Abstract
Background:
Ultrasound (US) has a high sensitivity in detecting monosodium urate (MSU) deposition in gout patients. However, the value of US in predicting gout flares has been reported only in a few monocentric studies.
Objective:
To investigate the association between gout flares in the previous year and US-detected MSU burden using two different US scores.
Design:
A retrospective study.
Methods:
Patients with gout were consecutively recruited to undergo musculoskeletal US examinations of their knees, ankles, and feet. The score derived from Outcome Measure in Rheumatology (hereinafter referred to as MSU score) and musculoskeletal US features-based (hereinafter referred to as MSKF score) were used to quantify the MSU burden of gout. Odds ratios for frequent gout flares were calculated.
Results:
We enrolled 1894 patients with gout (mean age: 45 years; gout duration: 5 years; males: 96.1%), experiencing a median of three flares over the past year. Of these, 428 (22.6%) patients reported frequent (⩾7) gout flares. The MSU and MSKF median scores were 6 and 9, respectively. For each five-point increase in MSU and MSKF score, the odds ratio of frequent gout flares increased 1.13-fold and 1.24-fold, respectively. The area under the curve (AUC) for the MSU and MSKF score was 0.635 [95% confidence interval (CI): 0.604–0.665] and 0.688 (95% CI: 0.659–0.718), respectively, (AUC difference 0.054, p value for AUC difference < 0.001).
Conclusion:
The MSU and MSKF scores were significantly associated with the number of gout flares in the previous year. The MSKF score outperformed the MSU score in terms of frequent gout flare discrimination.
Introduction
Gout is a common inflammatory arthritis that leads to progressive joint destruction and disability. The deposition of monosodium urate (MSU) crystals in articular and peri-articular structures can trigger gout flares, which are the main cause of joint injury and disease progression and are associated with disease severity and poor functional outcomes.1,2 Recurrent gout flares may further aggravate joint damage. Therefore, it is essential to identify patients with recurrent flares early to strengthen intervention and management strategies to avoid disability. Previous studies have identified risk factors for gout flares, including elevated serum urate levels,3,4 a long gout duration, 5 subcutaneous tophi, 6 meals high in purine, 7 alcohol consumption, 8 male sex, and advancing age. 9
Ultrasonography was recommended to aid in the diagnosis of gout owing to its low cost, widespread availability, and absence of radiation exposure in 2015. 10 Furthermore, the Outcome Measures in Rheumatology (OMERACT) ultrasound (US) group had defined three specific types of MSU deposition – double contour (DC), tophi, and aggregates, 11 based on US examination. Previous studies have reported the high sensitivity of US in detecting MSU deposition.12,13 To semiquantify MSU deposition, two recent studies introduced a scoring system (hereinafter referred to as MSU score) to obtain a sum score of MSU deposition and evaluated its sensitivity to changes in US-detected MSU deposition during urate-lowering therapy.14,15 Therefore, this MSU score is mainly used to monitor the treatment effect of gout. However, the association between this score (MSU score) and the risk of gout flares has not been well established. In a 12-month prospective observational study published in August 2022, Cipolletta et al. 16 enrolled 81 gout patients, using MSU score to quantify MSU deposition, and found that MSU deposition was an independent predictor of gout flares. Although this study provided a great strategy for gout prediction, more studies, with larger sample sizes, are needed to validate this score’s value in gout prediction.
In our previous study, 17 we developed a musculoskeletal US features-based score (hereafter referred to as the MSKF score) that could effectively evaluate the joint injury of gout using six elementary lesions (DC sign, aggregates, synovial hypertrophy, synovial effusion, tophi, and bone erosion). However, our previous study did not include an analysis of the correlation between US score and gout flares. Therefore, in this current study, we aimed to assess the MSU burden using the MSU score and MSKF score based on musculoskeletal US examinations. And furtherly explore the association between gout flares in the previous year and US score to provide clinical support for identifying frequent gout flares.
Methods
Study design and patients
In this retrospective, cross-sectional, single-center study, we consecutively included patients with a diagnosis of gout, in accordance with ACR/EULAR 2015 gout classification criteria, 10 from Outpatient Endocrinology Clinics in the Shanghai Tenth People’s Hospital from April 2015 to March 2021. Patients aged ⩾18 years and undergoing musculoskeletal US examination of their joints were included. We excluded patients with other types of inflammatory arthritis. Patients who had ever received uric acid-lowering agents before the enrollment were also excluded.
Clinical and laboratory assessments
All patients completed a unified questionnaire at enrollment that included demographic characteristics, gout history, the number of flares over the last year, comorbidities, and any history of previous or current smoking. Alcohol consumption was dichotomized into alcohol and non-alcohol drinkers. Gout flare was defined according to the ACR/EULAR 2015 gout classification criteria. 10 Pain in the joints or periarticular tissues can be described as stabbing, gnawing, or burning. The severity of pain, the time of pain from onset to peak intensity, is usually less than 24 h. It may be accompanied by swelling, warmth, and erythema. These symptoms are completely relieved between the intercritical period until another gout flare occurs. Our professional assistants in our clinic would introduce and confirm these details to patients to avoid misjudgment in medical history.
Physical examinations, including height, weight, waist circumference, hip circumference, and blood pressure, were performed according to standard protocols. Body mass index (BMI) was calculated as the weight in kilograms divided by the height in meters squared. All patients underwent standard laboratory evaluations of fasting plasma glucose (FBG), glycosylated hemoglobin A1c (HbA1c), alanine aminotransferase (ALT), aspartate aminotransferase (AST), serum creatinine (SCr), serum uric acid (SUA), C-reactive protein (CRP), the erythrocyte sedimentation rate (ESR) and lipid profiles.
Sonographic assessment
US assessments were conducted using a MyLab Twice (2014) US machine (ESAOTE GROUP, Italy) equipped with a 12–14 MHz transducer, operated by one of two trained musculoskeletal sonographers (JLX and CJ) who had 20 and 10 years of experience in ultrasonic examinations, respectively. Intra- and interobserver reproducibility of US scanning by the two sonographers was assessed in 60 randomly selected US images involving patients with gout. The intraclass correlation coefficients were 0.93 and 0.84, respectively.
US scanning encompassed the examination of articular structures and soft tissues of the knees, ankles, and first metatarsophalangeal (MTP) joints in terms of the following six features: DC sign, aggregates, synovial hypertrophy, synovial effusion, tophi, and bone erosion. 11 For each participant, previous comprehensive and qualified US images were identified and retrospectively scored. The MSU score for MSU deposition was evaluated according to OMERACT definitions as the sum score of each elementary lesion across the scanned sites (aggregate score, DC sign score, tophi score: score 0, none; score 1, possible; score 2, certain; and score 3, major).11,14
In addition, gout-related MSU burden was assessed using the MSKF score. In our previous study, 17 616 male patients were included: 245 with asymptomatic hyperuricemia and 371 with intercritical gout. The US signs which were significantly different between groups were subjected to binary logistic regression analysis to identify discriminative signs. The sifted US signs were scored based on their odds ratios, which were then used to evaluate the severity of gout and asymptomatic hyperuricemia. The performance of the US score was validated in an additional population including 163 patients with asymptomatic hyperuricemia and 196 patients with gout.
The MSKF score was assessed using a binary score (presence/absence). 17 The details were as follows: (i) knee joint: synovial effusion, two points; tophus, five points; (ii) ankle joint: synovial effusion, two points; synovial hypertrophy, five points; tophus, three points; bone erosion, seven points; and (iii) first MTP, DC sign; two points, synovial hypertrophy; three points, tophus; nine points, bone erosion; four points (Supplemental Material Table S1). The severity of the joint injury was classified into four grades according to the MSKF score (grade 1: 0–6, slightly impaired; grade 2: 7–12, moderately impaired; grade 3: 13–25, severely impaired; grade 4: 26–42, extremely severely impaired).
In the present study, we observed not only MSU deposition (DC sign, aggregates, tophi) but also synovial inflammation (synovial hypertrophy, synovial effusion) and structural damage (bone erosion) caused by MSU deposition. To distinguish this comprehensive assessment (MSKF score) from MSU deposition (MSU score) alone, we have chosen to use the term ‘burden’ to encompass MSU deposition, synovial inflammation, and structural damage.
Statistical analysis
Continuous variables were described as mean ± standard deviation or median (interquartile range), and Student’s t- or nonparametric tests were used for statistical comparisons. Categorical variables are presented as counts (percentages), and comparisons between groups were made using χ2 or Fisher’s exact tests.
Individuals with the highest quartile (⩾7) were defined as having frequent gout flares. Logistic regression analysis was then applied to determine the association between the US score and frequent gout flares, covariates including the significant variables (p < 0.1) in the univariable analysis, and potential clinically relevant parameters. Multicollinearity was examined in all logistic regression models using the variance inflation factor. We adjusted for variables including age, sex, BMI, waist circumference, alcohol consumption, family history of gout, hypertension, and renal stones; HbA1c, triglyceride, SCr, and SUA levels; and the ESR. Results are expressed as odds ratios (ORs) with 95% confidence intervals (95% CIs).
The value of the MSU and MSKF score in discriminating frequent gout flares was analyzed using the area under the receiver operating characteristic (ROC) curves (AUC) and compared as recommended by DeLong et al. 18 The optimal cutoff was defined as the value that maximized the Youden index. 19
In accordance with a previous study, 20 sensitivity analyses were performed by defining flares ⩾2 as having frequent gout flares.
A two-tailed p value of <0.05 was considered statistically significant. All statistical analyses were performed using SPSS (version 23.0; SPSS, Chicago, IL, USA) and R version 3.6.1 (R Foundation for Statistical Computing, Vienna, Austria) software.
Ethics and reporting standards
This study was approved by the Ethics Committee of Shanghai Tenth People’s Hospital (approval number: SHSY-IEC-5.0/22K265/P01) and registered at the Chinese Clinical Trial Registry (ChiCTR2200065728). The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology statement. 21
Results
Clinical and laboratory characteristics
We enrolled 1894 patients with a median of three flares over the past year. MSU burden was assessed using musculoskeletal US examinations. We categorized the included samples into four groups according to quartiles of gout flares (quartile 1, 0–1; quartile 2, 2–3; quartile 3, 4–6; and quartile 4, ⩾7). The demographic, clinical, and laboratory characteristics are described in Table 1.
Patient characteristics stratified by the quantiles of the declared number of flares over the past year.

Data are expressed as n (%), mean ± SD, or median (25th–75th percentiles).
ALT, alanine aminotransferase; AST, aspartate aminotransferase; BMI, body mass index; CRP, C-reactive protein; DBP, diastolic blood pressure; ESR, erythrocyte sedimentation rate; FPG, fasting plasma glucose; HbA1c, glycosylated hemoglobin; HC, hip circumference; HDL-c, high-density lipoprotein cholesterol; LDL-c, low-density lipoprotein cholesterol; SBP, systolic blood pressure; WC, waist circumference.
The mean patient age was 45 years, and 1820 (96.1%) patients were men. The mean duration of gout was 5 years. A total of 766 (40.4%) patients had renal stones, 653 (34.5%) had a family history of gout, and 456 (24.1%) had subcutaneous tophi. The median SUA and SCr levels were 508.7 and 89.1 μmol/L, respectively. Patients with more flares had a longer gout duration, a larger proportion of subcutaneous tophi, and higher SUA, ESR, and CRP levels (all p < 0.001). However, no difference was found between the quartiles in terms of a family history of gout.
Sonographic findings
The first MTP joint was the most frequent location for all signs, and synovial fluid was most frequently observed among all the signs, followed by the DC contour sign (Supplemental Material Figure S1).
Characteristics of the sonographic signs at different locations are presented in Table 2. The median MSU and MSKF scores were 6 and 9, respectively. The MSU and MSKF scores exhibited an upward trend corresponding to increasing quartiles of flares (quartiles 1–4: 4 versus 5 versus 8 versus 11; both p < 0.001; quartiles 1–4: 6 versus 7 versus 12 versus 17; Table 2). In addition, patients experiencing a higher frequency of flares demonstrated elevated MSU and MSKF scores (Figure 1).
Characteristics of sonographic signs at knee, ankle, and MTP joints.

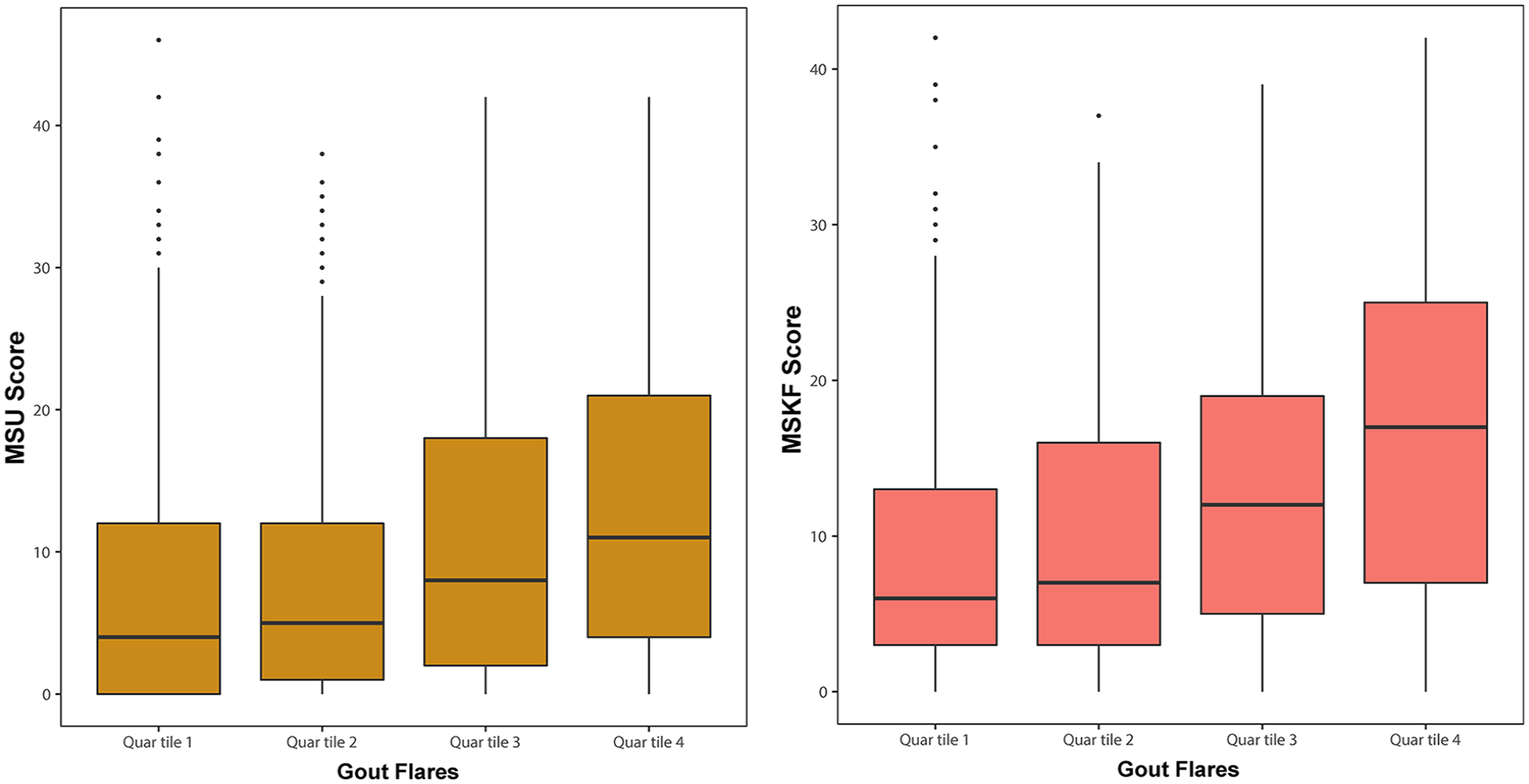
US score for gout flares in quartiles.
The association between gout flares in the previous year and the US score
The US score showed an increasing trend with quartiles of flares. Consequently, we further explored the association between the US score and gout flares to determine whether the US score was an independent risk factor for frequent gout flares.
When defining the patients with the highest quartile (⩾7) as having frequent gout flares, univariate regression analysis showed that the MSU and MSKF scores significantly correlated with frequent gout flares (both p < 0.001, Supplemental Material Table S2). Moreover, the significant discriminative value of the US score was preserved after adjusting for the following variables: age, sex, BMI, waist circumference, alcohol consumption, family history of gout, hypertension, and renal stones; HbA1C, triglycerides, SCr, SUA levels, and ESR.
In the fully adjusted model, the MSKF score was measured as a continuous variable, and a five-point increase in the MSKF score was found to be associated with a 1.24-fold OR of flare progression [OR 1.24 (95% CI: 1.13–1.37), p < 0.0001]. In the second model in which the MSKF score was described as a grading variable, the ORs (95% CIs) for flare progression when comparing MSKF scores in grades 2, 3, and 4 with that in grade 1 were 1.8 (95% CI: 1.10–2.95), 1.78 (95% CI: 1.10–2.86), and 3.49 (95% CI: 1.90–6.38), respectively. In Model 3, for every five points in the MSU score, there was an approximately 13% greater OR of frequent gout flares (95% CI: 1.04–1.23; Table 3).
Logistic regression analysis of the association between ultrasound score and frequent gout flares.

Logistic regression analysis also identified smoking (OR 1.56, 95% CI: 1.10–2.20; p = 0.012), duration of gout (OR 1.06, 95% CI: 1.04–1.09; p < 0.001), subcutaneous tophi (OR 2.06, 95% CI: 1.39–3.07; p < 0.001), AST (OR 1.02, 95% CI: 1.01–1.03; p < 0.001), and CRP (OR 1.01, 95% CI: 1.00–1.02; p = 0.038) as independent factors associated with frequent gout flares.
Spline curves between the continuous US score and the OR of frequent gout flares are presented in Figure 2. A continuous association was observed between the US score and the OR of frequent gout flares.

Spline curves between continuous US scores and the OR of frequent gout flares.
To compare the value of the MSU and MSKF scores in discriminating frequent gout flares, ROC analysis was performed (Figure 3). AUC values for the MSU and MSKF scores were 0.635 (95% CI: 0.604–0.665) and 0.688 (95% CI: 0.659–0.718), respectively (AUC difference, 0.054; p value for AUC difference < 0.001). The optimal threshold scores for MSU and MSKF that best discriminated patients with and without frequent gout flares were 12.5 (specificity, 0.74; sensitivity, 0.46) and 9.5 (specificity, 0.58; sensitivity, 0.71) points, respectively.

Comparison of ROC for the discrimination of patients with a declared number of flares ⩾7.
To assess the robustness of our findings, sensitivity analyses were performed by defining the number of flares ⩾2 as frequent gout flares in the fully adjusted model (Supplemental Material Table S3, Figure S2). The association remained robust across the sensitivity analyses.
Discussion
In this Chinese population-based study, we used a large sample size (n = 1894) to characterize the MSU burden in patients with gout, using semiquantitative scores with musculoskeletal US examinations. When comparing the MSU and MSKF scores, we found that both scores were associated with the number of frequent gout flares in the previous year and that the MSKF score performed better. Importantly, our data showed a surprising consequence, logistic regression analysis indicated that the MSKF score was an independent risk factor regardless of whether the cutoff point of frequent gout flares was set at 2 or 7 flares. The MSU score did not reach statistical significance in the sensitivity analyses where flares ⩾2 were defined as frequent gout flares. Moreover, the higher AUC value in the MSKF score than that in the MSU score suggested that the MSKF score had better discriminative ability than the MSU score in relation to frequent gout flares in the previous year. This is a novel finding, and the inclusion of additional US signs may explain this outcome. MSU deposition, soft tissue inflammation, and bone erosion are common manifestations of the MSU burden in patients with gout. Previous clinical and ultrasonic studies have considered MSU deposition, synovitis, and bone erosion separately, and no studies have combined them. Cipolletta et al. enrolled 81 gout patients, using the MSU score and Power Doppler score to quantify MSU deposition and synovitis, to assess the power of US findings (MSU deposition and synovitis) in predicting future gout flares. Their results suggested that baseline ultrasonography findings indicating MSU deposition and synovitis were independent predictors of gout flares. 16 Nicola Dalbeth et al. have applied CT scans to scoring the presence of bone erosions in a randomized controlled trial and observed that patients who had experienced a gout flare in the preceding year showed higher CT erosion scores, although the changes over time were small. 22 An important distinction was that, in our study, the weight of six ultrasonic signs in three joints was comprehensively evaluated to obtain the MSKF score, which can be used to describe the MSU burden, including MSU deposition, synovitis, and bone erosion.
Our statistical evaluations showed the superiority of MSKF over MSU score, with an AUC difference of 0.054 (p < 0.001). The optimal cutoff scores for the MSU and MSKF to discriminate patients with and without frequent gout flares were 12.5 and 9.5, according to the Youden index. The OR of frequent gout flares demonstrated an incremental increase with higher US score.
Unlike the MSKF score, the MSU score has been mainly applied to evaluate the severity of MSU deposition. Previous studies have not reached a consensus on the association between US-detected MSU deposition and gout flares; however, most studies have shown a significant association between US-detected MSU deposition and gout flares,16,23–25 indicating that US-detected MSU deposition may be a reliable predictor of gout flares. A 12-month observational study reported that the DC sign measured using US was not predictive of the risk of flares. 26 One reason for this negative finding may be that study did not count other ultrasonographic signs, such as aggregates and tophi. Our investigation revealed an association between the extent of MSU deposition and frequent gout flares using the MSU score, patients in the group with higher gout flares had elevated MSU scores.
In studies related to ultrasonography and gout, some have performed more comprehensive ultrasonography, including multiple joints of the upper and lower limbs.14,27 In our study, US scanning included only three joints (bilateral knees, ankles, and the first MTP joints); however, this did not affect the validity of our findings. Assessment of the three joints showed a predictive power comparable to the extended scanning of 12 joints of the upper and lower limbs. 16 In addition, it is time-consuming to perform multi-joint US without influencing its effectiveness, and a reduced three-joint examination could be more appropriate and helpful in saving time and reducing costs.
In contrast to prospective controlled studies, the current study utilized real-world data, yielding clinically significant results. There were few restrictions on the inclusion criteria, which were both an advantage and a disadvantage in this study. The data are therefore closely representative of real-world contemporary clinical practice.
Nevertheless, this study had certain limitations. First, it was a single-center study, and ultrasonography was conducted by one of two trained sonographers at a time. Second, the cross-sectional nature of our study involving retrospective flares over the past year did not allow for causal inferences. Third, recall bias may have influenced the reported number of flares over the past year. Finally, data on the specific joints involved in the flares were not collected. Further research that records the flares of a single joint would be useful to investigate the US score in terms of predicting flares at a single joint.
Conclusion
In conclusion, both MSU and MSKF scores were significantly associated with the number of gout flares in the previous year, and the MSKF score had better discriminative ability in relation to frequent gout flares. Our findings suggest that extra caution should be exercised in preventing flares in patients with initially high MSU burden scores to avoid poor outcomes.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X241240837 – Supplemental material for The association between gout flares and monosodium urate burden assessed using musculoskeletal ultrasound in patients with gout
Supplemental material, sj-docx-1-tab-10.1177_1759720X241240837 for The association between gout flares and monosodium urate burden assessed using musculoskeletal ultrasound in patients with gout by Shaoling Yang, Xiaojing Lin, Yining Gao, Nan Liang, Yali Han, Hang Sun, Shen Qu and Haibing Chen in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-docx-2-tab-10.1177_1759720X241240837 – Supplemental material for The association between gout flares and monosodium urate burden assessed using musculoskeletal ultrasound in patients with gout
Supplemental material, sj-docx-2-tab-10.1177_1759720X241240837 for The association between gout flares and monosodium urate burden assessed using musculoskeletal ultrasound in patients with gout by Shaoling Yang, Xiaojing Lin, Yining Gao, Nan Liang, Yali Han, Hang Sun, Shen Qu and Haibing Chen in Therapeutic Advances in Musculoskeletal Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
