Abstract
SARS-CoV-2 has caused a global pandemic and an unprecedented public health crisis, infecting more than 580 million people worldwide. Moreover, recent evidence has suggested the emergence of a new syndrome known as Long-COVID, a term used to describe a diverse set of physical and mental symptoms that persist after a diagnosed SARS-CoV-2 infection. Epidemiological data have identified myalgias, muscle and joint dysfunction, and bone fragility as common sequelae in patients with moderate and severe forms of this disease. Significant musculoskeletal dysfunction has also been detected in some healed patients, although knowledge about pathophysiological mechanisms of Long-COVID is still rather scarce. In this context, SARS-CoV-2 infection has been suggested to amplify the effects of aging on the musculoskeletal system by aggravating the osteosarcopenic state. Based on this evidence, our review focused on the muscle and bone tissue alterations induced by SARS-CoV-2 infection and Long-COVID, summarizing the current knowledge on the underlying biological mechanisms and highlighting the need for a multidisciplinary approach to predict the musculoskeletal targets and long-term consequences of COVID-19 disease.
Introduction
The new coronavirus SARS-CoV-2, which suddenly emerged in December 2019 in the city of Wuhan, is still haunting the entire human race, affecting not only health, but also global socioeconomic systems. According to the most recent estimates provided by the World Health Organization (WHO), there have been approximately 580 million confirmed cases and around 6.5 million confirmed deaths worldwide since the pandemic began, highlighting how the SARS-CoV-2 outbreak is a true health emergency of international significance. 1
The clinical manifestations of SARS-CoV-2 are not yet fully known, but symptoms vary from asymptomatic or mild to severe. Moreover, susceptibility to SARS-CoV-2 infection appears not to vary with age or gender. However, several evidence suggest that among all known patients, those who are older, male and have underlying comorbidities are more likely to have a more severe course of COVID-19, and a higher risk of developing complications. 2
Undoubtedly, aging has been associated with worse outcomes, due to pathophysiological changes in the respiratory system and, especially, the upper airways, considered the entry site for SARS-CoV-2 infection. 3 Coronavirus infectivity has been found to depend on its entry via the binding of its viral spike (S) protein to the receptor of angiotensin-converting enzyme 2 (ACE2) and the triggering of the S protein by the type II transmembrane serine protease (TMPRSS2). Interestingly, a decrease in ACE2 activity is known to increase the severity of lung lesions.4,5 Particularly, a strong association was found between reduced ACE2 levels in older patients and greater severity of disease, which in some cases progressed to acute respiratory distress syndrome (ARDS). 6 In fact, according to data provided by the Istituto Superiore di Sanità (ISS) in Italy, ARDS was observed in approximately 97% of patients who died in hospital, who were predominantly elderly and with acute heart, liver and kidney function disorders.7–9
Interestingly, a possible gender difference in the susceptibility and progression of COVID-19 has been proposed to depend on the ACE2 localization on the X chromosome. 10 In this regard, Fan et al. 11 recently observed that gender significantly influences the aberrant methylation of the ACE2 promoter, possibly making men more susceptible to the risk of COVID-19 infection. In contrast, other evidence has found no significant differences in ACE2 expression between males and females, making this hypothesis less likely. 12
The rapid identification of people at risk and the management of infected patients have been key to slowing the spread of COVID-19 worldwide, through a pathway based on early identification, immediate isolation, implementation of appropriate infection prevention and control measures, and provision of optimized supportive care.13,14 However, the emergence of many new variants of concern over time has increased the transmissibility or virulence of SARS-CoV-2, suggesting the need to develop efficient public health measures and vaccination programmes through a timely and science-based global response.15,16 Indeed, thanks to the relentless efforts of scientific research, a clear picture of the structure and genetic composition of SARS-CoV-2 is now known, and its host source, epidemiological characteristics and the histopathological changes that occur in lung tissue in response to infection.17–20 However, many questions remain unanswered, including the production of new strains and the mechanisms of infection spread to other target tissues, including the heart, intestine, kidney, bone, muscle, skin, liver, and some brain areas, with pathological changes very similar to those found in severe acute respiratory syndrome (SARS) and middle east respiratory syndrome (MERS) viral infections. 21
In this context, the aim of our review was to summarize the current scientific knowledge on how SARS-CoV-2 infection could amplify the effects of aging, especially on the musculoskeletal system, and exacerbate the osteosarcopenic state, characterized by concomitant bone loss and muscle atrophy, suggesting the need for a multidisciplinary approach to help predict the musculoskeletal targets and long-term consequences of COVID-19 disease.
Literature search strategy
For this narrative review, 98 articles were selected from the MEDLINE bibliographic database, published between 1945 (start date) and 2022. Articles concerning SARS-Cov-2 infection and, specifically, the effects of Long-COVID on the musculoskeletal system were included. The search strategy was based on the use or combination of the following keywords: ‘SARS-CoV-2 infection’; ‘Long-COVID’; ‘musculoskeletal system’; ‘osteoporosis’; ‘sarcopenia’; ‘osteosarcopenia’; ‘aging’; ‘fragility fractures’; ‘prevention’. The search process was carried out on a worldwide basis, without excluding specific geographical areas or different ethnic groups. Language and species filters were applied to the list of results to eliminate non-English articles.
The musculoskeletal system as a potential target
SARS-CoV-2 infection causes severe acute inflammation, triggering a storm of cytokines and inducing multi-organ damage that also involves the musculoskeletal system (Figure 1).22–24 According to Goshua et al., 25 such injury could be mediated by endothelial damage and a propensity for micro- and macro-thrombosis, through indirect mechanisms responsible for cytokine release and subsequent homeostatic disruption. Alternatively, Ferrandi and colleagues proposed that the inflammation of musculoskeletal tissue could depend on a direct interaction of skeletal muscle cells with SARS-CoV-2 via ACE2. 26 Indeed, the ACE2 expression in skeletal muscle is well documented and its potential susceptibility to coronavirus is partially supported by clinical evidence pointing to pain, weakness and skeletal muscle injury as common symptoms of COVID-19 disease.27–29 In addition, several pro-inflammatory cytokines, such as interferon gamma (IFN-γ), interleukin 1 beta (IL-1β), interleukin 6 (IL-6), interleukin 17 (IL-17) and tumour necrosis factor alpha (TNF-α), whose levels were found to be elevated in COVID-19 patients, could directly induce proteolysis of muscle fibres and decrease protein synthesis, with a significant impact on skeletal muscle.30,31 Particularly, IL-1β and TNF-α are well known to inhibit satellite cell proliferation and differentiation, just as IL-6 is known to promote muscle fibroblast activity and lead to fibrosis, impairing muscle function and increasing susceptibility to injury.32–34

SARS-CoV-2 infection is responsible for severe acute inflammation, triggering a storm of cytokines and inducing multi-organ damage. The musculoskeletal system is also one of the target tissues infected by the virus. The ACE2 expression in muscle and bone cells could promotes virus binding, causing myalgia, weakness, and injury in muscles, and demineralization and fragility in bones.
Among the possible damage caused by the cytokine storm, elevated c-reactive protein (CRP) concentration and subsequent mitochondrial damage have also been correlated with the onset of sarcopenia and frailty in COVID-19 patients. 35 In fact, a role for systemic inflammation has also been proposed in the involvement of bone and joint tissue in infected patients, although the underlying mechanisms are still largely unknown. 31 Specifically, the inflammatory cytokines detected in large quantities in COVID-19 patients with a severe disease course, such as IL-1β, IL-6, TNF-α, granulocyte colony-stimulating factor (G-CSF), interferon gamma-induced protein 10 (IP-10) and monocyte chemoattractant protein-1 (MCP-1), are known to be mediators normally involved in bone turnover, with a role in osteoclast formation and increased bone resorption capacity via the receptor activator of nuclear-factor κB (RANK)/RANK ligand (RANKL)/osteoprotegerin (OPG) pathway. 17 In this regard, Zheng et al. 36 suggested that the evolution of inflammation from acute to chronic could promote the recruitment of osteoclasts by inflammatory cytokines, reducing osteoblastogenesis and triggering bone loss. In addition, IL-1β, IL-6, IL-17 and TNF-α could induce chondrolysis, causing arthralgia and osteoarthritis, and impair the normal biological activity of tenocytes, altering matrix remodelling and exacerbating tendon degenerative disorders.
Long-COVID: epidemiology, pathogenesis and clinical manifestations
People infected with SARS-CoV-2 commonly develop symptoms a few days after exposure, and most show complete recovery within a few weeks after infection. However, ongoing follow-up of patients healed of COVID-19 has shown that one or more symptoms persist in a substantial proportion of people, even weeks or months after recovery.37,38 In this regard, an Italian patient in May 2020 used the term ‘Long-COVID’ to describe the persistence of symptoms weeks or months after the initial SARS-CoV-2 infection. Since then, it has passed through various social media outlets to clinical experts and formal policymakers, acquiring a reasonable consistency. Thus, the phenomenon of Long-COVID, or post-COVID syndrome, has become an emerging problem and, according to the most recent estimates, could produce another public health crisis like the pandemic itself.39,40
The follow-up of patients healed of infection allowed the identification of some factors commonly associated with the Long-COVID development. Among these, age and gender seem to play a key role, as patients with persistent symptoms were generally men about 4 years older than those without symptoms. 41 The presence of comorbidities and the number of symptoms characterizing the acute phase of the disease have also been suggested to increase the risk of Long-COVID. 42
Several scientific evidences have shown a prevalence of Long-COVID after acute SARS-CoV-2 infection of between 10% and 30%. 37 Specifically, Dennis et al. 43 conducted a prospective observational cohort study to assess medium-term organ impairment in individuals with a mean age of 45 years and persistent symptoms after initial recovery from acute SARS-CoV-2 infection compared to healthy age-matched controls. Of a total of 201 patients, 42% had 10 or more Long-COVID symptoms 4 months after initial SARS-CoV-2 infection, including fatigue, muscle pain, breathlessness and headache, while 60% had severe Long-COVID symptoms associated with multi-organ impairment. In agreement, other studies reported persistence of symptoms in 32% of COVID-19 outpatients from Switzerland and 83% of COVID-19 inpatients from Italy. 44 Daugherty and colleagues’ retrospective analysis of 193,113 patients also identified more than 50 clinical sequelae, or types of Long-COVID, during the 4 months following the acute illness. Interestingly, 14% of patients aged ⩽ 65 years developed a new clinical condition requiring medical care, suggesting that although older people with pre-existing conditions and hospitalized due to COVID-19 were at higher risk, younger people without pre-existing conditions or not hospitalized for the infection also had an increased risk of Long-COVID. 45 Finally, although most evidence reports the development of chronic persistent symptoms mostly in adult individuals above 40 years of age, the Long-COVID phenomenon has surprisingly also been found in asymptomatic children. In this regard, Buonsenso et al. 46 reported that out of a total of 129 children aged ⩽ 18 years with a diagnosis of COVID-19, of whom 33 were asymptomatic and 96 symptomatic, 2.3% developed multi-system inflammatory syndrome in children (MIS-C) and 1.6% developed myocarditis, 35.7% had one or two Long-COVID symptoms, 22.5% had three or more symptoms and 41.8% recovered completely.
The pathophysiological mechanisms of Long-COVID are still largely unknown, and several hypotheses have been put forward in this field. Among these, there is complete agreement that SARS-CoV-2 infects the organism after exploiting its tropism in several tissues. This results in immune system dysfunction, increased inflammatory processes and impairment of several other systems, leading to Long-COVID.47,48 Particularly, Khazaal and colleagues suggested that the virus persistence in the body after the acute phase is the main responsible, as the virus would appear not to be eliminated in Long-COVID patients, but rather could reactivate with harmful effects that persist over time. 49 The multi-organ and multi-system impact of the virus is closely dependent on the ACE2 expression in many cell types, including neurons, astrocytes, oligodendrocytes and microglial cells. 50 Such viral neurotropism could contribute to the Long-COVID pathophysiology, potentially explaining olfactory disorders or manifestations related to autonomic nervous system dysfunction. 51 Second, Baig 52 proposed that oxidative stress and inflammation are responsible for a weak immunological response and incomplete virus eradication. This hypothesis, confirmed by the identification of viral particles in various organs after acute infection, could explain the onset of deleterious disorders, including autoimmune manifestations, activation of the coagulation and fibrosis pathways and metabolic disorders.53,54 Finally, deconditioning and psychological problems, such as post-traumatic stress disorder (PTSD), may also contribute to the Long-COVID development. 55
Long-term consequences of COVID-19: a shortcut to osteosarcopenia
Osteosarcopenia is a newly defined syndrome that describes the coexistence of sarcopenia and osteoporosis, two age-related musculoskeletal disorders increasingly recognized to be associated with significant morbidity, mortality and socioeconomic costs. 56 The pathogenesis of osteosarcopenia is multifactorial, with several mechanical, biochemical, genetic and lifestyle factors all contributing to the regulation of the muscle-bone unit. 57 The balance between these factors is crucial, and a disruption of it leads to innumerable bone and muscle risks and complications, causing deterioration of bone microarchitecture in association with the loss of muscle mass, strength and function.58–60 In this context, SARS-CoV-2 infection has been suggested to amplify the effects of aging on the musculoskeletal system by aggravating the osteosarcopenic state. This is true both for COVID-19 patients with a severe disease course, where prolonged intensive care unit induced loss of muscle mass and strength, weakness and persistent pain, and for all other patients, where reduced physical activity and walking due to restriction measures increased muscle atrophy and the risk of fragility fractures. 61
Particularly, the COVID-19 pandemic has been suggested to have caused an increase in risk factors for the incidence and progression of sarcopenia, especially in the elderly, such as decreased physical activity and unhealthy eating habits caused by social isolation.62–65 The inflammatory reaction caused by COVID-19 has also been correlated with metabolic stress and muscle catabolism.66,67 However, as suggested by Wang and colleagues, the interaction between COVID-19 and sarcopenia could be bidirectional and trigger a vicious circle. 68 In fact, the sarcopenic condition is known to be associated with chronic inflammation, malnutrition, metabolic and endocrine dysregulation, and various systemic dysfunctions, thus representing a key risk factor in the increased vulnerability of the elderly to COVID-19.69,70
The negative impact of sarcopenia on the COVID-19 treatment was partially supported by some indirect evidence showing that sarcopenic patients had impaired respiratory muscle strength and respiratory function, which was detrimental to the treatment of severe pneumonia and ARDS. 71 In addition, Okazaki and colleagues observed that sarcopenia is a risk factor for aspiration pneumonia in the elderly adult population due to dysfunction of the swallowing muscles, worsening the condition of bedridden patients with SARS-CoV-2 infection. 72 Therefore, it is to be expected that sarcopenic patients have higher infection and mortality rates in association with greater disease severity.
Myalgias and generalized weakness were found in a quarter to a half of symptomatic COVID-19 patients.73,74 Although a correlation between the onset of muscle pain and disease severity has not been found, computed tomography (CT) and X-ray imaging investigations have shown that muscle pain on admission is associated with abnormal images of the lung and predicts a poor prognosis, especially in the elderly, suggesting myalgia as an important predictor for disease severity. 75 Furthermore, several evidences suggested that myalgia, which occurs at the onset of SARS-CoV-2 infection, may not disappear in the acute phase of the disease, becoming a predominant feature of Long-COVID.76–80 In this regard, longitudinal studies conducted in Turkey, France and Italy showed that 6 months after discharge, approximately 60% of patients still suffered from at least one SARS-CoV-2-related symptom, of which the most common were fatigue, myalgia and joint pain with an average prevalence of 30%, 20% and 15%, respectively.81,82 Noteworthy, recent surveys have suggested that between 4.6% and 12.1% of patients still suffer from joint pain during the first year after infection, and that Long-COVID pain is associated with fatigue and is more prevalent in women.83,84
An emerging feature of COVID-19 is a clinically relevant osteo-metabolic phenotype characterized by acute diffuse hypocalcemia and chronic hypovitaminosis D, and a high prevalence of morphometric vertebral fractures. 85 This phenotype has been associated with a more severe form of the disease and higher mortality, confirming a role for SARS-CoV-2 also on bone tissue and mineral metabolism. 86 Specifically, hypocalcemia was found in 62.6–87.2% of COVID-19 patients, in association with increased markers of inflammation and thrombosis, a greater need for hospitalization and a higher mortality rate. 87 Low calcium levels have been mainly related to malnutrition and hypovitaminosis D, which in turn is known to be associated with a significantly higher mortality risk in COVID-19 patients due to its immunomodulating action on the immune system. 88 Therefore, immobilization due to hospitalization related to SARS-CoV-2 infection or isolation and quarantine, in combination with vitamin D deficiency and hypocalcemia, are all factors that could contribute to bone demineralization and the subsequent onset of osteoporosis.
Table 1 summarizes all the proposed mechanisms that correlate with worsening osteosarcopenic status in COVID-19 patients.
A summary table on possible mechanisms of osteosarcopenia in Long-COVID and possible prevention and treatment strategies.

Long-COVID-related osteosarcopenia: possible prevention and treatment strategies
The clinical management of patients with osteosarcopenia is already complex and neglected under normal circumstances, as there is still no specific diagnostic and therapeutic protocol for the condition due to its recent definition. However, a new potential predictive model has recently been identified on the basis of the correlation between the variables T-score and Handgrip test, suggesting it as a new parameter for a faster and more targeted diagnosis of osteosarcopenia. 89
Undoubtedly, prevention is fundamental and represents a goal that, if achieved, makes it possible not only to improve individuals’ quality of life but also to reduce the socioeconomic and health costs associated with such a widespread disease. 90 The osteosarcopenia development can be prevented by acting on modifiable risk factors. Indeed, a healthy lifestyle and regular exercise are currently the first-line choices for the prevention and treatment of osteosarcopenia (Table 1). 91 These recommendations are the same as those provided by the clinical guidelines for the management of COVID-19 or post-COVID syndrome patients. Indeed, since SARS-CoV-2 infection has been suggested to cause muscle wasting and bone demineralization, several evidences agree that interventions targeting musculoskeletal tissue could break the vicious circle and benefit the treatment of both COVID-19 disease and osteoporosis and sarcopenia (Figure 2). 68 In this regard, aerobic and resistance exercises are known to improve muscle protein synthesis by sensitizing muscles to insulin- or amino acid-mediated anabolic actions.92,93 Furthermore, adequate protein support together with physical activity successfully preserves skeletal muscle mass.94,95 Finally, adequate rehabilitation has been recommended to prevent the development of Long-COVID already in intensive care units, as soon as sedation and clinical stability permit. Indeed, pulmonary rehabilitation is known to improve patients’ breathing, exercise capacity, muscle strength, quality of life and functional outcome. In association, early mobilization could improve respiratory, functional and cognitive conditions in these patients, even reducing hospital stay.96,97 Similarly, adequate physical rehabilitation is recommended for non-hospitalized Long-COVID patients to improve their ability to engage in activities of daily living. 98
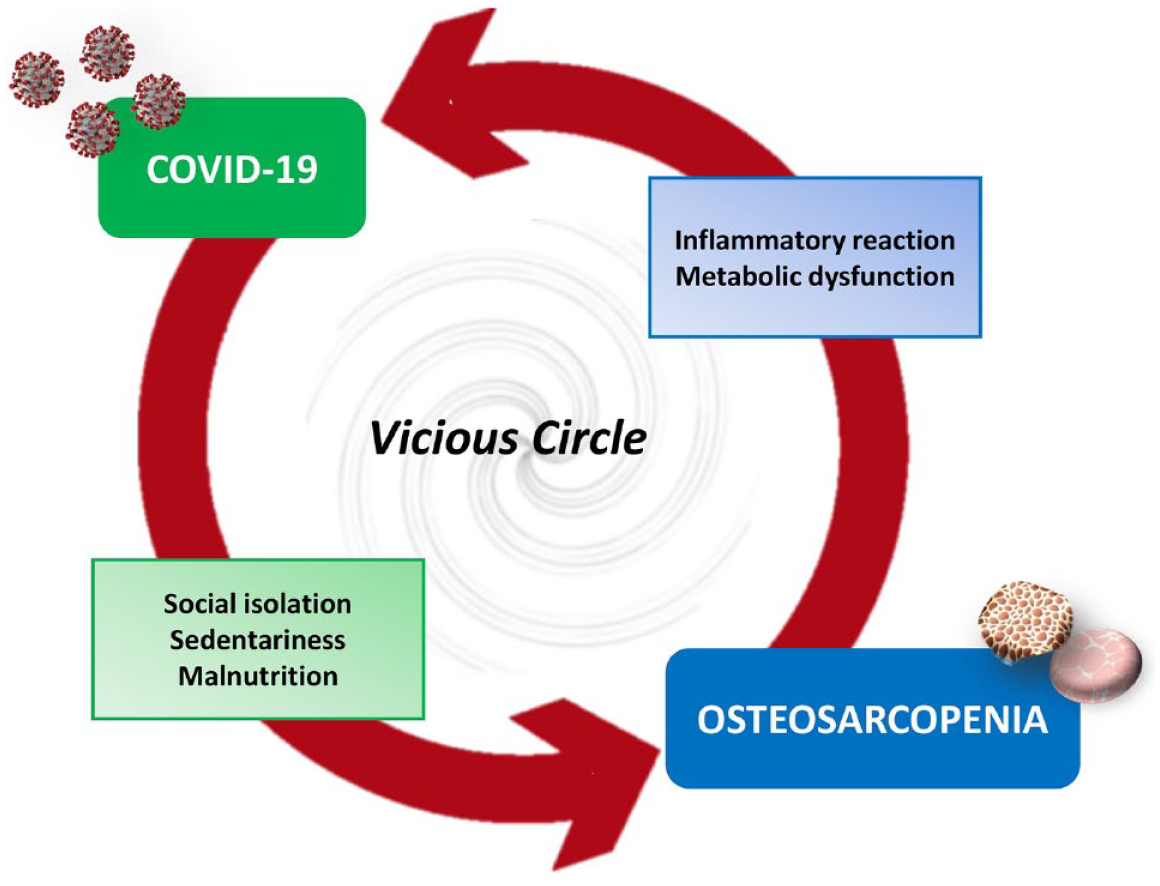
The interaction between COVID-19 and osteosarcopenia could be bidirectional and trigger a vicious circle. Indeed, factors associated with SARS-CoV-2 infection, such as social isolation, sedentariness and malnutrition, are known to promote osteosarcopenia. Conversely, events triggering osteosarcopenia, such as inflammatory reaction and metabolic dysfunction, are also factors associated with COVID-19 disease.
Conclusion
The continued growth of our experience with SARS-CoV-2 is closely associated with a better understanding of the short- and long-term complications caused by COVID-19 disease. Undoubtedly, the onset of musculoskeletal dysfunction as a sequelae of SARS-CoV-2 infection and the potential development of osteosarcopenia that follows may adversely affect the clinical course in older individuals, thus increasing the already high burden of the disease and underlining the need for appropriate patient assessment, including personalized rehabilitation and dietary approaches. Indeed, therapeutic options are currently limited, as the understanding of the biological mechanisms underlying Long-COVID is insufficient. However, most evidence agrees with the adoption of a multidisciplinary approach for long-term monitoring of ongoing symptoms, so that musculoskeletal targets can be predicted and potential complications can be identified. Further studies will be necessary for a deeper understanding of musculoskeletal changes and underlying determinants to alleviate clinical symptoms and improve quality of life in COVID-19 or Long-COVID patients.
