Abstract
Background:
Preclinical data suggest that tofacitinib would protect bone health in patients with rheumatoid arthritis (RA).
Objective:
To assess fracture risk in tofacitinib RA clinical trials.
Design:
Post hoc analysis.
Methods:
We analyzed pooled data of phase I/II/III and long-term extension studies (‘P123LTE cohort’), pooled data of placebo-controlled portions of phase III studies (phase III placebo-controlled cohort), and data from ORAL Surveillance [phase IIIb/IV randomized, open-label trial evaluating tofacitinib 5/10 mg twice daily (BID) vs tumor necrosis factor inhibitor (TNFi) in patients ⩾ 50 years with ⩾ 1 additional cardiovascular risk factor].
Results:
In the phase III placebo-controlled cohort, incidence rates (IRs) [95% confidence interval (CI)] of fracture were 2.11 (1.09–3.68), 2.56 (1.23–4.71), and 4.43 (1.78–9.12) per 100 patient-years (PYs) for tofacitinib 5 mg BID, tofacitinib 10 mg BID, and placebo, respectively [tofacitinib 5 mg BID vs placebo: hazard ratio (HR) (95% CI) = 0.55(0.18–1.65); tofacitinib 10 mg BID vs placebo: HR (95% CI) = 0.72 (0.26–2.01)]. In P123LTE, IRs (95% CI) were 2.62 (2.29–2.99) and 2.26 (2.02–2.52) per 100 PY for average tofacitinib 5 and 10 mg BID, respectively. In ORAL Surveillance, IRs (95% CI) were 2.79 (2.34–3.30), 2.87 (2.40–3.40), and 2.27 (1.87–2.74) per 100 PY for tofacitinib 5 mg BID, tofacitinib 10 mg BID, and TNFi, respectively. In ORAL Surveillance, the risk of fracture was numerically higher than TNFi for tofacitinib 5 mg BID [HR (95% CI) = 1.23 (0.96–1.58)] and tofacitinib 10 mg BID [HR (95% CI) = 1.26 (0.97–1.62)]. In ORAL Surveillance, independent predictors of all and osteoporotic fractures with tofacitinib or TNFi included age ⩾ 65, female sex, history of fracture/osteoporosis, and baseline oral corticosteroid use.
Conclusion:
This post hoc analysis showed numerically lower fracture risk with tofacitinib versus placebo and numerically greater risk versus TNFi. We did not identify any tofacitinib-specific predictors of fractures, and predictors of fracture were generally aligned with prior literature in the general population and patients with RA. Patients with fracture risk factors should be adequately monitored and treated.
Clinical trial registration:
NCT00960440, NCT00847613, NCT00814307, NCT00856544, NCT00853385, NCT01039688, NCT02187055, NCT02831855, NCT00413699, NCT00147498, NCT00413660, NCT00550446, NCT00603512, NCT00687193, NCT00661661, NCT01164579, NCT00976599, NCT01059864, NCT01359150, NCT01262118, NCT01484561, NCT02281552, NCT02147587, NCT02092467
Introduction
The inflammatory state associated with rheumatoid arthritis (RA) contributes to an elevated risk of osteoporosis and osteoporotic fractures.1–5 Patients with RA have approximately double the risk of osteoporosis2,3 and two- to three-fold higher rates of fracture1,4 compared with the general population; this increase in risk is most pronounced in patients with longer RA disease duration, chronically persistent inflammation, seropositive disease, use of corticosteroids, and low physical activity.6,7 Other known risk factors for fracture include older age, smoking, lower body mass index (BMI), low bone mineral density (BMD), female sex, history of prior fracture, and the use of opioids, proton pump inhibitors, and antidepressants.1,8–14
Some studies in patients with RA have suggested that treatment with biologic disease-modifying antirheumatic drugs (DMARDs) and targeted synthetic DMARDs can reduce the loss of BMD and/or fracture risk,10,15,16 potentially by controlling systemic inflammation and/or reducing use of glucocorticoid therapy. Other studies have found that patients with RA continue to have an elevated risk of fracture compared with the general population, despite recent improvements in the RA therapeutic landscape.7,17
Tofacitinib is an oral Janus kinase (JAK) inhibitor for the treatment of RA.18,19 Published preclinical data in animal models of RA suggest that tofacitinib may reduce osteoclastogenesis, stimulate osteoblast function and have a protective effect on BMD in the pro-inflammatory state of RA.20–22 There are several theoretical pathways by which JAK inhibition can potentially impact bone loss. One of these is via interleukin-7 (IL-7), a cytokine that signals through JAK1 and JAK3, leading to T-cell development and maturation. 23 T-cell activation induces expression of receptor activator of nuclear factor κ B (RANK)-ligand, a member of the tumor necrosis factor (TNF) cytokine family, which is a crucial regulator of osteoclastogenesis. 24 Other cytokines (such as IL-20), which signal via JAK1 and/or JAK2, also drive osteoclastogenesis and bone loss in the context of both chronic inflammation and osteoporosis.25–28 Therefore, by inhibiting JAK pathway-mediated signaling cytokines, tofacitinib would be expected to inhibit bone loss and stimulate osteoblast function. In fact, animal models of disease do support a protective effect of tofacitinib on bone, and no tofacitinib-related detrimental effects on bone growth and development have been noted in animal toxicity studies.20–22,29
In a pharmacovigilance database search, osteoporosis was identified as a potential risk for patients with RA on tofacitinib. 30 While the preclinical data regarding tofacitinib and bone health did not raise safety concerns, here, we report post hoc analyses of the tofacitinib clinical trial program to assess the impact of tofacitinib on fracture risk in patients with RA.
Methods
Clinical trial data
We assessed the risk of fracture in the tofacitinib RA clinical trial program across two different pooled cohorts, and separately in the ORAL Surveillance trial (NCT02092467). Based on differences in the patient populations compared and the inclusion of tumor necrosis factor inhibitor (TNFi) as an active control in the long-term ORAL Surveillance study, separate analyses were conducted for this data set.
The P123LTE cohort included integrated data from all phases I, II, III, and long-term extension (LTE) studies (full details in the Supplementary Appendix). The P123LTE cohort focused on patients who received ⩾ 1 dose of tofacitinib. Potential dose-related effects were assessed in two analysis groups based on the patient’s overall tofacitinib exposure: average tofacitinib 5 mg twice daily (BID; average total daily dose < 15 mg) and average tofacitinib 10 mg BID (average total daily dose ⩾ 15 mg).
To allow for comparisons between tofacitinib and placebo, the phase III placebo-controlled cohort included integrated data from the placebo-controlled portions of phase III randomized controlled trials (RCTs) (full details in the Supplementary Appendix). Data were analyzed for the randomized treatment groups of tofacitinib 5 mg BID, tofacitinib 10 mg BID, and placebo. For most of the placebo-controlled studies, patients continued treatment with methotrexate or a non-biologic DMARD regardless of treatment group (including placebo).
ORAL Surveillance (NCT02092467) was a phase IIIb/IV randomized, parallel-arm, open-label, safety endpoint study evaluating the safety of tofacitinib at two doses (5 mg BID and 10 mg BID) versus TNFi [adalimumab 40 mg every other week (North America), or etanercept 50 mg once weekly (rest of the world)]. 31 In contrast to other tofacitinib RA clinical studies, all patients enrolled in ORAL Surveillance were ⩾ 50 years of age with ⩾ 1 additional cardiovascular (CV) risk factor.
All studies were conducted in accordance with the Declaration of Helsinki, International Conference on Harmonization Guidelines for Good Clinical Practice, and local country regulations, and were approved by the Institutional Review Board and Independent Ethics Committee at each center. Patients provided written informed consent. No further ethical approval was required for these post hoc analyses in accordance with the policies of our institutions.
Study outcomes
Fracture events were identified via MedDRA preferred terms. Separate analyses were conducted for all fractures and for osteoporotic fractures only. 32 Falls were also captured as adverse events to contextualize the fracture data. Full details are available in the Supplementary Appendix.
Statistical analyses
Outcomes were presented as incidence rates [IRs; number of patients with events per 100 patient-years (PYs)] with hazard ratios (HRs) and 95% confidence intervals (CI) to compare treatment groups.
Potential baseline predictors of fractures in the P123LTE and ORAL Surveillance cohorts were analyzed using hierarchical regressions. First, potential baseline predictors were selected based on the existing literature1,6–14,33–40 and screened via simple Cox analyses which included treatment group and a single candidate baseline predictor in each model. Any baseline predictor with p < 0.10 entered the multivariable model. Next, a multivariable Cox proportional hazard model using a backward selection algorithm with selection criterion p < 0.05 was used to obtain the final set of baseline covariates. Treatment group was retained regardless of the p value. In addition, an interaction term between the treatment group and a predictor (whether it is a dichotomous or polytomous variable) was added to the simple Cox model to evaluate whether the treatment had a differential impact on that variable’s relationship with fracture risk. In ORAL Surveillance, subgroup IRs and HRs (vs TNFi) and corresponding 95% CIs were reported for the predictors retained in the final multivariable model. The risk of fracture between tofacitinib 5 or 10 mg BID versus TNFi within a level of predictor was then modeled using simple Cox models, with treatment as the only covariate.
All analyses were post hoc. Across these exploratory analyses, no multiplicity adjustments were applied; p values are provided as a descriptive measure only.
Results
Patients
The demographic and clinical characteristics of patients in the P123LTE cohort (tofacitinib treatment groups) and ORAL Surveillance are listed in Table 1.
Patient demographics and baseline characteristics.

BID, twice daily; BMI, body mass index; COPD, chronic obstructive pulmonary disease; ILD, interstitial lung disease; RF, rheumatoid factor; SD, standard deviation; TNFi, tumor necrosis factor inhibitor.
⩾ 50 to < 65 in ORAL Surveillance.
Includes post-menopausal osteoporosis.
Includes corticosteroids administered through any route of administration. In ORAL Surveillance, baseline use of oral corticosteroids was recorded in 776 (53.3 %) patients who received tofacitinib 5 mg BID, 773 (53.1%) patients who received tofacitinib 10 mg BID, and 774 (53.3%) patients who received TNFi. Route of administration for corticosteroids was not captured in the P123LTE cohort.
Incidence of fracture in patients with RA (pooled tofacitinib clinical trial data and ORAL Surveillance)
Figure 1(a) shows that the incidence of all fractures was numerically higher with placebo than with tofacitinib 5 mg BID or 10 mg BID in the phase III placebo-controlled cohort. The HR (95% CI) for tofacitinib 5 mg BID versus placebo was 0.55 (0.18, 1.65), and that for tofacitinib 10 mg BID versus placebo was 0.72 (0.26, 2.01). Similar results were observed for osteoporotic fractures.

IRs (patients with events/100 PY) for all fractures and osteoporotic fractures in (a) phase III placebo-controlled cohorta,b. (b) P123LTE cohortb. (c) ORAL Surveillance (⩾ 50 years of age with ⩾ 1 additional CV risk factor at baseline)c.
In the P123LTE cohort, IRs for all fractures and osteoporotic fractures were numerically higher in the average tofacitinib 5 mg BID group relative to the average tofacitinib 10 mg BID group [Figure 1(b)].
IRs in the tofacitinib groups in ORAL Surveillance were similar to the rates reported in the P123LTE cohort. The HRs (95% CIs) for all fractures with tofacitinib 5 or 10 mg BID versus TNFi were 1.23 (0.96, 1.58) and 1.26 (0.97, 1.62), respectively [Figure 1(c)]. When limiting the analysis to osteoporotic fractures, the HRs (95% CI) for tofacitinib 5 or 10 mg BID versus TNFi were 1.35 (0.91, 2.01) and 1.60 (1.09, 2.36), respectively. The comparison of osteoporotic fractures between tofacitinib 10 mg BID and TNFi was the only one for which the 95% CI of the HR excluded 1 [Figure 1(c)].
Details of the number and location of all fractures in P123LTE and ORAL Surveillance are presented in Table S1.
Predictors of fracture in patients with RA
In the P123LTE cohort, the following were identified as risk factors for both all fractures and osteoporotic fractures using the multivariable Cox regression analysis via backward selection: age ⩾ 65 years; history of osteoporosis, history of chronic lung disease [i.e. chronic obstructive pulmonary disease (COPD) or interstitial lung disease (ILD)], and baseline use of corticosteroids (Figure 2). In addition to these, Health Assessment Questionnaire–Disability Index (HAQ-DI), rheumatoid factor (RF) negativity, and baseline use of antidepressants, osteoporotic treatments, and opioids were identified as risk factors for all fractures. Moreover, history of fractures, anti-cyclic citrullinated peptide (anti-CCP) negativity, BMI > 25 to < 30 kg/m2 (ref.: ⩾ 18 to ⩽ 25 kg/m2), and baseline use of statins were also identified as a risk factor for osteoporotic fractures.
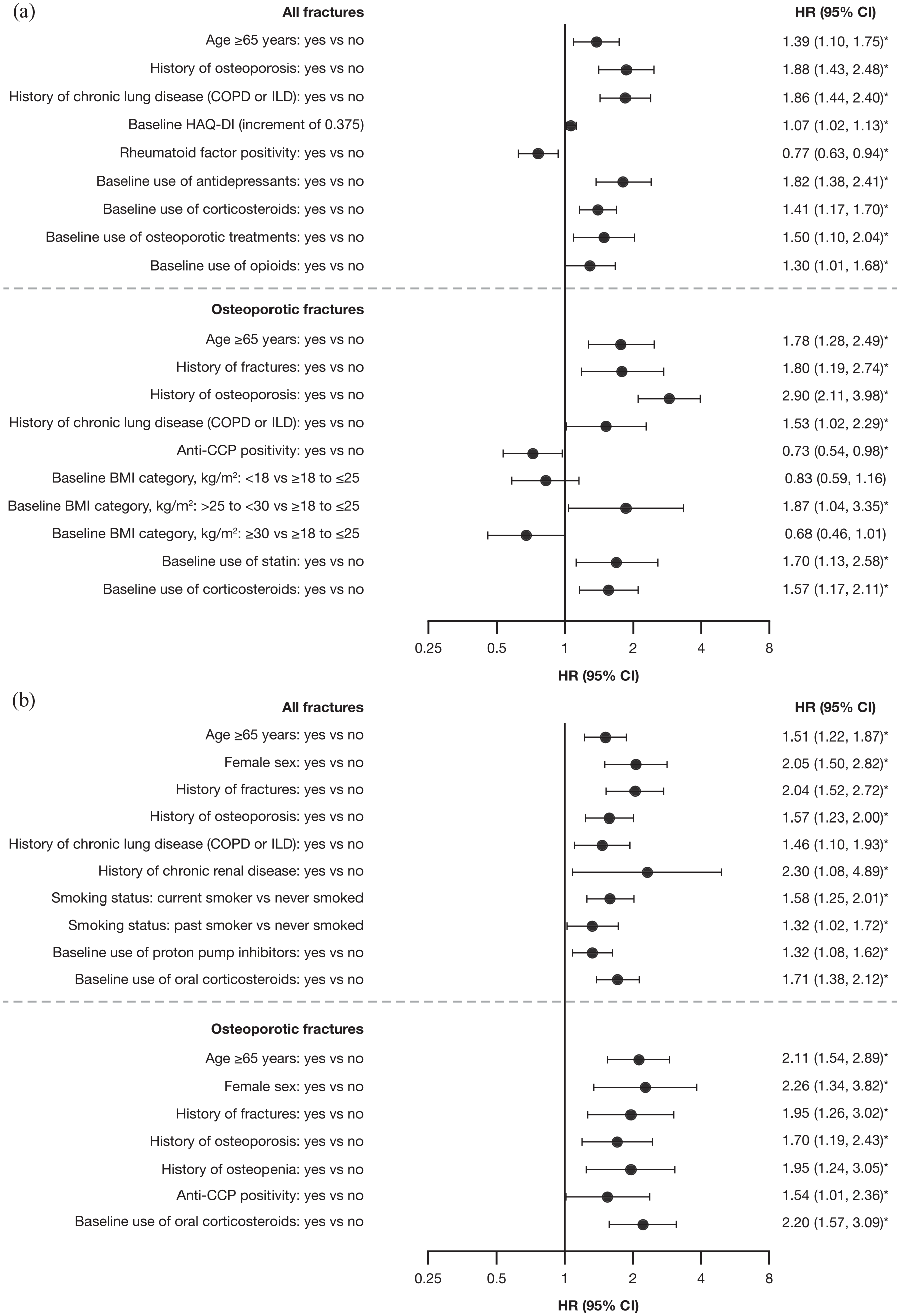
Predictors for all fractures and osteoporotic fractures from multivariable Cox models via backward selection in (a) P123LTE cohort and (b) ORAL Surveillance.
In the ORAL Surveillance cohort, the following were identified as risk factors for both all fractures and osteoporotic fractures using the multivariable Cox regression analysis via backward selection: age ⩾65 years, female sex; history of fractures, history of osteoporosis, and baseline use of oral corticosteroids (Figure 2). In addition to these, history of chronic lung disease, chronic renal disease, being a past or current smoker, and baseline use of proton pump inhibitors were identified as risk factors for all fractures. Moreover, history of osteopenia and anti-CCP positivity was also identified as risk factors for osteoporotic fractures. Univariate analyses of baseline predictors for all fractures and osteoporotic fractures are shown in Tables S2–S5.
Figure 3 shows subgroup analysis data for patients receiving either dose of tofacitinib or TNFi in ORAL Surveillance. The variables represented are those with a p value < 0.05 in the multivariable analysis. However, the p values for the interaction between the treatment groups and the retained risk factor – that is, whether the treatment effects differed by subgroup – were ⩾ 0.1009, which do not lend statistical support for differential effects.

IRs (patients with events/100 PY) for osteoporotic fractures in ORAL Surveillance stratified by independent predictors of fractures.
Discussion
Preclinical data from in vitro and animal studies suggest that tofacitinib would have a positive impact on bone health in patients with RA.20–22,29 To assess whether clinical data support these findings, we performed post hoc analyses of the tofacitinib clinical trial program. We assessed the relationship between use of tofacitinib and fracture risk in patients with RA in comparison with placebo and TNFi.
In line with preclinical findings, our analysis suggests that tofacitinib numerically reduced the rate of all fractures and osteoporotic fractures compared with placebo in patients with RA. As ethical considerations prevented prolonged treatment with placebo, it should be noted that the observation period for this group was relatively short and exposure was limited (158 PY for all fractures). Of note, the rates of fractures in the P123LTE cohort (for which the follow-up period was longer for patients on tofacitinib) were similar to those in the phase III placebo-controlled cohort. The observed differences between tofacitinib and placebo may be due to a rapid reduction in pain or changes in patients’ mobility, a reduced need for use of analgesic medications or opioids, or a reduced risk of falls.10,41,42 Tofacitinib may also reduce the risk of fracture relative to placebo due to reduced need for oral steroids with better RA disease control.43,44 Our findings align with the results of a small randomized clinical trial of 30 patients with RA which found that 12 months of tofacitinib treatment stabilized BMD, increased markers of bone formation and decreased markers of bone resorption. 15
Data from ORAL Surveillance allowed for comparison of fracture risks associated with tofacitinib versus TNFi in an older population with ⩾ 1 additional CV risk factors. Although the current post hoc analysis was not designed to evaluate fracture as a primary or secondary endpoint, it showed numerically higher risk for fractures with tofacitinib versus TNFi in this particular population. This safety study of tofacitinib included TNFi as an active comparator and no placebo arm; therefore, the difference seen in the risk for fractures may imply that TNFi reduce fracture risk in patients with RA to a greater extent than tofacitinib, rather than reflecting an increase in risk with tofacitinib. In the subgroup analysis of these data, patients who were 50–65 years old had a numerically higher risk of fractures in the tofacitinib-treated arms compared with the TNFi arm (a difference that was not observed for all fractures in patients > 65 years old). Likewise, in ORAL Surveillance, female patients who received tofacitinib had a numerically higher risk of fracture than those who received TNFi. However, the p values for the interaction between age group (⩾ 65 or < 65 years) and treatment group (tofacitinib 5 or 10 mg BID, or TNFi), and that for the interaction between sex and treatment group, were both ⩾ 0.1, which does not support differential effects. Previous studies have suggested that the decline in ovarian function with menopause is associated with an increase in pro-inflammatory cytokines (IL-1, IL-6, and TNF-α), 45 but our clinical trials did not collect data on onset of menopause.
Overall, these analyses did not provide evidence of clear dose effects with tofacitinib 5 BID versus 10 mg BID. The multivariable Cox regression analysis adjusting for baseline predictors showed no difference in fracture risk between the two doses of tofacitinib in the P123LTE cohort. Similarly, in ORAL Surveillance, no clear differences in fractures were noted between the two doses. The recommended dosage of tofacitinib for RA is 5 mg BID for the immediate-release formulation or 11 mg once daily for the extended-release formulation,18,19 but the latter was not evaluated separately in the analyses reported here (some patients received the extended-release formulation in P123LTE and were included in the average tofacitinib 5 and 10 mg BID groups based on their average daily dose).
Based on the results of multivariable regression analysis in the P123LTE cohort and in ORAL Surveillance, risk factors for all fracture risk with treatment (tofacitinib in P123LTE, and tofacitinib or TNFi in ORAL Surveillance) included female sex (ORAL Surveillance only), older age (⩾ 65 years), and use of corticosteroids at baseline. These are well-known risk factors for fractures in the general population. 46 In addition, history of chronic lung disease (COPD or ILD) and history of osteoporosis were risk factors for all fractures in both cohorts. The increased risk observed with history of chronic lung disease may be reflective of cumulative exposure to smoking or COPD-related steroid treatment,47,48 both of which were also identified as risk factors in this analysis. Other known risk factors for fracture in women include time since menopause and age at menopause; 49 however, detailed history of menopause was not collected in any of the clinical data sources used and, therefore, this variable could not be included in the analysis. In the P123LTE cohort, history of fractures, baseline HAQ-DI, BMI > 25 to < 30 kg/m2, osteoporotic treatment, and use of antidepressants and opioids at baseline were also noted as independent risk factors for all fractures and/or osteoporotic fractures, which is consistent with previous studies in RA.8–11 However, RF and anti-CCP positivity were independent protective factors for all fractures and/or osteoporotic fractures in P123LTE, whereas both were associated with osteoporosis and BMD loss in previous studies;9,50 these findings also contrast with those for ORAL Surveillance, where RF and anti-CCP positivity appeared to be risk factors (either in simple or multivariable Cox models). Furthermore, statin use was identified as a risk factor for osteoporotic fractures in P123LTE, whereas several previous studies identified it as a protective factor.10,51 However, one retrospective analysis of medical claims data found that low-dose statin use protected against osteoporosis, whereas high-dose statin use increased the risk of osteoporosis. 52 In ORAL Surveillance, history of fractures, chronic renal disease, current/past smoking and proton pump inhibitor use at baseline were additionally identified as risk factors of all and/or osteoporotic fractures. Overall, the lack of interaction between treatment groups (tofacitinib and TNFi) and risk factors in ORAL Surveillance suggests the risk factors for fractures are common to both tofacitinib and TNFi; these risk factors were already known for the general population and patients with RA.
Some limitations should be noted for the analyses presented here. To begin, none of the tofacitinib trials were designed to assess risk of fractures. As a result, clinical fractures and falls were captured as adverse events, data on BMD were unavailable, and silent compression fractures could not be detected. Furthermore, this study was subject to the general limitations of post hoc analyses, although potential confounders were well balanced among the treatment groups, reflecting the large sample size and randomization. Moreover, although ORAL Surveillance was a safety endpoint study, it was specifically powered and enriched to evaluate major adverse CV events and malignancies (excluding non-melanoma skin cancer) in patients receiving tofacitinib versus TNFi. 31 In addition, p values were not corrected for multiple comparisons. Finally, the analyses presented here were exploratory in nature, and the baseline predictors identified do not imply causation.
Conclusion
In this post hoc analysis, pooled clinical trial data in patients with RA showed numerically lower fracture rates with tofacitinib versus placebo (in the phase III placebo-controlled cohort) and numerically greater risk versus TNFi (in ORAL Surveillance), without a clear dose effect. We did not identify any tofacitinib-specific risk factors for fractures in these analyses, and predictors of fracture risk were generally aligned with prior literature on the general population and patients with RA. Patients with fracture risk factors should be monitored and treated per established standards of care.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X221142346 – Supplemental material for Fracture in clinical studies of tofacitinib in rheumatoid arthritis
Supplemental material, sj-docx-1-tab-10.1177_1759720X221142346 for Fracture in clinical studies of tofacitinib in rheumatoid arthritis by Karen E. Hansen, Mahta Mortezavi, Edward Nagy, Cunshan Wang, Carol A. Connell, Zaher Radi, Heather J. Litman, Giovanni Adami and Maurizio Rossini in Therapeutic Advances in Musculoskeletal Disease
Footnotes
Acknowledgements
The authors would like to thank the patients, investigators, and study teams involved in the tofacitinib development program. The authors would also like to acknowledge David Gold, PhD, and Joseph Wu, PhD, employees and shareholders of Pfizer Inc., for important contributions to this work. Medical writing support, under the guidance of the authors, was provided by Julia King, PhD, CMC Connect, a division of IPG Health Medical Communications, and was funded by Pfizer Inc., New York, NY, USA, in accordance with Good Publication Practice (GPP 2022) guidelines (Ann Intern Med 2022;175:1298-1304). Editorial assistance with submission was provided by Eva Dekker, CMC Connect, IPG Health Medical Communications, and was funded by Pfizer Inc, New York, NY, USA. The authors authorized the submission of their manuscript via a third party and approved all statements and declarations, for example, conflicts of interest and funding.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
