Abstract
Bone fragility is the susceptibility to fracture even for common loads because of structural, architectural, or material alterations of bone tissue that result in poor bone strength. In osteoporosis, quantitative and qualitative changes in density, geometry, and micro-architecture modify the internal stress state predisposing to fragility fractures. Bone fragility substantially depends on the structural behavior related to the size and shape of the bone characterized by different responses in the load–deformation curve and on the material behavior that reflects the intrinsic material properties of the bone itself, such as yield and fatigue. From a clinical perspective, the measurement of bone density by DXA remains the gold standard for defining the risk of fragility fracture in all population groups. However, non-quantitative parameters, such as macro-architecture, geometry, tissue material properties, and microcracks accumulation can modify the bone’s mechanical strength. This review provides an overview of the role of different contributors to bone fragility and how these factors might be influenced by the use of anti-osteoporotic drugs and by the COVID-19 pandemic.
Keywords
Introduction
In materials science, brittleness is defined as a material’s tendency to break in an unwanted and sudden way without previous deformation. 1
If a material, even biological such as bone, is subjected to external stress, it will undergo deformation and changes its internal stress state due to the interactive exchange of the molecules that compose it. 2 As the external stress progressively increases, the material will deform by modifying the internal stresses up to a point limit beyond which it will break, and the intensity value of the external stress applied at that moment will be defined as breaking load. 2 In summary, the break (or fracture) will occur when the ratio between the deforming force and the strength of the structure exceeds the unit value.
In physiological conditions, the load/strength ratio for the bone is always less than 1, indicating a ‘mechanical’ competence of the structure to perform the function for which it is intended.3–5 Under normal conditions, the bone must be subject not only to the force of gravity but also to common additional mechanical stresses such as those that occur in postural changes, walking, running, jumping, or other dynamic activities. 6 In all these conditions, a normally structured bone resists and does not break. If the mechanical load increases beyond a certain threshold, for example, due to a jump from an excessive height, the bone can fracture. On the contrary, if the bone has structural, architectural, or material alterations, it can present less strength and therefore fracture even for common loads that it should normally be able to tolerate, such as a sudden postural variation, a minimum jump, or a fall from a standing position to the ground. 7 This is the typical condition of pathological bone fractures, including fragility or osteoporotic fractures.8,9
Fragility substantially depends on the interaction of bone behavior considered globally as an organ, and therefore composed of several tissues that are anatomically connected (structural behavior) and the mechanical features of the bone tissue (mechanical behavior).10,11 The structural behavior is related to both the size and shape of the bone and is largely determined by the amount of bone tissue present, and therefore by its density. The material properties of the bone determine its ‘material behavior’ and are independent of the size and shape of the sample, thus reflecting the intrinsic characteristics of the bone itself. 12
The structural properties of biological tissue (but also of any other material) are characterized by the relationship that exists between a force applied to it and the resulting deformation, namely the load–deformation curve.13,14 Using this curve, the structural behavior of the bone can be defined. Generally, load and strain are linearly correlated, in the so-called elastic region, until the yield point is reached when the slope of the load–strain curve is reduced. If the load is removed at this stage, the bone can return to its original shape without deformation. If the load increases beyond the yield point, the bone will undergo permanent deformation and will not return to its original shape even if the load clears (plastic region). A further increase in the load would cause a break (fracture) of the bone (Figure 1).
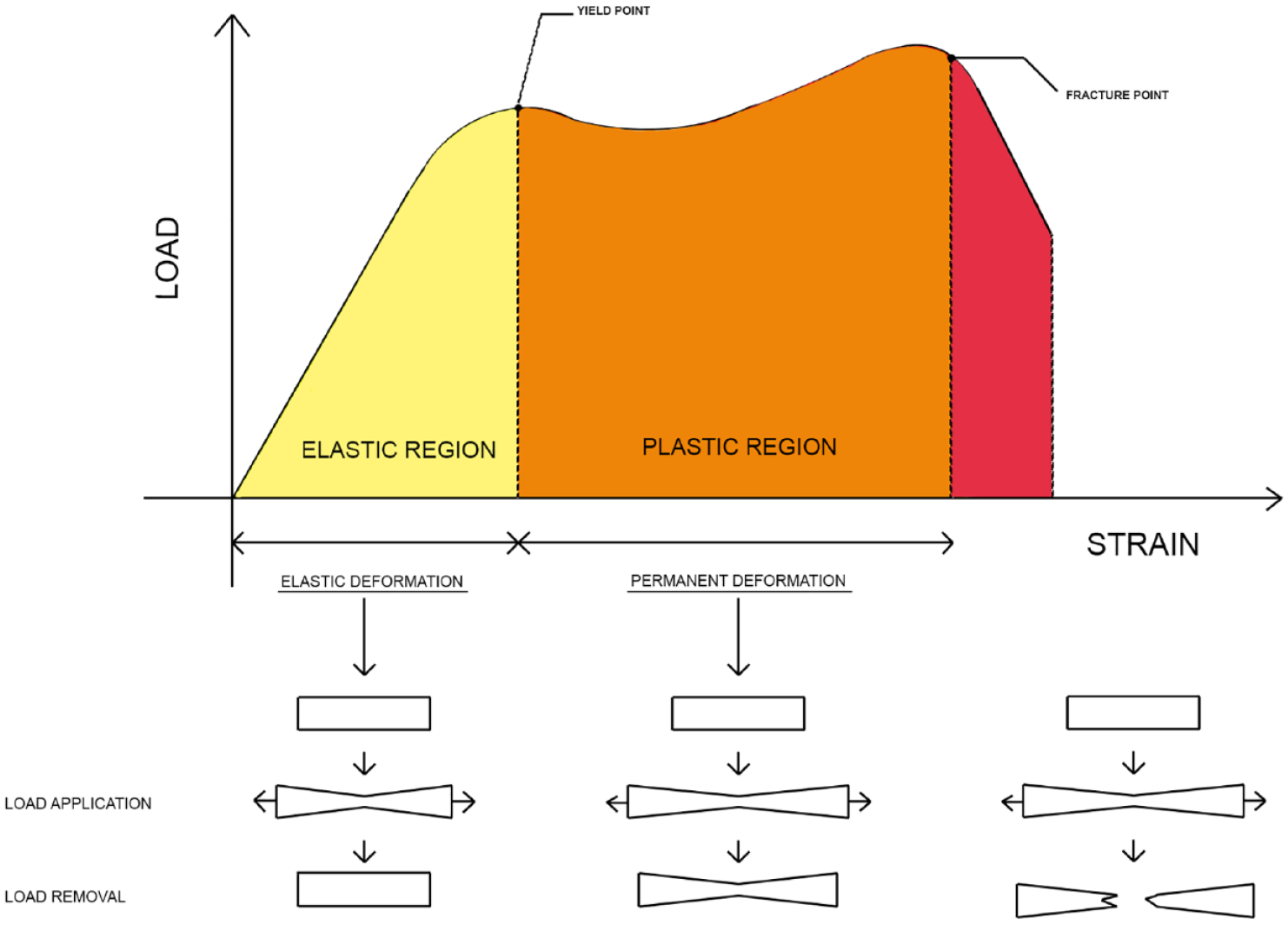
Load–deformation concept.
The material behavior of the bone tissue, on the contrary, depends on the geometry of the bone sample to be examined. 15 The applied force causes deformation of the sample by the forces generated within it. The deformation will be relative to that point and dependent on the ‘intensity’ of the internal forces (stress) at that point examined. The curve will then become of deformation for stress rather than to the load (stress/strain versus load/strain). 16
Using the stress/strain curve, we can determine the ‘toughness’ (modulus of toughness) of the bone, which is a biomechanical property that reflects the amount of work per unit volume of material required to fracture the bone identified as the area under the curve. 17 Hard (‘tough’) bone could be considered more resilient, although it can fracture at lower stress and, in this respect, be considered weaker. 18 Instead, fracture toughness is a measure of a material’s ability to resist the progression of an internal crack once it has already begun. The fracture toughness value of bone (3–6 MPa m½) is comparable to that of Plexiglas® or a ceramic such as alumina (Al2O3), while it is lower than that of aluminum alloys (from 20 to 30 MPa m½), of steel (from 70 140 MPa m½), or titanium alloys (from 70 to 110 MPa m½).19–21 Cortical bone has a relatively low fracture toughness due to its fragility, which makes it unsuitable for absorbing energy through plastic deformation similar to what happens with non-biological ceramic material. 11
Physiologically, bone possesses stress concentration points such as osteocyte lacunae, osteoclastic resorption loci, and points of passage of areas with different degrees of osteoid mineralization, which can represent starting points of the discontinuity that in the presence of low fracture toughness becomes an essential element of skeletal fragility.7,22
Another fragility fracture mechanism is ‘fatigue’, by which a structure subjected to repeated cyclic loads with mechanical stress levels lower than those necessary to overcome bone strength can break. This event occurs when the local stress concentrations within the material are much higher than the average stress of the tissues, causing a so-called fatigue failure which manifests itself as a stress fracture.23,24 These fractures can also occur with normal cyclic loading but are applied to pathologically weakened bone (e.g. osteoporosis). 25 On the contrary, in this case, there will also be stress concentrators such as the largest osteoclastic resorption gaps. 26
The stress/strain curve can also provide information on the tendency of a tissue to accumulate damage and on the mechanisms underlying its structural failure. 27 A material that fractures immediately after reaching the yield point is a material that has little plastic deformability and is defined as ‘brittle’ in biomaterial science. 28 It should be noted that even if the behavior of the material can be described as fragile, the material itself could be very resistant. 28
On the contrary, a material that has wide deformability in the plastic regime is defined as ductile. 29 The material properties of the bone vary depending on its orientation for the direction of the load to which it is subjected (anisotropy), so the shaft of a long bone is more resistant to compressive load than the transverse one. 30 Even a bone composed mainly of cancellous tissue such as the vertebra is much more resistant to a load in the vertical direction than transverse. 31
In the field of the analysis of the structural and material properties of bone, the measurement of the amount of bone, and therefore its density, is undoubtedly the most easily to be assessed through the instrumental approach. 32 The quantification of bone mass with the dual-energy X-ray absorptiometry (DXA) examination is a routine investigation to define the risk of fragility fracture in all population groups, from children to the elderly, reaching a diagnostic capacity in the latter and sometimes also providing a therapeutic threshold for drug treatment.33,34 On the contrary, as regards non-quantitative aspects, ‘bone quality’ is an umbrella concept under which numerous factors are identified that can modify the mechanical strength of the bone itself. 35 Among these, we can list the rough three-dimensional geometry of the bone (e.g. macro-architecture, percentage distribution of compact or cancellous tissue typology, periosteal thickness), micro-architecture (spatial arrangement of cancellous trabeculae and Haversian canals in the compact bone), the material properties of the tissues [arrangement of hydroxyapatite crystals, nature and three-dimensional structure of collagen, nature and arrangement of non-collagenic proteins (NCPs), age-related changes in the organic and inorganic molecules of the bone tissue], presence of microcrack, and accumulation of micro-traumatic injuries (Table 1).36,37
Glossary of bone fragility.

In this review, we will try to analyze the role that these various factors play in the genesis of skeletal fragility, including therapeutic implications.
Bone mass
Skeletal fragility is significantly associated with the reduction in bone density, which is currently measured by the DXA as the gold standard for clinical practice.33,34 DXA allows measuring bone mass and bone mineral density (BMD) in g/cm2 of the projected bone area accurately and precisely.38,39
According to the World Health Organization (WHO), the BMD value obtained by DXA and compared with the mean BMD of healthy and young adults of the same sex allows the densitometric diagnosis of osteoporosis. 40 The unit of measurement is represented by the standard deviations (SD) from the mean peak bone mass (T score). The risk of fracture begins to increase exponentially with densitometric T score values < –2.5 SD, which is the threshold beyond which a diagnosis of osteoporosis can be made (diagnostic threshold).41,42
Although it is still recognized as having a primary role in the diagnosis of bone fragility, DXA has some critical issues. The measurements obtained are not density but areal, with consequent underestimation of BMD in patients of small size and overestimation in those of greater size. 43 DXA fails to differentiate trabecular from cortical BMD, resulting in an inability to accurately analyze compartmental bone changes that may be common in patients with pathologies that induce cortical or juxta-articular porosity. 44
Recent advancement of the DXA method also allows for the evaluation of a series of geometric parameters of the bone examined that are related to its mechanical strength. The hip structural analysis (HSA) evaluates some geometric parameters of the proximal femur such as the cross-sectional area (CSA), the moment of inertia of the cross-section, the section modulus, and the buckling ratio. 45 The trabecular bone score (TBS) consists in processing the degrees of inhomogeneity in DXA scans at the lumbar spine, as indirect variables of the trabecular micro-architecture. 46 The TBS analysis seems to improve the quantification of fracture risk in subjects with mild BMD loss. 47
Pharmacotherapy of osteoporosis and BMD
DXA is used both in randomized controlled trials (RCTs) and in clinical practice as a surrogate measure of efficacy for the reduction of bone fragility and fracture risk since the association between increased BMD and reduced fracture risk has been demonstrated in subjects treated with anti-osteoporotic drugs. A meta-analysis of 13 RCTs of various antiresorptive drugs in postmenopausal women showed through a Poisson regression model that this therapy by increasing the BMD of the lumbar spine by 8% reduces the risk of spinal fractures by 54%. 48
Similar data are obtained by analyzing the association between the increase in BMD of the proximal femur and lumbar spine and the reduction of the risk of non-vertebral fractures. 49
It should be emphasized, however, that in serial DXA controls, we can consider substantial stability of BMD over time as an acceptable response to drug therapy, with a consequent reduction in the risk of fracture. Therefore, repeated measurements of BMD, in facilities that ensure optimal quality standards, can play a role in the clinical management of patients not only by increasing compliance with drug treatment but also by providing quantitative information on the efficacy that can be subject to reconsideration of the therapeutic approach. 50
BMD change obtained with teriparatide may not be correlated with the reduction of fracture risk since these drugs can cause a decrease in bone mass at the proximal femur (due to cortical porosis), which is, however, accompanied by a reduction in both vertebral and non-vertebral fracture risk.51,52
Denosumab, a monoclonal antibody against RANK-L, has been tested on cynomolgus monkeys in preclinical studies, showing that it can increase the BMD of the lumbar spine and proximal femur by 7–12%. 53 The drug would act not only by reducing osteoclast resorption in bone remodeling but also by increasing the deposition of new bone through bone modeling. 54 This combined action could explain the continuous increase in BMD observed during treatment with denosumab, unlike what was found with bisphosphonates (BPs), which do not have further significant effects on BMD after 2 years of treatment. 55
Osteoanabolic drugs and bone fragility
Osteoanabolic drugs, while globally increasing bone remodeling, substantially favor bone formation, causing an increase in BMD, which is also accompanied by an improvement in the structural and material properties of the bone tissue which overall reduces the risk of fracture. 56
In particular, parathyroid hormone (PTH) and its analogs stimulate the formation of cancellous bone at both the trabecular and endocortical sites with a consequent increase in BMD, with a limited effect on the formation of subperiosteal cortical bone. 57 The effect of PTH on bone turnover is, however, transitory and decreases over time due to presumed exhaustion of the remodeling surfaces. 58
More recently has been marketed a new osteoanabolic drug that acts to modulate the Wnt/beta-catenin signaling by inhibiting the activity of sclerostin. 56 This protein was known for two genetic diseases in which it is poorly expressed with consequent alteration of bone metabolism and osteosclerosis, namely sclerosteosis and van Buchem disease, both autosomal recessive diseases from mutations with loss of function of the SOST gene coding for sclerostin. 59 This depletion increases the differentiation of osteoblasts by activating the canonical Wnt pathway, with a decoupling between bone formation and bone resorption, and greater formation of both subperiosteal and endosteal trabecular and cortical bone. 60
Anti-sclerostin antibodies (Scl-Ab) have been shown, in preclinical studies, to be strong stimulators of short-term bone formation and suppressors of long-term bone resorption, resulting in marked increases in BMD. 61
In clinical trials, two monoclonal antibodies against sclerostin, romosozumab, and blomosozumab, were tested in osteoporotic patients, while setrusumab, a third neutralizing fully human IgG2l monoclonal antibody, is currently being studied only for osteogenesis imperfecta and hypophosphatasia.62,63
Of the two, only romosozumab is already used in clinical practice, having confirmed the strong anabolic potential in phase I and II clinical studies and documenting a marked increase in lumbar spine BMD much higher than that obtained with teriparatide or BPs.64,65
In the phase III registration study (FRAME), BMD in treated patients increased at the lumbar spine by 13.3% and by 6.9% at the total hip after 1 year of treatment.65–68
Even in men treated with romosozumab after 1 year of treatment, the BMD increased in the lumbar spine by 12.1% and by 2.5% in the hip. 69
In the FRAME study, 1 year of therapy with romosozumab was followed by another year of treatment with denosumab. 66 This sequential therapy resulted in an increase in BMD of 17.6% at the lumbar spine and 8.8% at the hip. In addition, in patients who continued an additional 12 months of denosumab treatment, there was an additional 4.3% increase in lumbar spine BMD and 1.7% hip BMD increase. The overall analysis of the effectiveness of the sequential therapy of romosozumab followed by antiresorptive shows that denosumab is superior in continuously increasing bone density and mechanical strength.
The discontinuation of treatment with Scl-Ab after 6 months, in animal models, resulted in a gradual decrease in BMD at baseline while the mechanical strength of the bone and its material properties appeared to be partly preserved, especially in cortical sites.70,71 Subsequent studies in humans seem to confirm this finding, with a gradual decrease in BMD after discontinuation of treatment with Scl-Ab. 72
It should be emphasized that treatment with romosozumab, unlike what happens for PTH analogs, can be repeated several times during the management of patients with severe osteoporosis. Indeed, in a phase II study, romosozumab was given for a second 12-month course, following an initial 24-month treatment period with romosozumab followed by either placebo or denosumab for a 12-month treatment period. 72 In the romosozumab–placebo–romosozumab group, lumbar spine BMD increased by 15.5% during the first course of treatment with romosozumab, followed by a loss of BMD of 9.1% during treatment with placebo and again an increase of 12.4% after the second course of treatment with romosozumab with comparable results for total hip BMD changes. In the romosozumab–denosumab–romosozumab group, the initial increase in lumbar spine BMD was 16.4% followed by a further 2.5% increase during denosumab treatment and again a 2.3% increase with the second course of treatment with romosozumab. Similar percentages of increase were seen for hip BMD. Romosozumab–denosumab–romosozumab sequential therapy resulted in an overall increase of 22.1% in lumbar spine BMD and a 7.3% increase in hip BMD.
Bone geometry and macro-architecture
The size and shape play an important role in the mechanical behavior of the bone and generally, large bones are more resistant than smaller ones. 73 In everyday life, bones undergo a combination of compressive and tensile loads with bending and torsional moments that occur in both the axial and appendicular skeletons.74,75 The mechanical efficiency for resistance to bending and torsional loads requires the distribution of the bone away from the neutral axis of bending or torsion and is determined by the moment of inertia of the area, which is, in a cylindrical bone, proportional to its diameter at the fourth power. 76 As a result, small increases in the outer diameter of a long bone can greatly improve its resistance to bending and torsional loading. The compressive strength of the same cylindrical bone, on the contrary, is directly proportional to the area of the transverse section. 10
Pharmacotherapy of osteoporosis and bone geometry, and macro-architecture
Placebo groups of trials investigating changes in bone geometry in women over 65 after treatment with anti-osteoporotic drugs have shown that with years in the proximal femur there is a significant increase in the diameter of the femoral neck (+1.4%) and section modulus (+3.5%), while there are no variations in the CSA of the bone or in cortical thickness. 77 On the contrary, subjects treated with antiresorptive drugs [hormone replacement therapy (HRT) or alendronate] showed significant increases in bone CSA (from 3.5% to 7%), cortical thickness (from 1.5% to 5%), and section modulus (from 6.5% to 10.5%) compared with both baseline and placebo. The femoral neck diameter was increased from baseline (1.3% to 2.0%) but was no different from the placebo. 78
Poole et al., 79 in a three-dimensional (3D) mapping study of the proximal femur analyzed on computed tomography (CT) scans of postmenopausal women, reported that treatment with denosumab led to significant increases in surface density and thickness of the cortical mass already after 1 year, which reached +5.4% in 3 years, largely due to the increase in cortical thickness. At 3 years, the increases in thickness and density reached 12% in some critical points particularly subject to traumatic stress such as the lateral portion of the trochanter. According to the authors, this activity of denosumab could partly justify its speed and power in terms of reduced fragility fracture risk.
After 18 months of teriparatide treatment, in the proximal femur, there was a significant increase in cortical thickness and section modulus compared to baseline (from 4% to 7%) with an unchanged internal diameter, while this drug did not have any effect on the structure of the femoral shaft. 80
Romosozumab, thanks to its dual action on bone modeling and remodeling, has an important impact on the mechanical strength of the bone by significantly changing its density and geometry. The rapid initial effects based on modeling consist in depositing an additional layer of osteoid on the periosteal surface, without any increase in cortical porosity unlike what happens with teriparatide. 81 At the same time, new osteoid is deposited, thanks to the remodeling activity, also at the endocortical site. Recently Poole et al. 82 analyzed the lumbar 3D CTs of postmenopausal women treated with romosozumab, placebo, or open-label teriparatide, showing a small but statistically significant increase in BMD Ct (+2.1%) in the romosozumab group and a decrease in the teriparatide group (–0.1%). Furthermore, the authors were able to separate the thickness variations resulting from the therapy in an external cortical component and an endocortical one, that is, in the transition region from cortical bone to trabecular bone, showing a significant increase of 137.6% and 47.5% for romosozumab and teriparatide, respectively, mostly due to endocortical apposition, although the existence of periosteal apposition cannot be excluded.
Bone micro-architecture
The introduction of high-resolution imaging techniques has allowed a non-invasive evaluation of bone micro-architecture. High-resolution peripheral quantitative computed tomography (HR-pQCT) and magnetic resonance imaging (MRI) were used to analyze bone microstructure under normal and pathological conditions. The HR-pQCT allows the simultaneous acquisition of a stack of parallel CT slices with a nominal resolution (voxel size) of 82 μm. 83 Thanks to this method, 110 slices are obtained at the distal radius and the tibia during a temporal scan of 3 min, thus providing a 3D representation of approximately 9 mm in the axial direction. 84 The region of interest (ROI) is automatically separated into a cortical and a trabecular region using a threshold-based algorithm. Of this, the total volumetric, trabecular, and cortical bone density are measured, along with the number and distribution of the trabeculae and the CSA. Trabecular bone volume, trabecular thickness, and separation are calculated with standard formulae from histomorphometry. In particular, cortical porosity can be evaluated with this technique. Large case–control and cross-sectional observational studies documented that the micro-architecture of the bone significantly influenced the fracture risk.85,86 In these studies, it was shown that among osteopenic women those with fragility fractures had a lower trabecular density and a more heterogeneous trabecular distribution compared with non-fractured women with the same BMD. Similarly, HR-pQCT measurements of the cortical parameters of the distal tibia, and in particular its porosity, can distinguish osteopenic women with fractured wrists from non-fractured women.
We do not yet have robust data to demonstrate with certainty that the micro-architectural changes evaluated at HR-pQCT can predict an incident fracture in postmenopausal women, although in a prospective cohort of French postmenopausal women, after adjustment for hip areal BMD (aBMD), total volumetric BMD (vBMD), trabecular vBMD, trabecular number and connectivity to the distal radius, this imaging technique was found to be predictive of incident fracture. 87
Pharmacotherapy of osteoporosis and bone micro-architecture
Anti-osteoporotic drugs can modify some bone micro-architectural parameters as documented in various RCTs. 88 Treatment with alendronate would result in significant improvement in HR-pQCT parameters from baseline in the distal tibia. 89 Risedronate would also counteract the decline of vBMD and cortical thickness (Ct.Th) at the distal tibia, 90 as would oral ibandronate after 24 months of therapy. 91 In an RCT of 247 postmenopausal women, denosumab was superior to alendronate, which, in turn, was superior to placebo, after 12 months of treatment in preventing micro-architectural deterioration assessed with HR-pQCT, with even improvement in the denosumab group. 92
The effects of romosozumab on bone strength and micro-architecture parameters were examined in ovariectomized (OVX) cynos. 71 After 6 months of therapy, romosozumab resulted in a rapid increase in bone mass at both the spongy and cortical bone tissue. This drug transiently increases intracortical activation without significant increase of the cortical porosity at the distal radius.
In preclinical studies, romosozumab increased bone strength in the lumbar vertebrae, femoral neck, and femoral shaft to a much greater extent than with other anti-osteoporotic drugs and mainly as a result of activation of bone formation.93–95
Molecular composition
Bone tissue is a heterogeneous composite material consisting of an inorganic mineral phase, hydroxyapatite (Ca10(PO4)6 (OH)2), and an organic phase composed of about 90% of type I collagen, 5% NCP, 2% lipid and water. 96
The strength of the bone depends on various factors, which are globally identified in two large categories of the quantity and quality of the bone itself. Among the ‘qualitative’ factors, there is the percentage distribution of each component of the bone tissue (cellular and extracellular), 97 the distribution and interaction between collagen and NCPs including the quantity and type of bonds among collagen fibers,98,99 the state of mineralization intended as the type, 100 quantity, and geometric arrangement of mineral crystals, the level of homogeneity of the mineral structure, 101 and finally the presence of microcracks. 102
All these factors are changing over time because of bone growth, modeling, and remodeling. They can also be negatively influenced by the person’s health condition and the use of some drugs.
The mineral component of bone can be studied with various imaging techniques, including BMDD (bone mineral density distribution), the Raman method, and Fourier infrared spectroscopy.103,104 The parameters obtained through these techniques are a measure of the heterogeneity of mineralization and the alteration of the normal distribution of calcium (BMDD) and phosphate [Fourier transform infrared spectroscopy (FTIR), Raman] in bone tissue as occurs in osteoporosis but also osteomalacia and osteogenesis imperfecta. 105 In this latter condition, the mineral content (mineral/matrix ratio) is increased due to a lower amount of collagen as a result of improper collagen matrix production. 106 In osteoporosis, the ratio is increased due to an unbalanced reduction in bone formation. 107 In osteomalacia, the mineral/matrix ratio is unchanged when only the mineralized tissue is examined, while, as is known, in this pathology the osteoid tissue produced is often not mineralized.108–111
It should be emphasized that the number of osteoclasts can be unchanged, but their activity has increased or decreased. Furthermore, in osteoporosis, there is usually a reduction in W.Wi (average wall width), which is a variable related to the depth of erosion from previous remodeling activity and is a measure of the amount of bone formed in a given basic multicellular unit (BMU), driven by either increased osteoclast activity or reduced osteoblast activity (or a combination of both).112,113
From a histological point of view, osteomalacia is characterized by a newly formed bone matrix that does not mineralize promptly, resulting in the accumulation of thick osteoid junctions and, in some cases, a volume of osteoid tissue that can represent up to 40% of the total bone volume. 114 As it also appears in imaging, osteomalacia is characterized by low bone mass in the presence of a normal bone volume, but largely non-mineralized. To diagnose osteomalacia, however, it is necessary to evaluate not only the volume of the osteoid but also its width in some areas. 115 Large volumes of osteoid can also occur due to high bone formation with normal mineralization rates but in this situation, there are no more thickened junctional points. From the histological point of view, an osteoid tissue with a width >12 μm is, however, to be considered pathognomonic of osteomalacia.108,116
Pharmacotherapy of osteoporosis and molecular composition
The mechanisms of action of the antiresorptive drugs might cause a progressive aging of hydroxyapatite, which undergoes prolonged secondary mineralization. This phenomenon might lead to lower ductility, and therefore a lower capacity to absorb the load resulting in propensity to bone fragility in the long term. The increase of the mineral maturity at both trabecular and cortical bone was confirmed in a recent study on OVX rats treated with oral BPs, 117 where their antiresorptive effect was followed by a prolonged secondary mineralization. Also denosumab significantly enhances secondary bone mineralization due to its potent inhibition of bone remodeling at both the osteonal and interstitial levels. However, the mineral features (maturity, crystallinity, and carbonation) do not seem to undergo significant changes after treatment with denosumab, thanks to its ability to partly favor modeling, allowing deposition of new crystals on the mature hydroxyapatite. Indeed, the organic matrix in treated patients is not significantly modified at least after a short treatment (2–3 years) with denosumab. 118
The anabolic stimulation of osteoblasts’ activity due to teriparatide causes a marked increase in the synthesis of collagenic and NCPs and the formation of new crystals, which are typical characteristics of young bone. The mineral matrix appears to be quantitatively lower with low maturity of mineral crystals, while the organic part has higher water content and a lower content of glycosaminoglycans, with a lower ratio of pyridinoline/divalent enzymatic collagen cross-link. 119
In animal and human models of sclerostin deficiency, enhanced bone formation led to increased proteoglycan content, lower matrix mineralization, and lower maturity/crystallinity of hydroxyapatite suggesting that romosozumab might improve bone strength also through molecular composition changes. 120
Figure 2 shows different pathogenic mechanisms of bone fragility and relative influence of anti-osteoporotic drugs.

Determinants of bone strength and possible mechanism-based treatment for bone fragility.
Bone fragility and age-related bone alterations in the COVID-19 scenario
Aging is a multifactorial biological process that involves the whole organism and that alters the mechanical strength of the bone making it more fragile. 121 The main age-related factors that reduce skeletal resistance include the following:
The decrease in the autophagic mechanism and the consequent accumulation of unwanted substances; 122
The hyperproduction of pro-inflammatory factors such as interleukin (IL)-1β (IL-1β), with deleterious effects on skeletal remodeling; 123
The inflammaging; 124
The numerical and functional depletion of stem cells, with the prevalent differentiation of residual stem cells in adipoblastic rather than osteoblastic precursors, resulting in a significant reduction in bone mass and an increase in intraosseous adipose tissue; 125
The increase of reactive oxygen species (ROS), which may be related to the blocking of the Wnt signaling and to the transfer of β-catenin with consequent transcriptional block; 126
Immunosenescence characterized by a decline in the vitality and precision of the adaptive immune system in old age, with reduced diversity of T lymphocytes which would negatively affect skeletal remodeling; 127
Significant reduction in the endogenous production of vitamin D due to a decrease in the activity of both 25-hydroxylase and 1α-hydroxylase. In addition, aging affects the skin’s ability to produce vitamin D. 128
SARS-CoV-2 infection and subsequent COVID-19 can damage many organs. Multisystem involvement can occur acutely and go to remission within weeks, but it can also increase the risk of long-term consequences that characterize long-COVID syndrome. 129 Since SARS-CoV-2 infection mainly affects older people, one of the things to consider shortly is its negative interaction with bone turnover. To our knowledge, it is still unclear whether the incidence of osteoporosis and the risk of fracture may increase in patients after recovery from the infection, due to the potentiation of the negative effects of the well-known risk factors (aging, smoking, bedrest for a prolonged time, hypovitaminosis D, corticosteroids) in combination with the inflammatory process during COVID-19. In patients after recovery from severe acute respiratory syndrome (SARS), osteonecrosis and other bone changes with reduced bone density were observed, 130 which were partly but not fully explained by corticosteroid treatment, the severity of infection, or several comorbidities that markedly increase the production of inflammatory mediators. 131 Significantly higher serum concentrations of pro- and anti-inflammatory cytokines, including IL-6, tumor necrosis factor-α, and IL-10, suggest that disease severity may be associated with a ‘cytokine storm’. Several cytokines, in particular RANKL, play a fundamental role in osteoclastogenesis in physiological and pathological conditions. Although the long-term effects of COVID-19 are still unknown, the presumed consequences of the disease would likely be similar to those of SARS, with viral infection–mediated effects stimulating osteoclastogenesis. Similarly, SARS-Cov-2 could have direct and indirect effects on osteoclastogenesis and, consequently, on bone resorption. In particular, the infection-related osteoimmunological response might directly stimulate the differentiation and osteoclastic activation, while the cytokine storm might indirectly favor bone resorption by upregulating osteoclasts. 130
COVID-19 pandemic has further complicated the clinical condition and the management of people with bone fragility for several reasons. Immobilization drives rapid muscle wasting that will contribute to increased fall risk and fracture incidence.132,133 Also, National Health Systems prioritized urgent visits and delayed elective care, leading to the underutilization of orthopedic services, including osteoporosis outpatient clinics. 134 It is key to note that patients with fragility fractures are at the highest risk of another fracture in the following year, which for many patients coincided with the period of poor access to healthcare services due to the COVID-19 pandemic. 135 Moreover, patients with hip fractures were quickly discharged after surgery, often without anti-osteoporotic therapy, post-surgical rehabilitation, or follow-up planning. This is a dramatic scenario, considering that bone fragility is rising globally with foreseeable consequences in terms of mortality, disability, and financial burden. Furthermore, the treatment gap will presumably spread considering that also before the COVID-19 pandemic only 20% of hip fracture patients received anti-osteoporotic drugs. 136 The multidisciplinary approach advocated by the Fracture Liaison Service programs was demonstrated to be effective to guarantee the appropriateness of care after fragility fractures. However, the CDC COVID-19 guidelines led to the closure of routine primary or specialty care, including fracture liaison service (FLS) and rehabilitation services. 137 Taken together, these issues might lead to a lack of attention and funding for the care of people with bone fragility.
Conclusion
Bone fragility is a common and multifaced pathological condition depending on qualitative and quantitative changes in bone tissue predisposing to fracture. Factors determining bone strength are positively influenced by anti-osteoporotic drugs through different mechanisms of action. Most conditions might adversely affect bone health causing a reduced resistance of the skeleton to mechanical stress and increased risk of fragility fractures. In this scenario, COVID-19 could be a new player even if we cannot exactly define what and how the pathogenic mechanism of SARS-CoV-2 infection and related therapies could interact with the key components of bone strength. However, COVID 19 pandemic just seems to have severely affected management strategies for bone fragility and fractures, whose implications need to be quantified in the next years.
