned in the same articles. Therefore, during data extraction, we screened each article for individual metabolites that were reported. A comprehensive review of these articles ensured that metabolites were recorded based on their frequency of mention and the context in which they were associated with signaling pathways. By doing so, we avoided redundancy while ensuring that no significant metabolites were overlooked.
The relatively low number of articles retrieved could indeed be attributed to the specificity of the search terms and their combinations. However, this search strategy was intentionally designed to focus on high-relevance studies that specifically address the metabolomics aspects of OA and signaling pathways, rather than general articles that might not provide pertinent insights.
Standard terms used
In the systematic review, standard terms were utilized to ensure a comprehensive and consistent search strategy. The search terms included variations and combinations of keywords related to metabolomics, OA, signaling pathways, and biomarkers. Specific terms used were “metabolomics,” “metabolic profiling,” “osteoarthritis,” “signaling pathways,” and “biomarkers.” These terms were selected based on their relevance to the study objectives and their prevalence in the literature. A detailed search strategy incorporating these terms was employed across multiple databases to capture a broad range of relevant studies.
Evaluation of journal quality and impact factors
To ensure the reliability and credibility of the sources included in the review, journal quality and impact factors were evaluated. Journal quality was assessed based on several criteria, including the reputation of the journal, the rigor of the peer-review process, and the relevance of the journal to the field of study. Impact factors, which reflect the average number of citations to recent articles published in a journal, were used as one measure of journal quality. Journals with higher impact factors were preferred as they typically indicate higher scholarly influence and quality.
Limitations of impact factor consideration
While impact factors are a useful indicator of journal quality, they have limitations. Impact factors do not always reflect the specific relevance or quality of individual articles and may be influenced by factors unrelated to the study’s focus. In addition, some high-quality journals may have lower impact factors if they are newer or cover niche areas. Therefore, in this review, impact factors were considered alongside other quality indicators, such as the journal’s reputation, editorial board, and relevance to the study topic. This approach helped to balance the limitations of impact factors and ensured that a diverse range of high-quality sources was included.
Eligibility criteria
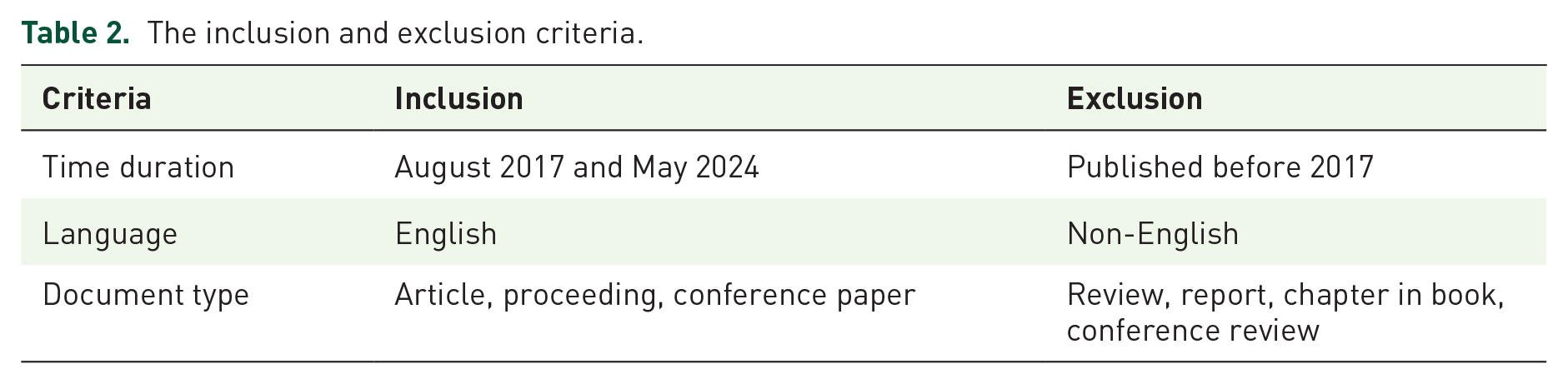
We searched for and included only original articles focusing on the metabolomics markers associated with dysregulated signaling pathways involved in OA. We excluded articles in a language other than English and articles that do not focus on the area of interest in the inclusion criteria, especially those related to surgery, physical therapy outcomes, and clinical studies. Articles in reviews, reports, book chapters, and conference proceedings were also excluded (Table 2).
The inclusion and exclusion criteria.
Article selection and data extraction
Two steps were used for article selection based on eligibility criteria. The first step involved screening abstracts, and the second step involved reviewing the full-text articles. The final selection of articles was independently validated by the three co-authors. For the second part of the review, we will outline our process for screening the literature to identify which metabolites were reported and how frequently they appeared. This will ensure comprehensive coverage and help avoid redundancy. Data were extracted using a CSV file from the NCBI database. The use of CSV files was crucial for organizing and managing the data extracted from the NCBI database, supporting accurate and efficient analysis while ensuring transparency and reproducibility in the systematic review process. For each article, we collected the following (Figure 1).

Flow diagram of information through the different phases of a systematic review.
Results
The extracted articles focusing on metabolomics studies investigating biomarkers for OA reveal a diverse array of sample types (Figure 2), including human serum, 13 synovial fluid, 14 and plasma, 15 and animal samples like rabbit serum and rat plasma 16 and synovial fluid 17 (Table 3). Sample sizes varied significantly, from small cohorts of 6 individuals to larger studies involving over 3000 participants. Across these studies, a range of biomarkers was identified, including fatty acids (such as stearic acid and oleic acid), amino acids (including glutamine, valine, and arginine), lipids (like phosphatidylcholines (PCs)), and other metabolites (such as phenylalanine). Compared to controls, these biomarkers showed variable degrees of upregulation or downregulation in OA patients. Crucially, the discovered metabolites were linked to important metabolic pathways and processes such as inflammation, oxidative stress, collagen deterioration, and cartilage breakdown that are connected to the pathogenesis of OA. Numerous research works suggested the potential clinical use of these biomarkers in OA diagnosis, disease progression tracking, and treatment result prediction. Moreover, while the majority of studies concentrated on human subjects, several investigations employed animal models, including rats and rabbits, to elucidate the pathophysiology of OA and identify potential treatment targets. Taken together, these findings underscore the complexity of metabolic dysregulation in OA and the promise of metabolomics analysis in uncovering novel avenues for diagnosing and treating this prevalent musculoskeletal disorder.

Type of sample used in literature for metabolomics analysis in discovering biomarkers for osteoarthritis.
Summary of major metabolomics biomarkers identified in human and animal samples across various studies.
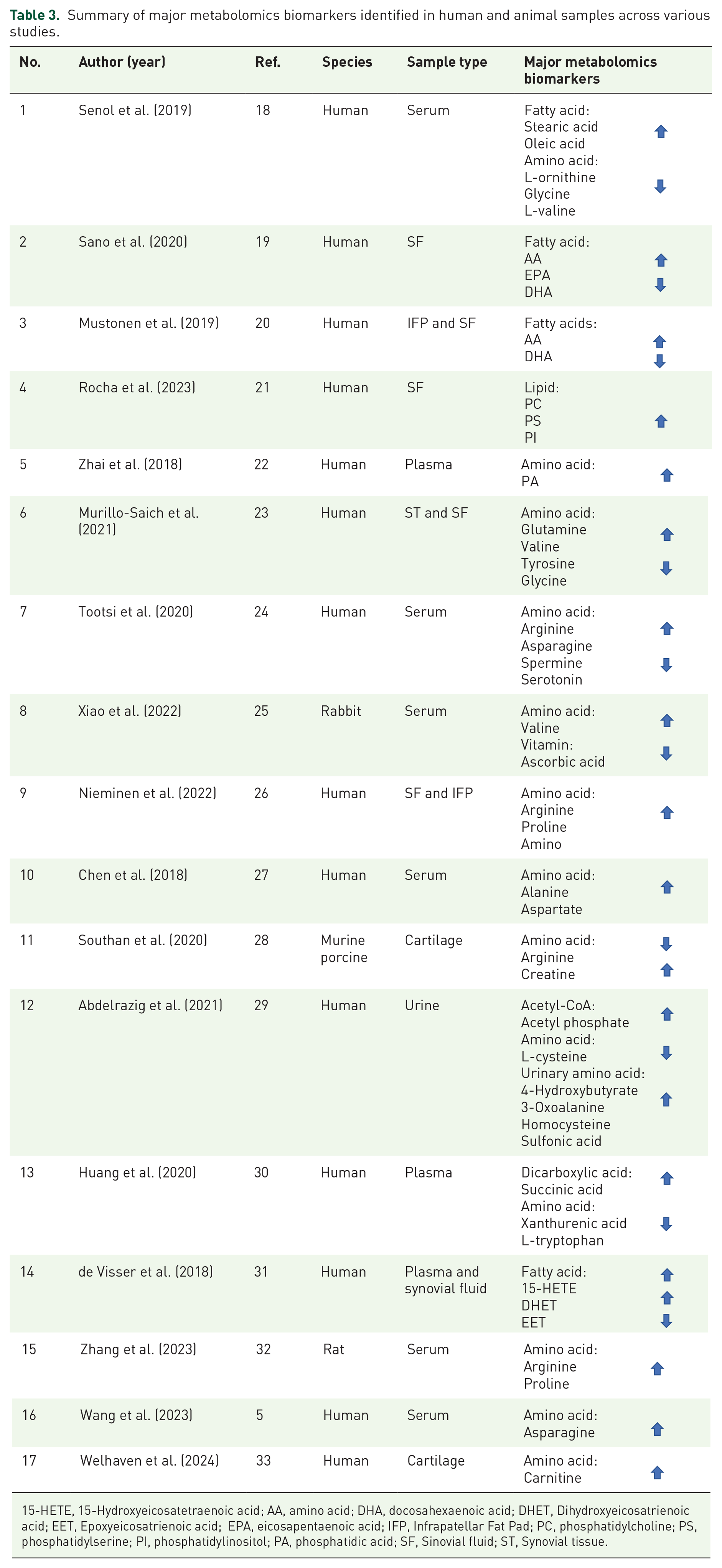
15-HETE, 15-Hydroxyeicosatetraenoic acid; AA, amino acid; DHA, docosahexaenoic acid; DHET, Dihydroxyeicosatrienoic acid; EET, Epoxyeicosatrienoic acid; EPA, eicosapentaenoic acid; IFP, Infrapatellar Fat Pad; PC, phosphatidylcholine; PS, phosphatidylserine; PI, phosphatidylinositol; PA, phosphatidic acid; SF, Sinovial fluid; ST, Synovial tissue.
Discussion
Metabolomics is an emerging and promising field that offers the opportunity to identify metabolites involved in key biological and pathological processes. It offers the opportunity to understand disease pathophysiology comprehensively by utilizing metabolic biomarkers to detect diseases early, monitor their progression, and evaluate treatment efficacy. In the context of OA, metabolomics research has identified dysregulated metabolites associated with altered metabolic pathways and profiles. These dysregulated metabolites, such as Lys phosphatidylcholines (LPCs), PCs, sphingolipids, amino acids, glucose, lactate, ATP, prostaglandins, and oxidized lipids, play critical roles in inflammation, energy metabolism, lipid metabolism, amino acid metabolism, and oxidative stress. Their dysregulation contributes to OA pathogenesis, leading to cartilage degradation, ECM breakdown, inflammatory responses, and altered cellular functions.
OA is a highly prevalent degenerative joint disease affecting more than 500 million people worldwide and is characterized by cartilage breakdown and changes in joint structure. Unfortunately, no effective therapies are available to halt or reverse disease progression. 34 Although the exact cause of OA remains unclear, dysregulated signaling pathways and metabolic disturbances are believed to be significant factors. Metabolomics, studying small molecule metabolites in biological systems, provides a promising approach to identify potential biomarkers and gain insights into the molecular mechanisms underlying OA.
Therefore, understanding the dysregulated signaling pathways involved in OA is crucial for developing effective treatments and slowing disease progression.35,36 This systematic review summarizes metabolomics biomarkers associated with dysregulated signaling pathways in OA, covering studies analyzing various biological samples such as serum, plasma, synovial fluid, synovial tissue, urine, infrapatellar fat pad, and cartilage. Both animal models (rabbits, rats, and pigs) and human subjects were included in the study. Consistently, several metabolites were found to be altered in OA, including amino acids, fatty acids, lipids, and carbohydrates. Notably, alanine, lysine, arginine, glutamine, tyrosine, and proline were commonly identified metabolites linked to signaling pathways involved in inflammation, pain, cartilage degradation, and apoptosis in OA, such as the NFκB, MAPKs, tumor necrosis factor (TNF), Wnt/β-catenin, and mammalian target of rapamycin (mTOR) pathway. In the following sections, the dysregulated metabolites and signaling mechanisms will be discussed.
Metabolomics and signaling pathway dysregulation in OA and dynamic interaction
Metabolic and signaling pathways together share a complex relationship. Understanding the coordination of metabolic activities and signaling pathways in mammalian cells remains challenging and poorly understood. The interplay between dysregulated metabolomics and signaling pathways is crucial in OA pathogenesis. Metabolites can directly influence cellular signaling pathways, and conversely, dysregulated signaling pathways can alter metabolic processes, creating a bidirectional relationship.
Many studies have reported that dysregulated metabolomics impact signaling pathways. This indicates certain metabolites can act as signaling molecules and modulate various cellular processes in joint tissues. For instance, alterations in metabolites like reactive oxygen species (ROS), nitric oxide (NO), prostaglandins, and certain lipid species can activate inflammatory signaling pathways such as NFκB.37 –41 Likewise, signaling pathways can also affect metabolic pathways. Dysregulated signaling pathways in OA can directly influence metabolic processes in joint tissues. For example, the NFκB pathway, a central player in inflammation, can impact the expression of various enzymes involved in glycolysis, lipid metabolism, and amino acid metabolism.42,43
Dysregulated metabolomics in OA
Dysregulated metabolomics in OA involves changes in various metabolic pathways that reflect changes in the osteoarthritic joint microenvironment. Several key metabolomics dysregulations have been identified in OA studies, including amino acid metabolism, lipid metabolism, oxidative stress markers, and energy metabolism (Tables 2 and 4).
A summary of dysregulated metabolomics pathways and biomarkers in OA pathophysiology.

AhR, aryl hydrocarbon receptor; BCAAs, branched-chain amino acids; DKK-1, Dickkopf-1; ECM, extracellular matrix; FFA, free fatty acids; MAPK, Mitogen-activated protein kinase; MMPs, matrix metalloproteinases; mTOR, mammalian target of rapamycin; NFκB, nuclear factor kappa B; NO, nitric oxide; OA, osteoarthritis; ROS, reactive oxygen species; TCA, tricarboxylic acid.
Amino acid metabolism
Dysregulation of amino acid metabolism has been observed in OA, with altered levels of key amino acids such as arginine, proline, glutamate, and tyrosine.27,29 –31,36 –39,40 –42,44 –48 These dysregulated amino acids can contribute to the dysregulation of signaling pathways and affect cartilage homeostasis, inflammation, and matrix degradation. 64 Our findings reported that increased levels of glutamine, valine, arginine, and tyrosine in OA may reflect changed metabolic processes in the joint. 23 While increased proline levels are related to OA-induced fibrosis. 44 Thus, understanding the connections between dysregulated amino acid metabolites and signaling pathways can provide valuable insights into OA’s molecular mechanisms and potential therapeutic targets.
BCAAs, tryptophan–kynurenine pathway, arginine metabolism, glycine metabolism, and serine metabolism are some dysregulated amino acids in OA (Figure 3).

Amino acids regulate the NFκB signaling pathway in OA.
BCAAs are groups of leucine, isoleucine, and valine. These three essential amino acids are critical in protein synthesis and energy production. 49 Dysregulation of BCAA metabolism in OA is associated with increased catabolism and altered balance between catabolism and anabolism. Elevated levels of BCAAs especially have been observed in OA patients’ synovial fluid and cartilage. 50 Similarly, elevation of valine has been observed in human serum, synovial fluid, synovial tissue, and rabbit serum in our findings.18,23,25 These increased BCAAs are thought to activate the mTOR, signaling pathway, which regulates cell growth, metabolism, and inflammation. mTOR activation can further activate NFκB signaling, leading to an enhanced inflammatory response in OA. 51 This dysregulated BCAA metabolism and its downstream effects on mTOR and NFκB may contribute to OA’s chronic inflammation and cartilage degradation.
Tryptophan is an essential amino acid that serves as a precursor for synthesizing various metabolites, including kynurenine. Dysregulation of tryptophan metabolism in OA has been linked to activating the aryl hydrocarbon receptor (AhR) pathway. Kynurenine, a tryptophan metabolite, activates AhR, potentiating NFκB signaling and driving inflammation. 52 In addition, kynurenine metabolism has been associated with increased production of ROS and oxidative stress, which further contributes to OA pathogenesis. 52
Arginine is a semi-essential amino acid that plays a role in various cellular processes, including NO production by nitric oxide synthases (NOS). Dysregulated arginine metabolism in OA can increase NO production, particularly by inducible NOS, activating NFκB, and upregulating proinflammatory genes. 53 In addition, elevated NO levels can contribute to oxidative stress and cartilage degradation in OA. 53
Furthermore, our finding suggests that arginine contributes to undesirable collagen synthesis and relates to fibrosis-related factors.24,26,28
Glycine is a non-essential amino acid involved in various cellular functions, including antioxidant defense. Dysregulated glycine metabolism has been associated with increased ROS production and oxidative stress in OA. 54 This oxidative stress can activate NFκB and drive the inflammatory response, contributing to cartilage degradation. 54 In addition, our finding suggests that glycine levels are lower in obesity 18 indicating that obesity can also cause oxidative stress in arthritis patients. Collagen breakdown in the joint causes cartilage degeneration and lowers glycine levels since it is required to repair or replace damaged collagen. 23
Serine is a crucial amino acid involved in various biological processes, including nucleotide synthesis and redox balance. 65 Dysregulation of serine metabolism has been linked to increased oxidative stress and activation of NFκB in OA. 55 Moreover, alterations in serine metabolism can impact the availability of glutathione, a potent antioxidant, further contributing to oxidative stress and inflammation. 55
Lipid metabolism
Lipid metabolism plays a crucial role in the pathogenesis of OA, and dysregulation of lipid metabolism may contribute to inflammation, oxidative stress, and alterations in cellular function in OA joints. 66 Understanding the connections between dysregulated lipid metabolites and signaling pathways can shed light on OA’s molecular mechanisms and potentially lead to the development of novel biomarkers for disease diagnosis and therapeutic interventions.
One of the key aspects of dysregulated lipid metabolism in OA is the increased production of proinflammatory lipid metabolites. Inflammatory lipids, such as prostaglandins, leukotrienes, and platelet-activating factors, are generated from arachidonic acid, a polyunsaturated fatty acid. 56 Dysregulation of enzymes involved in arachidonic acid metabolism, such as cyclooxygenases and lipoxygenases, leads to an imbalance in proinflammatory lipid production. These lipid mediators can activate NFκB and other inflammatory signaling pathways, promoting a chronic inflammatory state in the joint. 47
In addition, lipid signaling molecules from lipid metabolism, such as prostaglandins and sphingolipids, can also be measured as metabolite counters in OA. Prostaglandins, including prostaglandin E2 (PGE2), are inflammatory mediators derived from arachidonic acid metabolism. 19 Elevated levels of PGE2 have been detected in OA joints and may contribute to inflammation and pain. Sphingolipids, such as ceramides and sphingosine-1-phosphate (S1P), are involved in cell signaling and inflammation. 67 This is similar to our findings, where elevated levels of PGE2 and PGD2 metabolites in joints can stimulate the release of enzymes and cytokines that contribute to cartilage breakdown and inflammation. 19
Free fatty acids (FFAs) are essential intermediates in lipid metabolism and may be dysregulated in OA. Increased levels of FFAs, such as palmitic acid and oleic acid, have been reported in OA patients’ synovial fluid and serum. 18 FFAs may act as inflammatory mediators and contribute to OA cartilage degradation and joint inflammation. 68
Another factor of lipid metabolism in OA is lipid peroxidation and oxidative stress, which plays a critical role in OA pathogenesis. Lipid peroxidation, a process involving polyunsaturated FFA oxidation in cell membranes, generates ROS and lipid peroxidation products. 39 These oxidative products, such as malondialdehyde (MDA) and 4-hydroxynonenal (4-HNE), can induce cellular damage and promote inflammatory responses. Elevated levels of lipid peroxidation products have been detected in OA joints and are associated with increased cartilage degradation and synovial inflammation. 59 Sphingolipids, such as ceramides and S1P, are involved in cell signaling and inflammation. 67 Dysregulation of sphingolipid metabolism and altered levels of sphingolipid metabolites have been associated with OA pathogenesis.
In our finding, PC, phosphatidylserines, and phosphatidylinositol have great potential as a biomarker for knee OA progression since they were found to be substantially related to cartilage volume decrease in knee OA patients. 21
Energy metabolism
In OA, changes in energy metabolism may affect the concentration of certain metabolites involved in energy-related metabolic pathways. OA is associated with alterations in energy metabolism, including glucose utilization disorders, mitochondrial dysfunction, and impaired energy production. These alterations may affect cellular energy balance and contribute to the pathogenesis of OA.
Glucose is a key metabolite in energy metabolism and serves as a fuel source for cells. Dysregulated glucose metabolism may affect glucose utilization and energy production in OA. Measurement of glucose levels and its metabolite lactate may reflect the dysregulation of glucose metabolism and the switch to anaerobic glycolysis observed in OA. 46 The tricarboxylic acid (TCA), or Krebs cycle, is an important metabolic pathway in energy production by oxidizing carbohydrates, fats, and amino acids. In OA, dysregulation of the TCA cycle may occur, affecting the concentration of metabolites such as citrate, α-ketoglutarate, succinate, and malate.57,58 Measurement of these metabolites may provide insight into the dysregulation of the TCA cycle and altered energy metabolism in OA.
An α-ketoglutarate is an essential metabolite from the TCA cycle with multiple functions, including antioxidant and anti-inflammatory properties. In a recent study, Liu et al. 69 reported that the content of α-ketoglutarate decreased in damaged Cartilage and interleukin 1-beta (IL-1β) induced chondrocytes in humans. Further supplementation with α-ketoglutarate has shown the potential to alleviate the osteoarthritic phenotype by modulating mitophagy and reducing oxidative stress. 69
Mickiewicz et al. 70 reported increased levels of citrate and fructose and decreased malate levels in SF samples from OA patients. The higher citrate content indicates a high energy demand in OA patients, while a decrease in malate content indicates a specific disturbance and dysregulation of energy production by OA. 70
Dysregulated signaling pathways in OA
Dysregulated signaling pathways in the pathogenesis of OA can have profound effects on metabolomics, leading to the dysregulation of various metabolites involved in key metabolic pathways. These altered metabolites contribute to the metabolic changes observed in OA and can serve as potential biomarkers to study disease progression and identify therapeutic targets.
Several key signaling pathways have been implicated in OA pathogenesis, including the NFκB pathway, MAPKs, Wnt/β-catenin pathway, and mammalian target of the mTOR pathway.
Dysregulated NFκB signaling and alteration in metabolism
Chronic inflammation is a hallmark of OA, and dysregulated signaling pathways, such as NFκB activation, play a central role in driving inflammation. The activation of NFκB leads to increased expression of enzymes involved in arachidonic acid metabolism, leading to the generation of proinflammatory lipid mediators like prostaglandins and leukotrienes. These lipid metabolites can further exacerbate the inflammatory response and contribute to cartilage degradation. 19 Metabolomic profiling in OA joints often reveals elevated levels of proinflammatory lipid metabolites, reflecting the dysregulation of lipid metabolism driven by inflammatory signaling pathways. 47
NFκB is a transcription factor that regulates the expression of numerous genes involved in the inflammatory response, immune activation, and tissue remodeling. Dysregulated NFκB activity in OA can lead to alterations in various metabolites, contributing to the pathogenesis of the disease. The following are further details into how NFκB dysregulation affects metabolites in OA (Figure 4).

Molecules affected by NFκB activation in OA.
One of the primary effects of NFκB dysregulation in OA is the upregulation of enzymes involved in arachidonic acid metabolism. This leads to increased production of proinflammatory lipid mediators, such as prostaglandins and leukotrienes, crucial in driving inflammation and cartilage degradation. Elevated levels of these lipid metabolites have been detected in OA joints, reflecting the impact of NFκB dysregulation on lipid metabolism and inflammation. 47
NFκB activation in OA is often associated with the expression of proinflammatory cytokines, such as IL-1β. These cytokines can alter the metabolism of certain amino acids, particularly tryptophan. Dysregulated tryptophan metabolism leads to increased kynurenine production, a tryptophan metabolite. Kynurenine activates the AhR pathway, further potentiating inflammation in OA. 52 The elevation of kynurenine levels in OA joints indicates the impact of NFκB dysregulation on amino acid metabolism and its connection to inflammation. 52
NFκB dysregulation can lead to oxidative stress, an imbalance between ROS production and antioxidant defense. Increased NFκB activity induces the expression of genes involved in the production of ROS. Excessive ROS can initiate lipid peroxidation, leading to the generation of lipid peroxidation products, such as MDA and 4-HNE. These lipid peroxidation products are altered metabolites that reflect the impact of NFκB dysregulation on oxidative stress and lipid metabolism in OA. 59
NFκB activation in OA can also influence energy metabolism and glycolysis. 46 Dysregulated NFκB signaling has been shown to increase glycolytic activity in chondrocytes, leading to elevated levels of lactate and decreased pH in OA joints. 70 This shift toward glycolysis and altered energy metabolism can affect various metabolites involved in cellular energy production and contribute to the pathogenesis of OA.
Therefore, dysregulation of the NFκB signaling pathway in OA has wide-ranging effects on metabolites, including proinflammatory lipid metabolites, amino acid metabolites like kynurenine, lipid peroxidation products, altered glycolytic intermediates, and changes in cellular stress-related metabolites. These altered metabolites collectively contribute to the inflammatory and oxidative stress environment in OA joints, driving cartilage degradation and disease progression. Understanding the impact of NFκB dysregulation on metabolites is essential for identifying potential therapeutic targets to intervene and halt the inflammatory cascade in OA.
Dysregulated MAPK signaling and alteration in metabolism
MAPK signaling pathways, including extracellular signal-regulated kinase, c-Jun N-terminal kinase, and p38 MAPK, are dysregulated in OA and closely interconnected with dysregulated metabolites. 60 The dysregulation of these signaling pathways can influence various metabolic processes within the joint, leading to altered levels of specific metabolites. Activation of these pathways increases proinflammatory cytokine production (e.g., IL-1β, TNFα) and other inflammatory mediators. 71 These cytokines alter cellular metabolism and promote the synthesis of specific metabolites involved in inflammation and oxidative stress. For example, increased cytokine levels can lead to the production of ROS, which can oxidize lipids and generate oxidized lipids as dysregulated metabolites. 72
Dysregulated MAPK pathways can upregulate the expression and activity of MMPs. MMPs are enzymes responsible for degrading the ECM components in cartilage. 61 The breakdown of ECM results in the release of metabolites such as amino acids and oligopeptides that would otherwise be sequestered within the cartilage structure. MAPK dysregulation can also impact amino acid metabolism in chondrocytes, the cells responsible for maintaining cartilage. For example, the increased expression of MMPs due to MAPK activation can promote the breakdown of cartilage components, releasing amino acids like arginine and proline into the joint space. 44 The altered activity of MAPK pathways can influence gene expression in glycolysis, the TCA cycle, and oxidative phosphorylation. 73 Therefore, the levels of various metabolites related to glucose metabolism, such as glucose and lactate, can be dysregulated.
The dysregulated MAPK pathways are also associated with the altered expression and activity of enzymes involved in lipid metabolism. This can lead to changes in the levels of various lipid metabolites, such as LPCs and PCs, which are essential components of cell membranes and play roles in signaling and inflammation.74,75
Aberrant MAPK signaling pathways in OA lead to altered gene expression and cellular responses, resulting in changes in the levels of various metabolites. These dysregulated metabolites, such as oxidized lipids, amino acids, glucose, and LPCs, significantly promote inflammation, cartilage degradation, and other pathological processes associated with OA. Our finding suggests that excess proinflammatory eicosanoids (signaling molecules derived from polyunsaturated fatty acids. They play key roles in various physiological processes, including inflammation, immunity, and platelet aggregation) produced by amino acids can aggravate the inflammatory process and contribute to the development of OA. 76 Meanwhile, a high level of oxidative stress in OA reduced the docosahexaenoic acid level, which possesses antioxidant properties leading to damage to joint tissues, including cartilage. 20 So, understanding these interconnected pathways and their effects on metabolites is essential for identifying potential biomarkers and developing targeted therapies for OA.
Wnt/β-catenin pathway
Dysregulation of the Wnt/β-catenin pathway has been implicated in OA pathogenesis. Dysregulated metabolites, such as Dickkopf-1 and Wnt inhibitors, can inhibit Wnt/β-catenin signaling, leading to altered chondrocyte differentiation, matrix synthesis, and cartilage integrity. 74
mTOR pathway
The mTOR pathway, a crucial regulator of various cellular processes, is dysregulated in OA. This dysregulation impacts several key functions including autophagy, cell proliferation, and metabolism within the joint environment. Specifically, dysregulated metabolites, such as energy substrates and amino acids, exert significant influence over mTOR signaling. Consequently, this disruption in signaling pathways can have profound effects on critical aspects of OA pathology, including chondrocyte viability, maintenance of the ECM, and the inflammatory response. This interconnection underscores the complex relationship between metabolic dysregulation and the progression of OA, offering potential avenues for therapeutic intervention targeting these metabolic pathways to mitigate the impact of the disease. 75
Comorbidities with other diseases
OA is not only a significant cause of disability but also frequently occurs alongside various comorbidities that can exacerbate the disease’s impact and complicate management. Obesity is a notable comorbidity, contributing to the initiation and progression of OA through increased mechanical stress on joints and by promoting inflammatory pathways and releasing inflammatory cytokines such as IL-1β, IL-6, and TNFα, which can deteriorate joint degradation. 77 This correlation has been discussed extensively in the literature, highlighting the biomechanical and biochemical pathways influenced by obesity. 77 Metabolic syndrome, another common comorbidity, involves a cluster of conditions, including increased blood pressure, 78 high blood sugar, excess body fat around the waist, and abnormal cholesterol levels, which intensify the risk of chronic inflammation, further influencing OA pathogenesis. Studies have shown that these metabolic factors significantly contribute to the inflammatory processes that exacerbate OA. 79 The emerging field of metabolomics has provided insights into these complex interactions by identifying specific metabolites, such as certain amino acids and lipids, implicated in key signaling pathways like NFκB, MAPK, Wnt/β-catenin, and mTOR. These pathways are crucial in regulating inflammatory processes and metabolic dysfunctions associated with OA, highlighting the interconnected nature of metabolic syndromes, obesity, and OA. Research in this area points to potential metabolic targets for mitigating the impact of these comorbidities on OA. 80 Furthermore, chronic pain from OA often leads to psychological stress and mood disorders like depression and anxiety. A study reported that one-fifth of people with OA experience symptoms of depression and anxiety. 81 This comprehensive understanding presents new opportunities for targeted therapeutic interventions that could ameliorate the effects of these comorbidities on OA.
Conclusion
In OA, dysregulated metabolomics and dysregulated signaling pathways are intertwined and contribute to the pathophysiology of this debilitating joint disorder. Dysregulated metabolites and signaling pathways interact to promote cartilage degradation, inflammation, and other pathological processes.
Several metabolites such as amino acids, fatty acids, and lipids were found to be altered in OA and linked to signaling pathways involved in inflammation, cartilage degradation, oxidative stress, and apoptosis. Such notable pathways are NFκB, MAPKs, Toll-like receptors, mTOR, and the TCA cycle. Understanding the complex interplay between dysregulated metabolomics and dysregulated signaling in OA may pave the way for developing targeted therapies to restore metabolic and signaling homeostasis, ultimately alleviating symptoms and improving patient outcomes. However, it is essential to acknowledge that more research is needed to uncover the specific molecular pathways behind these dysregulations. This continuous research will improve our understanding of OA pathogenesis and help us design more focused and effective therapy options. The intricacy of OA needs ongoing study to unravel the intricate molecular landscape, eventually improving our capacity to treat the healthcare cost caused by this devastating degenerative joint disease.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X241299535 – Supplemental material for The interplay between dysregulated metabolites and signaling pathway alterations involved in osteoarthritis: a systematic review
Supplemental material, sj-docx-1-tab-10.1177_1759720X241299535 for The interplay between dysregulated metabolites and signaling pathway alterations involved in osteoarthritis: a systematic review by Atiqah Aziz, Kavitha Ganesan Nathan, Tunku Kamarul, Ali Mobasheri and Alimohammad Sharifi in Therapeutic Advances in Musculoskeletal Disease
Footnotes
Acknowledgements
The authors would like to thank the National Orthopaedic Centre of Excellence for Research and Learning (NOCERAL) for providing resources to prepare this article.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
