Abstract
Bone marrow is a highly cellular tissue undergoing significant developmental and physiologic changes with age. Indeed, with maturation from pediatric to the adult age there is a progressive, centrifugal conversion from red to yellow bone marrow. Histological characteristics of bone marrow are reflected in MR image signal. MR is therefore extremely sensitive in detecting pathological changes which are mostly characterized by increased free water causing high signal intensity on T2. Among the numerous diseases causing bone marrow edema in children chronic nonbacterial osteomyelitis (CNO) certainly has to be mentioned. This idiopathic inflammatory disorder is characterized by nonspecific migrating symptoms like skeletal pain with phases of exacerbations and relapses with alternating acute and chronic MR signs and it is often a diagnosis of exclusion. Hence, with bone marrow edema, various features at imaging should be considered to differentiate malignancies such as osseous lymphoma, osteosarcoma, and Ewing’s sarcoma as well as benign lesions like osteomyelitis, post-traumatic, or post-treatment bone marrow edema. The aim of this review is to recall the main characteristics of CNO and provide an overview of its main mimickers highlighting similarities and differences.
Introduction
Bone marrow is a highly cellular tissue with a predominant hematopoietic function and undergoing developmental and physiologic changes with age.1–3 Indeed, hematopoiesis starts in the fourth fetal month with granulocytic and megakaryocytic proliferation and at birth bone marrow becomes the major site of red cell production. 1 On gross examination, bone marrow is composed of red and yellow elements, containing respectively hemoglobin of erythrocytes with their precursors, and fat. Based on requirements to regulate hematopoietic activity, the space for hematopoiesis or fat increases or decreases consequently. The trabeculae constituting the cancellous bone represent the architectural support for the marrow and act as scaffold for mineral deposition. With maturation from the pediatric to the adult age groups, there is a progressive, centrifugal conversion from red to yellow bone marrow.1,2,4
Histological characteristics of bone marrow are reflected in MR image signal. MR is therefore extremely sensitive in detecting pathological changes which are mostly characterized by increased free water causing high signal intensity on T2.1,2 This, according to its distribution and association with other features in the cortex, periosteum, and surrounding soft tissue, can imply specific diseases not only in adults but also in children, with numerous etiologies including chronic nonbacterial osteomyelitis (CNO). CNO is an idiopathic inflammatory disorder characterized by nonspecific migrating symptoms and phases of exacerbations and relapses5,6 with alternating acute and chronic signs. CNO is often a diagnosis of exclusion both clinically as well as radiologically since it may mimic other malignant and benign conditions like bone tumors, osteomyelitis, and metabolic disorders. 6
Thus, our aim is to shortly recall the main characteristics of CNO and provide an overview of its main malignant and benign mimickers causing bone marrow edema in children, highlighting their similarities and differences.
Chronic nonbacterial osteomyelitis
Nowadays CNO is considered an umbrella term including chronic recurrent multifocal osteomyelitis and Synovitis, Acne, Pustulosis, Hyperostosis and Osteitis (SAPHO) syndrome. Although some researchers consider both acronyms as interchangeable, the term SAPHO should be applied only for patients with cutaneous features, including acne and pustulosis. 7 As previously mentioned, this idiopathic inflammatory disorder is typically characterized by alternating phases of exacerbations and relapses. In the diagnostic work-flow of patients with CNO, especially at onset and when the symptoms are localized in one area, radiographs are utilized for screening and to rule out fractures. 7 According to the phase of the disease, lytic lesions, sclerosis, and hyperostosis are seen. 7 Historically, bone scintigraphy was used to detect non-symptomatic lesions and monitor disease course and activity but to-date it has been replaced by Whole Body MR Imaging (WBMRI).5–8 In fact, this technique avoids radiation exposure and provides accurate information regarding the multifocal behavior and activity of the disease. On MR images, active lesions are characterized by bone marrow edema, periostitis, soft tissue inflammation, transphyseal involvement and in up to 30% of the cases, joint effusion; sclerotic changes of the chronic phase are hypointense on T1- and T2-weighted sequences. 5 In terms of location, it typically affects the metaphyseal area of long bones, especially of the lower extremities. The spine, pelvis, and mandible are often involved; overall lesions can be symmetric (Figure 1).5–8 Especially at diagnosis, unifocal lesions can be identified in the mandible and clavicle, representing a confounding factor and a diagnostic challenge. 6

Whole-body magnetic resonance of a 12-year-old girl with chronic nonbacterial osteomyelitis demonstrating multiple areas of bone marrow edema in the right sacrum (arrowheads in a and b) and in the left acetabulum (arrow in c), the latter being the only site of referred pain.
The protocol for CNO usually includes head-to-toe coronal T1 and short tau inversion recovery or T2 fat-sat. 9 Sagittal images of the spine and/or axial of specific areas might be added if needed. At diagnosis, especially if malignancies have to be ruled out, the inclusion of whole-body diffusion-weighted imaging (DWI) may support the differentiation since inflammatory lesions typical of CNO have high apparent diffusion coefficient (ADC) values. 10
Malignant mimickers
Osseous lymphoma
Osseous involvement in lymphomas may be due to the skeletal spread of a systemic disease or to a primary lesion. Primary pediatric bone lymphoma is a rare type of non-Hodgkin lymphomas representing around 3% of all primary bone tumors and being even less common in children.11–13 Patients usually present with pain without any previous trauma. Swelling, mass, fever, and weight loss are other common symptoms. 12 Given the nonspecific and indolent clinical presentation, a delay in the diagnosis may occur. 11 Around half of the patients have multifocal involvement at diagnosis with metaphyseal and diaphyseal locations been more common than epiphyseal lesions. 11 Femur, tibia, and pelvis are often affected.11,12
Radiographs, that often are the first imaging modality, may show lucent and/or sclerotic areas or permeative lesions, with or without periosteal reaction, and pathologic fractures (Figure 2). However, it cannot be overlooked that X-rays may be negative.11–14 Similar findings can be detected at Computed Tomography (CT). Given the degree of bone marrow infiltration, MR is a very suitable modality for this type of disease demonstrating areas of bone marrow edema with or without soft tissue swelling 13 (Figures 2 and 3). Moreover, the tumor’s high cellularity can be well characterized by DWI while CNO usually has high ADC values (Figure 4).10,15 Recently, Duffy and Ecklund 11 in 8 out of 10 children with primary osseous lymphoma described the occurrence of an inhomogeneous pattern of contrast enhancement resembling osteomyelitis or bone infarcts which may favor a distinction from CNO (Figure 2). During follow-up, this type of pattern should also be distinguished from post-chemotherapy ischemic phenomena. 16
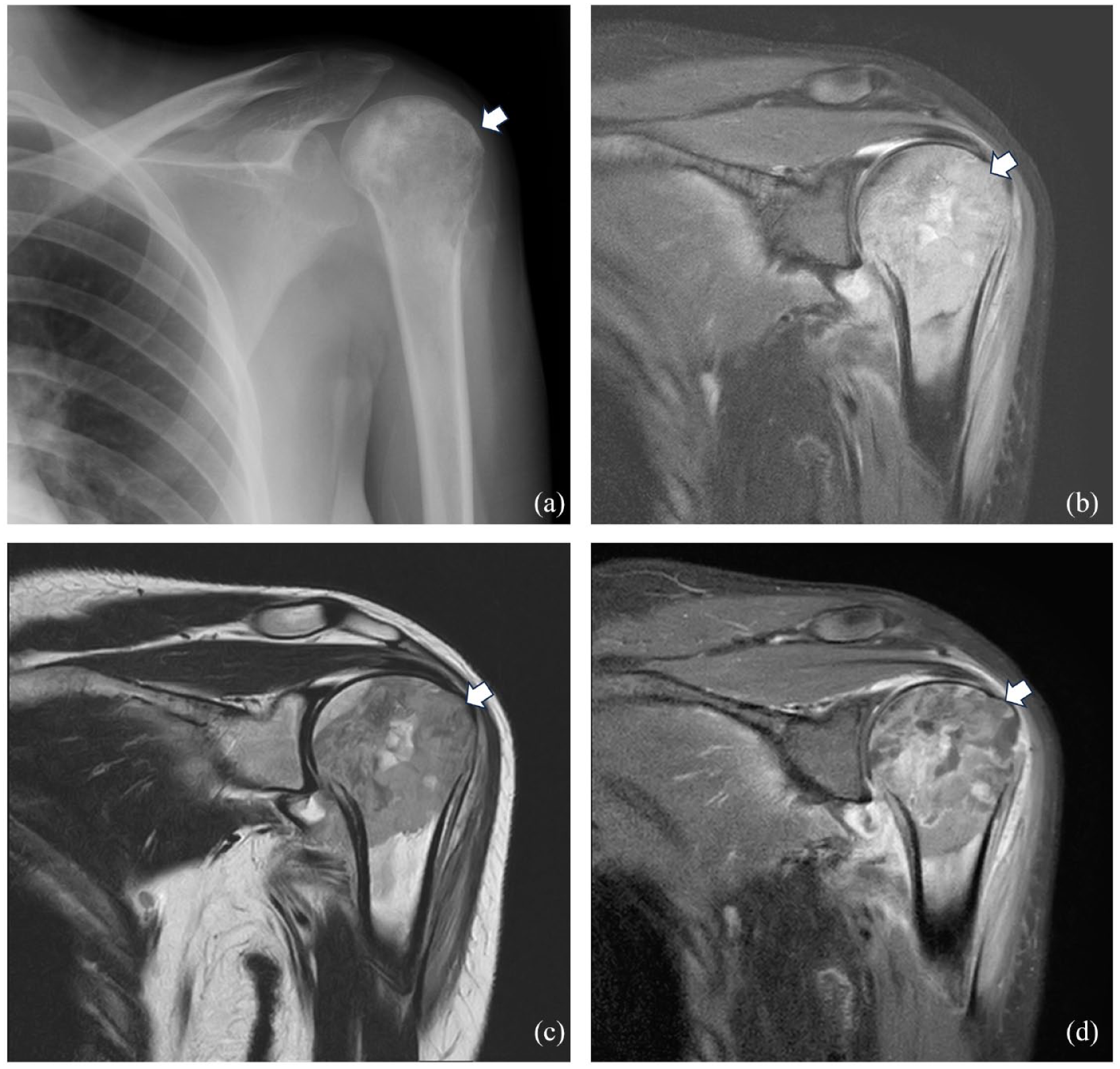
Seventeen-year-old girl with anaplastic large cell lymphoma of the left humerus. The radiograph showed a permeative lesion (white arrow in a) that corresponded to severe bone marrow edema on MR (white arrows pointing the hyperintensity on coronal STIR and the corresponding hypointensity on T1w, respectively, in b and c). After contrast medium injection a inhomogeneous pattern of enhancement is evident (white arrow in d).

Fourteen-year-old boy with diffuse large B-cell lymphoma of the left tibia characterized by metaphyseal and diaphyseal inhomogeneous bone marrow edema (black asterisk) with periosteal reaction on the axial (white arrow in a) and coronal STIR (b); heterogeneous hypointense bone marrow signal is evident on the coronal T1w in c (black asterisk in c).

Twelve-year-old boy with diffuse large B-cell lymphoma of the right mandible characterized by severe bone marrow edema (axial STIR in a) with restricted diffusion (high signal on b800 and low signal on ADC map, respectively, in b and c).
Whole-body MR and hybrid imaging (i.e., [18F]-fluorodeoxyglucose (FDG)-Positron Emission Tomography (PET)/CT and -PET/MR) are especially useful to distinguish between CNO and osseous involvement in systemic lymphoma since they would reveal the presence of nodal or other extra-nodal lesions (Figure 5). In particular, PET/MR carries advantages of allowing a precise characterization of bone marrow given the MR component and provides information about extra-nodal and nodal involvement in FDG-avid lymphomas given the PET part, all with a lower radiation dose than PET/CT. 17 Overall, in case of a single area of bone marrow edema corresponding to a lytic or permeative lesion without sclerosis at radiographs, showing FDG uptake, a primary osseous lymphoma should be ruled out.
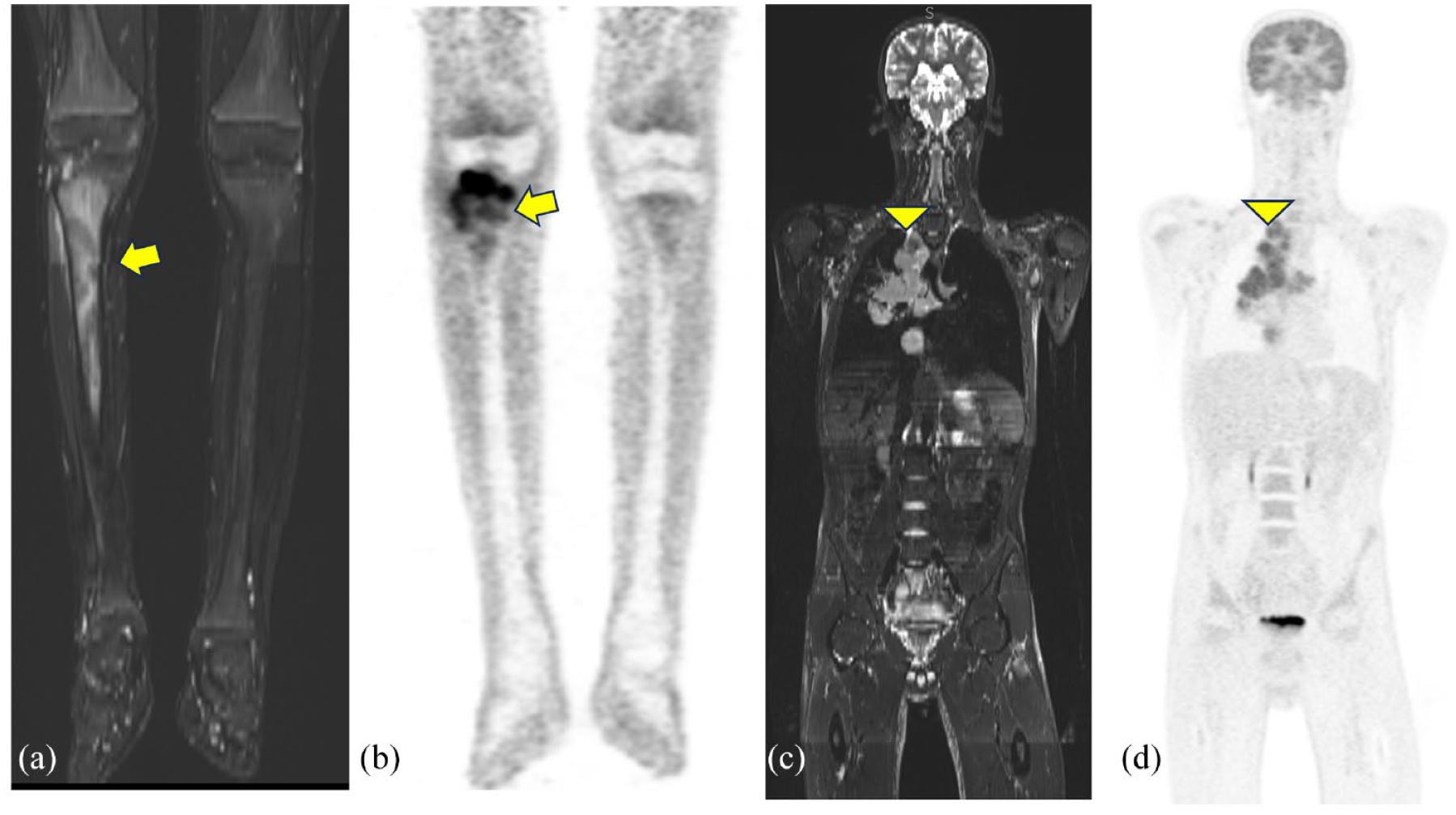
Eleven-year-old boy with anaplastic large cell lymphoma infiltrating the metaphyseal region of the right tibia (bone marrow edema on the coronal STIR in a and high [18F]-FDG uptake on the PET map in b). Nodal involvement is evident in the mediastinum on the coronal STIR in c and coronal PET map in d.
Unlike other more common primary osseous tumors, surgery does not represent typical treatment but chemotherapy, often in association with radiotherapy, is usually applied. 11 The prognosis is good with >90% 5-year overall survival.11,18 Single bone involvement, older age, and large-cell histology seem to be associated with a better prognosis. 12
Leukemia
Leukemia is the most common pediatric malignancy with leukemic cells proliferating in the red marrow. 19 Musculoskeletal symptoms such as bone, joint, and muscular pain as well as limping occur as initial clinical manifestations of the disease in 20%–40% of children with acute leukemia. 20 Similarly to CNO it affects the metaphyseal area of long bones. 21 In radiographs, osteopenia and lucent transverse bands are common but in contrast to CNO these are not associated with areas of sclerosis. On MR despite similar locations to CNO, leukemia can be distinguished as it causes more frequently marrow replacement rather than bone marrow edema (Figure 6). 21 Moreover, as highlighted in a Japanese case series, diffuse bone marrow low T1 signal intensity in children older than 1 year, even without blood abnormalities, may be very suggestive of acute leukemia. 20

Diffuse hyperintense signal on the sagittal STIR (white asterisks in a) and hypointense signal on the sagittal T1 (white asterisks in b) images reflecting the bone marrow replacement in a 13-year-old boy affected by leukemia.
Primary bone tumors (osteosarcoma and Ewing’s sarcoma) and skeletal metastases
Bone sarcomas are rare, accounting for around 6% of all malignant tumors in children. 22 Osteosarcoma is the most common bone sarcoma followed by Ewing’s sarcomas. 23 Like CNO, the former has a predilection for the metaphysis of long bones (Figure 7), while Ewing’s sarcomas often affect the pelvis and the diaphysis of long bones of the lower extremities. 23 These malignancies usually have an aggressive pattern at imaging with sunburst periosteal reaction, cortical breakthrough, and soft tissue mass (Figure 8). 21 In CNO, the periosteal reaction is commonly solid or single-layered and rarely the onion-skin pattern can be seen, especially in the clavicle; soft tissue involvement is also uncommon. 21 Even if the distinction between these aggressive, malignant tumors and CNO is usually straightforward, in some patients it might be more challenging, and a biopsy is required for accurate diagnosis. 24 In line with this evidence, Nepal et al. 24 described the case of a 11-year-old patient affected by a lesion in the right iliac bone with radiological features resembling an aggressive tumor which was then benign at histology.

Histologically proven osteosarcoma of the metaphyseal and epiphyseal region of the right femur in a 16-year-old girl, characterized by severe bone marrow edema (black asterisk in a) and highly hypointense signal on T1 (white asterisk in b).
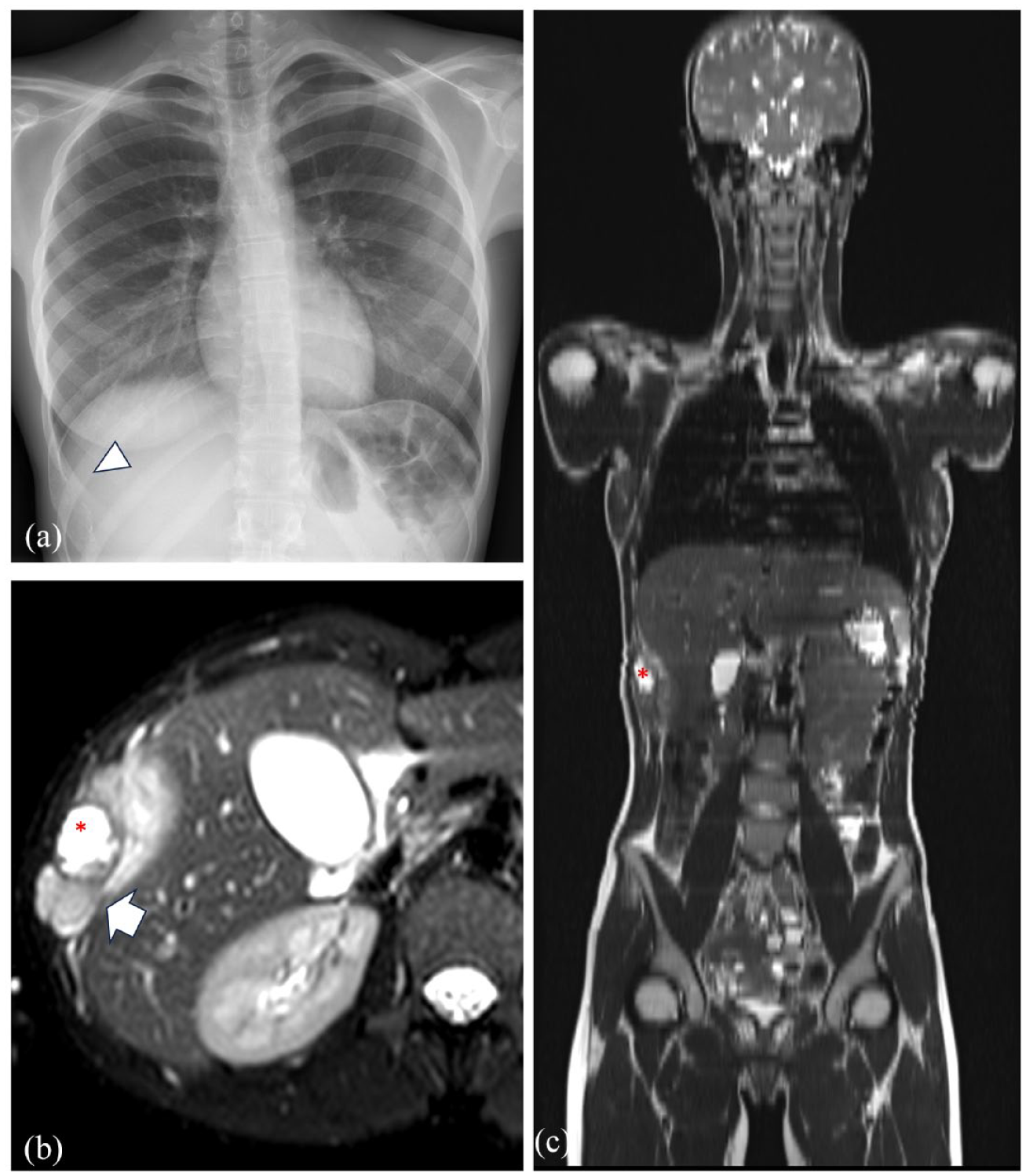
Single lytic lesion of the ninth right rib (white arrowhead on the radiograph in a) corresponding to the lesion hyperintense in T2 (red asterisk in b and c) with a soft tissue mass (white arrow in b) and a wide transition zone (axial T2fs in b and coronal Half-Fourier Acquisition Single-shot Turbo spin Echo (HASTE) in c) due to Ewing sarcoma in a 17-year-old girl.
In case of multiple lesions, skip lesions due to osteosarcoma and osseous metastases should be excluded.
As highlighted by a recent systematic review on the role of WBMRI for skeletal metastases, Ewing’s sarcomas/Primitive Neuro-Ectodermal Tumors (PNET), neuroblastomas, and rhabdomyosarcoma are among the most frequent tumors metastasizing in bones (Figure 9). 25 Bone metastases may cause cortical destruction, and this favors the distinction from CNO at imaging.

Thirteen-year-old boy with Ewing sarcoma of the left iliac bone (white arrow in a) and skeletal metastases in the left humerus (axial STIR in b) and in spine (sagittal STIR in c).
Langerhans cell histiocytosis
Langerhans cell histiocytosis (LCH) is a rare disease characterized by high proliferation and deposit of Langerhans cells in various organs (e.g., skin, lung, liver, and bone marrow). 26 Currently, it is classified as a single-system, with single or multiple sites, or a multi-system disease according to the extent of involvement at diagnosis.27–29
In children, the skeleton is the most affected system (ca 75%–80%) being skull, ribs, and pelvis involved in more than 50% of patients (Figure 10). A single lesion is more frequent than multiple localizations. 30 Although the diagnosis is based on histology, imaging plays a significant role and to-date the skeletal survey is still recommended. In fact, LCH usually causes lytic lesions with or without periosteal reaction that can be identified at radiographs. 30 Nevertheless, the radiation exposure and the fact that lytic lesions become detectable on radiographs only when 30%–50% of bone mineral density is already lost should not be overlooked. Therefore, the use of MR should be further promoted.27,30 As for osseous lymphoma and other primary skeletal tumors, restricted DWI and low ADC values support the diagnosis (Figure 9).10,31 In general, LCH lesions are lytic and round or oval-shaped with blurry margins corresponding to areas of edema on MR. 30 Periosteal reaction can be present, and sclerosis may occur during the healing phase, but it is usually limited. In the spine, LCH, similarly to CNO has a predilection for the thoracic vertebrae where both may cause vertebral collapse with a vertebra plana appearance or involvement of the posterior elements.30,32,33
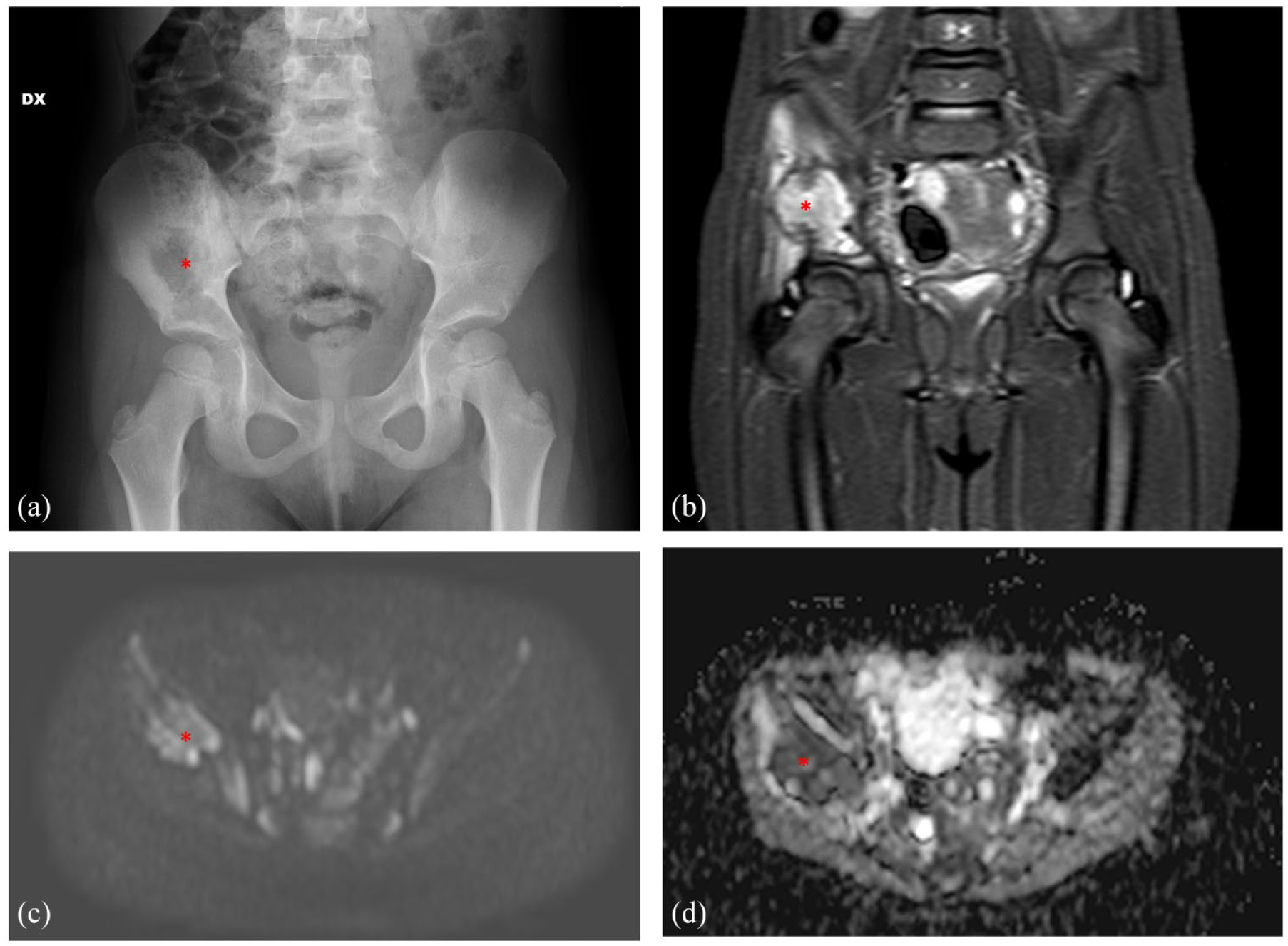
Five-year-old girl with histologically proven Langerhans cell histiocytosis of the right iliac bone. The lytic lesion on radiographs (red asterisk in a) corresponded to a hyperintense lesion on coronal STIR (red asterisk in b) and restricted diffusion (red asterisks in b800 in c and ADC map in d).
In long bones, LCH may affect the diaphysis and the metaphysis, while CNO has a predilection for the metaphyseal area.30,32 Moreover, in LCH, the lesions usually do not cross the growth plate whereas in CNO lytic metaphyseal destruction about the growth plate.30,32
If in patients with a single lesion in the skull or in children with multi-system disease, the differentiation from CNO is easier, in case of single-system disease affecting the bone in multiple sites, imaging might not be decisive.
Regarding the treatment of skeletal LCH, spontaneous resolution has been described, but usually single lesions are treated with curettage and/or corticosteroid injection and in case of vertebral foci without collapse, radiotherapy can be considered.34,35 In these patients, the risk of recurrence is around 10%. Multiple bone lesions with or without involvement of other systems, usually require chemotherapy. 35
Benign mimickers
Infectious osteomyelitis
Osteomyelitis is an infection of the bone involving the medullary canal due to a contiguous spread of soft tissue infection (e.g., penetrating injury, puncture, post-surgical wounds) or to a hematogenous spread.36,37 The most common organisms causing hematogenous osteomyelitis in children include Staphylococcus aureus, Streptococcus pyogenes, and pneumonia; Kingella kingae is becoming frequent in children under 3 years of age while infections due to Haemophilus influenzae are rare in countries with high immunization rates. 38 In pediatric patients with comorbidities such as sickle cell disease, Salmonella and Escherichia coli are common infectious agents.36,38
MR is considered the gold standard in case of osteomyelitis because, in contrast to ultrasound which provide only an assessment of soft tissue involvement, may reveal bone marrow edema which is the main feature and is already detectable 1–2 days after the onset of the infection. 39
Given the abundance of vessels in the metaphysis, children are predisposed to an hematogenous spread causing metaphyseal osteomyelitis. This localization, especially in the early phases, may mimic CNO. The Brodie’s abscess, which is a subacute form of osteomyelitis, has also a predilection for the metaphyseal region of long bones (especially femur and tibia) (Figure 11), but the typical fluid collection with peripheral contrast enhancement, the target appearance with four layers (i.e., central abscess cavity, one inner ring due to granulation, one outer ring due to reactive sclerosis, and a peripheral halo due to bone marrow edema), and the “penumbra sign” (transition zone hyperintense in T1 between the abscess and the sclerosis) enable the distinction from CNO.36,40 Overall, additional findings that can support the differentiation of osteomyelitis from CNO include cellulitis, adjacent soft tissue ulcers, soft tissue abscess, phlegmon, sinus tracts, and cortical bone destruction (Figure 12). 41 In infants, vessel traversing the physis may cause an epiphyseal and even an intra-articular spread of the infection. 36

Brodie’s abscess in the left tibia of a 14-year-old girl. The multiple layers typical of this subacute form of osteomyelitis can be seen on the sagittal STIR and T1w images (white arrows in a and b, respectively).
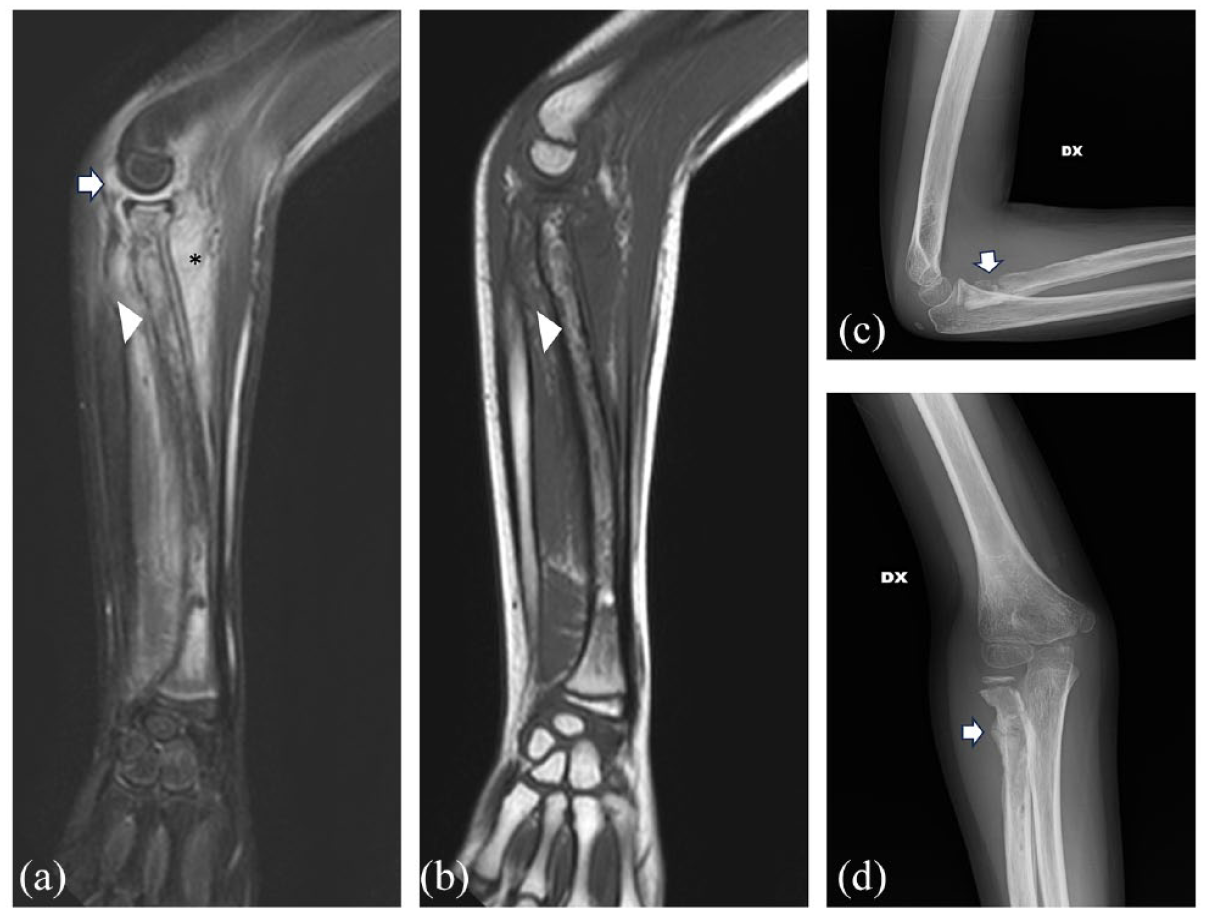
Osteomyelitis of the right forearm due to Staphylococcus aureus in a 10-year-old boy well demonstrated by the extensive bone marrow edema in the radius (arrowhead in a and b), soft tissue involvement (black asterisk in a), and joint effusion (arrow in a). The confluent bone marrow pattern on T1w contributes to the diagnosis (b). The corresponding radiographs in c and d nicely demonstrate the cortical destruction (white arrows in c and d).
Not only the hyperintense signal on T2-weighted sequence due to bone marrow edema plays a crucial role for the diagnosis of osteomyelitis but also the characteristics of T1-weighted features can contribute. For instance, Collins et al. 42 showed that T1 marrow replacement with a confluent pattern and a medullary distribution was perfectly matching with pedal osteomyelitis. Similar features were found by Howe et al. 43 also in non-pedal osteomyelitis (Figure 11).
The typical involvement of a single bone contributes to the distinction from CNO. Nevertheless, the differentiation might be more challenging in patients with multiple foci. In particular, children with Panton-Valentine Leucocidin (a cytotoxin that causes leukocyte destruction and tissue necrosis) positive Staphylococcal aureus infections are more prone to a multifocal spread. Similarly, osteomyelitis due to tuberculosis may cause multiple lytic lesions (aka cystic tuberculosis).44,45
COVID-19
While at the beginning of the Coronavirus Disease 2019 (COVID-19) pandemic, all attention was focused on the respiratory disease caused by Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-COV2), as time passed by, it became clear the systemic action of this infection with involvement also of the musculoskeletal system even beyond the spectrum of the Multysistem Inflammatory syndrome in children (MIS-c) syndrome or Pediatric Multisystem Inflammatory Syndrome (PMIS). 46 While in adults the occurrence of bone marrow edema due to SARS-COV2 has been often reported, in children, only recently, a study described the presence of the so-called phalangeal microgeodic syndrome.46,47 Indeed, in this case series including five children with phalangeal swelling and erythema (four with involvement of the hand and one of the feet), the MR revealed bone marrow edema primarily affecting the metaphysis and the epiphysis (Figure 13). 47 Among other signs, in two children, erosions were also identified. 47 Such a peripheral location and the concomitant or previous COVID-19 infection enable the distinction of this vascular phenomena from CNO.
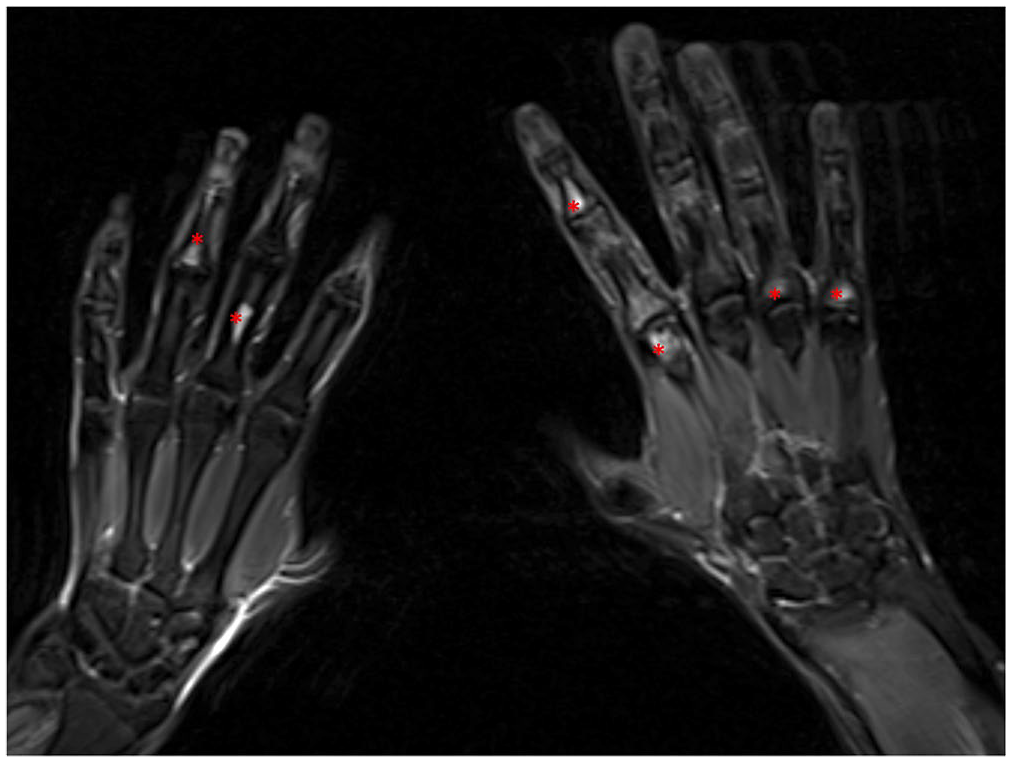
Multiple areas of bone marrow edema affecting the metaphysis and the epiphysis of both hands of a 16-year-old boy with persistent pain in the hands, after COVID-19 infection, expression of the so-called phalangeal microgeodic syndrome (red asterisks).
Vitamin deficiency
Vitamin deficiency may cause areas of bone marrow edema in children and adults. A retrospective study on 13 children with persistent foot pain showed that environmental factors associated with vitamin D deficiency can be responsible of the clinical symptoms and the so-called bone marrow edema syndrome of the foot and ankle. 48 Therefore, in case of multiple areas of bone marrow edema affecting these areas, vitamin D levels should be measured.
Scurvy is caused by low vitamin C levels (clinical symptoms occur with vitamin C values <2 mg/dl and total body values <300 mg) and, although unusual in high-income countries, it may occur in children with restricted diets such as those with neurodevelopmental disorders or physical disabilities.38,39,49 In case of malnutrition, simultaneous vitamin C and D deficiency may occur.
Children with scurvy may have heterogeneous symptoms including musculoskeletal, dermatological, dental, and systemic manifestations. 38 Even if the diagnosis of scurvy is based on clinical and laboratory findings, especially in case of pain in the extremities, imaging is performed and may significantly contribute to the diagnostic process. Being radiographs often the first level imaging technique, radiographic signs of scurvy have been broadly described in the literature. Typically, osteopenia with cortical thinning, periosteal reaction due to hemorrhage, the Frankel’s line (i.e., widening of the zone of provisional calcification), the Trümmerfeld’s zone (i.e., lucency on the diaphyseal side of Frankel’s line due to unorganized osteoid), the Pelkan spur (i.e., healing metaphyseal fractures), and the Wimberger ring (i.e., a circular calcific, sclerotic rim surrounding a lucent, osteoporotic epiphyseal center of ossification) can be seen. 50
Nevertheless, it should not be overlooked that the typical radiographic findings become evident after 3–6 months of vitamin deficiency; therefore in the early phases, MR could be more sensitive. Only a few studies described MR findings of scurvy. Multiple, bilateral metaphyseal areas of bone marrow edema with periosteal reaction and adjacent soft tissue edema especially in the lower limbs have been reported38,50,51 (Figure 14). Recent case reports using whole-body MR showed the occurrence of multiple lesions also in the pelvis and in the upper extremities.52,53 Given this MR pattern very similar to CNO, laboratory analyses are crucial to differentiate these two diseases.

Bone marrow edema of the metaphyseal region of the femur and tibia due to vitamin C deficiency in a 15-year-old boy with dumb pain for months (white arrows).
Osteoid osteoma
Osteoid osteoma is a benign neoplasm accounting for around 10%–12% of all benign tumors.54,55 It usually occurs in adolescents and young adults and is characterized by deep and intense pain worse at night responsive to non-steroid anti-inflammatory drugs.54,55 It is characterized by a nidus (the core) which represents the tumor itself and surrounded by a cortico-periosteal thickening.54,55
According to the locations, osteoid osteoma can be classified as cortical (around 75% of the cases), medullary (20%), and subperiosteal (5%). 54 The typical cortical lesion appears as an intracortical lucency surrounded by sclerosis on radiograph. CT is the modality of choice, nicely showing the nidus even when not depictable on radiographs, 55 and it is usually applied as guidance for percutaneous radiofrequency which is the recommended treatment 56 (Figure 15). On MR, the nidus may have variable signal intensity, being often moderately hyperintense on T2. Surrounding inflammatory changes may obscure the nidus, but typically the peripheral component is hypointense on T1- and T2-weighted images.54,55

Thirteen-year-old boy with an osteoid osteoma of the right hip. Coronal STIR (a) and T1 turbo spin echo (b) MR sequences demonstrate cortical exuberant hyperostosis and sclerosis (white arrows in a and b). The sagittal STIR nicely shows the hyperintense nidus (white arrowhead in c). Treatment with CT-guided LASER thermoablation targeting the nidus (image in d acquired during the procedure).
Osteoid osteoma in its typical appearance can be easily distinguished by CNO being a single cortical lesion with distinctive symptoms.
Juvenile idiopathic arthritis
According to the International League of Associations for Rheumatology, juvenile idiopathic arthritis (JIA) is classified in seven types and enthesitis-related arthritis (ERA) mainly represents juvenile spondyloarthritis (jSpA).57,58 ERA accounts for around 5%–10% of JIA.58,59
Patients satisfy the diagnostic criteria if affected by arthritis and enthesitis or if they have one of the two symptoms plus at least two of the following: sacroiliac joint tenderness and/or inflammatory lumbosacral pain, test positive for Human Leukocyte Antigen (HLA)-B27 antigen, are boy over 6 years of age, are affected by acute, symptomatic, anterior uveitis, have a history of ankylosing spondylitis, ERA, sacroiliitis with inflammatory bowel disease, reactive arthritis, or acute anterior uveitis in a first-degree relative.57–59 Overall, signs and symptoms of jSpA are different than in adults with a less prominent involvement of the sacroiliac and vertebral joints. 58 The main feature is represented by enthesitis, especially at the distal attachment of the Achilles and quadriceps tendons which is not encountered in CNO. 59
In case of sacroiliitis due to jSpA, the association of bone marrow edema with other findings such as capsulitis, synovitis/joint space inflammation, enthesitis, and erosions favors the distinction from bone marrow edema due to CNO (Figure 16). 60

Eleven-year-old boy, HLA-B27 positive, with inflammatory lumbosacral pain then diagnosed with juvenile spondyloarthritis. The axial STIR in a and b nicely show the bone marrow edema in the left sacroiliac joint (white arrows in a) and the pubic enthesitis (white arrowheads in b). The axial T1 out of phase Dixon demonstrates the presence of erosions (red asterisks in c) then confirmed at CT (red asterisks in d).
Focal periphyseal edema
In adolescents, focal areas of periphyseal bone marrow edema due to the physiologic physeal closure, the so-called focal periphyseal edema (FOPE) may occur. This self-limiting entity can be associated with pain and given its higher prevalence in children doing athletic activities, additional stress due to intense sport probably favor its occurrence.61,62 FOPE commonly affects the knee, in particular the femur, has a central location, and rarely it is multifocal. 61 Radiographs are usually unremarkable. The presentation, typical location, self-resolution, and a single location support its differentiation from CNO.
Bone marrow reconversion
Bone marrow composition changes throughout the course of life. Indeed, at birth the hematopoietic marrow is predominant in the whole skeleton. Then there is a progressive, centripetal transition to yellow bone marrow reaching a mature state in the late third decade.1,2 Conditions causing an increased hematopoiesis, such as chronic anemias, sickle cell diseases, treatment with granulocyte colony-stimulating factors (see also section “Post-traumatic bone marrow edema” below), stress, endurance sports, and obesity induce a reconversion of yellow to red marrow (Figure 17). On T2 fat sat images, the red marrow is slightly hyperintense to muscles and on T1w the signal is diffusely hypointense. Bone marrow reconversion, in contrast to CNO, is usually symmetric and rarely crosses the growth plate, sparing the epiphysis. 63

Diffuse reconversion of yellow to red marrow due to increased hematopoiesis in a 10-year-old boy with sickle cells disease (coronal T1w and coronal STIR in a and b, respectively).
Post-traumatic bone marrow edema
In children, injuries to the extremities often involve the physis. In contrast to CNO, Salter–Harris fractures affect a single site, and especially in case of lesions of type II–IV, a fracture line extending to the metaphyseal and/or epiphyseal region is visible on MR. 64 Certainly, the clinical history, reporting a recent trauma, is essential to guide toward the correct diagnosis mostly in case of lesions of type I and V. 21
Traumatic events or prolonged immobilization may also cause the so-called complex regional pain syndrome (CRPS; aka as reflex sympathetic dystrophy or Sudeck atrophy). Patients refer edema, hyperalgesia, altered skin color, and temperature. 65 The MR findings are quite unspecific including skin enhancement, muscle edema, and bone marrow edema (Figure 18). Especially the latter may mimic CNO. As for the above-mentioned fractures, the clinical history, the single location, as well as the predilection for the feet are supportive of CRPS rather than CNO.
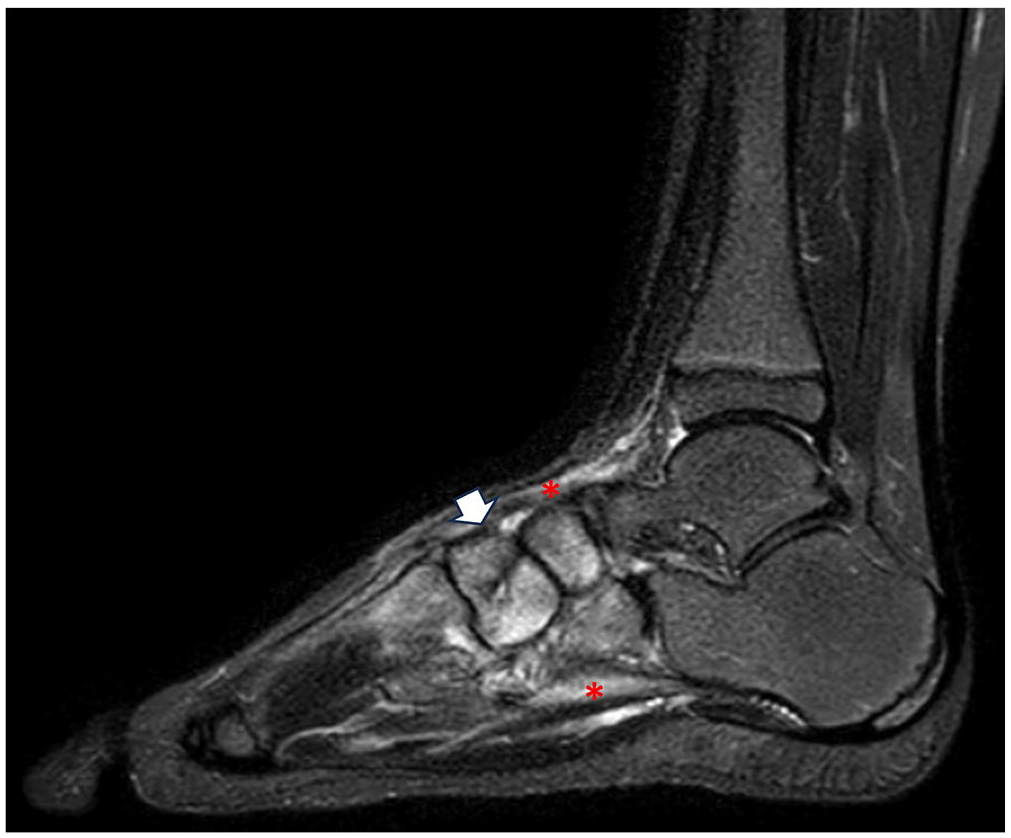
Bone marrow edema (white arrow) and soft tissue edema (red asterisks) in the midfoot of a 10-year-old 2 months after a sprain.
Post-treatment bone marrow edema
MR is often applied to assess the effects of treatment in patients with cancer or chronic inflammatory/autoimmune diseases and some therapies may induce changes and or complications in the skeletal system that should not be misinterpreted. 1 While methotrexate, a folate antagonist commonly applied for cancer and for inflammatory diseases, such as rheumatoid arthritis, may cause stress fractures which should be easily distinguished from CNO, the effects of other drugs on bones may mimic other diseases.66,67
For example, the cellular and vascular changes caused by chemotherapy for leukemia cause edema during the first week of treatment, especially in case of acute lymphoid leukemia rather than acute myeloid leukemia probably because of the higher bone marrow toxicity of some drugs used for the former. 68
Recently, it has been shown that treatment with immune checkpoint inhibitor may induce inflammatory arthritis. In a case series, three out of eight patients with this adverse event had also bone marrow edema at MR. 69 Another study including six patients, reported for instance extensive bone marrow edema in the shoulder of a 59-year-old man with stage IV melanoma after 8 months since the beginning of treatment with ipilimumab and nivolumab. 70 To rule out potential metastatic spread, the patient underwent also two biopsies which revealed a mixed inflammatory infiltrate. 70
After bone marrow cell transplantation, high T2-weighted bone marrow areas occur and within 3 months a typical “band pattern,” consisting of peripheral T1-hypointense zone (corresponding to repopulating hematopoietic marrow at histology) and a central T1-hyperintense zone (corresponding to marrow fat), occurs in the spine.68,71,72
In case of radiotherapy, the changes in the bone marrow depend on the time and dose of irradiation. 68 Bone marrow edema is very common in the early phase after treatment (1–3 days) and in the following days, there might be areas of hemorrhage (hyperintense on T1- and T2-weighted images). 68 Typically, as time passes by, there is a progressive fatty reconversion, but in some cases bone marrow edema may persist even months after irradiation (Figure 19). 68 Similar changes, although less severe, may occur also outside the irradiation field causing a multifocal distribution.

Ewing’s sarcoma of the left fibula in a 7-year-old boy (white arrows coronal and axial STIR in a and b). After radiotherapy, a complete resolution of the extraosseous component of the tumor is evident. Diffuse, patchy bone marrow edema on STIR, corresponding to hypointense areas on T1, due to treatment occurred in the fibula (red asterisks c and d) and tibia (red asterisks e and f).
As previously mentioned (see section “Bone marrow reconversion”), the therapy with granulocyte colony-stimulating factor may cause red marrow hyperplasia and induce patchy or diffuse, usually symmetric, marrow changes on fluid-sensitive MR sequences.21,73
Moreover, as shortly described above (see section “Osseous lymphoma”), areas of bone marrow edema especially if associated with serpiginous low-intensity areas in oncological children under treatment (i.e., steroid, chemotherapy, and/or radiotherapy) should suggest ischemic phenomena (Figure 20).17,73

Multiple serpiginous ischemic areas (white arrows) in the pelvis after chemotherapy in a 17-year-old girl treated for mediastinal Hodgkin’s lymphoma.
The clinical history as well as the timing and duration of treatment are essential to correctly distinguish CNO from all described post-treatment changes.
Conclusion
In conclusion, numerous diseases, inclusive of malignant and benign conditions, may cause bone marrow edema and mimic CNO. Certainly, the use of WBMRI and a detailed knowledge of typical signs and sites as well as overall characteristics of bone marrow edema in addition to the patients’ clinical history are crucial to reach the correct diagnosis.
