Abstract
The fracture liaison service (FLS) care is considered the most appropriate organizational approach for secondary fracture prevention. We performed a literature search to evaluate to what extent the introduction of a FLS reduced subsequent fracture rates. We identified five studies that compared subsequent fracture rates. These studies varied in study design, proportion of women, baseline and subsequent fracture type [vertebral fracture (VF), non-VF (NVF) or hip fractures], duration of follow-up, response rates of attending the FLS, as well as variables included in adjusted analyses (age, sex, baseline fracture, time dependency). In two studies comparing hospitals with and without a FLS, the adjusted hazard ratio (HR) for subsequent fractures was significantly lower in the FLS hospitals (HR: 0.84 during the first year, 0.44 during the second year for subsequent NVFs after baseline NVF, and 0.67 during the third year for subsequent VFs + NVFs after baseline VFs + NVFs). When comparing fracture rates before (pre-FLS) and after (post-FLS) introduction of a FLS, the adjusted HR for subsequent NVFs after baseline NVF was significantly lower in the post-FLS group after 2 years in one study (HR = 0.65) and nonsignificant in another study for subsequent hip fractures after baseline hip fracture. One study comparing pre-FLS and post-FLS with a follow-up of less than a year did not demonstrate a significant difference in subsequent fracture risk. In conclusion, only five FLS studies with heterogeneous study designs are available, three of them reported a lower subsequent fracture rate related to FLS care. Larger and long-term studies will be needed to further quantify the effect of FLS care on subsequent fracture risk.
Introduction
Patients with a recent fragility fracture after the age of 50 years are at increased risk of sustaining a subsequent fracture. Patients with a nonvertebral fracture (non-VF) have a twofold higher risk of subsequent fractures as compared with individuals without a fracture. 1 Peri- and postmenopausal women with vertebral fractures (VF) even have a four-to-fivefold higher risk of subsequent VF.1,2 Subsequent fracture risk is highest within the first 2 years after a fracture3–7 and subsequent fractures are also associated with an increased morbidity and mortality resulting in a high financial burden for healthcare. 8
Despite the availability of good treatment options for osteoporosis that have proven to be effective in reducing subsequent fracture risk, patients who recently sustained a fracture often do not receive appropriate treatment. This large treatment gap is a problem in many countries. Studies have shown that as few as 10% of women with fragility fractures receive osteoporosis therapy.9,10 In 1999, McLellan and colleagues initiated the first fracture liaison service (FLS) in the UK, aiming at reduction of subsequent fracture risk in high-risk patients, namely those who sustained a recent fracture. 11 Since then, several FLSs have been initiated from different medical departments (i.e. internal medicine and orthopedic surgery) in hospitals all over the world.12,13 As supported by the International Osteoporosis Foundation (IOF), European League Against Rheumatism (EULAR)/European Federation of National Associations of Orthopaedics and Traumatology (EFORT) and American Society for Bone and Mineral Research (ASBMR) task force, FLSs are proposed as the most successful approach for secondary fracture prevention.14–16 This FLS approach starts with the identification of patients with a fragility fracture (case finding), followed by assessment of clinical risk factors for subsequent fractures and secondary osteoporosis (including bone density, imaging of spine, laboratory assessment), treatment initiation (if indicated) and adequate long-term follow-up to improve treatment adherence.14,15,17
The presence of a FLS increases diagnosis and treatment initiation and therefore it is assumed to reduce subsequent fracture rates.12,13 However, although FLSs are introduced and implemented worldwide, limited data are available on the effectiveness of FLS in reducing subsequent fracture rates. Therefore, we aimed to provide an overview of the efficacy of FLS care in reducing subsequent fracture rates.
Methods
An extensive literature search was conducted in PubMed and Embase to identify relevant manuscripts up to September 2016 using following keywords: ‘Fracture Liaison Service’ or ‘secondary fracture prevention’ combined with ‘re-fracture,’ ‘subsequent fracture,’ ‘response rate,’ ‘osteoporosis,’ ‘effectiveness.’ The search was limited to English-language articles. In addition, manual searches through the reference lists were performed. We selected only manuscripts that reported subsequent fracture rates related to FLS care. Studies in primary care or nursing home settings were excluded, as well as studies presenting community-based strategies, primary prevention or universal prevention strategies. In addition, manuscripts presenting studies of treatment persistence in a FLS cohort were also excluded.
Results
Search results
As shown in Figure 1, our literature search resulted in 114 articles, of which 92 were excluded based on title and abstract. We assessed 22 full-text articles and 17 were excluded because of several reasons: study outcome (n = 6), only description of FLS organization (n = 3), interview/letter about FLS (n = 3), study design (n = 2), duplicate data (n = 1), no FLS intervention (n = 1) and a review (n = 1). Finally, five papers remained to be included in this overview.18–22 The studies were conducted in three European countries (the Netherlands,20,21 Sweden 18 and United Kingdom 19 ) and one study was conducted in Australia. 22
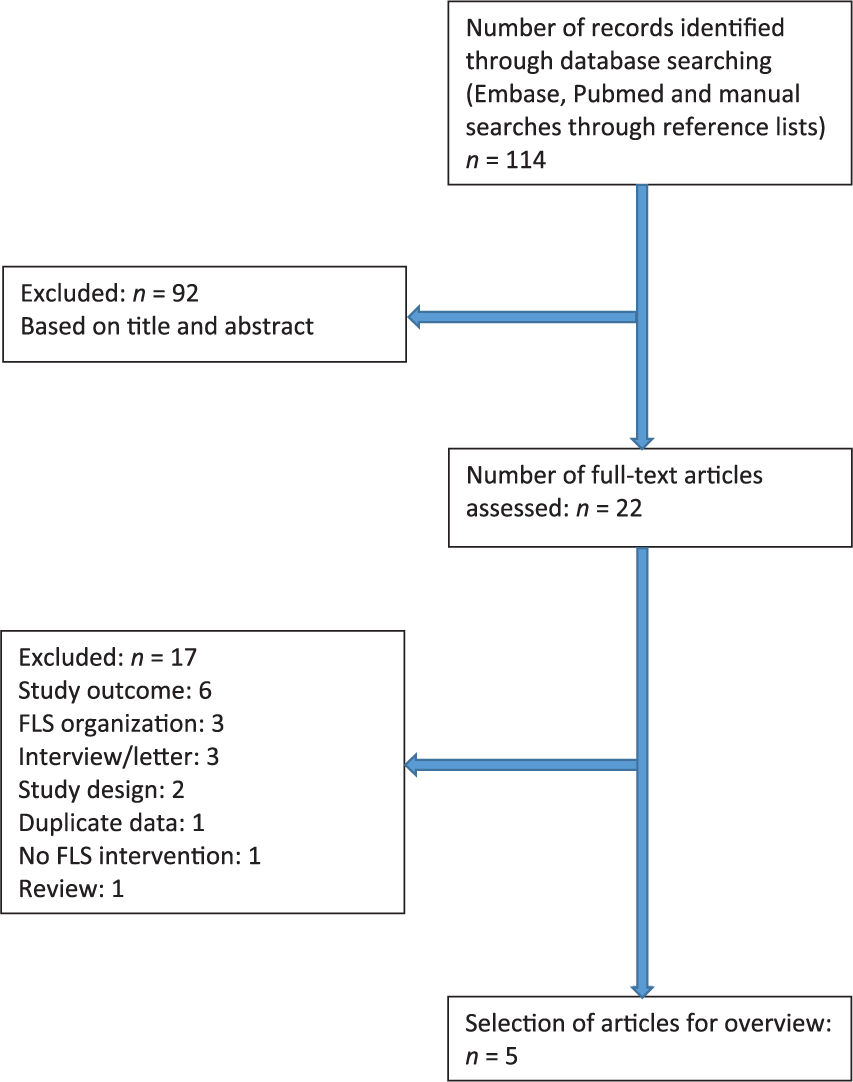
Flowchart of literature search.
Study design
As shown in Table 1, all five studies were designed as cohort studies, three with a prospective design18,20,21 and two with a historical/retrospective design,19,22 with follow-up duration varying from 341 days 18 to 3 years. 22
Characteristics of studies that assessed the effect of fracture liaison service care on subsequent fracture rates.

Prospective design, **nonsignificant, $significant, §calculated.
HR, hazard ratio; CI, confidence interval; VF, vertebral fracture; NVF, nonvertebral fracture; FLS, fracture liaison service.
Authors of three studies assessed subsequent fracture rates before (pre-FLS) and after (post-FLS) introduction of FLS.18,19,21 Huntjens et al. 20 and Nakayama et al. 22 assessed subsequent fracture rates in patients visiting a FLS hospital versus a hospital without FLS (no-FLS hospital). Hawley et al. 19 reported the results form a post-hip fracture care model in 11 hospitals, of which eight hospitals changed to a nurse-led FLS or orthogeriatrician model of care. Each hospital was analyzed separately and acted as its own control in a before-after time-series design, in which the appointment of an orthogeriatrician or set-up/expansion of a FLS was evaluated. 19
Fracture liaison service organization
Table 2 presents the FLS organization models. In the two studies of Huntjens et al.,20,21 case finding was performed by a fracture nurse as central coordinator, in contrast to Axelsson et al., 18 where screening was not performed by a central coordinator. Nakayama, et al. 22 did not specify whether there was a central coordinator responsible for case finding. Axelsson et al. 18 and Nakayama et al. 22 reported that patients were identified by the secretaries at the emergency department or were automatically invited.
Organization of fracture liaison service care in studies that assessed subsequent fracture rates.

BMD, bone mineral density; DXA, dual energy X-ray absorptiometry; FRAX, fracture risk assessment tool; MTF, minor trauma fracture; FLS, fracture liaison service.
Not all FLSs reported systematic laboratory testing for evaluation of secondary osteoporosis or metabolic bone disorders. In the paper of Axelsson et al., 18 primary care physicians were responsible for ruling out secondary osteoporosis. Huntjens et al. 21 did not specify whether laboratory testing was undertaken; Huntjens et al. 20 specified the laboratory tests; Nakayama et al. 22 only described that a laboratory assessment was performed. Treatment was initiated in accordance to local, national or international guidelines, as reported by Huntjens et al., 21 Huntjens et al. 20 and Axelsson et al., 18 and Nakayama et al. 22 did not report if the initiated treatment was in accordance with guidelines. Axelsson et al. 18 only gave a treatment advice to the general practitioner. None of the papers reported how follow-up of treatment was organized or adherence with anti-osteoporosis medication was assessed.
Hawley, et al. 19 did not specify, either, how case finding was performed, which assessments were conducted, nor how treatment was initiated in the different hospitals that were included in the longitudinal study.
Attendance rate
Attendance rate is defined as the number of patients actually attending the FLS divided by the number of the eligible patients being invited for the FLS. As shown in Table 1, Huntjens et al.20,21 reported an attendance rate of 67.8% and 68.4%, respectively. In the study of Nakayama et al., 22 an attendance rate of 20% could be calculated. Attendance rate was not reported in the studies of Axelsson et al. 18 and Hawley et al. 19
Fracture liaison service population
As presented in Table 1, all studies included both sexes, with 70% 20 up to 75%19, 22 women. With respect to age, four studies included patients aged 50 years and older,18,20–22 and one study, patients older than 60 years. 19 Mean age ranged from 70 20 to 83 years. 19 Axelsson et al. 18 and Nakayama et al. 22 included patients with non-VFs and VFs as baseline fractures and investigated non-VFs and VFs as subsequent fractures. Huntjens et al.21,20 included only patients with non-VFs and studied non-VFs as subsequent fractures of interest, while Hawley et al. 19 included only patients with hip fractures and also, only hip fractures as subsequent fractures. Samples sizes ranged from 931 22 to 33,152. 19
Subsequent fractures
Subsequent fracture rate is reported in all studies, and in the FLS cohorts, the rate was 6.7% in the patients with subsequent NVFs after baseline NVFs during a follow-up period of 2 years, 8.3% and 12.2% in patients with subsequent VFs and NVFs after baseline VFs and NVFs during a follow-up of 2 years and 3 years, respectively (Table 1).18,20–22 The subsequent fracture rate in the no-FLS cohorts was 6.8% and 9.9% in patients with subsequent NVFs after baseline NVFs during of follow-up of 2 years, and 8.4% and 16.8% in patients with subsequent VFs and NVFs after baseline VFs and NVFs during 2 and 3 years’ follow-up, respectively.18,20–22 Hawley et al. 19 reported a subsequent fracture rate of 4.2% for the whole cohort of hip fractures (Table 1).
Axelsson et al. 18 used a time-dependent Cox model adjusting for age, sex, previous fracture, index fracture, prevalent medical treatment, prevalent calcium or vitamin D treatment, prevalent glucocorticoid treatment, prevalent osteoporosis diagnosis, rheumatoid arthritis, secondary osteoporosis according to FRAX and a modified Charlson morbidity index. The fully adjusted hazard ratio (HR) was 0.95, 0.79–1.14, p = 0.60 (Table 1). 18
Besides adjustments for age and sex, Hawley et al. 19 adjusted the analyses for index of multiple deprivation score and Charlson comorbidity index. The analyses showed no significant difference in the pre- and post-FLS care [HR = 1.03 (0.85–1.26)] and no significant difference in the pre- versus postorthogeriatric service model [HR = 0.95 (0.79–1.15)]. 19
Both studies of Huntjens et al. performed adjustments for age, sex and baseline fracture.20,21 Huntjens et al. 20 showed no significant difference in the proportion of subsequent fractures between the FLS and no-FLS cohort. The time-dependent Cox model showed a significantly lower time-dependent subsequent fracture risk in the FLS group compared with the no-FLS group: HR time dependency 24 months, 0.44 (0.25–0.79). The subgroup analysis of baseline hip fracture patients showed a time-dependent significant lower HR of 0.09 (0.02–0.48) after 24 months in the FLS group compared with the no-FLS group. 20 In addition, Huntjens et al. 21 showed with multivariate analysis a significant lower risk for subsequent fractures in the post-FLS cohort in comparison with the pre-FLS cohort: HR = 0.65 (0.51–0.84).
Nakayama et al. 22 used a Cox model with adjustments for age (<65 and ⩾65 years), sex, baseline fracture and socioeconomic status, and reported a significantly lower risk of subsequent fractures in the FLS group compared with the no-FLS group (HR FLS versus no-FLS group: 0.67 (0.47–0.95), p = 0.025). Moreover, Nakayama et al. 22 demonstrated an increased risk for major subsequent fractures in the patients aged ⩾ 65 years in comparison with patients aged < 65 years [HR = 1.79 (1.00–3.18, p = 0.049)]. The risk for any subsequent fracture and a major subsequent fracture was also increased in the cohort with a baseline major fracture compared with minor fracture [HR = 1.60 (1.05–2.44, p = 0.029)]. 22
Time to subsequent fracture
Only Axelsson et al. 18 reported the time to subsequent fracture (294 days in the treated patients versus 185 days in the untreated patients, p < 0.001, respectively). Time to subsequent fracture was not significantly different between the pre- and post-FLS cohort (207 days +/− 168 days in pre-FLS versus 200 +/− 163 days in post-FLS, p = ns). 18
Treatment rate
Axelsson et al. 18 and Huntjens et al. 21 described the different treatment rates in the pre- and post-FLS cohorts. Before the introduction of FLS treatment rates were respectively 13% and 22%. The post-FLS cohorts had increased treatment rates of 32% and 51%.18,21 Huntjens et al. 20 only described an estimated treatment rate of approximately 50% in the FLS cohort, there were no data available from the no-FLS hospital. 20 Hawley et al. 19 and Nakayama et al. 22 also did not report treatment rates.
Discussion
In this literature overview, we evaluated the evidence regarding the effect of FLS care on subsequent fracture risk reduction. We excluded other study designs. As an example, one study showed fracture reduction at the population level after implementing secondary fracture prevention after hip fracture. This and other studies (n = 6) did not mention to what degree, specifically, a FLS had an effect on subsequent fractures. Our literature search resulted in a total of five studies; in three studies subsequent fracture rates before and after the introduction of a FLS (pre-FLS versus post-FLS) and in two studies subsequent fracture rates between hospitals with and without a FLS were compared.
When comparing fracture rates before (pre-FLS) and after (post-FLS) introduction of a FLS, the adjusted HR for subsequent NVFs after baseline NVF was significantly lower in the post-FLS group after 2 years in one study (HR = 0.65, 95% CI: 0.51–0.84) and nonsignificant in another study for subsequent hip fractures after baseline hip fracture (HR = 0.95, 95% CI: 0.79–1.15 for the orthogeriatric care model and HR = 1.03, 95% CI: 0.85–1.26 for a nurse-led FLS). One study with a follow-up of less than a year did not demonstrate a significant difference in subsequent fracture risk pre- and post-FLS (HR = 0.95, 95% CI: 0.79–1.14). In two studies comparing hospitals with and without a FLS, the adjusted HR for subsequent fractures was significantly lower in the FLS hospitals (HR = 0.84, 95% CI: 0.64–1.10, during the first year, 0.44, 95% CI: 0.25–0.79, during the second year for subsequent NVFs after baseline NVF, and 0.67, 95% CI: 0.47–0.95, during the third year for subsequent VFs + NVFs after baseline VFs + NVFs). In all five studies, HRs were adjusted for baseline fractures. Only Nakayama et al. 22 analyzed the original fracture type as predictor for any re-fracture using a proportional hazard regression model. The subdistribution HR for any re-fracture after major versus minor baseline fracture was 1.60 (95% CI: 1.05–2.44; p = 0.029). The proportional hazards regression results for major re-fracture showed a subdistribution HR for baseline fracture major versus minor of 2.31 (95% CI: 1.33–4.01; p = 0.003).
Although these findings are of great interest and point at a positive effect of FLS care on subsequent fracture risk, the number of studies is limited. In addition, there were important differences between the five available studies. Firstly, three studies had a prospective, and two a retrospective, design. Second, the studies differed with regard to baseline and subsequent fracture types included, with two studies focusing on subsequent NVF after baseline NVF, two on VF + NVF after VF and NVF and one study on hip fractures after a baseline hip fracture.
Thirdly, attendance rates were reported in only two studies, and treatment rates in three. In addition, none of the studies reported if and how persistence with antiosteoporosis medication was evaluated. Fourth, none of the included studies mentioned a systematic evaluation of prevalent VFs. Axelsson et al. 18 and Nakayama et al. 22 included patients with a clinical VF. Since the presence of VFs increased subsequent fracture risk, this important information is lacking. Lastly, based on studies showing that the effectiveness of antiosteoporosis mediations for reduction of VFs and NVFs is achieved after 12, and respectively, 18–24 months,23–26 the follow-up period needs to be at least 2 years to demonstrate a positive effect of treatment in a FLS setting on NVFs, and even longer for hip fractures. In addition, there may be a time dependency when studying the relationship of FLS care and subsequent fracture rate, as shown in two of the five studies.18,20
When comparing FLS versus no FLS care, either in a pre- versus post-FLS or between hospital comparisons, it may be important to take into account the possibility of immortal time bias. Compared with a situation without FLS, patients have to be alive to attend the FLS. Hence, when comparing a population consisting of FLS attenders with no attenders, or a population without FLS care, the immortal time between the time of fracture and FLS attendance should be corrected for. None of the five studies in this overview mentioned immortal time bias. Another possible bias is the healthy compliance bias, that is, that more healthy patients are attending the FLS, so that a comparison with nonattenders should be adjusted for differences in patient characteristics, which is difficult because the level of health is not known in the nonattenders.
Within the constraint of the limited number, heterogeneity and relatively short follow-up period of studies, FLS care reduces subsequent fracture risk in comparison with no FLS care. Future studies are needed to establish a more robust effect of FLS care. Since randomization is not ethical, bias in these types of studies is almost inevitable. Nevertheless, it is still possible to correct for some types of bias. Future studies should provide detailed information on the total FLS cohort studied, including attendance rates, comorbidity, medication, additional assessments such as dual energy X-ray absorptiometry, Vertebral Fracture Assessment (VFA), laboratory testing, intervention strategy, treatment rates, persistence and the organization of patient follow-up. Also, studies with longer follow-up periods are needed. When comparing FLS with no-FLS care, it is important to compare the total populations in both groups. Although the effect of FLS care can only be achieved in the attenders, healthcare providers and other regulatory organizations will point at the necessity of demonstrating an effect of FLS care on the total group of patients with a fracture, and not only in the sometimes small group of FLS attenders. Advanced time-dependent analyses with adjustment for confounding factors should be performed and probable bias, including immortal time bias, should be taken into account and reported.
In conclusion, three FLS studies with heterogeneous designs and study populations, have shown a reduction in subsequent fracture rates after the introduction of a FLS. Larger and long-term studies are needed to further quantify the effect of FLS care on subsequent fracture risk.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
Irma J. A. de Bruin and Caroline E. Wyers that there is no conflict of interest.
Joop P. W. van den Bergh:
Research funding: Amgen, Will Pharma, Eli Lilly, MSD. Advisory board/honorarium speaker: Eli Lilly, Amgen.
Piet P. M. M. Geusens:
Clinical studies, advisory board, speakers fee: Pfizer, Abbott, Lilly, Amgen, MSD, Will, Roche, UCB, BMS and Novartis.
