Abstract
Musculoskeletal disorders are characterized by several impairments, including pain, affecting muscles, bones, joints and adjacent connective tissue, resulting in temporary or permanent functional limitations and disability. Musculoskeletal pain is particularly prevalent worldwide and greatly impacts the quality of life, social participation and economic burden. To date, several issues persist about the classification of musculoskeletal pain and its management strategies and resources. The treatment of musculoskeletal pain conditions is complex and often requires a multimodal approach, including pharmacological and non-pharmacological therapy that might be ineffective in many cases, resulting in poor patient satisfaction and controversial expectations about the potential benefits of available interventions. This manuscript provides an overview of unmet needs in managing musculoskeletal pain, particularly focusing on pharmacotherapeutic pitfalls in this context.
Epidemiological insights into musculoskeletal pain
Musculoskeletal (MSK) pain is an unpleasant sensory and emotional experience that can be attributable to disorders affecting bones, muscles, ligaments, tendons and/or related soft tissues. 1
According to the International Association for the Study of Pain (IASP), ‘more people around the world experience MSK pain than any other type of pain’. 2 Altogether, MSK pain conditions are the main contributor to disability worldwide, 3 led by low back pain (LBP) that affects about 7.5% of the global population (i.e. age-standardized point prevalence), 4 followed by neck and shoulder pain (6.1%), 5 and knee pain. 6 Musculoskeletal pain patterns vary greatly by age and sex. Age is an important risk factor, considering that older people are more likely to have chronic MSK pain. Osteoarthritis (OA), the most common arthritis, is present in 43% of older people. This clinical insight seems to be attributable to age-related joint deterioration, muscle weakness and a sedentary lifestyle. However, MSK pain can affect all ages since in the paediatric, adolescent and young adult population of the United States, MSK pain was responsible for more than 3 million visits annually.
Other risk factors for developing MSK pain are smoking, low education level, limited social interactions, low income, sleep disorders, anxiety, depression, manual labour, being separated or divorced and recent immigration. 7 Although some studies show that there is no difference in the onset of MSK pain conditions between urban and rural populations, it is difficult to estimate the prevalence of these disorders in countries such as Central and South America and sub-Saharan Africa, given the poor availability of epidemiological data. 8 However, some data indicate that MSK pain in general, and more specifically for some conditions such as knee and hip OA, is more frequent in industrialized countries such as Europe and the United States. 9 In Italy, the age-standardized prevalence of people with musculoskeletal disorders, including rheumatoid arthritis, osteoarthritis, LBP, neck pain and gout per 100,000 people ranged from 4400 to 4900 in 2020, according to The Global Burden of Diseases, Injuries and Risk Factors Study. 10
While patients with acute MSK pain are usually treated by general practitioners, those with chronic pain are commonly managed by both general practioners (GPs) and specialist doctors, such as orthopaedists, physiatrists and rheumatologists, but clinicians in all fields may be involved in the management of these conditions.
The most prescribed drugs are non-opioid analgesics (38.5–48.8%), followed by opioids, especially in young adults (23%). These data are in line with international guidelines, such as those for OA of the American College of Rheumatology that strongly recommend non-steroidal anti-inflammatory drugs (NSAIDs) for patients with hand, knee and hip OA. 11 Moreover, European Alliance of Associations for Rheumatology (EULAR) recommended paracetamol and topical NSAIDs as first-line treatment for knee OA as well as European Society for Clinical and Economic Aspects of Osteoporosis and Osteoarthritis and Musculoskeletal Diseases (ESCEO) guidelines. 12 This latter considers oral NSAIDs and opioids as second and third steps, respectively. 13 However, awareness of different MSK pain phenotypes is growing, such as in the case of OA, where the definition of several phenotypes has been proposed (i.e. intra-articular phenotype, extraarticular phenotype, secondary phenotype, age-related and systemic phenotypes) supporting a personalized approach, both pharmacological and non-pharmacological. 14 In the elderly population (>75 years), according to the Helsinki Aging Study, more than 60% of patients suffer from daily or intermittent MSK pain. Of these, up to 16% were prescribed analgesics, especially paracetamol and NSAIDs. 15 This data is probably associated with an underestimation of pain both by the patient himself and by the doctor, who can sometimes prefer to avoid prescribing drugs, in particular NSAIDs, considering the comorbidities of this population, although appropriate.
On the other side, health education, counselling and physical therapies were prescribed less frequently (<20%). This is in contrast with available guidelines for the management of MSK disorders with high prevalence, such as LBP and OA, where non-pharmacological interventions are recommended as first-line treatment strategies. 16
Issues in musculoskeletal pain classification
The definition and classification of MSK pain are still debated topics. Considering the different aetiologies and underlying physiopathological mechanisms, the IASP identified three categories of pain 17 :
Nociceptive, also known as inflammatory or physiological, is associated with tissue damage. For this reason, it is also defined as ‘protective’; it is often acute, well-localized, sharp and throbbing. It can be defined as a physiological sensory experience secondary to the activation of peripheral nociceptors. It can be further subdivided according to the site of nociceptor stimulation in somatic, if associated with soft MSK tissue damage or inflammation, sometimes intermittent, often constant; bone pain, associated with damage of this tissue, with deep characteristics and sensation of tenderness or visceral, from activation of nociceptors of internal organs, often poorly localized or referred to other sites.
Neuropathic: frankly pathological as it is associated with a primary lesion or dysfunction of the somatosensory nervous system. It presents peculiar clinical features such as burning, tingling, electric shock sensation and alterations of sensitivity with dermatomal distribution.
Nociplastic: resulting from an alteration of nociceptive mechanisms in the absence of current or previous tissue damage that could trigger the onset of nociceptive or neuropathic pain.
Two other categories of pain that do not exist in the IASP taxonomy are described in the literature:
Mixed: the co-presence of nociceptive and neuropathic symptoms. 18
Idiopathic: in the absence of a definite cause or disproportionate to the type or extent of any tissue damage. 19
It is important to highlight that the IASP classification of pain includes three descriptors, which are valuable for directing pharmacological interventions towards the specific pathogenic mechanisms while disregarding the psychological and social aspects of pain, which cannot be effectively treated with medications.
For a better characterization of chronic pain, and in particular for codifying its diagnosis and documenting investigations or therapeutic measures within the health care systems of many countries, a new classification has been introduced in the International Classification of Diseases Edition 11 (ICD-11) by the IASP. 20 This classification has identified chronic primary and secondary pain syndromes, including MKS ones. Chronic primary pain is defined as pain in one or more anatomical regions persisting or recurring for more than 3 months and is associated with emotional distress or functional disability and cannot be explained better from another clinical condition, thus resulting in a condition in its own right. Therefore, in the case of chronic primary MSK pain, it is not attributable to other pathological conditions. Instead, chronic secondary MSK pain is a symptom attributable to a specific disease. Primary forms include chronic non-specific LBP and chronic widespread pain, like fibromyalgia. Chronic secondary MSK pain is pragmatically subdivided into three main forms that are, respectively, associated with (1) persistent inflammation (e.g. infections, crystal deposits, or autoimmune or autoinflammatory disorders), (2) structural changes (e.g. OA, and MSK injuries) and (3) diseases of the nervous system (e.g. Parkinson’s disease, multiple sclerosis, peripheral neurologic disease). 21
The new ICD-11 aims to implement and make pain research and therapy more accessible, facilitate patient care and improve health policy decisions. However, this classification is not based on new pain pathogenesis insights but is rather pragmatic to increase awareness about pain management needs and to contrast its under-treatment. 22 Although this classification strengthens the representation of chronic pain conditions in clinical practice, the distinction between chronic primary and secondary pain is somewhat challenging because it is difficult to prove that chronic pain is causally related to a specific disease (i.e. LBP due to sacroiliac joint disorder or facet joint syndrome), or that pain is a persistent symptom of a disease or a trauma that has been cured and healed. The word ‘chronic’ in secondary pain conditions is conceptually time-based but not pathomechanics-based as the definition of chronic pain would require (i.e. a disease in its own right), thus resulting in misunderstanding versus acute pain definition as a symptom of biological warning for dangerous stimuli (disease or tissue injury). Also, on one hand, socioeconomic implications of the new ICD-11 implementation might be advantageous for chronic pain patients to recognize their complaints, enhance social participation and receive financial support, whereas on the other hand, once symptoms receive a diagnostic label as chronic pain, some patients might experience no clinical benefit because of the diagnosis itself, fostering stigmatization, overtreatment and passive coping behaviours. 23
Another issue to be addressed is the pragmatic model used for chronic pain classification and decision-making purposes that might be unable to estimate the complexity and multidimensional nature of this condition, leading to neglect for psychosocial components of subjective patient experience that might be better assessed by a holistic model. Conversely, emphasizing the biopsychosocial components of chronic pain might reduce the scientific legitimacy of the classification as well as increase overdiagnosis and overtreatment. 24 Therefore, considering the dynamic nature of chronic pain, its classification should be intended as a conceptual pragmatic tool and its wide use in clinical practice should be viewed with caution.
Issues in the management of musculoskeletal pain
One of the main issues in the field of MSK pain is the diagnosis, considering that it is often challenging to correlate pathophysiological mechanisms to the individual experience of the pain. In this context, in addition to a mechanistic approach, a biopsychosocial, multidisciplinary and multimodal (i.e. pharmacological and non-pharmacological) approach is usually appropriate in most painful MSK diseases. 25
The mechanistic approach is based on the identification of the mechanism underlying the cause of the pain (e.g. nociceptive, neuropathic, nociplastic) to undertake a targeted therapy. 26 This approach must be integrated with the biopsychosocial one, which should consider the impact of the disease on the patient’s disability, changes in employment status and quality of life to personalize the treatment as well as to meet the patient’s needs.27,28 Indeed, one of the limits of mechanistic model could be the underestimation of the multidimensional nature of MSK pain, not allowing to predict the possible failures of pharmacotherapy. For example, trials usually do not consider some critical issues, such as predisposition to pain chronicization, comorbidities and/or cognitive-behavioural features (e.g. pain catastrophizing).
The initial therapeutic approach is the patient’s education on his condition through self-management and the explanation of the therapeutic options. Non-pharmacological therapy, including exercise and multidisciplinary rehabilitation protocols, should be initiated for patients with chronic MSK pain. 29 When no adequate response is reported, pharmacological therapy can be prescribed in combination with these approaches.
Pharmacological therapy is a cornerstone of the management of MSK pain. In choosing the analgesic therapy for different painful conditions, including MSK disorders, the World Health Organization (WHO) initially proposed a three-step ladder, recently modified into four steps, based on the intensity of the symptom perceived by the patient and on the response to therapy. The analgesics should be taken by the simplest route of administration, at the lowest effective dose for the shortest possible time to minimize side effects on the patient’s health status and comorbidities. 30 The first step involves the use of non-opioid analgesics possibly associated with adjuvants, which can enhance the effect; the second step provides for the combination of weak opioids with the drugs of the first step; from the third step, the introduction of minimally invasive interventions is recommended; the last step provides the use of strong opioids. It is possible to pass from one step to another depending on the pain intensity during the disease course (Figure 1 and Table 1).
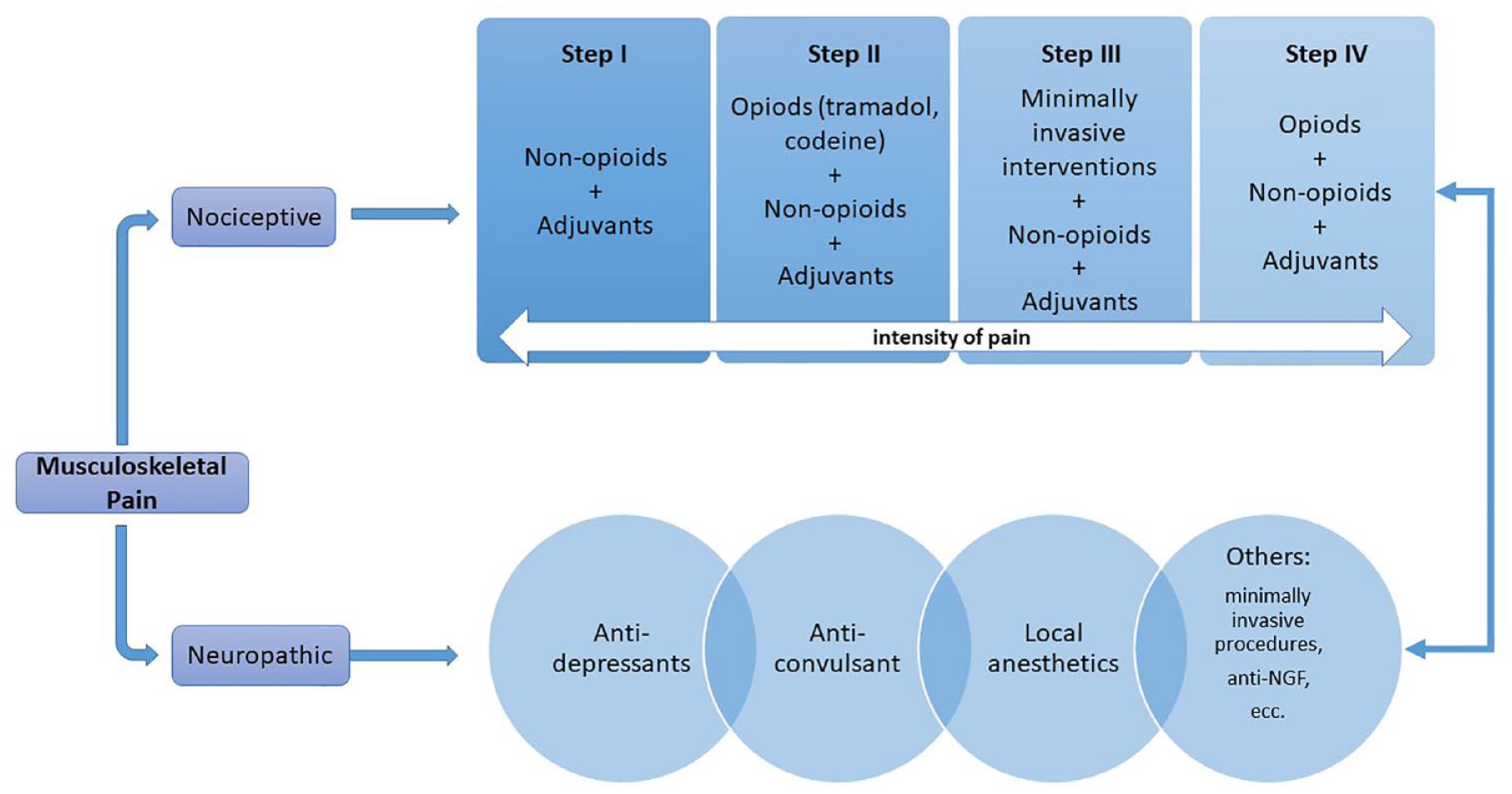
WHO analgesic ladder and mechanistic approach to musculoskeletal pain.
Pharmacotherapy of MSK pain.

COXIBs, COX-2 inhibitors; MSK, musculoskeletal; NSAIDs, non-steroidal anti-inflammatory drugs; SYSADOA, slow-acting drugs for osteoarthritis; WHO, World Health Organization.
Pharmacological therapy of musculoskeletal pain: Opportunities and pitfalls
Acetaminophen
Also known as paracetamol, acetaminophen is an analgesic with a predominantly central nervous system effect. 33 In particular, this drug inactivates COX through the reduction of the hydroperoxide tone, thus reducing the production of prostaglandins. Moreover, a bioactive metabolite of acetaminophen in the central nervous system (CNS), AM404, activates TRPV1 channels modulating neuronal response to pain. 34 Alone or in combination with NSAIDs/opioids, it is used in a wide range of indications (conditionally recommended for OA). However, a meta-analysis shows that its efficacy in chronic pain, when used alone, appears to be limited.31,32 On the contrary, a recent meta-analysis found that paracetamol monotherapy might be a first-line analgesic for acute musculoskeletal injuries. 35 The maximum daily dose is 3000 mg, but it should be used with caution in patients with liver disease due to its hepatotoxicity. 11
Despite a huge increase in paracetamol use in the last decades, particularly in elderly patients with MSK pain, 15 both uncertain benefits and growing evidence about safety issues (also in children) 36 require careful consideration for clinical practice. 47 In our opinion, considering the small reduction of pain in MSK conditions, this drug could be used as an alternative treatment strategy for both acute and chronic MSK pain in patients with contraindications to oral NSAIDs, despite the risk of hepatotoxicity due to the overdose or previous liver diseases, we consider acetaminophen as quite safe in general population, including older people.
Non-steroidal anti-inflammatory drugs
NSAIDs are a wide class of drugs that inhibit the cyclooxygenase enzyme (COX), which catalyses the metabolism of arachidonic acid-producing prostanoids, substances involved in the inflammatory process. These drugs have different selectivity for COX 1 and 2 isoforms and are burdened by several side effects such as gastrointestinal (GI), cardiovascular (CV) and renal side effects and potential drug interactions. Selective inhibition for COX-2 results in fewer GI side effects but is burdened by a poor CV risk profile if used in the long term. Both traditional NSAIDs and COXIBs are indicated for nociceptive and inflammatory pain. These drugs are commonly used as monotherapy, as the first line of treatment for OA, both of the knee and of other joints, as well as for LBP, for the shortest possible time at the lowest effective dose.11,28,31 NSAIDs can also be used in combination with opioids, having a synergistic effect and allowing the dose of both drugs to be reduced, although this therapeutic strategy is not widely supported by available evidence and international guidelines for MSK pain conditions and potentially increases the risk for adverse events of both drug classes. 44
Even if several guidelines for MSK pain management recommend NSAIDs as first-line pharmacological options, these drugs are among the most frequently overused/misused medications in older patients. 37 According to a recent meta-analysis, the administration of diclofenac 150 mg/day and Etoricoxib 60 mg/day are effective for pain in patients with OA (MD −0.56 and −0.65, respectively). 48
Safety of NSAIDS
All NSAIDs are burdened by GI adverse effects, mainly attributable to COX-1 inhibition, 44 such as dyspepsia, risk of bleeding or developing GI ulcers. Concomitant use of proton pump inhibitors or H2-receptor blockers is commonly recommended in patients at risk of GI adverse events. 49 However, some NSAIDs seem to have a better GI risk profile than others, along with comparable efficacy (e.g. aceclofenac). 50 These drugs also cause an increase in blood pressure and potential kidney damage. Finally, NSAIDs increase the risk of CV events such as myocardial or cerebrovascular infarction, especially in patients with a positive history of such events or who receive acetylsalicylic acid for the prevention of CV diseases. Therefore, in patients with a high CV risk, this drug class should be avoided, although it seems that the use of naproxen for a short time is burdened by fewer adverse CV events than other NSAIDs.44,51
Topical NSAIDs
Besides the systemic formulations of NSAIDs, there are also topical formulations of different compounds, such as diclofenac, ibuprofen and flurbiprofen. These interventions are recommended by international guidelines as first-line drugs for the management of MSK pain associated with OA of the knee, hands or feet. 16 These formulations could be considered as a first line of treatment in these conditions because they are effective and associated with the lowest systemic exposure to the active agent, minimizing the risk of adverse reactions. 11 A recent Cochrane systematic review highlights that some of the most used topical NSAIDs, such as diclofenac and ketoprofen, significantly reduce pain in about two-thirds of people with chronic musculoskeletal pain. 51 They are less effective in treating OA of deep joints such as the hip.11,51
Generally, we encourage the use of NSAIDs on MSK pain as a first-line treatment for their good risk–benefit ratio; however, it is crucial to provide the patient with explicit instructions regarding the duration of therapy and recommended dosage to prevent adverse events.
Opioids
Opioids are a large class of drugs that act by binding specific receptors of the endogenous opioid system widely distributed in the brain, spinal cord and outside the CNS. Several opioids are available (e.g. codeine, tramadol, tapentadol, buprenorphine, methadone, oxycodone, fentanyl, morphine) with different formulations and administration routes, such as oral, sublingual, intravenous (IV), intramuscular (IM), subcutaneous (SC) and transdermal. 46 Opioids are indicated for the treatment of moderate-to-severe pain, not controlled by NSAIDs or other first-line drugs. However, evidence supporting their effectiveness is limited, with relatively small clinical benefits demonstrated by trials on opioids for the management of chronic pain 38 with adverse effects such as respiratory depression, nausea and vomiting, drowsiness, constipation and addiction. 52 A recent systematic review and network meta-analysis of RCT reported no significant improvement in using tramadol [MD 0.95 (−0.80 to 2.70; 95% confidence interval, CI)] for the treatment of pain in patients with acute musculoskeletal injuries. 39 Considering these issues, their use must be provided after a careful assessment of the risk–benefit ratio for each patient, 53 particularly in terms of harm reduction strategies for patients receiving narcotics (e.g. gabapentinoids or benzodiazepines), 40 also considering that evidence-based strategies for opioids, tapering are still lacking. 54 Personal factors should also be taken into account, such as the level of physical activity of patients, considering that unaccustomed exercise may enhance pain through the modulation of central mechanisms (increasing in serotonin transporter expression) in the rostral ventromedial medulla; at the same time engaging in regular physical activity could be considered a possible non-pharmacological approach towards reducing or preventing chronic pain probably attributable to the modulation of mu-opioid receptors. 55
Another common debate in the field of opioid use is their classification as weak or strong according to the WHO analgesic ladder and not to pharmacodynamic properties, contributing to opioid misuse and opioid-related adverse events. 56 Indeed, all opioids might increase the risk of opioid use disorder, and codeine might be even more addictive than other opioids. 57 Therefore, this arbitrary categorization of opioids should be discontinued because of both limited support to clinicians to adopt a mechanistic approach and the increased risk for opioid-related harms. Therefore, when managing chronic moderate-to-severe MSK pain, we suggest using opioids only if other pharmacological therapies have proven ineffective or if surgery is contraindicated or refused by the patients (e.g. in the case of joint replacement for OA). However, it is crucial to thoroughly evaluate risk factors for opioid-related adverse events, such as family or personal history of substance abuse, history of major depression or anxiety, chronic obstructive pulmonary diseases, GI disorders (i.e. constipation, bowel obstruction), seizure disorders as well as co-prescriptions of some drugs (e.g. anticonvulsants, benzodiazepines, antidepressants).
Mixed non-opioid–opioid
Different associations of acetaminophen or NSAIDs with opioids such as tramadol, oxycodone and codeine are available in clinical practice. These combined formulations perform a putative synergistic analgesic effect, where the end results of the combined drug are greater than the sum of the parts, 58 due to the different mechanisms of action as well as reduce the dose of both active ingredients to limit the incidence of potential side effects. 44 The combinations of non-opioid–opioid drugs seem to be useful in the management of both acute and chronic MSK pain conditions, such as LBP, or in cancer pain. 59 On the other side, evidence about the effectiveness of this intervention is conflicting, considering that analgesic monotherapies may be more effective in MSK pain relief. Also, the combined formulations of non-opioid–opioid drugs might hamper effective opioid titration and tapering as well as increase the risk of adverse events (e.g. hepatotoxicity).
Adjuvants
Adjuvants include several drugs belonging to different classes with different mechanisms of action that can be used in association with analgesics to enhance their effect when the latter are not adequately effective in monotherapy. 46 Adjuvants might improve MSK pain management by modulating different pathogenic pathways compared to opioids or other non-opioid analgesics (e.g. NSAIDs). However, evidence of their effectiveness is quite limited, also considering that the type of pain might be not well characterized in MSK pain conditions (e.g. neuropathic pain component). Moreover, adjuvants are used as off-label drugs in most cases for the treatment of MSK painful disorders. Therefore, any adverse events, which may occur quite frequently in their use might expose physicians to inappropriateness and legal issues.
Slow-acting drugs for osteoarthritis
Slow-acting drugs for osteoarthritis (SYSADOA), including glucosamine sulfate and chondroitin sulfate, might have joint tissue structural changes resulting in potential analgesic effects in OA patients. These substances are glycosaminoglycans that are found in human tissues, including cartilage and bone. The hypothesized effect of their administration in OA patients is the prevention of cartilage damage by restoring the extracellular matrix, thus resulting in pain relief. ESCEO recommends chronic use of SYSADOA (more than 3 years) as the first step in symptomatic knee OA in association with acetaminophen or topical NSAIDs. 60 Moreover, glucosamine sulfate and/or chondroitin sulfate are widely used and are considered safe medications. 43
Anticonvulsants
Anticonvulsants are recommended by international guidelines as first-line drugs for the management of neuropathic pain, particularly gabapentin and pregabalin. 61 Pregabalin is also indicated in the treatment of fibromyalgia syndrome. 62 These drugs are analogues of gamma-aminobutyric acid (GABA) able to bind α2δ subunit of voltage-gated calcium channels in the CNS, causing a reduction in the release of glutamate, noradrenaline and substance P.44,63 A recent systematic review and network meta-analysis on the effectiveness of pregabalin found a significant improvement of pain in adults with acute non-specific LBP [MD −24.7 (−34.6 to −14.7), low confidence]. 64 Anticonvulsants have an acceptable safety profile, although their dose should be reduced in the presence of renal insufficiency and they can cause respiratory depression when used together with opioids. 44 The main issues in the use of gabapentinoids for MSK pain are their misperception as not addictive compared to opioids, their common use as off-label drugs as well as the recommendation for their use in guidelines for the management of MSK pain in different populations. 45
In our opinion, these drugs might be useful for the treatment of the neuropathic component of mixed pain which may be involved in chronic MSK conditions such as OA and LBP.
Antidepressants
Antidepressants are a heterogeneous class of drugs whose primary indication is major depression. These include tricyclic antidepressants (amitriptyline, nortriptyline, etc.), serotonin and norepinephrine reuptake inhibitors-SNRIs (duloxetine, etc.) and selective serotonin reuptake inhibitors-SSRIs (fluoxetine, etc.). 46 The ability to act on pain seems independent of their antidepressant action. Specifically for tricyclic antidepressants, the analgesic action seems to be attributable to their action as antagonists of calcium sodium channels as well as N-methyl-D-aspartate (NMDA) glutamate receptors. Instead, the action of SRNIs is attributable to the presynaptic inhibition of norepinephrine and serotonin uptake, capable of enhancing the action of the descending endogenous antinociceptive pathways. 65 Antidepressants are indicated in pathological conditions characterized by neuropathic, nociplastic or mixed pain, 41 but are not considered first-line drugs in MSK pain, as reported by a recent meta-analysis. 42 Also, more recently, reliable evidence suggests no clinically significant efficacy for most of these drugs in the management of MSK pain.66,67 This issue should be carefully considered for clinical practice because antidepressants are usually prescribed as off-label drugs for MSK pain and are burdened by several side effects. 68 According to available evidence, we suggest low dosages of duloxetine as a pain reliever for some chronic MSK conditions like LBP, OA or fibromyalgia, also due to some benefits on psychological issues.
Muscle relaxants
Muscle relaxants are divided into antispasticity agents (tizanidine, dantrolene) and antispasmodic agents (diazepam, cyclobenzaprine). The first group works on the spinal cord to reduce hypertonicity and stiffness and is widely used in some conditions, such as multiple sclerosis or cerebral palsy. Instead, the antispasmodic drugs reduce muscle spasms by acting through the alterations of specific neurotransmitter pathways in the CNS; they are classified as benzodiazepines and non-benzodiazepines.44,69 Muscle relaxants are indicated in the management of acute MSK pain (e.g. acute LBP) because of their effect in terms of muscle spasm reduction, particularly cyclobenzaprine and tizanidine, but have poor evidence of effectiveness in the treatment of chronic MSK pain.44,70 A recent systematic review reported a significant improvement in administering tizanidine plus aceclofenac in adults with acute non-specific LBP [MD: −26.1 (−38.5 to −13.6)]. 45 For what concern the safety profile, these drugs might have side effects such as sedation and the possible development of addiction. 71 According to available evidence, 72 muscle relaxants could be used as first-line drugs for the short-term treatment of acute LBP, and their use may contribute to the reduction of the dosage and timing of administration of NSAIDs in order to avoid adverse events.
Topical agents (other than NSAIDs)
Outside of topical NSAIDs, there are different drugs that may have several advantages when used as topical formulations over systemic administration. For example, local administration of an active agent reduces systemic absorption and related side effects. 46 One of the main topical agents for MSK pain relief is lidocaine, a local anaesthetic available as plasters or creams with a concentration of up to 5%.44,46 This drug blocks voltage-gated sodium channels, preventing the propagation of the action potential in the sensory nerves. 73 However, the effectiveness of lidocaine plasters for MSK pain is limited. No clear evidence is available for patients with chronic LBP, 74 while an open-label RCT found no difference between 5% lidocaine plaster and celecoxib in terms of effectiveness and tolerability for patients with knee OA (KOA). 75 However, this drug is indicated as a second-line drug for localized neuropathic pain, such as post-herpetic neuralgia. 76 It can be associated with local side effects, such as irritation and redness. Although systemic absorption is negligible, topical lidocaine should be used with caution in patients receiving first-class antiarrhythmic or patients with liver and kidney disease.46,77
Another substance used in topical formulations is capsaicin, an agonist of the transient receptor potential vanilloid member 1 (TRPV1), a receptor prominent in small nerve fibres involved in pain transmission. 78 Analgesia obtained through capsaicin administration seems to be attributable to a short-term desensitization of small nerve fibres, and a long-term inhibition of the TRPV1. 79 A Cochrane systematic review reported that high concentration of topical capsaicin significantly improves neuropathic pain; although with moderate quality of evidence. 51 It is marketed in the form of cream or medicated patches with a concentration of up to 8%. 51 Given the limited evidence about its efficacy, it is used as a third-line drug for some forms of neuropathic pain. Topical capsaicin administration can lead to local side effects, such as burning, redness and local erythema. 44
Glucocorticoids
Glucocorticoids are potent anti-inflammatories acting through multiple mechanisms, including inhibition of phospholipase A2, thereby preventing the activation of the inflammatory cascade. 80 The most used molecules of this drug class are prednisone and methylprednisolone, which are often used for the treatment of acute radicular pain of the upper and lower limbs, 44 despite limited data about their efficacy in terms of pain relief and functional improvement available.81,82 Glucocorticoid use might result in numerous side effects such as an increase in blood glucose and blood pressure, insomnia and water retention. 44 These drugs are the mainstay for minimally invasive procedures to manage MSK pain, providing short-term pain relief and functional improvement as reported by guidelines for OA and LBP.9,83,84 A recent systematic review reported that corticosteroids significantly reduced pain when compared to placebo in patients with hand OA (MD −5.75; 95% CI, −10.08 to −1.42). 85
Bisphosphonates
Bisphosphonates (BPs) might be useful in specific conditions of chronic MSK pain such as OA, rheumatoid arthritis, complex regional pain syndrome (CRPS), and bone pain due to multiple myeloma, osteogenesis imperfecta and Paget’s disease of bone. 86 These drugs are analogues of pyrophosphates and are mainly used for the treatment of osteoporosis due to their anti-resorptive effect by inhibiting osteoclasts.87,88 Specifically, neridronate has been shown to be effective in pain symptoms compared to placebo in the treatment of CRPS type 1 both by intravenous and intramuscular administration routes89–91 as well as in the treatment of knee OA and bone marrow lesions. 92 A recent Cochrane systematic review reported a significant improvement of pain in using neridronate in patient with CRPS (MD −21.80; 95% CI, −30.28 to −13.32). 93 The mechanism underlying pain relief is not yet clear, but it seems to be attributable to the reduction of osteoclast activity resulting in less acid-induced activation of primary afferent nociceptors (acid-sensing ion channels and transient receptor potential cation channel subfamily V member 1 channels). 94
From a clinical perspective, parenteral use of BPs might be paradoxically associated with MSK pain, due to flu-like syndrome (acute phase reaction). This side effect might usually occur within 72 h from the first injection, lasts a few hours and is effectively managed with paracetamol. 95
Failure of anti-nerve growth factor
Nerve growth factor (NGF) antibodies are an interesting and promising class of drugs for the management of chronic MSK pain resistant to other therapies. The NGF is an essential growth factor for the development of sensory and sympathetic neurons during embryogenesis, but by binding to an appropriate tropomyosin receptor kinase A receptor (TrkA), it modulates the pain pathway through the enhancement of the neurotrophic and nociceptor functions. 96 Different phase II and III clinical studies on fasinumab and tanezumab have demonstrated their efficacy in significantly reducing pain symptoms and improving function in patients with knee and hip OA. Conversely, adverse events concerning rapidly progressive OA in the groups of patients treated with higher doses have led regulatory agencies to refuse market authorization, although the underlying mechanism is not yet fully understood. These drugs appear to be very promising for the management of chronic MSK pain, but there is a huge need to clarify their safety profile. 97
Resiniferatoxin
Resiniferatoxin (RTX) is a capsaicin analogue derived from the Euphorbia resinifera plant, capable of binding the TRPV1 receptor 98 a ligand-gated, non-selective cation channel expressed on sensory neurons. This receptor can be activated by thermal, mechanical and chemical nociceptive stimuli. 99 TRPV1 stimulation by specific molecules can lead to a reduction in the function of the sensitive nerve fibres and, consequently, pain relief through nociceptor desensitization, neuropeptide depletion and reversible nerve fibre degeneration. 100 RTX appears to be a promising drug in the management of chronic OA pain, and currently, two active trials aiming to investigate the efficacy and safety of its intra-articular administration are ongoing. The use of this substance can lead to local side effects such as a temporary increase in pain, tachycardia and hypertension. 101
Drug administration through minimally invasive procedures
To improve the effectiveness and pharmacokinetics of some drugs, it has been proposed their local administration through minimally invasive procedures. These are a set of techniques used in both acute and chronic MSK pain which provide for the local administration of a pharmacological minimizing their systemic effects. These procedures can be performed blindly or using imaging methods such as ultrasound or computed tomography (CT) and can be administered in an outpatient or day hospital setting. 102 The most common minimally invasive procedures for the management of MSK pain are injection of therapeutic substances into the joint or ligament (e.g. injection of the facet joints 103 or the sacroiliac joint 104 ), injection into the trigger points, 105 blocks of peripheral nerves, 106 and ablation of nerve endings by chemical neurolysis or radiofrequency.44,107 Different therapeutic agents can be used in this context, particularly corticosteroids (e.g. betamethasone, dexamethasone, methylprednisolone and triamcinolone) for the management of acute phases of the intra- and peri-articular inflammatory process (e.g. OA and tenosynovitis).44,108 These drugs should not be used for more than four injections per site for year to avoid serious side effects. Corticosteroids lead to pain relief and functional improvement in the short-to-medium term in OA, 16 and their use as intra-articular or peri-articular injection is characterized by minor systemic adverse effects but potential local adverse effects such as infection or atrophy at the injection site, accelerated OA progression, subchondral insufficiency fracture and osteonecrosis.44,109
During minimally invasive procedures, local anaesthetics are commonly used both for intra-articular injections, particularly lidocaine, bupivacaine or ropivacaine. 110 These drugs reduce MSK pain by blocking the excitation and conduction of the peripheral nerve fibres targeting Na+ channels. However, local anaesthetics should be used with caution due to both their potential systemic adverse effects and their chondrotoxicity. 111
Hyaluronic acid is a polysaccharide normally contained in synovial fluid and articular cartilage with trophic as well as shock-absorbing effects. 44 The rationale for its intra-articular use is given by the possibility of ‘lubricating’ the joint and inducing the endogenous production of hyaluronate. Available formulations of this medical device differ according to the structure and molecular weight as well as the production process (i.e. extraction from animal tissues or bacterial fermentation). 112 Their use in MSK pain is controversial and is indicated mainly for OA. Although several clinical studies support the use of hyaluronic acid as it is effective on pain in the medium term, only a few international guidelines recommend its use for knee OA.11,16
In the last decade, botulinum toxin type A, a potent neurotoxin capable of inhibiting the presynaptic release of acetylcholine at the level of neuromuscular plaques to manage focal spasticity, has been studied for the management of different MSK pain conditions, such as myofascial pain and OA. 113 However, limited evidence supports its use in this context.31,105
Although the number of studies about minimally invasive procedures is increasing, limited evidence is available about this topic, especially for some agents (e.g. hyaluronic acid and platelet-rich plasma). 83
In our opinion, minimally invasive procedures are useful techniques for the treatment of MSK pain, aiming to improve efficacy and tolerability by reducing systemic side effects, although the risk of local adverse events might be increased. This approach needs adequate experience and a learning curve with the scope of improving accuracy and avoiding mistakes and adverse events. Finally, minimally invasive procedures should be included in a broader treatment plan along with other pharmacological and non-pharmacological therapies.
The role of placebo and nocebo effects in musculoskeletal pain
The management of MSK pain is one of the major fields of interest for the application of placebo and nocebo in medicine, whose effects have been studied in several conditions, such as low back and neck pain, shoulder pain and OA. 114 The Society for Interdisciplinary Studies (SIPS) on Placebo defines the placebo or nocebo effect as the changes attributable to neurobiological and psychological mechanisms produced by verbal instruction or nonverbal situations. 115 Therefore, the placebo response includes the placebo effect and is influenced by contextual factors in which the treatment is delivered and not by the specific action of the administered substance. 116
The effectiveness of a multimodal approach to MSK pain consisting of pharmacological and non-pharmacological interventions is partly attributable to the placebo effect.16,114
Randomized controlled trials suggest that physiological and psychological reactions are triggered in patients treated with a placebo linked to the patient’s personality type, the therapist–patient relationship and the type of environment in which the treatment is provided. Psychological reactions can evoke a therapeutic effect in conditions of chronic pain through three mechanisms: (1) conscious anticipation of the beneficial effect of the therapy, (2) conscious conditioning linked to the sight of specific equipment and (3) unconditioned stimulus following the administration of the therapy. 117 The contextual factors (doctor’s uniform, patient expectations, verbal communication, etc.) have recently been proposed as responsible for the initiation of the placebo and nocebo effects in patients suffering from MSK pain. According to a survey conducted in Italy, patients with MSK pain expressed themselves positively about the adoption of contextual factors in clinical practice, hoping for their greater adoption in the medical field. 118
The placebo effect has been studied in various MSK pain conditions.119,120
Unlike the placebo effect, there are relatively few studies investigating the nocebo effect in relation to the treatment of MSK pain. The importance of the nocebo effect in patients with MSK pain lies in the possible impact it can have on treatment adherence, clinical outcomes and healthcare policy.117,121 Meta-analyses of RCTs involving patients with MSK disorders showed that, due to the nocebo effect, discontinuation of treatment by participants in the placebo arm is very common. 122 A typical example of a nocebo effect in clinical practice concerns the use of statins and the occurrence of MSK pain after intake. In a study conducted in 2021, it emerged that negative expectations regarding the occurrence of adverse events could lead patients to erroneously attribute the appearance of muscle symptoms to treatment with statins. 123
The neurophysiological basis of a placebo effect in the management of MSK pain conditions is complex and concerns psycho-neuro-endocrine-immunology. 124 Different psychological mechanisms have been proposed as responsible for the placebo effect in the treatment of MSK pain, including conscious anticipation of the benefit induced by the therapy, conscious conditioning due to the sight of equipment used for minimally invasive procedures, and the unconditioned stimulus due to the administration of the therapy. 125 Placebo analgesia results from several pain modulation mechanisms, such as the activation of descending circuits involving the dorsolateral prefrontal cortex and periaqueductal grey matter; the inhibition of the middle and posterior cingulate cortex, the insula and the thalamus; cholecystokinin inhibition (CCK); the activation of the endocannabinoid system and modulation of prostaglandin synthesis. 125
In the therapeutic context of MSK pain, particularly for OA, a placebo would appear to be a very effective resource, justifying a significant reduction in pain, stiffness and functional limitation (up to 75%, 71% and 83%, respectively), even when administered by intra-articular route. 126 This approach could be useful in the context of a personalized treatment strategy, according to the stage of the disease and taking into account the contribution of the peripheral and central mechanisms underlying OA pain. For example, in the early stages of OA, exercise therapy and NSAID use might be more beneficial considering the major role of peripheral risk factors, such as synovial radiographic changes and subchondral bone, on joint pain, whereas in the intermediate and advanced stages of OA the use of intra-articular agents could be useful in consideration of the greater analgesic effect linked to the placebo (−47%) and the contribution of both peripheral and central risk factors (depression, anxiety and pain catastrophizing). 127
Despite the potential benefits of a placebo, its use is often limited by ethical issues because it is an inert substance or procedure for treating patients. 126 This aspect is particularly of interest, making us wonder why a placebo is not used in clinical practice. The ethical problem with placebo treatment is not that the patient is receiving ineffective medicine that often is equally effective as drugs with no risk of adverse events. The critical issue is that the doctor is deceiving the patient. 128 In clinical research, a placebo should be preferred as a comparator in clinical trials, according to the main pharmaceutical regulatory agencies. On the other hand, studies with smaller placebo effects (studies with positive results) are more likely to be published, even in the field of OA. 127
In patients with OA, the placebo effect should be considered an integral part of the effects of one or more treatments in clinical practice (‘efficacy paradox’), especially in the context of intra-articular therapy.126,129 With efficacy paradox one means the difference in the effect of a given treatment when tested in an RCT versus when tested in clinical practice. This gap arises because in RCTs, treatment efficacy is assessed by the separation between the treatment group and the placebo group (treatment-specific effect). For example, in several RCTs, including OA patients, the efficacy of hyaluronic acid is comparable to that of placebo, while in the case of NSAIDs, the efficacy is superior to placebo, but it is often not considered that the placebo effect is greater for interventional procedures than for systemic pharmacological intervention. 130 It is, therefore, clear that the placebo effect in the treatment of patients with OA is conditioned by several elements, in particular contextual factors. The weight of these factors can be assessed by measuring the proportion of the contextual effect (PCE), 131 which consists of the ratio between the changes in an outcome measure (from the baseline) in the placebo group to those obtained in the intervention group for the same outcome. In the case of OA, for example, the PCE for pain is 0.75; that is, 75% of the pain reduction is attributable to contextual factors, while for joint stiffness, it even reaches 100% if one proceeds to joint washing (Figure 2).

Key issues concerning pharmacotherapy of musculoskeletal pain.
Conclusion
Musculoskeletal pain is an umbrella term for a variety of conditions of different etiologies and different disease trajectories but taken together, they represent a substantial burden on patients, society and the healthcare system. It can be secondary to (or exacerbated by) multiple etiologies and often responds to a multimodal therapeutic approach by targeting different pathogenic mechanisms. However, the mechanistic approach might be ineffective; thus, the pharmacotherapy of MSK pain conditions should be included in a biopsychosocial approach that requires the evaluation of clinical and contextual factors (comorbidity, lifestyle, patient preferences and expectations, mental health). In this scenario, pharmacotherapy of MSK pain should be prescribed through a shared decision-making model where both patient and prescriber evaluate the risks and benefits of various therapeutic choices.
