Abstract
Background:
Early diagnosis of primary Sjögren’s syndrome (pSS) remains difficult due to its insidious onset.
Objectives:
To identify whether meibomian gland dropout (MGD) is a sensitive and noninvasive predictor of pSS by studying its association with histopathology in labial salivary gland biopsy in patients with clinically suspected pSS.
Design:
Prospective, randomized, multicenter, comparative effectiveness study.
Methods:
The study was conducted from July 2022 to July 2023. In all, 56 eligible participants with clinically suspected pSS were recruited from three combined ophthalmology medicine/rheumatology SS clinics. All participants with suspected pSS were evaluated and diagnosed by ophthalmology and rheumatology consultants and underwent infrared imaging of the meibomian glands using Keratograph 5M and histopathological evaluation of labial salivary gland biopsies. The length, width, and tortuosity of the meibomian glands were measured; the dropout rate in the nasal, temporal, and total eyelids was analyzed; and the dropout score was calculated using meibography grading scales.
Results:
Among the 56 participants, 34 were identified with pSS, and 22 were diagnosed with non-SS dry eye (NSSDE) and served as the control group. We recorded significant differences in the temporal and total MGD rates of the upper eyelids between the pSS and NSSDE groups (all p < 0.01). Improved prediction accuracy was achieved with the temporal and total MGD rates in the upper eyelids, with area under the curve values of 0.94 and 0.91, and optimal cutoff points of 0.78 and 0.75, respectively.
Conclusion:
MGD in the upper eyelids, especially in the temporal portion, is strongly associated with the histopathological outcome of labial salivary gland biopsy in pSS and is proposed as a highly predictive and noninvasive biomarker for the early diagnosis of pSS.
Trial registration:
ClinicalTrials.gov identifier: ChiCTR2000038911.
Introduction
Sjögren’s syndrome (SS) is a chronic systemic autoimmune disorder characterized by lymphocyte infiltration and destruction of the exocrine glands, accompanied by signs and symptoms of dry mouth and keratoconjunctivitis sicca.1,2 SS is the second most prevalent systemic autoimmune disease after rheumatoid arthritis (RA) and affects 0.3%–3% of the general population. Women are more prone to experience SS than men, with a sex ratio of approximately 9:1. 3 The syndrome occurs in two forms: primary SS (pSS) and secondary SS. The former occurs alone, while the latter often occurs in combination with other chronic inflammatory autoimmune diseases, such as RA or systemic lupus erythematosus. The onset of pSS remains frequently insidious, presenting with an accumulation of different subtle changes in the exocrine glands for an average of 7 years prior to receiving a diagnosis. 4
The diagnosis of pSS requires objective symptoms of dryness, including the characteristic features of minor salivary gland biopsy in combination with subsequent histopathological evaluation and/or specific serum autoantibody. According to the 2016 American College of Rheumatology (ACR)/European League Against Rheumatism (EULAR) Classification Criteria for pSS, 5 the final classification criteria are based on the weighted sum of five items: anti-SSA or Ro antibody positivity and focal lymphocytic sialadenitis with a focus score (FS) of ⩾1 foci/4 mm2, each scoring 3; an abnormal ocular staining score of ⩾5 (or van Bijsterveld score of ⩾4), a Schirmer’s test result of ⩽5 mm/5 min, and an unstimulated salivary flow rate of ⩽0.1 ml/min, each scoring 1. Individuals with signs and/or symptoms suggestive of SS who have a total score of ⩾4 for the above items meet the criteria for pSS. FS, defined as a set of 50 lymphocytic cells per 4 mm2, is regarded as the “gold standard” in diagnosing pSS due to its high specificity. Testing is necessary for patients with suspected SS, especially for those with negative anti-SSA and/or anti-SSB antibodies. However, salivary gland biopsy is an invasive procedure with several shortcomings, including pain, anxiety, hematoma, bleeding, and granuloma. 6
Increasing evidence demonstrates that progressive meibomian gland dysfunction is significantly more common in pSS than in non-SS dry eye (NSSDE).7,8 The meibomian glands are modified sebaceous glands embedded in the tarsal plate that stabilize the tear film and slow the evaporation rate. These glands comprise long central ducts surrounded by secretory acini units, with orifices terminating posterior to the mucocutaneous junction of the eyelid. Tracking and assessing the loss or truncation of these structures are presumed to reflect the degree of meibomian gland dropout (MGD) or atrophy.
MGD may play an important role in patients with pSS but is currently not included as a parameter in the classification criteria for pSS. With the advancement in the imaging technology of meibography, a method of mapping the morphology of meibomian glands, the glands can appear as an array of “string-like” structures. The OCULUS Keratograph 5M (Oculus, Wetzlar, Germany) is used as a “true” meibography device, with the advantages of being noninvasive, fast, and generating reproducible features for assessing meibomian gland function. 9 It uses both white light and infrared (IR) illumination to detect the architecture of meibomian glands, producing post-processed images that highlight the structure.
Since MGD may contribute to pSS and can be detected noninvasively in clinical practice, the correlation between MGD and pSS may be of great importance for quick and accurate prediction or diagnosis of pSS, besides the “gold standard” FS in labial salivary gland biopsy. This study aimed to explore the correlation between MGD, evaluated using Keratograph 5M, and FS in the pathohistology of labial salivary gland biopsies in pSS. In particular, it would be highly meaningful to investigate the reliability of estimating the scores of MGD in patients negative for extractable nuclear antigens to determine whether this parameter could help stratify patients with pSS and serve as a biomarker to diagnose pSS at its initial stage.
Methods
Study population
This was a prospective, multicenter, comparative effectiveness study, that included 56 participants with clinically suspected pSS at the combined ophthalmology medicine/rheumatology SS clinic in three hospitals: Tongren Hospital affiliated to Shanghai Jiao Tong University School of Medicine, Guanghua Hospital of Integrated Traditional Chinese and Western Medicine affiliated with Shanghai University of Traditional Chinese Medicine, and Wuhu Eye Hospital. The reporting of this study conforms to the Updated List of Essential Items for Reporting Diagnostic Accuracy Studies. 10
Patients with a history of dry eye, recurrent parotidomegaly, or extra glandular symptoms were enrolled between July 2022 and July 2023. Written informed consent was obtained for the use of participant medical data for clinical research purposes. All patients with suspected pSS were assessed and diagnosed by ophthalmology and rheumatology consultants. The patients were diagnosed with pSS if they met the ACR/EULAR diagnostic criteria. 5 Patients who had symptoms of dryness but did not fulfill the diagnostic criteria for pSS were classified as NSSDE and served as the control group. Data from the right eye were selected for the statistical analyses. All patients underwent evaluation for MGD and labial salivary gland biopsies. 11
The exclusion criteria were as follows: (a) eyelid abnormalities or inflammation, (b) history of ocular surgery or trauma that may affect ocular morphology, (c) contact lenses worn within 2 weeks, (d) history of head and neck radiation treatment, (e) active hepatitis C injection, (f) acquired immunodeficiency syndrome, (g) sarcoidosis, (h) amyloidosis, (i) graft-versus-host disease, (j) IgG4-related disease, and (k) history of other systemic diseases.
Labial salivary gland biopsy and histopathological study
A standard procedure was followed to excise six to eight glands from the lower labial mucosa under topical anesthesia. The labial salivary glands were immersed in 4% formalin and embedded in paraffin. The sections were stained with hematoxylin–eosin (H&E). Histological evaluation was performed independently by two pathologists who were blinded to the results of other examinations, including ocular investigations and blood tests. A positive outcome was considered for FS ⩾ 1 (Figure 1(a) and (b)).
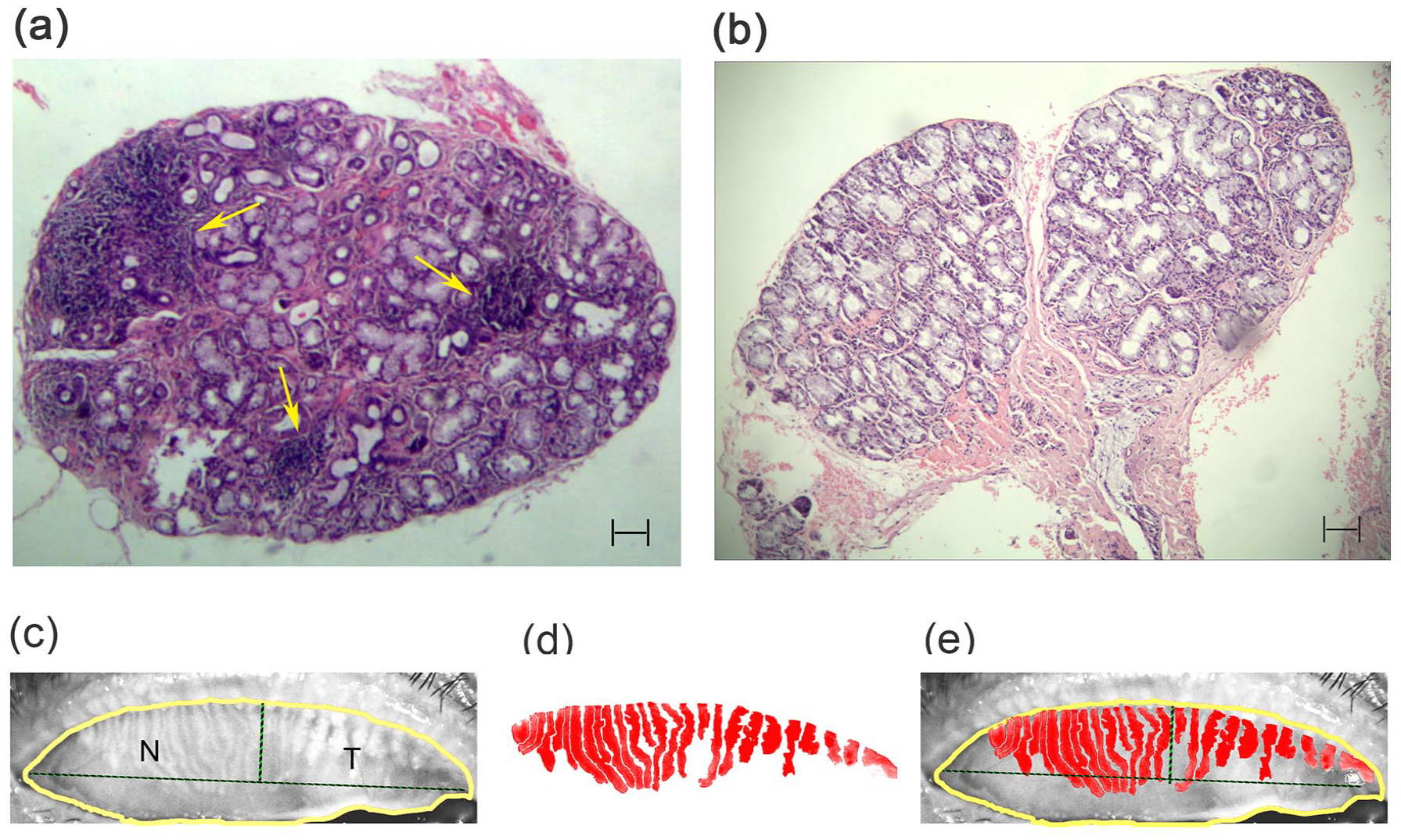
Histopathological view of the chronic inflammation in labial glands, and the meibography with severe MGD of the upper eyelids. (a) Labial glands stained by H&E with variously sized lymphocytic foci of mononuclear inflammatory cells. The entire specimen had a FS >1/4 mm2. The yellow arrow represents lymphocytic foci. (b) Original image of labial glands stained by H&E. (c) Original image of the upper meibomian glands using Keratograph 5M; (d) skeletonized image of the upper meibomian glands showing meibomian glands loss; (e) overlay of the original and skeletonized image of the meibomian gland. The yellow line indicates the total outline of the upper eyelid plate. The dotted line indicates the nasal (N) and temporal (T) outlines of meibography images from the upper eyelids margin to the lateral angle of the eye. Bar equals 100 μm.
Dry eye evaluation
The subjective severity of dry eye symptoms was scored using a questionnaire, the Ocular Surface Disease Index (OSDI), completed by the participants, 12 and ranged from 0 to 100. The Schirmer I test (SIT) was performed without anesthesia. A small strip (Tianjin Jingming, China) was inserted at the junction of the lateral and middle lower eyelids, and the moistened length of the strip was measured after 5 min. Tear meniscus height (TMH), noninvasive keratographic first and average break-up times (noninvasive first break-up time (f-NIKBUT) and noninvasive average break-up time (av-NIKBUT), respectively), and meibomian gland architecture were sequentially measured for each participant using Keratograph 5M.
Meibomian gland evaluation
The meibomian glands in the upper and lower eyelids were detected using IR images, which were captured using the Oculus TF-Scan module of Keratograph 5 M. The meibography images were analyzed using the ImageJ software (1.46r; Rawak Software Inc., Stuttgart, Germany). To analyze the nasal (N) and temporal (T) MGD, the meibography images were divided into N and T regions from the angulus oculi medialis to the lateral angle of the upper eyelids, and the corresponding dropout rates for the N, T, sum of N and T, and total regions were calculated (Figure 1(c)–(e)). The numbers of tortuous meibomian glands were measured, with a tortuous gland being defined as having at least one angle greater than 45°. 13 The dropout score ranged from 0 to 3 as follows: 0, no meibomian glands loss; 1, ⩽33% meibomian glands loss; 2, 33%–67% meibomian glands loss; and 3, >67% meibomian glands loss.14,15
Statistical analyses
The data were analyzed using the SPSS software (v25; IBM Corp., New York, USA). Descriptive parameters are expressed as counts for categorical data (%) and as the mean ± standard deviation. One-way analysis of variance was used to compare parameter values between the pSS and NSSDE groups. Receiver operating characteristic analysis was performed to determine the accuracy of the dropout rate in predicting labial gland biopsy results and classification as pSS. Sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), and cutoff points were calculated. A p-value < 0.05 was considered to indicate statistical significance.
Results
Patient characteristics
The baseline characteristics of participants are shown in Table 1, including age, sex, ocular staining score, and antibody test results (anti-SSA, anti-SSB, ANA, and RF antibodies). The mean age of the enrolled patients was 50.98 ± 9.36 years, and 87.5% were female (49/56). Among 56 participants, 34 patients were diagnosed with pSS, and the remaining 22 patients were diagnosed with NSSDE, which served as the control group. The absolute agreement between the histopathological outcome of the labial salivary glands biopsy and pSS classification was 85.71% (48/56), with a sensitivity of 96.43% (27/28), specificity of 75.00% (21/28), PPV of 79.41% (27/34), and NPV of 95.45% (21/22).
The characteristics of the patients between the pSS and NSSDE groups.

ANA, antinuclear antibody; av-NIKBUT, noninvasive average break-up time; FLS, focal lymphocytic sialadenitis; f-NIKBUT, noninvasive first break-up time; FS, focus score; LL, lower eyelids; MG, meibomian glands; N, nasal region; NSSDE, non-SS dry eye; N&T, nasal and temporal regions; OSDI, Ocular Surface Disease Index; OSS, ocular staining score; pSS, primary Sjögren’s syndrome; RF, rheumatoid factor; SIT, Schirmer I test; SSA, Sjogren syndrome-related antigen A; SSB, Sjogren syndrome-related antigen B; T, temporal region; TMH, tear meniscus height; UL, upper eyelids.
means p value <0.01 comparing pSS and NSSDE.
pSS had significantly lower SIT and f-NIKBUT than the NSSDE (3.12 ± 1.25 vs 4.76 ± 1.38, 2.40 ± 0.87 vs 3.20 ± 0.92, all p < 0.01). There were no statistically significant differences in the TMH, av-NIKBUT, and OSDI between the pSS and NSSDE groups (p = 0.08, 0.23, 0.08, respectively).
MGD analysis of upper or lower eyelids
The MGD rate in the upper eyelids was significantly higher in the pSS group than in the NSSDE group (70.21 ± 12.45 vs 44.85 ± 12.56, p < 0.01) (Figure 2(a)). However, there was no significant difference in MGD rate in the lower eyelids between the pSS group and the NSSDE group (41.26 ± 11.62 vs 39.68 ± 13.26, p = 0.64) (Figure 2(b)). For subsequent analyses, the length of meibomian glands in the upper eyelids was significantly lower in the pSS than in the NSSDE (3.06 ± 0.77 vs 3.98 ± 0.70 mm, p < 0.01), with no difference in the width and tortuosity of meibomian glands in the upper eyelids (p = 0.32 and 0.12, respectively).

Comparison of MGD between the pSS and NSSDE groups or positive versus negative labial glands biopsy. (a) MGD of the upper eyelids between the pSS and NSSDE groups. (b) MGD of the lower eyelids between the pSS and NSSDE groups. (c) MGD of the upper eyelids in positive versus negative labial glands biopsy. (d) MGD of the lower eyelids in positive versus negative labial glands biopsy.
The total dropout rate in the upper eyelids was significantly higher in patients with positive outcomes of the labial salivary glands biopsies than those with negative outcomes (74.52 ± 8.59 vs 45.97 ± 11.57, p < 0.01) (Figure 2(c)). Similarly, no significant difference was found in the lower eyelids in patients with positive outcomes of the labial salivary glands biopsies and those with negative outcomes (41.82 ± 12.76 vs 39.46 ± 11.74, p = 0.48) (Figure 2(d)).
To be in detail, the dropout rates in the T and N&T of upper eyelids were higher in the pSS group than that in the NSSDE group using violin plot (14.74 ± 4.78 vs 5.17 ± 3.58, 25.19 ± 8.37 vs 14.04 ± 5.32, all p < 0.01) (Figure 3). Of 34 patients in the pSS group, 7 patients belonged to grade 2 (20.6%) and 27 patients belonged to grade 3 (79.4%). Of 22 patients in the NSSDE group, 2 patients belonged to grade 1 (9.1%), 18 patients belonged to grade 2 (81.8%), and 2 patients belonged to grade 3 (9.1%).
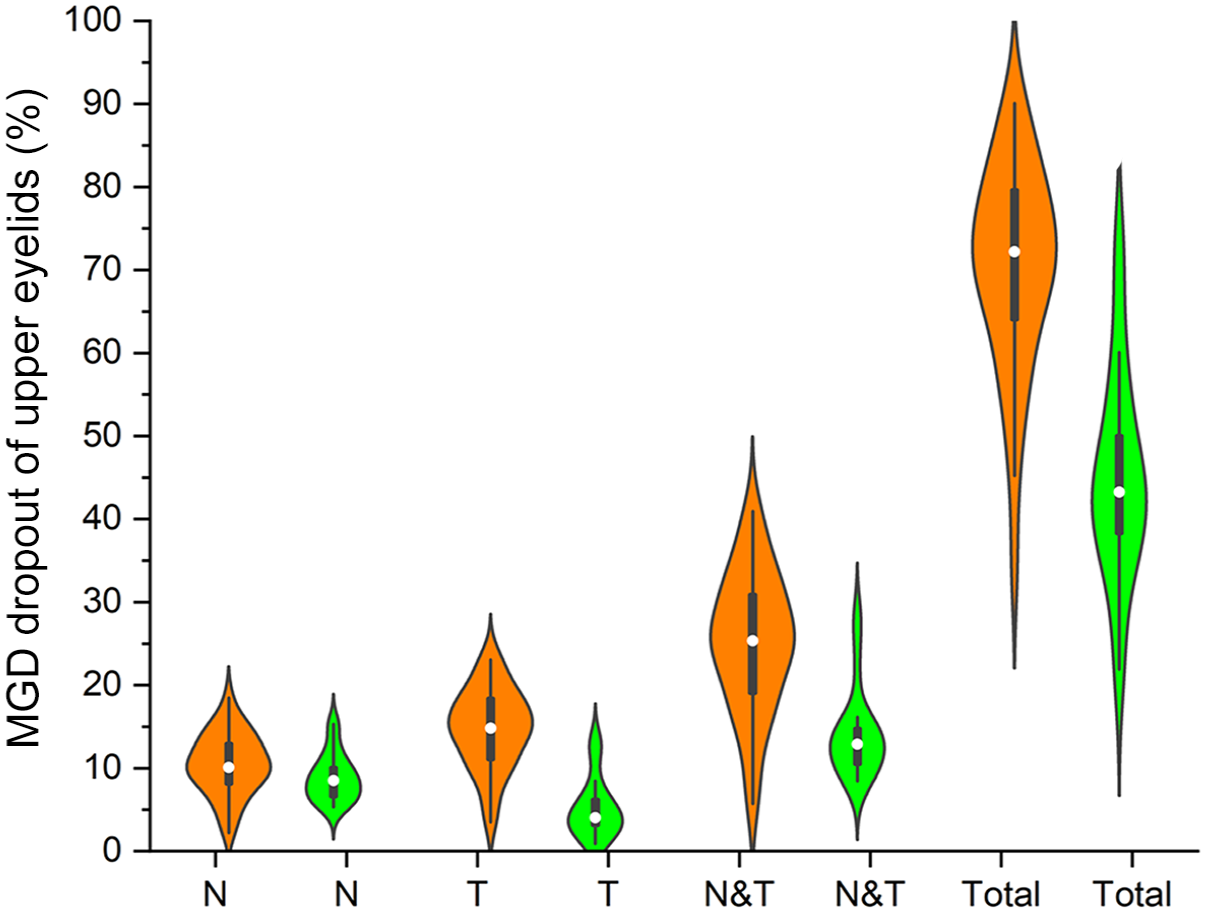
Comparison of meibomian glands of the upper eyelids between the pSS and NSSDE groups. Dropout of nasal (N), temporal (T), the sum of N and T, and total regions in the upper eyelids between the pSS and NSSDE groups using Violin chart, orange figures represent the pSS group, green figures represent the NSSDE group.
MGD to predict pSS classification or histopathological outcome of the labial salivary glands biopsy
The accuracy of temporal and total dropout rates in the upper eyelids to predict pSS classification was good, with an area under the curve (AUC) of 0.94 and 0.91, and an optimal cutoff point of 0.78 and 0.75, respectively. While the accuracy of the total dropout rate in the lower eyelids to predict pSS classification was poor, with an AUC of 0.57, and an optimal cutoff point of 0.24 (Figure 4(a)). The accuracy of temporal and total MGD rate in the upper eyelids to predict the positive histopathological outcome of the labial salivary glands biopsy was good, with an AUC of 0.96 and 0.98, and optimal cutoff point of 0.82 and 0.89, respectively (Figure 4(b)).
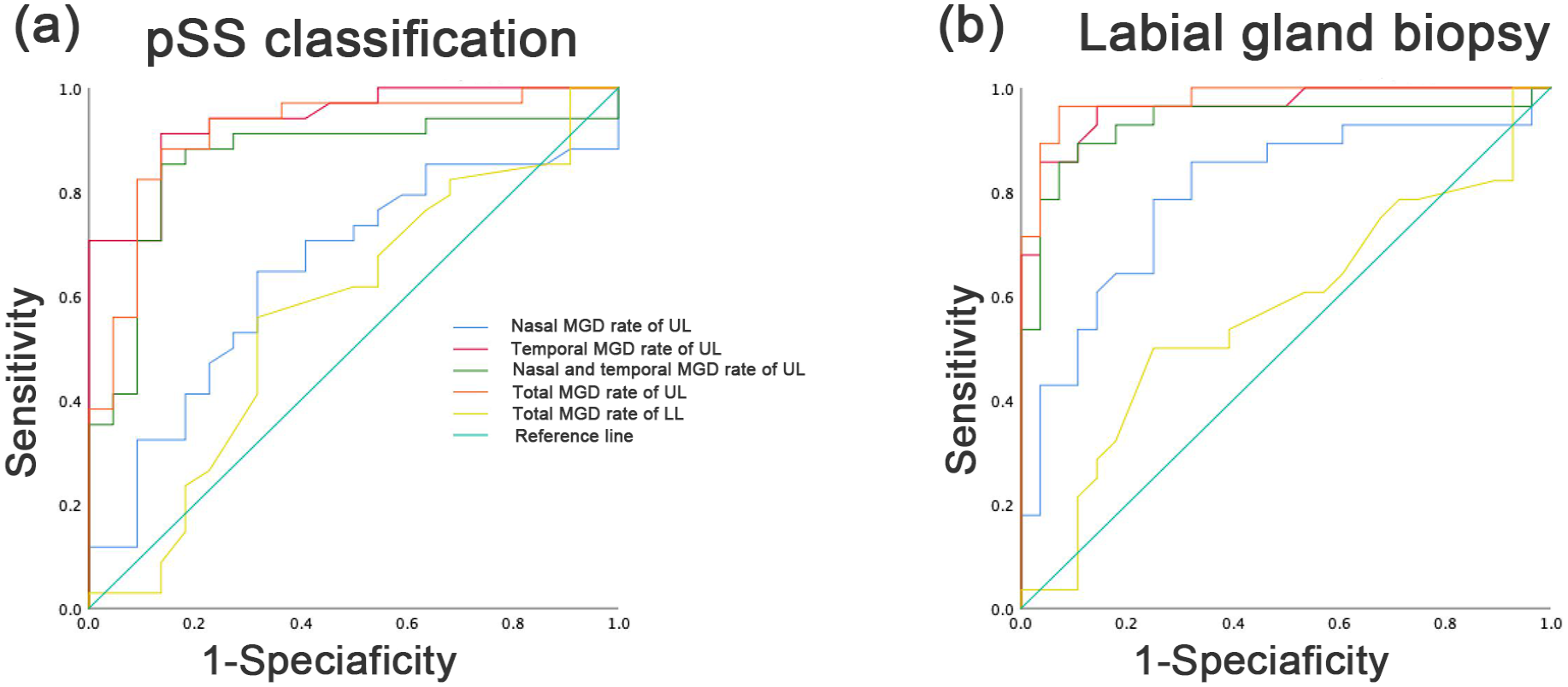
The comparison of area under the ROC curves for MGD of nasal, temporal, the sum of nasal and temporal, total in upper and lower eyelids between the pSS and NSSDE groups. (a) ROC curves illustrate the ability of the algorithm to discriminate between pSS and NSSDE according to the MGD of the upper or lower eyelids. (b) ROC curves illustrate the ability of the algorithm to discriminate between positive and negative labial glands biopsy according to MGD of the upper or lower eyelids.
Discussion
This study suggests that assessment of MGD in the upper eyelids in a representative population of patients with clinically suspected pSS may offer valuable insights. A positive association between MGD in the upper eyelids and positive histopathological outcome in the labial salivary gland biopsy was observed in the pSS group. The dropout rate in the upper rather than in the lower eyelids was a more accurate predictor of both pSS classification and a positive histopathological outcome in labial salivary gland biopsy. Our results indicated that MGD may serve as a sensitive and non-invasive biomarker for the prediction and early diagnosis of pSS.
Meibomian gland dropout is a vital cause of pSS-associated dry eye,16,17 and with the development of meibography, it provides new insights into the structure and status of meibomian glands.1,18,19 Meibomian gland detection using Keratograph 5M has gradually become a relatively accurate and noninvasive method for exploring morphological and common pathological characteristics. Our study observed that the total dropout rate and shortened length of the meibomian glands in the upper eyelids were more severe in the pSS than in the NSSDE group. These outcomes may be due to several reasons. First, diffuse lymphocytic infiltration into the lacrimal glands and the ocular surface inflammatory process are regarded as the central pathological mechanisms in the pathogenesis of pSS3,20; these processes mediate the production of acinar and periglandular inflammatory cells, and lower secretion reflectivity of the meibomian glands.21,22 Additionally, eyelids’ inflammation is associated with meibomian gland dropout. 23
Meibomian gland loss is mainly characterized by the shortening of the meibomian gland and a reduction in meibum volume. Several possible theories may explain the more obvious findings in the upper than in the lower eyelids in pSS. First, sheering stress generated by inadequate lubrication between the moving upper eyelids and keratoconjunctiva may explain the higher ocular staining score and mechanical harm to the meibomian glands. 24 Second, in terms of anatomical differences between the upper eyelids and the lower eyelids, the meibomian glands are embedded in parallel rows in the tarsal plates of the eyelids, with approximately 30–40 and 20–30 meibomian glands in the upper and lower eyelids, respectively. 25 Healthy meibomian glands are approximately 5.5 mm in length in the middle of the upper eyelids and 2 mm in the lower eyelids. 26 Third, in terms of meibography images, due to the large fold of conjunctiva that covers the meibomian glands in the lower eyelids, only a close-up view reveals the glands near the margin of the lower eyelids; in contrast, almost the entire meibomian gland population in the upper eyelids is easily observed throughout the tarsal region. 27
Notably, we found that MGD was significantly severe in the temporal rather than in the nasal region of the upper eyelids in patients with pSS. This distinction can be attributed to lacrimal gland dysfunction caused by pSS that may worsen the loss of meibomian glands from the temporal to the nasal region in the upper eyelids. As the inflammatory reaction is a critical factor in meibomian gland dystrophy, the release of inflammatory cytokines from lacrimal glands located in the temporal eyelids can result in the dysfunction of the temporal meibomian glands. This observation of temporal dropout of meibomian glands reveals an early stage event in the progress of pSS; morphological analysis of the temporal region in the upper meibomian glands may thus be a more sensitive indicator of dysfunction in the early stage of pSS.
Furthermore, our results indicate an association between MGD and the results of the diagnostic biopsy of the labial salivary glands in patients with pSS; this is important, considering that labial biopsy is prioritized in the pSS diagnostic criteria set. According to a previous study, the sensitivity of labial minor salivary gland biopsy ranged between 63.5% and 93.7%, and specificity between 61.2% and 100%, respectively. 28
Our study is innovative in that we assessed the diagnostic value of invasive labial biopsy as well as a new noninvasive diagnostic method using meibography. We showed that the total and temporal MGD rates in the upper eyelids were more sensitive in predicting pSS classification and positive labial gland biopsies. Importantly, a lower MGD rate in the upper lid may well be predictive of negative labial biopsy histopathology in patients with suspected pSS. Several explanations may be plausible regarding the association between MGD rate and the results of labial gland biopsy. First, sebaceous glands are present in both the lip (labial gland) and eyelid margin (meibomian gland); they are targeted by the autoimmune exocrinopathy, which is characterized by lymphocytic infiltration of the exocrine glandular epithelia in pSS,29,30 leading to a combination of dry eye and dry mouth in pSS. Alternatively, this association may be attributed to common histopathology, as inflammatory reactions could be responsible for both meibomian gland dysfunction and positive labial gland biopsy in patients with pSS. 31 In addition, estrogen and androgen receptors on exocrine glands are crucial regulators of gland function, and reduced androgen and increased estrogen levels are detected in the serum of patients with SS. These could contribute to the dysfunction and atrophy of the meibomian and labial glands, with females becoming susceptible to pSS.32,33 Interestingly, androgen deficiency may also result in decreased secretion of insulin-like growth factor 1 (IGF-1), which plays an important role in maintaining the normal function of meibomian gland epithelial cells. 34 Another study found that IGF-1 receptor levels decline in the labial salivary glands in SS. 35 Thus, to some extent, the dysfunction of the meibomian and labial salivary glands progresses synchronously.
Several limitations of the current study should be addressed in future research. First, a relatively small sample size of patients with clinically suspected pSS was enrolled. Thus, study enrollment only represents circumstances in daily clinical practice, and large-scale and multicenter studies are warranted to verify the results. Second, the underlying mechanisms of the MGD in pSS should be investigated; for example, whether the meibomian gland is a target of ocular surface autoantigens or whether the effect is secondary to the inflammatory process in pSS.
Conclusion
Overall, our study focused on the state of meibomian gland morphology on IR meibography as an imaging biomarker for pSS, we propose a clear role for the dropout rate in the upper eyelids in the diagnostic prediction of pSS. To the best of our knowledge, this is the first study to explore the association between MGD features and labial gland biopsy outcomes in patients with suspected pSS.
The dropout rate of the meibomian glands in the upper eyelids, especially in the temporal region, was significantly predominant in patients with a positive histopathological outcome in the labial salivary gland biopsies. We suggest that it may be an alternative parameter to the “gold standard” FS obtained in biopsies and that it can serve as a sensitive and easy-to-obtain predictive biomarker for pSS.
