Abstract
Background:
Takayasu arteritis (TA) is associated with an increased risk of developing complicated comorbidities, which can bring both psychological and physical burdens to the patients.
Objective:
TA is found to carry a high risk of developing depression. This research aimed to investigate the risk factors and prognosis of depression in TA patients.
Design:
A longitudinal observation cohort was conducted on TA patients with or without depression to explore the clinical characteristics.
Methods:
In this cohort study, 90 TA patients were split into two groups with or without depression. Depression was evaluated by the Hospital Anxiety and Depression Scale (HADS) in TA patients. TA patients with depression were followed up for at least 3 months. We used multivariate logistic regression analysis to find the risk factors and Kaplan–Meier curve analysis to determine the prognosis.
Results:
We concluded 90 TA patients in this research, 29 of whom were in depression. Indian Takayasu’s Arteritis Activity Score (ITAS2010) ⩾2 (odds ratio (OR) (95% confidence interval, CI) 26.664 (2.004–354.741), p = 0.013), interleukin-6 (IL-6) (OR (95% CI) 1.070 (1.022–1.121), p = 0.004), prednisone equivalents (OR (95% CI) 1.101 (1.030–1.177), p = 0.005), and carotidynia (OR (95% CI) 5.829 (1.142–29.751), p = 0.034) have been shown independent risk factors for depression in TA patients. We also identified the association between disease remission with the improvement of HADS-D score (Log-rank p = 0.005, hazard ratio (HR) 0.25) and depression (Log-rank p = 0.043, HR 0.28).
Conclusion:
Aggressive treatment to achieve remission can promote improvement of depression in patients with TA. Screening for depression should also be performed in patients with elevated disease activity, IL-6, glucocorticoid use, and carotidynia.
Introduction
Takayasu arteritis (TA) is a rare autoimmune chronic inflammatory disease, characterized by granulomatous full-thickness arteritis that mainly affects the aorta and branch vessels. 1 Inflammation in the arteries can lead to severe vascular damage and eventually to the involvement of several vital organs. 2 TA is recognized to have a strong association with an increased risk of developing complicated comorbidities, which can bring both psychological and physical burdens to the patients. 3 Most cases are young female patients with an onset age of 10–40 years. 4 It is vital to recognize the mental health impact of the disease as early as possible.
Psychological disorders have been recognized as important comorbidities of immune diseases for a long time. 5 A cross-sectional study in Europe involving 1800 patients with autoimmune disease, discovered that 45.9% of participants tended to develop depression, based on the Hospital Anxiety and Depression Scale (HADS). 6 Beurel et al. 7 recognized the immune system regulated mood and the potential causes of the dysregulated inflammatory responses in depressed patients. The bidirectional relationship between depression and rheumatic diseases is noteworthy. Depression is putatively implicated in high levels of pain, fatigue, disability, and higher suicide attempts.8–10 Previous studies have identified that depression could increase the likelihood of hospitalization and longer hospital stays of patients with autoimmune diseases.11,12
Yilmaz et al. 3 had proved that mental health parameters were impaired in TA. However, there was no definite association between patients with depressive states and the evolution of TA.13–16 The emotional state generated by the TA should not be ignored. Therefore, we collected the medical data and follow-up records of the TA patients admitted to our hospital. The major idea of this research was to explore the risk factors of comorbid depressive state in patients with TA and figure out the association of depression improvement with remission of the disease.
Methods
Patients
A total of 90 patients aged 16–65 years with TA in Beijing Anzhen Hospital between April 2021 and December 2022 (Figure 1), who fulfilled the criteria for classification of the American College of Rheumatology in 2022 17 were included in this study. The exclusion criteria were patients who had chronic or current infections, tumors, other autoimmune diseases, a history of psychiatric diseases, and any anti-depressant use. At the time of first admission, we assessed patients as treatment-naïve or treated, based on whether they had been treated regularly with TA. According to the past medical history and treatment, relapse was defined as the recurrence of active disease, at least one of the following: (a) current activity on imaging or biopsy, (b) ischemic complications attributed to TA, and (c) persistently elevated inflammatory markers (after other causes have been excluded). 18 Refractory disease was defined as inability to attain absence of all clinical signs and symptoms attributable to active TA and normalization of erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) despite the use of standard care therapy. 18 This study was conducted in accordance with the ethical principles Helsinki Declaration and was approved by the Ethics Committee of Beijing Anzhen Hospital (Approval Number: 2022228X). The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology: explanation and elaboration. 19

Flowchart of study participants.
Collection of sociodemographic characteristics, clinical data, and laboratory parameters
Sociodemographic characteristics include age, sex, duration of symptoms, marital status, educational time, and body mass index (BMI). We collected the disease activity, clinical symptoms, history, treatment paradigm, and Numano type as clinical data at baseline. All patients have carried the duplex ultrasound, magnetic resonance angiography, and computed tomography angiography to evaluate the involvement of vessels. 20 We used the National Institutes of Health (NIH) criteria, 21 the Indian Takayasu’s Arteritis Activity Score (ITAS2010), 22 and ITAS with acute-phase reactants (ITAS.A) 23 to assess the disease activity. Clinical symptoms during the course of the disease were recorded, such as chest pain, chest tightness, dizziness, headache, fever, limb pain, carotidynia, upper limb blood pressure asymmetry, pulselessness, and intermittent claudication. We also kept a detailed record of the medication used by patients at the time of enrollment, including glucocorticoids, conventional synthetic disease-modifying antirheumatic drugs (csDMARDs), and biologic disease-modifying antirheumatic drugs (bDMARDs). We documented the glucocorticoid dosage and converted it to prednisone equivalents. The dosages and methods of administration of DMARDs were Methotrexate 15 mg Qw (quaque week/every week), Cyclophosphamide 100 mg Qod (quaque omni die/every other day), Mycophenolate mofetil 0.75 g Bid, Azathioprine 100 mg Qd (quaque die/every morning), Hydroxychloroquine Sulfate 0.2 g Qd, Leflunomide 20 mg Qd, Tocilizumab 8 mg/kg Qm, Adalimumab 40 mg Q2w. The vascular type of TA was categorized by Hata and Numano. 21 We also recorded whether the patients’ coronary, pulmonary, and intracranial arteries were involved. Laboratory parameters included complete blood count, ESR, CRP, immunoglobulin, complement component, cytokines (interleukin-6 (IL-6) and tumor necrosis factor-alpha), which were assessed at the time of enrollment by the laboratory medicine of Anzhen Hospital.
The evaluation of depression
After all imaging and blood tests and disease evaluation had been completed, the patients included were all asked to fill in a HADS questionnaire 24 when they visited our department for the first time. The HADS was used to assess the depressive state. 25 The questionnaires contained 14 subscales for anxiety and depression symptoms. Each item is used on a four-point scale to make ratings, ranging from 0 to 3 in total. The HADS depression score (HADS-D) of 0–7 points indicated no or few depression symptoms, 8–10 points indicated mild depression, and ⩾11 points indicated severe depression. In our study, we defined a HADS depression score ⩾8 points as depressive state.
Follow-up and outcome
All patients were followed for 3 months to assess their disease remission status. Twenty-eight of these patients with depression at baseline continued to be followed from the date of visits to January 2023. One patient was lost to follow-up and refused to continue participating in the study. Three months after the first visit, we evaluated the disease condition of each individual. We defined TA disease remission according to 2018 EULAR recommendations for the management of large vessel vasculitis, 18 which met persistent normal ESR or CRP, symptom stabilization, NIH < 2, and the presence of a stable or improved lesion on follow-up angiography at the same time. Patients were split into remission group and non-remission group. They were examined every 3 months until their depressive state improved, or they were followed up for 15 months. At follow-up, we asked patients to complete the HADS questionnaire again and defined the improvement of depression as the HADS-D at follow-up of less or equal to 7, while the improvement of HADS-D score as the HADS-D at follow-up was less than baseline. The follow-up data, including HADS-D, laboratory parameters, disease activity, clinical presentation, imaging indicators, and medication, were obtained by either re-hospitalization records or telephone interviews.
Statistical analysis
Continuous variables were presented as mean ± SD for data of normal distribution and as median (Q1, Q3) for non-normal distribution. Categorical variables are presented in counts (percentages). The discrepancy between measured parameters of non-normal distribution in patients and controls was calculated by the Mann–Whitney test. The χ2 test was applied for the assessment of qualitative parameters. To quantitate the correlation between continuous variables, Spearman’s approach was used. The independent risk factors associated with depressive state involvement were identified by multivariate logistic regression. Differences between patient-measured parameters before and after treatment were assessed by paired sample Wilcoxon signed-rank test. Cumulative event rates were determined by Kaplan–Meier curve analysis. Statistical significance was defined as p-values < 0.05 and all statistical tests were two-tailed. All statistical analysis was operated with SPSS software (version 26.0; IBM Corp., Armonk, NY, USA), and we used GraphPad Prism 9 (GraphPad Software, San Diego, CA, USA) to create the figures.
Results
Sociodemographic characteristics and clinical differences in the depressed TA group versus the non-depressed group.
The study included 90 TA patients, of whom 29 (32.2%) had comorbid depression, with a female/male ratio of 11.86:1 (83/7). The median time between the diagnosis of depression and the diagnosis of TA was 14 months (14.00 (0.00–65.00) months). Patients with depression were younger (34.00 (24.50–48.00) vs 45.00 (33.50–49.00) years, p = 0.026) than patients without depression. There was also less married proportion in patients with depression (18 (62.1)% vs 55 (90.2)%, p = 0.001). No significant differences were found in education level, disease duration, and BMI between the two groups. Moreover, we found a higher proportion of carotidynia in TA patients with depression than those without depression (9 (31.0)% vs 4 (6.2)%, p = 0.001). No heterogeneity was observed in the frequency of other clinical presentations, histories, Numano types, and vascular damage between the two groups (Table 1, Supplemental Tables 1 and 2).
Clinical features of TA patients with or without depression.

Type is divided according to Hata and Numano’s criteria.
p < 0.05. **p < 0.010.
ADA, adalimumab; AZA, azathioprine; bDMARDs, biologic disease-modifying antirheumatic drugs; BMI, body mass index; csDMARD, conventional synthetic disease-modifying antirheumatic drugs; CTX, cyclophosphamide; HCQ, hydroxychloroquine sulfate; LEF, leflunomide; MMF, mycophenolate mofetil; MTX, methotrexate; TA, Takayasu arteritis; TCZ, tocilizumab.
Regarding the medication of TA patients at their first admission, 44 (48.9%) patients were treated with glucocorticoids, 50 (55.6%) with csDMARDs, and 21 (23.3%) with biologic disease-modifying antirheumatic drugs (bDMARDs). However, no difference was observed in the above three drug comparisons between the two groups. In a more detailed comparison of drug regimens, we found that TA patients with depression were used more prednisone equivalents (12.50 (0.00–20.00) vs 0.00 (0.00–10.00), p = 0.006) and less commonly used MMF (1 (3.4)% vs 14 (23.0)%, p = 0.016) (Table 1).
Laboratory parameters and disease activity between two groups of TA patients
In the comparison of blood routines, we found that the hemoglobin in the depression group was lower (113.00 (101.00–127.00) vs 125.00 (113.50–132.50) g/L, p = 0.016), while the White blood cell, neutrophils count, monocyte count, and platelet count were higher than those in the non-depression group (8.16 (6.55–10.10) vs 6.38 (5.06–8.66) 109/L, p = 0.020; 5.72 (4.04–7.59) vs 4.23 (2.98–5.80) 109/L, 0.023; 0.42 (0.31–0.62) vs 0.35 (0.28–0.51) 109/L, p = 0.024; 280.00 (234.00–373.00) vs 234 (191.00–328.00) 109/L, p = 0.015) (Table 2).
Laboratory parameters of TA patients with or without depression.
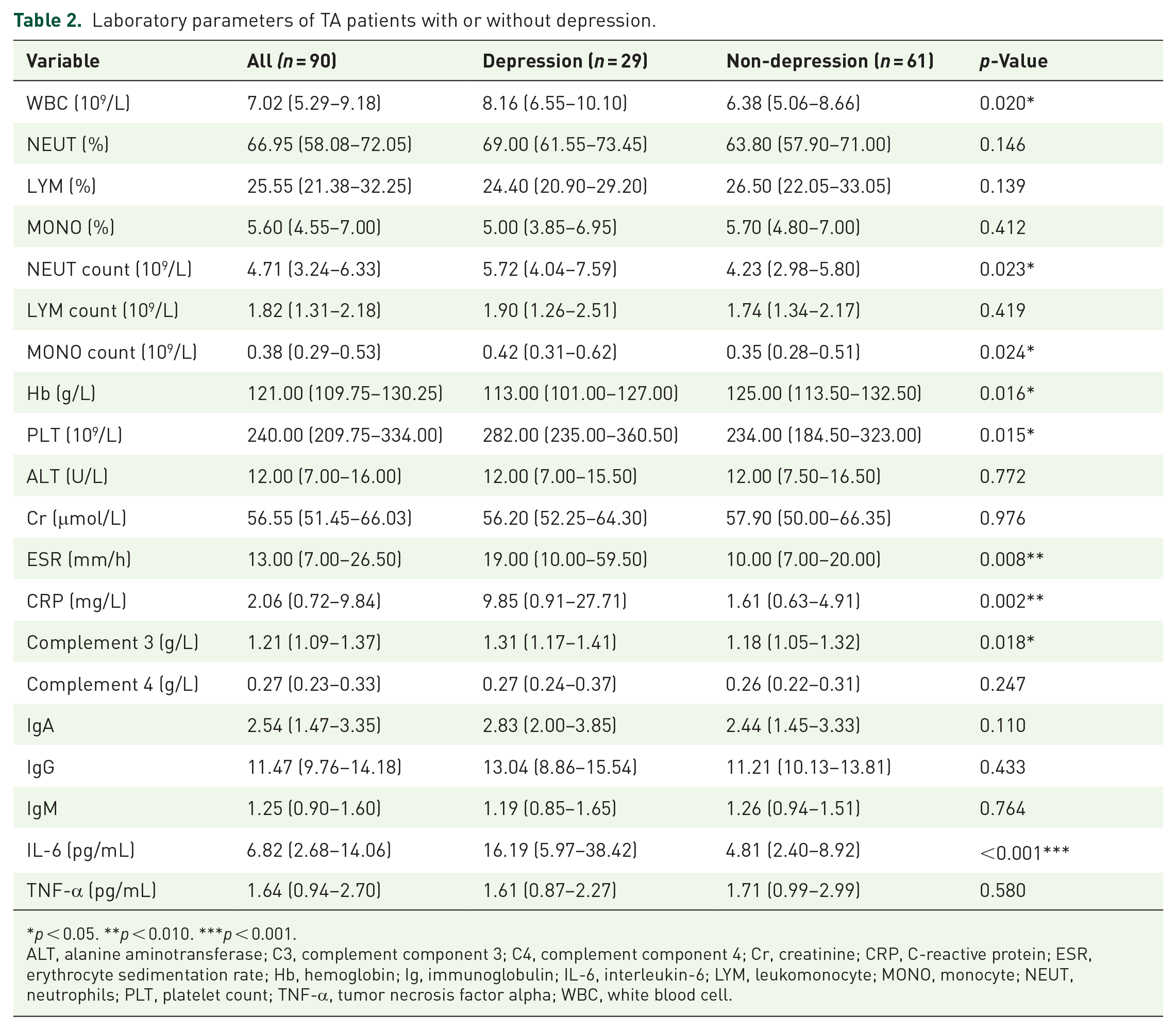
p < 0.05. **p < 0.010. ***p < 0.001.
ALT, alanine aminotransferase; C3, complement component 3; C4, complement component 4; Cr, creatinine; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; Hb, hemoglobin; Ig, immunoglobulin; IL-6, interleukin-6; LYM, leukomonocyte; MONO, monocyte; NEUT, neutrophils; PLT, platelet count; TNF-α, tumor necrosis factor alpha; WBC, white blood cell.
Both ESR and CRP were found higher in the depression group (19.00 (10.00–59.50) vs 10.00 (7.00–20.00) mm/h, p = 0.008; 9.85 (0.91–27.71) vs 1.61 (0.63–4.91) mg/L, p = 0.002). In the comparison with the non-depression group, IL-6, a classical inflammatory cytokine, showed a significantly higher level in the depression group (16.19 (5.97–38.42) vs 4.81 (2.40–8.92) pg/L, p < 0.001). C3 was observed relatively higher in the patients with depression (1.31 (1.17–1.41) vs 1.18 (1.05–1.32) mg/L, p = 0.018), while no difference was observed in C4 and immunoglobulin between two groups. We also compared the disease activity with the depression in TA patients and found NIH, ITAS.A, and ITAS2010 were higher in the depressed group (2.00 (2.00–3.00) vs 2.00 (0.50–2.00), p < 0.001; 9.00 (6.00–13.00) vs 4.00 (1.00–8.00), p < 0.001; 7.00 (4.00–10.00) vs 3.00 (0.00–6.00), p < 0.001) (Figure 2).

Comparison of disease activity between TA patients with depression and without depression. (a) The values of NIH, ITAS.A, and ITAS2010 in the depressed group were significantly higher than those in the non-depressed group. The proportion of NIH ⩾ 2 (b), ITAS.A ⩾ 5 (c), and ITAS2010 ⩾ 2 (d) were significantly higher than that in the non-depressed group.
Risk factors of depression in TA patients
We used univariate logistic analysis to identify the meaningful factors for depression in TA patients. We found age (odds ratio (OR) (95% confidence interval, CI) 0.955 (0.919–0.993), p = 0.022) and MMF (OR (95% CI) 0.120 (0.015–0.962), p = 0.046) were protective factors for depression in TA patients, while carotidynia (OR (95% CI) 6.412 (1.777–23.138), p = 0.005), NIH ⩾ 2 (OR (95% CI) 8.171 (1.775–37.615), p = 0.007), ITAS.A ⩾ 5 (OR (95% CI) 5.160 (1.839–14.481), p = 0.002), ITAS2010 ⩾ 2 (OR (95% CI) 9.375 (2.042–43.048), p = 0.004), ESR (OR (95% CI) 1.025 (1.006–1.045), p = 0.011), CRP (OR (95% CI) 1.046 (1.009–1.084), p = 0.013), IL-6 (OR (95% CI) 1.073 (1.026–1.122), p = 0.002), prednisone equivalents (OR (95% CI) 1.065 (1.021–1.110), p = 0.003), and refractory (OR (95% CI) 3.600 (1.321–9.813), p = 0.012) were significantly associated with the depression in TA patients (Table 3). We included the above clinical variables in the multivariate logistic regression analysis. Carotidynia (OR (95% CI) 5.829 (1.142–29.751), p = 0.034), ITAS2010 ⩾ 2 (OR (95% CI) 26.664 (2.004–354.741), p = 0.013), IL-6 (OR (95% CI) 1.070 (1.022–1.121), p = 0.004), and prednisone equivalents (OR (95% CI) 1.101 (1.030–1.177), p = 0.005) have been shown independent risk factors for depression in TA patients (Table 3, Figure 3). To ensure the accuracy of these results, we also examined the comparison of disease activity, and the involvement of blood vessels and medications between the patients aged <40 and aged ⩾40 groups. There were no significant differences between the two groups (Supplemental Table 3).
Logistics regression analysis of risk factors in TA patients with depression.

p < 0.05. **p < 0.010. ***p < 0.001.
CI, confidence interval; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; IL-6, interleukin-6; ITAS.A, ITAS with acute-phase reactants; ITAS2010, Indian Takayasu’s Arteritis Activity Score; MMF, mycophenolate mofetil; NIH, National Institutes of Health; TA, Takayasu arteritis.

Independent risk factors for TA patients with depression. Multivariate logistic analysis showed that IL-6, prednisone equivalent, ITAS2010, and carotidynia were independent risk factors for depression in TA patients.
Association between depression and TA disease remission
Twenty-eight TA patients participated in the follow-up. The median follow-up time was 10.25 months in the depressed group. After 3-month treatments, 67.9% (19/28) of TA patients with depression were assessed for remission. We used Kaplan–Meier curve analysis to identify the association between the disease remission with the improvement of HADS-D score (Log-rank p = 0.005, hazard ratio (HR) 0.25) (Figure 4(a)) and depression (Log-rank p = 0.043, hazard ratio (HR) 0.28) (Figure 4(b)). To explore whether the improvement of carotidynia would change the progression of depression, we further screened 10 patients in the follow-up cohort with carotidynia at baseline and grouped the patients according to the improvement. However, there was no significant association between the improvement of carotidynia and the improvement of HADS-D score and depression (Supplemental Figure 1). Compared to the baseline, NIH, ITAS.A, ITAS2010, and HADS-D after treatment were found to meaningfully decrease by paired sample Wilcoxon signed-rank test (2.50 (2.00–3.00) vs 0.00 (0.00–1.00), p < 0.001; 10.00 (7.00–13.00) vs 0.00 (0.00–2.50), p < 0.001; 7.50 (4.00–10.00) vs 0.00 (0.00–1.00), p < 0.001; 10.00 (9.00–10.00) vs 8.00 (7.00–9.00), p = 0.003) (Figure 5).
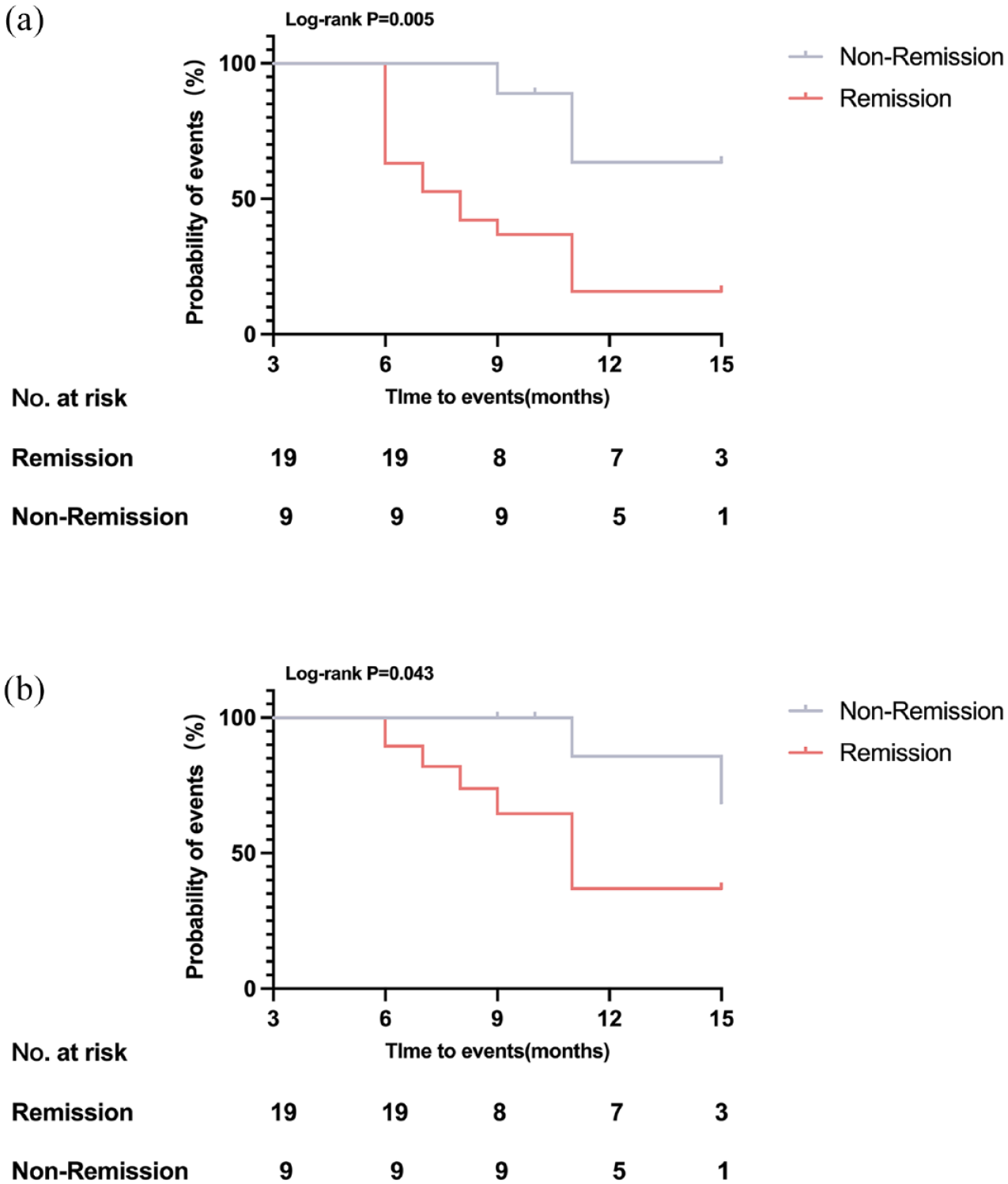
Association between the improvement of HADS-D score and depression with disease remission. Kaplan–Meier curve analysis identified that (a) the improvement of HADS-D score was associated with disease remission (Log-rank p = 0.005, HR 0.25) and (b) the improvement of depression was associated with disease remission (Log-rank p = 0.040, HR 0.28).

Changes in NIH, ITAS.A, ITAS2010, and HADS-D before and after treatment. After treatment, paired sample Wilcoxon signed-rank test identified that (a) NIH (p < 0.001), (b) ITAS.A (p < 0.001), (c) ITAS2010 (p < 0.001), and (d) HADS-D (p = 0.003) were significantly lower than those before treatment.
Discussion
This cohort study documented that 32.2% of TA patients had comorbid depression. This article focused on the clinical features of depression in TA patients and found that the IL-6, prednisone equivalent, ITAS2010 ⩾ 2, and carotidynia might be risk factors for the depression of TA patients. This study also followed up on the treatment and prognosis of TA patients with depression and identified that the improvement of depression was associated with disease remission.
In the comparison of sociodemographic parameters, we found that depressed TA patients were younger and less married. A similar result on age with negative emotion was also seen in a study on quality of life, disability, and mental health in TA patients. 3 This might be related to the fact that young people had to bear more social responsibility and work pressure, while the adverse effects of TA on their work and life aggravated these burdens, resulting in greater mental stress even depression for patients. 26 We also found the importance of being married for depression in TA patients. A similar association could be seen in previous research. A study conducted by Cairney et al. 27 found that single mothers, compared to married mothers, were prone to suffer an episode of depression and higher levels of chronic stress, which could account for differences in exposure to stress and social support.
In the comparison of clinical symptoms, the ratio of carotidynia was much higher in the depression of TA patients than non-depression and was identified as the independent risk factor of depression. Carotidynia was recognized as a typical feature of vascular pain, 28 which was reportedly associated with a higher rate of relapsing and active disease in the previous research. 29–31 In our results, we found the depression group had significantly higher disease activity, and a higher proportion of refractory and active disease, even ITAS2010 ⩾ 2 was shown to be an independent risk factor for depression in TA. Even though the p-value was close to 0.05, we could see the same trend that relapsing disease accounted for a greater proportion of depressed TA patients. Erdal et al. 32 also found, according to ITAS2010, the active TA group had a significantly higher HADS-D score. Although there were currently no studies with similar results in autoimmune disease, refractory and relapsing TA also appeared to have a greater impact on depression, which was due to an individual’s perception of the consequences of the disease. 33 The instability of the disease, which caused the patients to have a negative impact on the disease, is an important reason for this outcome. In addition, the inflammation brought about by the unstable phase of the disease was a predisposing factor for depression.
In the analysis of laboratory indicators, ESR, CRP, C3, and IL-6, the markers of inflammation, were higher in the depression of TA patients. Previous studies have shown that controlling inflammation may provide benefits in the treatment of depression, whether inflammation was secondary to early life trauma, acute stress response, or other factors. 7 Moreover, IL-6 was the independent risk factor for TA patients with depression in our research. Another study we conducted on the cytokines profile of treated TA patients with depression, also found the same trend of IL-6, although the p-value was greater than 0.05, which should be related to the small sample size. 34 Increased release of IL-6 in depression has been found to be a factor associated with the prognosis and therapeutic response and may affect a wide range of depressive symptomatology. 35 Kelly et al. 36 verified the relationship between the IL-6 signaling pathway and depression symptoms. A large body of evidence supports the entry of IL-6 into the central nervous system (CNS) via humoral and neural pathways. 13 The humoral pathways included circulating mediators activating the blood–brain barrier endothelium29,37 and IL-6 entering the CNS through the periventricular organs. 37 So far, IL-6 has been a key target for the treatment of TA. 38 In previous experiments, IL-6 was directly positive throughout the vascular wall in most specimens in inflammatory sites of active TA patients. 39 Due to the important role of IL-6 in TA and depression, future studies should focus more on whether targeting IL-6 would also be beneficial in depressed TA.
The role of medication in depression of TA patients should also be considered. We found TA patients with depression used more glucocorticoids and less MMF. Prednisone equivalents have also been shown to be an independent risk factor for depression in TA. Glucocorticoids have been implicated in the pathogenesis of depression and their side effects can lead to negative emotions. The increase in cortisol in the blood always triggered a negatively regulated cycle that restricted further release of adrenocorticotropin-releasing hormone and adrenocorticotropin. Under chronic stress caused by depression, the negative feedback loop is disrupted, resulting in a persistent increase in glucocorticoids in the blood. 40 The abnormality of hypothalamic–pituitary–adrenal axis regulation and elevated blood cortisol levels has been observed in patients with depression.41,42 In the association of MMF and depression, Mazumder et al. 43 conducted a study and found depression-like behaviors had substantially decreased in the MMF-treated epileptic rats. In previous studies, MMF had shown neuroprotective effects to inhibit the secretion of neurotoxic substances by microglia. 44 Our article provided direct evidence that MMF could be considered more often, and glucocorticoids should be reduced as soon as possible in the treatment of TA patients with depression.
During follow-up, we found that disease remission helped the improvement of depression in TA patients. After treatment, the changes of NIH, ITAS.A, ITAS2010, and HADS-D were significantly decreased. Abularrage et al. 26 also identified the only factor to have a positive impact on the quality of physical and mental life is disease remission. Like previous findings, which confirmed the association between depression in rheumatoid arthritis and less frequency of remission,45,46 our study also suggested that depression in TA should be paid more attention. Although inflammation was proved necessary for depression, there were a lot of mechanisms, such as the neural, physiologic, molecular, and genomic mechanisms, were also relevant for depression. 47 Sigmund Freud wrote in 1917 that “the complex of melancholia behaves like an open wound.” 48 We should devote to lightening both the physical and mental burden of TA patients, which proved to be associated with the disease outcomes. 49
Despite these findings, our study still had a few limitations. First, the study used only one single-center trial with a limited sample size. Additionally, because of the pandemic, the follow-up patients were in a tough medical environment, which led to missing partial data, such as cytokine and immunoglobulin. In the future, we will design perspective studies to get more compelling findings.
Conclusion
In conclusion, this study presented that ITAS2010 ⩾ 2, IL-6, prednisone equivalents, and carotidynia might be independent risk factors for depression in TA patients. During follow-up, the improvement of depression was associated with disease remission. It is necessary to control the disease activity and achieve disease remission in TA to promote the improvement of depression.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X241296414 – Supplemental material for Risk factors and prognosis of depression in Takayasu arteritis patients
Supplemental material, sj-docx-1-tab-10.1177_1759720X241296414 for Risk factors and prognosis of depression in Takayasu arteritis patients by Yaxin Zhang, Anyuyang Fan, Juan Du, Xuemei Shi, Shiyu Yang, Na Gao, Lili Pan and Taotao Li in Therapeutic Advances in Musculoskeletal Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
