Abstract
Background:
Studies have proposed that nutritional and immune-related markers are relevant with patient outcomes of various medical conditions and could be a useful indicator of patient prognostication.
Objectives:
This study investigated whether a prognostic immune nutritional index (PINI) at diagnosis could predict adverse clinical outcomes in patients with antineutrophil cytoplasmic antibody-associated vasculitis (AAV).
Design:
A retrospective, single-centre observational cohort analysis of patients with AAV.
Methods:
All-cause mortality and end-stage renal disease (ESRD) were investigated outcomes during the observation period. PINI was calculated by serum albumin (g/mL) × 0.9 − monocyte count (/mm3) × 0.0007, and the optimal cut-off of PINI was obtained using a Youden index-based bootstrapping method. Cox hazard analyses were performed to identify independent predictors of patient outcomes.
Results:
Of the 250 eligible patients, the median age of patients was 60.0 years, and 34.0% were men. During the disease course, 33 (13.2%) died and 42 (16.8%) developed ESRD, respectively. The ideal PINI cut-offs for all-cause mortality and ESRD were set as ⩽2.47 and ⩽3.12 (sensitivity and specificity of 75.1% and 60.6% for mortality and 46.2% and 78.6% for ESRD). AAV patients with PINI ⩽2.47 and those with PINI ⩽3.12 exhibited significantly higher rates for all-cause mortality and ESRD compared to those with PINI >2.47 and >3.12. In the multivariable Cox analysis, PINI ⩽2.47 (hazard ratio [HR]: 3.173, 95% confidence interval [CI]: 1.129, 8.916, p = 0.029) was independently associated with all-cause patient mortality; however, PINI ⩽3.12 was not independently associated with ESRD (HR: 1.097, 95% CI: 0.419, 2.870, p = 0.850).
Conclusion:
Findings from this study demonstrated PINI could predict all-cause patient mortality in AAV, and a higher clinical attention is warranted in those with PINI ⩽2.47 at initial diagnosis.
Keywords
Introduction
Systemic vasculitides are categorised based on the affected vessel size, primary organs involved and the presence/absence of underlying diseases. Among these factors, the affected size of the vasculatures has been the most widely used for the classification of vasculitides. 1 Among these vasculitides, small vessel vasculitis primarily affects capillaries and adjacent arterioles and venules, and occasionally arteries, and is characterised into two types, immune complex vasculitis and antineutrophil cytoplasmic antibody (ANCA)-associated vasculitis (AAV).1,2 AAV is an immune-mediated inflammatory disorder, and a classical laboratory feature is the detection of ANCAs against myeloperoxidase (MPO) and proteinase 3, and the increase of acute phase reactants is commonly found. 3 AAV is associated with life and organ-threatening outcomes such as death and major organ failure in a considerable number of patients.4–6 In general, the Five-Factor Score (FFS) is the most widely accepted objective measure in the prediction of patients’ prognosis, and Birmingham Vasculitis Activity Score (BVAS) is related to disease severity and outcomes7–9; however, there are limitations in FFS that it is calculated based on the presence of certain clinical manifestations in AAV without a consideration of protean disease features that may overlook overall disease status in organs and systems affected. Meanwhile, BVAS relies on the clinician’s interpretation, which can lead to inter-observer variability, and an issue of discordance is raised if extensive training is not provided. Therefore, although discovering indices for predicting patient outcomes in AAV is imperative, biomarkers that could be simply applicable are still lacking.
To date, several nutritional and immune-related markers for predicting the prognosis of various medical conditions have been proposed.10–13 These indices are thought to be associated with patient prognosis as they reflect the immune status of a certain individual, enabling the anticipation of outcomes. Of note, application of these indices to AAV patients demonstrated that they might also possess clinical significance in predicting adverse outcome of patients.14,15 In this context, a prognostic immune nutritional index (PINI) was recently introduced and demonstrated to be useful in predicting the prognosis of patients with colon cancer and in a haemodialysis population.16,17 PINI is composed of two variables, serum albumin and monocyte count, and therefore has advantages in clinical application as it is a readily available measure that could be simply assessed during routine laboratory testing. However, there has been no study on the clinical significance of PINI in AAV patients. Therefore, this study investigated whether PINI at the time of AAV diagnosis could predict outcomes during the disease course in patients with AAV.
Patients and methods
Study population
We selected 250 AAV patients from the Severance Hospital ANCA-associated vasculitis cohort during the period of 2000–2022, which is an observational cohort of Korean patients with AAV. After identifying patients who had the diagnostic code for AAV (microscopic polyangiitis [MPA], granulomatosis with polyangiitis [GPA] and eosinophilic granulomatosis with polyangiitis [EGPA]) in the hospital database, patients were further excluded for the following reasons: (a) final diagnosis was not compatible for AAV; (b) insufficient patient records to estimate BVAS and FFS7,18; (c) who could not classified as AAV according to the 2007 European Medicines Agency and 2012 Chapel Hill Consensus Conference definitions1,2; (d) initial laboratory data unavailable; (e) follow-up duration of less than 3 months without experiencing the outcomes of interest in the hospital and (f) those who did not receive glucocorticoids >20 mg or immunosuppressive drugs for AAV treatment within the first month before AAV diagnosis (Supplemental Figure 1). The findings presented in this study adhere to the guidelines outlined in the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 19
Clinical and laboratory data at the time of AAV
Age, sex, body mass index (BMI) and smoking history were collected as demographic data. Information on AAV subtype, ANCA type, AAV-specific indices of BVAS and FFS, acute phase reactants of erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP), and comorbid disorders were also collected. Laboratory data including monocyte count and serum albumin level were recorded.
Patient outcomes during the disease course and medications
The occurrence of all-cause mortality and end-stage renal disease (ESRD) during the disease course were investigated outcomes of the patients. 20 The follow-up duration based on each outcome was defined as the period from the time of AAV diagnosis to the time of the occurrence of each adverse outcome in patients with the corresponding events, whereas it was defined as the period from the time of AAV diagnosis to the last visit to the clinic in outcome-free patients.
In addition, the numbers of AAV patients who received glucocorticoids and immunosuppressive drugs of cyclophosphamide, rituximab, mycophenolate mofetil, azathioprine, tacrolimus and methotrexate during the disease course were also queried.
PINI calculation
Calculation of PINI was done using the baseline laboratory result of serum albumin and monocyte count, as previously described: PINI = serum albumin (g/mL) × 0.9 − monocyte count (/mm3) × 0.0007. 16
Statistical analyses
All statistical analyses were performed using SPSS Statistics for Windows, version 26 (IBM Corp., Armonk, NY, USA) and R software (version 4.2.0; R Foundation for Statistical Computing, Vienna, Austria). Continuous variables are expressed as medians with interquartile ranges, whereas categorical variables are expressed as numbers (percentages). The correlation coefficient (r) between the two variables was obtained using either the Pearson or the Spearman correlation analysis. The standardised correlation coefficient (β) was obtained by the multivariable linear regression analysis using variables with statistical significance in the univariable analysis. The receiver operating characteristic (ROC) curve analysis was performed to calculate area under the curve (AUC) with 95% confidence interval (CI). Given that there is a risk of overfitting in our study population, the Youden index-based optimal cut-off was obtained by a bootstrapping method (using 1000 samples) to reduce this risk. Comparison of the cumulative survival rates between the two groups was analysed by the Kaplan–Meier survival analysis with the log-rank test. The multivariable Cox hazard analyses was performed using variables with statistical significance in the univariable Cox hazard model. Since serum albumin level and monocyte count were the parameters comprising the formula of PINI, they were excluded from the correlation, linear regression and Cox analyses, and for all analyses, p-values <0.05 were considered statistically significant.
Results
Characteristics at the time of AAV diagnosis
The median age of patients was 60.0 years, and 34.0% were men. Most patients had a normal BMI (22.3 kg/m2), and 12 patients were ex-smokers. Among the 250 AAV patients, 135, 69 and 46 patients were diagnosed with MPA, GPA and EGPA, respectively, and 167 and 46 patients had MPO-ANCA (or perinuclear (P)-ANCA) and PR3-ANCA (cytoplasmic (C)-ANCA), respectively. The median BVAS, FFS, ESR and CRP levels were 12.0, 1.0, 59.5 mm/h, and 14.5 mg/L, respectively. The most common concomitant chronic disease was hypertension (40.0%), followed by diabetes mellitus (26.0%). The median PINI of the patients was 3.0 (Table 1).
Characteristics of AAV patients at disease diagnosis.
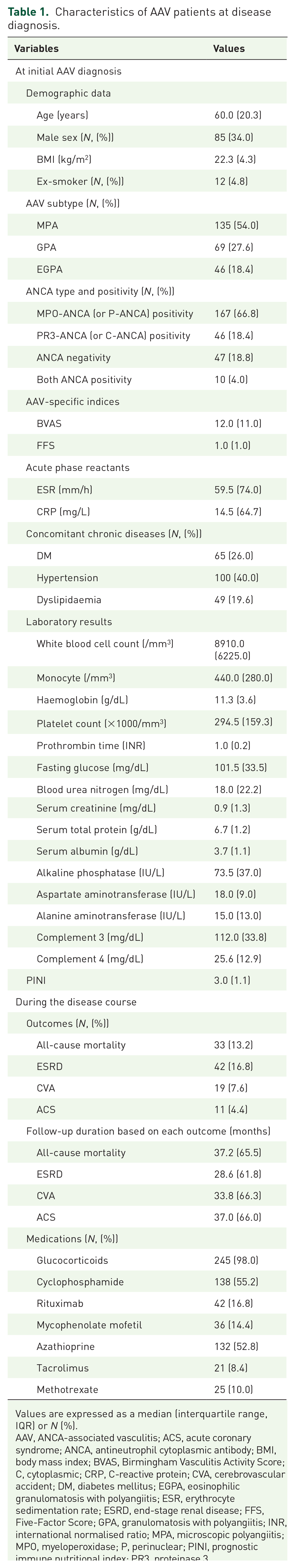
Values are expressed as a median (interquartile range, IQR) or N (%).
AAV, ANCA-associated vasculitis; ACS, acute coronary syndrome; ANCA, antineutrophil cytoplasmic antibody; BMI, body mass index; BVAS, Birmingham Vasculitis Activity Score; C, cytoplasmic; CRP, C-reactive protein; CVA, cerebrovascular accident; DM, diabetes mellitus; EGPA, eosinophilic granulomatosis with polyangiitis; ESR, erythrocyte sedimentation rate; ESRD, end-stage renal disease; FFS, Five-Factor Score; GPA, granulomatosis with polyangiitis; INR, international normalised ratio; MPA, microscopic polyangiitis; MPO, myeloperoxidase; P, perinuclear; PINI, prognostic immune nutritional index; PR3, proteinase 3.
Outcomes and medication prescription during the disease course
Among the 250 AAV patients, 33 (13.2%) died with a median follow-up duration of 37.2 months, and 42 (16.8%) developed ESRD during the median follow-up of 28.6 months. Glucocorticoids were administered to 98.0% of patients, and the most frequently administered immunosuppressive drug was cyclophosphamide (55.2%), followed by azathioprine (52.8%) (Table 1). The most common cause of death in patients were pneumonia, followed by disease aggravation and interstitial lung disease (ILD) (Supplemental Table 1).
Correlation analysis between PINI and continuous variables at initial diagnosis
PINI exhibited a significantly inverse correlation with age (r = −0.380, p < 0.001), ESR (r = –0.546, p < 0.001), CRP (r = −0.611, p < 0.001), white blood cell (r = −0.433, p < 0.001) and platelet (r = −0.413, p < 0.001) counts, prothrombin time (r = −0.500, p < 0.001), blood urea nitrogen (r = −0.215, p = 0.001), serum creatinine (r = −0.163, p = 0.010), alkaline phosphatase (r = −0.309, p < 0.001) and aspartate aminotransferase (r = −0.134, p = 0.035). Conversely, PINI showed a significantly positive correlation with haemoglobin (r = 0.570, p < 0.001) (Supplemental Table 2).
Multivariable linear regression analysis of continuous variables for estimating the cross-sectional BVAS
In the univariable linear regression analysis for initial BVAS, age, ESR, CRP, white blood cell and platelet counts, haemoglobin, prothrombin time, fasting glucose, blood urea nitrogen, serum creatinine and PINI were significantly correlated with BVAS. In the multivariable analysis, white blood cell count (β = 0.151, p = 0.033), haemoglobin (β = −0.354, p < 0.001), fasting glucose (β = 0.145, p = 0.011) and blood nitrogen (β = 0.239, p = 0.010) were significantly correlated with BVAS. However, PINI was not significantly correlated with BVAS (β = −0.107, p = 0.232) (Table 2).
Linear regression analysis of continuous variables the time of AAV diagnosis for estimating initial BVAS.
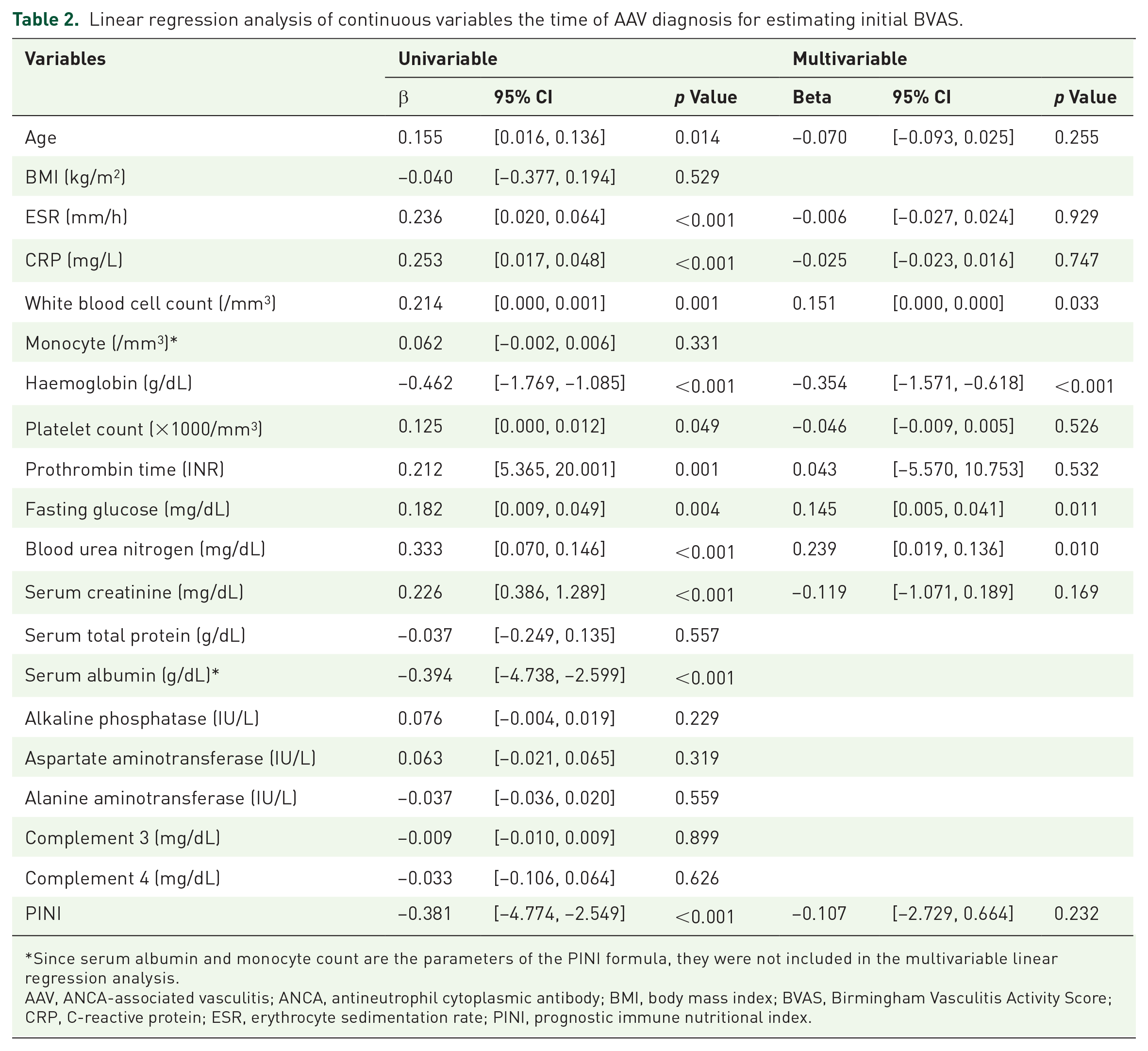
Since serum albumin and monocyte count are the parameters of the PINI formula, they were not included in the multivariable linear regression analysis.
AAV, ANCA-associated vasculitis; ANCA, antineutrophil cytoplasmic antibody; BMI, body mass index; BVAS, Birmingham Vasculitis Activity Score; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; PINI, prognostic immune nutritional index.
Optimal cut-offs of PINI for predicting outcomes
Due to the negative association between PINI and acute phase reactants, ROC curve analyses with dependent variables of the absence of outcomes of interest were conducted. In the ROC curve analysis, PINI exhibited a significant AUC for all-cause mortality (AUC 0.732, 95% CI: 0.639, 0.824), and the occurrence of ESRD (AUC 0.610, 95% CI: 0.521, 0.699) (Supplemental Figure 2).
When the PINI cut-off for all-cause mortality was set as ⩽2.47 using a bootstrapping method, the sensitivity and specificity were 75.1% and 60.6%, respectively, and this model exhibited the hazard ratio (HR) of 4.482 for the estimation of patient mortality. In addition, when a PINI cut-off for the occurrence of ESRD was set as ⩽3.12, the sensitivity and specificity were 46.2% and 78.6%, respectively, and those with PINI ⩽3.12 had HR of 2.885 for ESRD (Supplemental Table 3).
Comparison of patient outcomes according to PINI cut-off values
AAV patients with PINI ⩽2.47 exhibited a significantly lower cumulative patients’ survival rate than those with PINI >2.47 (p < 0.001). Similarly, AAV patients with PINI ⩽3.12 exhibited a significantly lower cumulative ESRD-free survival rate than those with PINI >3.12 (p = 0.003) (Figure 1).
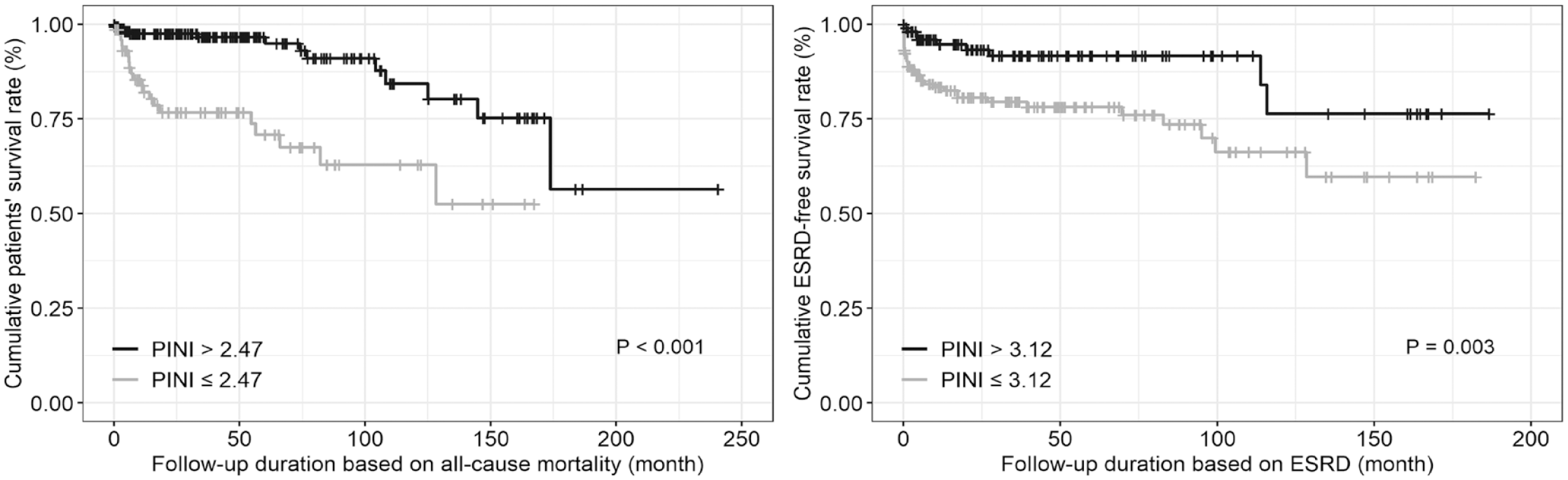
Comparison of cumulative survival rates during follow-up.
Cox hazards model analysis for all-cause mortality
In the univariable Cox analysis, age, male sex, BVAS, FFS, ESR, CRP, haemoglobin, prothrombin time, blood urea nitrogen, serum creatinine, serum total protein, alkaline phosphatase and aspartate aminotransferase were significantly associated with all-cause mortality. In particular, PINI ⩽2.47 was also significantly associated with all-cause mortality. Multivariable Cox analysis indicated that age (HR: 1.038, 95% CI: 1.003, 1.073), male sex (HR: 2.595, 95% CI: 1.154, 5.832), BVAS (HR: 1.100, 95% CI: 1.032, 1.172), FFS (HR: 1.580, 95% CI: 1.029, 2.426), alkaline phosphatase (HR: 1.006, 95% CI: 1.000, 1.011) and PINI ⩽2.47 (HR: 3.173, 95% CI: 1.129, 8.916) were independently associated with all-cause mortality in AAV patients (Table 3). Similar results were obtained even when a different model only including (a) age, sex, ESR, CRP and PINI ⩽2.47 or (b) age, sex, BVAS, FFS and PINI ⩽2.47 as a covariate was created (Supplemental Tables 4 and 5).
Cox hazards model analysis using variables at AAV diagnosis for all-cause mortality during the disease course.

AAV, ANCA-associated vasculitis; ANCA, antineutrophil cytoplasmic antibody; BMI, body mass index; BVAS, Birmingham Vasculitis Activity Score; C, cytoplasmic; CRP, C-reactive protein; DM, diabetes mellitus; ESR, erythrocyte sedimentation rate; FFS, Five-Factor Score; INR, international normalised ratio; MPO, myeloperoxidase; P, perinuclear; PINI, prognostic immune nutritional index; PR3, proteinase 3.
Since serum albumin and monocyte count are the parameters of the PINI formula, they were not included in the multivariable linear regression analysis.
Cox hazards model analysis for the occurrence of ESRD
In the univariable Cox analysis, BMI, MPO-ANCA (or P-ANCA) positivity, BVAS, FFS, hypertension, haemoglobin, blood urea nitrogen, serum creatinine, aspartate aminotransferase, alanine aminotransferase, complement 3 and PINI ⩽3.12 were significantly associated with the occurrence of ESRD. In the multivariable Cox analysis, both FFS (HR: 1.581, 95% CI: 1.041, 2.402) and serum creatinine (HR: 1.452, 95% CI: 1.247, 1.691) were independently associated with the occurrence of ESRD in AAV patients. However, PINI ⩽3.12 was not independently associated with the occurrence of ESRD (HR: 1.097, 95% CI: 0.419, 2.870) (Table 4).
Cox hazards model analysis of variables for predicting ESRD during follow-up.

Since serum albumin and monocyte count are the parameters of the PINI formula, they were not included in the multivariable linear regression analysis.
AAV, ANCA-associated vasculitis; ANCA, antineutrophil cytoplasmic antibody; BMI, body mass index; BVAS, Birmingham Vasculitis Activity Score; C, cytoplasmic; CRP, C-reactive protein; DM, diabetes mellitus; ESR, erythrocyte sedimentation rate; ESRD, end-stage renal disease; FFS, Five-Factor Score; INR, international normalised ratio; MPO, myeloperoxidase; P, perinuclear; PINI, prognostic immune nutritional index; PR3, proteinase 3.
Discussion
In this study, we investigated whether PINI at AAV diagnosis could predict outcomes during disease course and observed an association with all-cause patient mortality. It was appeared that PINI may be clinically useful, as the Cox-proportional analysis revealed that PINI was associated with patient mortality even when BVAS and FFS – a traditional measure that is considered to have an association with patient prognosis in AAV – was included as a covariate. The exact mechanism underlying the association between PINI and all-cause mortality in AAV patients remains unclear; however, we attempted to consider that the role of serum albumin and monocyte counts in inflammation could partly explain our results. In terms of serum albumin, it is not only an index for nutritional status but also a negatively correlated biomarker for systemic inflammation.21–23 Firstly, regarding malnutrition represented by hypoalbuminaemia, it was reported that malnutrition is likely to increase the mortality rate in patients with various illnesses. 24 Similarly, it was demonstrated that hypoalbuminaemia and being underweight are significant risk factors for all-cause mortality in AAV patients.14,25 Secondly, the decrease of albumin could indicate heightened inflammation in AAV, which appears to be associated with all-cause mortality in AAV. In line with this hypothesis, previous studies have suggested that albumin is associated with disease severity or with the burden of inflammation in AAV.25–28
On the other hand, monocyte count may increase in active inflammatory conditions because monocyte recruitment is a key process during inflammation, and chemokines can mobilise monocytes from the bone marrow to the bloodstream. 29 Previous studies demonstrated that circulating monocyte count increased in patients with active autoimmune diseases such as rheumatoid arthritis, Crohn’s disease and Sjogren syndrome, compared to those with inactive diseases.30–32 Since PINI is obtained by subtracting monocyte count from serum albumin level, greater inflammatory burden of higher serum monocyte count, could result in a decrease in PINI. Therefore, it may be reasonable to assume that PINI consisting of serum albumin and monocyte count is associated with all-cause mortality, even when considering the effects of multiple confounding variables.
Findings from our study suggest that using PINI could be a clinically relevant indicator in the prognostication of patients with AAV and aid in providing optimal management. On evaluating the cause of death of patients with AAV, we found that the majority of death in our cohort of patients were attributed by pneumonia, disease activity, ILD and there was no disease unrelated mortality. Accordingly, among patients with AAV and low PINI, it is possible that an individualised approach according to PINI is feasible in order to improve patient outcome of mortality. For example, regular monitoring of chest X-rays and inquiry of the presence of respiratory symptoms could enable early identification of those with infections in the respiratory tract or deterioration of ILD, while a prudent monitoring of disease aggravation and recommendation of short-term follow-up might be required in those having signs raising a clinical suspicion of disease relapse.
Limitations
This study has several limitations. Firstly, despite the fact that AAV is a rare disease, the number of patients was insufficient to generalise the results in patients with AAV. Secondly, due to the retrospective study design, even though we included patients with incident case of AAV and those who were not prescribed with immunosuppressive agents prior to disease diagnosis, the potential confounding factors that might influence patient outcomes, albumin and monocyte counts could not be evaluated in detail. Thirdly, since this was a single-centre study, an issue of reproducibility and validation exists. We believe that an external validation by its application in independent cohorts and prospective studies would better verify our results and provide insights in whether PINI are useful in predicting outcomes of patients with AAV.
Conclusions
This study demonstrated that PINI at the time of AAV diagnosis could predict all-cause mortality during the disease course in AAV patients. PINI could be a potential laboratory marker indicating adverse clinical outcomes, suggesting a precaution is needed in those with PINI ⩽2.47 at diagnosis.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X231188818 – Supplemental material for A prognostic immune nutritional index can predict all-cause mortality in patients with antineutrophil cytoplasmic antibody-associated vasculitis
Supplemental material, sj-docx-1-tab-10.1177_1759720X231188818 for A prognostic immune nutritional index can predict all-cause mortality in patients with antineutrophil cytoplasmic antibody-associated vasculitis by Sung Soo Ahn, Jung Yoon Pyo, Jason Jungsik Song, Yong-Beom Park and Sang-Won Lee in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-docx-2-tab-10.1177_1759720X231188818 – Supplemental material for A prognostic immune nutritional index can predict all-cause mortality in patients with antineutrophil cytoplasmic antibody-associated vasculitis
Supplemental material, sj-docx-2-tab-10.1177_1759720X231188818 for A prognostic immune nutritional index can predict all-cause mortality in patients with antineutrophil cytoplasmic antibody-associated vasculitis by Sung Soo Ahn, Jung Yoon Pyo, Jason Jungsik Song, Yong-Beom Park and Sang-Won Lee in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-docx-3-tab-10.1177_1759720X231188818 – Supplemental material for A prognostic immune nutritional index can predict all-cause mortality in patients with antineutrophil cytoplasmic antibody-associated vasculitis
Supplemental material, sj-docx-3-tab-10.1177_1759720X231188818 for A prognostic immune nutritional index can predict all-cause mortality in patients with antineutrophil cytoplasmic antibody-associated vasculitis by Sung Soo Ahn, Jung Yoon Pyo, Jason Jungsik Song, Yong-Beom Park and Sang-Won Lee in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-docx-4-tab-10.1177_1759720X231188818 – Supplemental material for A prognostic immune nutritional index can predict all-cause mortality in patients with antineutrophil cytoplasmic antibody-associated vasculitis
Supplemental material, sj-docx-4-tab-10.1177_1759720X231188818 for A prognostic immune nutritional index can predict all-cause mortality in patients with antineutrophil cytoplasmic antibody-associated vasculitis by Sung Soo Ahn, Jung Yoon Pyo, Jason Jungsik Song, Yong-Beom Park and Sang-Won Lee in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-docx-5-tab-10.1177_1759720X231188818 – Supplemental material for A prognostic immune nutritional index can predict all-cause mortality in patients with antineutrophil cytoplasmic antibody-associated vasculitis
Supplemental material, sj-docx-5-tab-10.1177_1759720X231188818 for A prognostic immune nutritional index can predict all-cause mortality in patients with antineutrophil cytoplasmic antibody-associated vasculitis by Sung Soo Ahn, Jung Yoon Pyo, Jason Jungsik Song, Yong-Beom Park and Sang-Won Lee in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-jpg-6-tab-10.1177_1759720X231188818 – Supplemental material for A prognostic immune nutritional index can predict all-cause mortality in patients with antineutrophil cytoplasmic antibody-associated vasculitis
Supplemental material, sj-jpg-6-tab-10.1177_1759720X231188818 for A prognostic immune nutritional index can predict all-cause mortality in patients with antineutrophil cytoplasmic antibody-associated vasculitis by Sung Soo Ahn, Jung Yoon Pyo, Jason Jungsik Song, Yong-Beom Park and Sang-Won Lee in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-tif-7-tab-10.1177_1759720X231188818 – Supplemental material for A prognostic immune nutritional index can predict all-cause mortality in patients with antineutrophil cytoplasmic antibody-associated vasculitis
Supplemental material, sj-tif-7-tab-10.1177_1759720X231188818 for A prognostic immune nutritional index can predict all-cause mortality in patients with antineutrophil cytoplasmic antibody-associated vasculitis by Sung Soo Ahn, Jung Yoon Pyo, Jason Jungsik Song, Yong-Beom Park and Sang-Won Lee in Therapeutic Advances in Musculoskeletal Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
