Abstract
Osteoporosis and fragility fractures (FFs) are considered critical health problems by the World Health Organization (WHO) because of high morbidity, mortality, and healthcare costs. The occurrence of a FF raises the risk of a subsequent fracture (refracture). The hip is the most common site of fragility refracture, and its onset is associated with a further increase in patient’s morbidity, mortality, and socioeconomic burden. Therefore, the prevention of refracture is essential. In this context, fracture liaison service (FLS) demonstrated to be able to reduce FF risk and also improve patients’ adherence to anti-osteoporotic treatments, particularly for bisphosphonates (BPs). However, long-term and high adherence to BPs may lead to atypical femoral fractures (AFFs). These latter are tensile side stress fractures of the femur, with high rates of complications, including delayed and non-healing. An effective FLS should be able to prevent both FF and AFF. A comprehensive and interdisciplinary approach, through the involvement and education of a dedicated team of healthcare professionals (i.e. orthopedic, geriatrician, primary care physician, rehabilitation team, and bone nurse) for evaluating both FF and AFF risks might be useful to improve the standard of care.
Keywords
Introduction
Osteoporosis and fragility fractures (FFs) are considered critical health problems by the World Health Organization (WHO) because of the high incidence, costs, and effects on the patient’s quality of life.1,2 Indeed, FFs are responsible for high Disability Adjusted Life Years (DALYs) and increased mortality. 3 Although the risk is exceptionally high in hip fractures, an increased mortality rate has been reported in all types of FF.4,5 Most commonly, FFs could be observed in the spine, distal radius, proximal humerus, and proximal femur.4,6–8 The latter is associated with some of the most relevant consequences of osteoporosis because of their incidence, the high level of disability, and the financial burden that is estimated to be equivalent to cardiovascular disease. 9 The incidence of hip fractures is expected to constantly increase worldwide,3,7,10 and the hip is the most common site of a second FF. 11 Most of these refractures could be observed early in the 3–4 years after the first FF. 11 The occurrence of a second FF further affects patients’ disability and DALYs, and therefore their prevention must be a priority for healthcare systems.11,12 To reduce the incidence of a second FF, several prevention programs have been proposed, and good outcomes were reported in terms of both mortality and treatment adherence.12–32 The fracture liaison service (FLS) is one of the proposed models to effectively manage a patient with an FF. Generally, FLS refers to a multidisciplinary functional coordination structure established in a third-level hospital. 32 The FLS is generally a virtual structure, bridging the various hospital units that could take on the charge of the patient with a surgically treated FF at various levels.32,33 The FLS model involves various physicians (orthopedist, internist, endocrinologist, rheumatologist, geriatrician, physiatrist, radiologist, and pain physician) dedicated to the management of FFs and eventually coordinated by a skilled nurse.32,34 This kind of approach has been proven to be cost-effective, with an average reduction of 20% in the treatment gap, a 20% increase in adherence to anti-fracture treatment, and a 5% reduction in the rate of re-fracture and mortality.12–32 Adherence to anti-fracture treatment is one of the pivotal points in the prevention of FFs. 35
Bisphosphonates are the most common drugs used to reduce bone resorption and prevent osteoporotic fractures.36–38 Long-term use of these drugs has been associated with some adverse events, such as osteonecrosis of the jaw and atypical femoral fractures (AFFs). These latter are stress fractures with defined diagnostic criteria as proposed by the American Society for Bone and Mineral Research (ASBMR). 39 AFFs may be complete or incomplete, with the latter that could be treated either surgically or non-operatively.40,41 The occurrence of an AFF may be a dramatic event considering the huge challenge for the orthopedic surgeon to achieve bone healing.41–44 Therefore, an effective FLS model should pay attention not only to the adherence to the anti-osteoporotic treatment but also to the appropriate management of antiresorptive drugs and the early detection of an AFF. This perspective review aimed to propose a practical approach for implementing the FLS model to prevent and/or early treat AFFs.
AFFs: Prevention and early diagnosis
AFF is a stress fracture involving the tensile side of the femur from the subtrochanteric to the supracondylar flare, with a mean incidence of 2.2 per 100,000 per year.39,41,45 The diagnosis of AFF is based on the ASBMR criteria emended in 2014 (see Table 1). 39 It has been proposed that one of the underlying mechanisms of BPs-related AFF was the accumulation of microcracks in a bone ‘frozen’ by the long-term and highly compliant use of the drug. 46 Therefore, the mechanism of action of BPs, as well as other antiresorptive drugs (i.e. denosumab), might be responsible not only for the occurrence of AFF but also for the difficulties in their treatment and bad outcomes. 41 In fact, AFF healing is generally delayed and the fixation of these fractures is prone to fail.41,44 It seems that some BPs (i.e. Alendronic and risedronic acids) are more prone to lead to an AFF. 47 This could be related to a different degree of impairment of bone vascularization due to a supposed action of BPs on endothelial cells. 48 Of note, in a recent pharmacovigilance study, the prolonged use of denosumab was associated with a risk of AFF even higher than alendronate and risedronic acids. 47 This could be probably related to the exceptionally high prevalence of AFF observed among denosumab high-dosage users for bone metastases.47,49 However, antiresorptive long-term therapy is not the only pathogenetic mechanism underlying AFF. 41 In fact, some observations lead to give a more relevant role to the changes in femoral geometry. 41 In particular, varus neck-shaft angle, and bowing femur, through a modification of the biomechanics of the femoral shaft, may lead to AFF not related to antiresorptive drugs.50–55 Koeppen et al. hypothesized that the distribution of tensile strain had a prominent role in the pathogenesis of subtrochanteric, whereas age-related cortical thinning in diaphyseal AFF. 56 These observations lead both Oh et al. and Toro et al. to identify two different types of AFF: BPs-related (BRAFF) and AFF not related to bone turnover suppression.41,53–55 Use of proton pump inhibitors, glucocorticoids, collagen diseases, chronic pulmonary disease, and asthma are among the other factors associated with AFF.57,58
ASBMR task force criteria of atypical femoral fractures. Fractures of the femoral neck, intertrochanteric fractures with spiral subtrochanteric extension, periprosthetic fractures, and pathological fractures associated with primary or metastatic bone tumors and miscellaneous bone diseases (e.g. Paget’s disease, fibrous dysplasia) are excluded. 39

ASBMR, American Society for Bone and Mineral Research.
A prompt diagnosis of AFF is relevant to start the most appropriate treatment.40,41 To effectively diagnose AFF, an appropriate clinical and radiographical evaluation is needed.
Adequate patient history and physical examination are essential to make the diagnosis of AFF, particularly in cases of incomplete fractures. Prodromal thigh or groin pain is extremely common, 59 so much so that both the Food and Drugs Administration and the European Medicines Agency recommended paying extreme attention to the appearance of such pain in long-term BP users.57,59–61 Obviously, a complete fracture should be characterized by severe pain that appeared suddenly after a history of chronic thigh or groin pain. In clinical examination, a lower limb alignment assessment might be of aid, considering that varus deformity and femoral bowing are considered risk factors for AFF. 41
Radiographically, the AFFs are characterized by some specific features, which generally lead to their diagnosis following the ASBMR task force criteria (see Tables 1 and 2 for further details). 39 Of note, the focal cortical thickening and the transverse fracture on the lateral side are considered the elements with the highest accuracy for the diagnosis of AFF. 62
Imaging modalities and common findings in AFF.
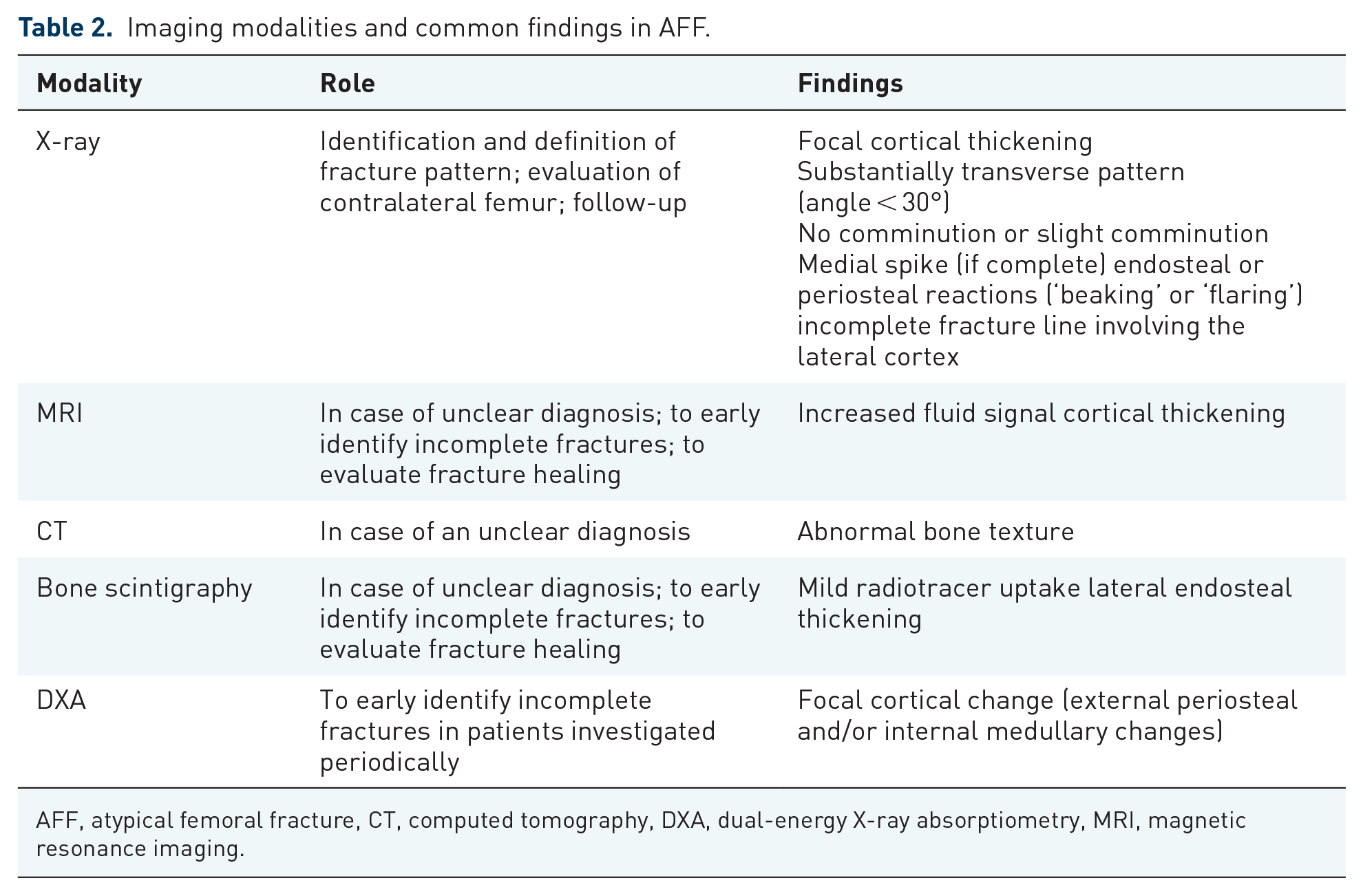
AFF, atypical femoral fracture, CT, computed tomography, DXA, dual-energy X-ray absorptiometry, MRI, magnetic resonance imaging.
Computed tomography (CT), magnetic resonance imaging, and other imaging modalities may be useful in doubtful cases, particularly in incomplete AFF. 63 The CT can demonstrate the presence of the abnormal bone texture underlying the fracture. 64 However, MRI is considered the most sensitive and specific imaging modality to identify stress and unknown fractures. 64 A fluid-intense signal (identifying the fracture line) and the lateral cortical thickening can be observed on the MRI of patients affected by AFF. 59
Bone scintigraphy demonstrated a high ability to early diagnose AFF. 65 Mild radiotracer uptake with endosteal thickening along the lateral proximal diaphysis is considered a relatively specific finding of AFF.41,66
Several authors suggested that dual-energy X-ray absorptiometry (DXA) scans could be useful for the early identification of AFFs.67–71 Generally, in these fractures, the DXA scan shows focal cortical changes (beaking) in the lateral side of the femur.70,72 Considering the promising results, McKenna et al. recommended performing extended femur DXA scans in all BP long-term users. 69 Although approximately one-fourth of DXA scans could be false negative, their reliability is still higher when compared with clinical evaluation alone. 70 The combination of a positive DXA scan with prodromal signs presents an exceptionally high detection rate of AFFs. 70 Considering the limits of extended femur DXA and the possible anxiety of patients continuously undergoing these evaluations, van de Laarschot et al. recommended carefully evaluating the lateral cortical bone of an extended femur DXA scan in case of (1) patients who have already sustained an AFF in the past; (2) bisphosphonate users who (upon specific inquiry) report pain in the hips, and groin, or upper legs; and (3) patients who have used antiresorptive treatment for more than 5 years or have other risk factors of developing an AFF such as long-term use of glucocorticoids. 72
Considering that long-term use of Bps is still one of the most important risk factors of AFF, appropriate management of antiresorptive therapy should be considered the most effective way to prevent their occurrence.
AFF risk drops by about 70% 1 year after BP discontinuation. 73 Interestingly, during this period, the BPs’ effects on fracture risk reduction are still observable. 74 Indeed, both the FLEX and the HORIZON trials reported a carry-over effect of BPs in fracture prevention, at 2 and 3 years after alendronate and zoledronic acid discontinuation, respectively. 74 Therefore, a drug holiday may be a reasonable compromise to effectively reduce AFF risk while still protecting our patients against other FFs.
The United Kingdom National Osteoporosis Guideline Group (NOGG) recommended a BP treatment re-assessment after 3 years in case of zoledronic acid use or 5 years in case of other drugs. The NOGG recommends that at this time point, the treatment should be continued only in high-risk patients (i.e. 75 years or older patients, patients with previous femoral neck or vertebral fracture, prednisone equivalent dose ⩾ 7.5 mg per day) or in case of T-score < 2.5. 75
A clearer approach to BP holiday was proposed in the update of the guidelines on postmenopausal osteoporosis emanated in 2020 by the American Association of Clinical Endocrinologists (ACCE) and American College of Endocrinology (ACE) (see Table 3). 76
ACCE/ACE recommendations for BP discontinuation.

Source: Adapted from Camacho et al.76
ACCE, American Association of Clinical Endocrinologists; ACE, American College of Endocrinology; AFF, atypical femoral fracture; BP, bisphosphonate; DXA, dual-energy X-ray absorptiometry.
According to ACCE and ACE guidelines, the ending of BP holidays should be based on specific circumstances, such as an increased risk of fractures, a decrease in Bone mineral density (BMD), or an increase in bone turnover markers. 76
Anyway, according to the ASBMR, if BPs are used for longer than 5 years, a yearly patient’s AFF and FF risk assessment should be done. 77 Particularly, they recommend a median BP treatment period of 7 years, and to start a drug holiday for patients with no history of any recent fractures and femoral neck T-scores above −2.5. Similar recommendations have been expressed by the Endocrine Society 78 and National Osteoporosis Foundation, 79 and both suggested a drug holiday after 3–5 years of BP use, for women at low to moderate risk of fractures.
However, as also suggested by ACCE and ACE guidelines, a safer way to identify those patients who most could benefit from drug holidays might be the evaluation of bone metabolism biomarkers. 80 Particularly, Statham et al. identify a subset of BP users with a suboptimal suppression of cross-linked C-telopeptide of type I collage (CTX) in which the drug holiday should be avoided. 81
Nayak and Greenspan, in a recent meta-analysis, analyzed the effect of BP holiday in terms of BMD and FF risk. 82 The authors reported that patients who discontinued BPs did not present a higher risk of hip fracture, compared to those who continued BPs, clearly suggesting a tail effect that could be used to prevent FF while lowering BRAFF risk. 82
Dell and Greene proposed a practical approach to prevent AFFs based on a global fracture risk assessment, including other FFs. 73 In particular, a BP holiday should be evaluated: (1) after at least 3–5 years of antiresorptive therapy in patients with a high risk of AFF and low-to-moderate risk of other FFs; (2) the occurrence of a new groin or tight pain; and (3) the occurrence of an AFF. 73
Although some other specific risk factors for AFF were reported (see Table 4),41,73,83–86 a tool to stratify AFF risk is not available. Although the scoring system proposed by Min et al. was not specifically developed for antiresorptive treatment discontinuation, it could be of aid in the decision-making in case of incomplete AFF. 87
Known risk factors for AFF.

AFF, atypical femoral fracture; BP, bisphosphonate.
Anyway, as a general rule, in the absence of high fracture risk and after a specific treatment duration (3–7 years, depending on the BPs used), drug holidays are safe and useful to reduce AFF risk. 80 However, considering the general safety of BPs and that anti-resorptive effects constantly reduce over time, a careful patient evaluation and drug resumption are advisable as soon as the risk of a new FF increases. 88
The rules of the BP drug holiday could not be applied to denosumab. 89 In fact, denosumab discontinuation is associated with a rapid reversal of its effects on bone remodeling resulting in an increased fracture risk.89,90 Interestingly, the detrimental effect of denosumab discontinuation is correlated with its time of use.89,91 In practice, the longer is the denosumab used, the worse the detrimental effect of its discontinuation on fracture risk. 91 However, the European Calcified Tissue Society (ECTS) emanated some recommendations in the case of AFF following denosumab use (Table 5). 89
ECTS recommendations for AFF in denosumab users (adapted from Noble et al. 89 ).

AFF, atypical femoral fracture; ECTS, European Calcified Tissue Society; FF, fragility fracture.
It is to underline that surgery may act in preventing the propagation of an incomplete AFF. 41 AFFs are typically surgically managed using intramedullary (IM) nailing. The preference toward IM nailing over plating is related to the endochondral fracture healing achievable with the nail. However, a high rate of intraoperative fracture was reported with the nailing of an AFF. 44 To reduce their incidence, some technical pearls have been proposed including overreaming, careful identification of the correct entry point, and thinner nails use.92,93 Plate fixation and elastic IM nailing are among the proposed techniques in case of very bowed or narrow femora.41,46,94–98 The use of prophylactic surgery in incomplete fractures seems to be associated with more reliable outcomes compared to the conservative treatment (mainly based on no weight bearing, supplementation, and/or anabolic drugs), especially in case of high risk of fracture progression (i.e. patients long time and/or high compliant BPs user; Proton-pump inhibitors (PPI) or glucocorticoid user; patients with a varus femur or a contralateral progress fracture). 41
The role of fracture liaison services in preventing FFs
The occurrence of an FF is a multifactorial event related to an osteoporotic bone with poor biomechanical characteristics, patients’ drug side effects, and motor function impairment.99,100 The FLS was shown to be effective in several countries in the reduction rate of second FF (also known as re-fractures), thanks to an integrated and multidisciplinary fracture care pathway. 99 FLS has been proven to be efficient and cost-effective.12–32 The efficacy in reducing the treatment gap, and increasing the adherence to the anti-osteoporotic treatment might partly explain the reduced mortality rate reported by González-Quevedo et al. after 1 and 2 years of the implementation of their FLS program.20,101 These observations were further confirmed by Li et al. in their recent meta-analysis. 102 The authors, analyzing the results of 16 studies, reported a reduction rate in both refracture and mortality with the implementation of FLS programs. 102 The clinical effects of FLS pathways are responsible also for the cost-saving reported with their implementation. In fact, it was estimated that the FLS program was associated with a cost-saving of £21,000 over the lifetimes of 1000 patients in the UK, and 617,275 per 1000 patients over a 3-year period in Australia.103–105 An effective FLS program improves the health of the elderly through a stepwise approach based on case findings, assessment of the patient’s bone quality and general health status, fall prevention, and physical exercises. 106 The FLS is considered a coordinated, multidisciplinary approach for secondary prevention that integrates inpatient (i.e. patients admitted to a hospital or rehabilitation center after an FF) and outpatient (i.e. patients recovered from the acute fracture who are seen in the outpatient service) settings. The final aim is to improve the healthcare systems’ quality in preventing FF, thanks to the effective use of diagnostic screening modalities [i.e. DXA, Radiofrequency echographic multi-spectrometry (REMS)], laboratory evaluation, patient education, therapeutic options, and adequate follow-up by devoted physicians and nurses.12,32,107 Based on the intensity of care, Ganda et al. identified four FLS models and classified them from A to D. 108 Type A is the most intensive and comprehensive model, consisting of a coordinated approach in which, following a fracture, the patient is identified, assessed, and treated by an all-encompassing service. 108 The main difference between Type A and Type B FLS models is related to the anti-osteoporotic treatment starting and primary care physician (PCP) involvement. 108 In fact, in Type B FLS, the PCP is delegated to start the anti-osteoporotic treatment. 108 The Type C model is a less intensive model, in which the pathway for the prevention of FF consists of the identification of the FF patients and their education on the diagnosis and lifestyle modifications needed. The need for further diagnostic work-up and anti-osteoporotic treatment is communicated with the PCP. 108 Finally, Type D is the least intensive model in which fractured patients received only education about osteoporosis, with no diagnostic or treatment initiation by the FLS and no involvement of or communication with the PCP. 108 Although more expensive, Type A FLS demonstrated the best outcomes in terms of case finding, treatment adherence, as well as refracture prevention. 108
A fundamental requirement for Type A FLS programs is the availability of a robust information technology service and network, to aid in the case finding, capturing key information, and allowing reliable and efficient follow-up.108,109 In fact, as formerly underlined, an effective FLS program should include several phases: case finding; patient general health assessment; bone quality assessment and osteoporosis treatment; fall prevention; patient education; and physical exercises.12–32 The reported most effective approaches in case finding are as follows: manual abstraction of cases; electronic reminder systems in the patient medical record; and electronic case finding diagnosis that automatically refers the patient to the FLS. 110 The patient assessment needs an accurate collection of the present fracture history, including the mechanism of injury, the general bone health, and the prior anti-osteoporotic treatment received, as well as medication history. In addition, a physical examination based on posture and balance observation, gait analysis, and identification of deformed or painful joints should be made when possible. 111 Obviously, a specific assessment of bone health is an essential task of an FLS program. The availability of the novel REMS method (an innovative non-ionizing approach based on the analysis of the raw unfiltered ultrasound signals acquired during an echographic scan of the lumbar spine and/or femoral neck) potentially allows evaluating patients’ BMD as they are hospitalized for a FF.112–115 When REMS is not available, DXA should be the modality of choice for bone quality assessment. Theoretically, a reliable assessment of the patient’s BMD should be based on DXA performed every 2 years using the same machine. 76 Specific laboratory examinations, including bone turnover biomarkers, should also be regularly evaluated.12,76 A fall prevention program should be started with the identification of all causes of falling, including treatable disease, drugs, or motor function impairments with the assistance of a physical therapist. An essential element of any high-performing FLS program is the patient and/or caregivers’ continuing education. This is generally made by the FLS case coordinator, commonly identified by a nurse. 12 A careful discussion on the relevance of fall and fracture prevention should be done, underlying the potential loss of quality of life, motivating constantly the patient to adhere to the treatment and the follow-up with the FLS provider. 116 Although there is no pharmacologic treatment specifically approved for sarcopenia, some evidence exists on the role of low vitamin D in muscle weakness and fall prevention,117,118 as well as in antiresorptive treatment response. 119
The role of fracture liaison services in preventing AFFs
An effective FLS should be able to prevent any further FF, including the AFFs. Therefore, a continuous evaluation of the patient’s fracture risk to adjust antiresorptive is advisable. We propose an FLS Type A model in which a pivotal role throughout the entire program should be played by both the orthopedic surgeon and a nurse devoted to FFs (bone nurse). This kind of approach might be able to assure an extremely high adherence to osteoporosis treatment.12,32,120 However, the starting point of an effective FLS program must be based on the case finding of FF. Therefore, the involvement of the emergency physician, the ward geriatrician, the PCP, and/or other bone specialists is advisable (Figures 1 and 2).

Healthcare staff involved and stepwise approach for FF case finding and entry into the FLS program.

Comprehensive Type A FLS model for FF and AFF prevention.
During the patient follow-up, the bone nurse, together with the orthopedic surgeon and/or other bone specialists, should constantly evaluate both the patient’s adherence to the therapy and any modification in fracture risk. This latter might be assessed using the same tools used for FF case identification (i.e. blood test; DXA scan; spine X-ray). However, to early identify an AFF, some further evaluations should be performed, including the extended femur DXA scan, clinical assessment of any prodromal sign, and other second-level imaging modalities (i.e. MRI or bone scan).
Finally, any modification in antiresorptive treatment should be done according to a comprehensive evaluation of both FF and AFF risk, paying attention to the timely start of anabolic treatment and performing prophylactic surgery when needed.
Conclusion
FF incidence is constantly increasing worldwide, and the occurrence of a single FF significantly raises the risk of a refracture. Both first FF and refractures are associated with high morbidity, mortality, and healthcare costs. The application of FLS has been proposed as a valuable model to prevent refractures. One of the main effects of FLS is the high patient adherence to anti-osteoporotic treatment. However, prolonged and highly compliant antiresorptive treatment might be associated with AFF. These latter might be dreadful events, considering the difficulties in achieving fracture healing. Therefore, an effective FLS should also prevent AFF. A comprehensive model that embraces also specific tools useful for preventing and early identifying AFFs might be preferable in the prevention of refracture. The constant monitoring of both FF and AFF risks by the multidisciplinary team would aid in enhancing FLS ability in the management of osteoporotic patients.
