Abstract
Background:
Magnetic resonance imaging (MRI) is important in the management of axial spondyloarthritis (SpA). However, many MRI lesions are not exclusive to axial SpA. Further characterization of these lesions may lead to better clinical decisions.
Objective:
The objective of this study was to compare the frequency of individual spinal MRI lesions between axial SpA and noninflammatory back pain. The factors associated with individual lesions in participants with axial SpA were also determined.
Design:
This was a cross-sectional observational study.
Methods:
MRI lesions in 447 participants with axial SpA and 122 participants with noninflammatory back pain were compared using the propensity score adjustment method. Individual lesions included discovertebral lesions (DVL), Modic type 1 lesions, DVL without Modic type 1 lesions, facet joint lesions, costovertebral joint lesions, corner inflammatory lesions (CIL), and fatty corner lesions (FCL). The factors associated with the lesions were determined using regression analyses.
Results:
Among participants with axial SpA, 81.9% were HLA-B27-positive, 55.0% had radiographic axial SpA, and 60.5% had radiographic features of spinal damage (mSASSS >2). Almost half (48.6% in axial SpA versus 31.1% in noninflammatory back pain) had inflammatory lesions on spinal MRI. In propensity score matching with noninflammatory back pain, axial SpA had an increased occurrence of DVL without Modic type 1 lesion (OR = 3.43, p = 0.01), costovertebral lesion (OR = 11.89, p = 0.02), number of CIL (B = 1.19, p < 0.001), and number of FCL (B = 3.33, p < 0.001). Similar associations were found in the regression models in the radiographic axial SpA subgroup: DVL without Modic type 1 lesion (OR = 2.46, p = 0.001), costovertebral lesion (OR = 3.86, p < 0.001), number of CIL (B = 1.13, p < 0.001), and FCL (B = 2.29, p < 0.01).
Conclusion:
MRI lesions including DVL without Modic type 1, costovertebral joint lesions, CIL, and FCL were more specific in axial SpA.
Introduction
Magnetic resonance imaging (MRI) has become important in the management of axial spondyloarthritis (SpA) because of its unique role in the early diagnosis, classification, and monitoring of the disease.1,2 It is the only disease assessment tool that has been validated with histological inflammatory cellularity in tissue biopsies of the sacroiliac (SI) joint. 3 Many MRI lesions are, however, not exclusive to axial SpA and may occur in other conditions such as infection, degeneration, and malignancy. Further characterization of these lesions may guide the development of targeted therapies.
The Assessment in SpondyloArthritis international Society–Outcome Measures in Rheumatology (ASAS/OMERACT) MRI working group described several inflammatory and structural lesions on spinal MRI. 4 These include corner inflammatory lesions (CIL), discovertebral lesions (DVL), spondylitis, facet joint lesions, costovertebral lesions, and fatty corner lesions (FCL). 5 Among these, CIL, FCL, facet joint, and costovertebral lesions are specific for axial SpA.4,5 The DVL is associated with degeneration. 6 Further studies are needed to delineate the types of lesions specific to axial SpA.
This study aimed to compare the frequency of individual MRI spinal lesions in patients with axial SpA and noninflammatory back pain (controls). The factors associated with individual MRI lesions in participants with axial SpA were also determined.
Methods
This cross-sectional study used data from a multicenter MRI cohort that was initially designed to evaluate the usefulness of diffusion-weighted imaging (DWI) in axial SpA. 6 It has been registered in the clinical trial registry of the University of Hong Kong (HKUCTR-2087) with details of the design and data collection described in previous publications.6,7 In addition to clinical and radiographic data, MRI data included only short tau inversion recovery (STIR) sequences and T1-weighted images for analysis. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 8
Recruitment
In total, 447 participants with axial SpA and 122 participants with noninflammatory back pain (controls) were recruited from 11 hospitals (Queen Mary, Grantham, Tung Wah, Pamela Youde Nethersole Eastern, Caritas Medical Center, Tseung Kwan O, Kwong Wah, Prince of Wales, Prince Margaret, Hong Kong Eye, and the University of Hong Kong, Shenzhen) from April 2014 to November 2020. Inclusion criteria for participants with axial SpA were as follows: (1) axial SpA diagnosed by a specialist in rheumatology, (2) age >18 years, (3) current back pain, (4) biologics-naïve, (5) steroid therapy <10 mg prednisolone (or equivalent) per day, and (6) ability to provide written consent. The exclusion criteria were as follows: (1) inability to undergo MRI and (b) pregnancy. The inclusion criteria for participants with noninflammatory back pain (controls) were as follows: (1) noninflammatory back pain, (2) age >18 years, (3) current back pain, (4) steroid therapy <10 mg prednisolone (or equivalent) per day, and (5) ability to provide written consent. The exclusion criteria were as follows: (1) rheumatology assessment for suspected axial SpA, (2) inability to undergo MRI examination, and (3) pregnancy. All the participants provided written informed consent.
Clinical assessment
All recruited participants underwent clinical assessments, blood tests, radiographs, and MRI examinations on the same day. Clinical data included age, sex, ethnicity, smoking status, alcohol use, age at the onset of back pain, duration of back pain, family history of SpA, history of psoriasis (PsO), history of inflammatory bowel disease (IBD), history of dysentery or sexually transmitted disease preceding SpA, and history of uveitis. Physical examination was conducted to determine body weight, tender and swollen joint count, and spinal mobility for the Bath Ankylosing Spondylitis Metrology Index (BASMI). 9 The following self-assessment questionnaires were completed: the Bath Ankylosing Spondylitis Disease Activity Index (BASDAI), 10 Bath Ankylosing Spondylitis Functional Index (BASFI), 11 and Bath Ankylosing Spondylitis Patient Global score (BAS-G). 12 Blood tests for C-reactive protein (CRP) level, erythrocyte sedimentation rate (ESR), and HLA-B27 status were performed. The ankylosing spondylitis disease activity index (ASDAS) was calculated based on the CRP (ASDAS-CRP) and ESR (ASDAS-ESR). 13
Radiographic assessment and interpretation
Radiographs of the lumbosacral spine (anteroposterior and lateral views) were performed in all participants to distinguish between those with radiographic axial SpA. Cervical spine (lateral view) radiographs were obtained only in the participants with axial SpA. One investigator (H.H.L.T., a specialist in rheumatology with 7 years of experience in radiographic interpretation in axial SpA) blinded to the clinical data read and scored the radiographs of participants with SpA according to the modified Stoke Ankylosing Spondylitis Spinal Score (mSASSS) 14 for degree of ankylosis and the modified New York (mNY) criteria for ankylosing spondylitis (AS) for radiographic axial SpA. 15
MRI assessment and interpretation
Whole-spine (STIR and T1-weighted, sagittal view, C2 to S1) and bilateral SI joint (STIR only, oblique coronal view) MRI were performed using a 3.0-T imaging unit with inclusion of a torso coil. STIR and T1-weighted images were obtained consecutively with the participants in the supine position on the same MRI examination. Details of the MRI parameters have been reported previously.6,7
SI joint MRI was independently assessed by two investigators (S.C.W.C. and H.Y.C., with 5 and 9 years of experience in axial SpA, respectively), and positive sacroiliitis was defined according to the Assessment of SpondyloArthritis International Society (ASAS) handbook. 16 Spinal MRI images were also read by both investigators according to the Spondyloarthritis Research Consortium of Canada (SPARCC) scoring system, 17 with average SPARCC scores used for analyses.
All lesions on whole-spine MRI were inflammatory, except for FCL. The inflammatory lesions were hyperintense on STIR imaging. A DVL was defined in the Canada-Denmark scoring system 18 as hyperintense bone marrow contiguous with the vertebral endplate and intervertebral disk, with or without involvement of the vertebral corner in any central sagittal section. Modic type 1 lesions in the subchondral bone marrow of the vertebral endplate with disk degeneration are hyperintense on STIR, but hypointense on T1-weighted imaging. 19 CIL are hyperintense in the vertebral corners. Facet joint and costovertebral lesions are hyperintensities in the respective anatomical areas. An FCL is a structural lesion defined as a well-demarcated triangular lesion in the corner of any vertebral body in at least one sagittal slice that is hyperintense on T1-weighted images and hypointense on STIR. 20
The presence of individual MRI lesions was determined on the basis of the consensus of the principal investigator (H.Y.C.) and another investigator with variable expertise in MRI interpretation. S.C.W.C. and K.H.L. both had 5 years of experience, and R.S.W.Y. had 2 years of experience. CIL, facet joint, and costovertebral joint lesions were read by S.C.W.C.; DVL and Modic type 1 lesions by K.H.L.; and FCL by R.S.W.Y. All lesions were independently scored as either 0 (absent) or 1 (present). Except for the costovertebral joint lesions, all lesions were scored at the cervical, thoracic, and lumbar spinal levels. A lesion was considered a true positive if both investigators scored ‘1’. All readers were blinded to the clinical, blood, and radiographic data during the MRI interpretation.
Sample size calculation
The same size was calculated based on an estimation that spinal MRI lesions are found in 50% of patients with axial SpA based on our previous publication. 21 We assumed that 0.35% of the population (7.5 million) had axial SpA. 22 Using a confidence level of 95% and 5% margin of error, the calculated sample size was 379. The final recruited number of participants with axial SpA was 447. The frequency of spinal MRI lesions in noninflammatory back pain is not known. The recruited number of participants with noninflammatory back pain was 122, which was similar to our previous study. 21
Statistical analyses
Independent-samples t tests and chi-square tests were used to compare continuous and categorical variables, respectively. Inter-reader agreements of the MRI scores, number of CIL and FCL were all determined by the intraclass correlation coefficient, while that of MRI lesions by Cohen’s kappa test. The agreement was interpreted as slight (0.00–0.20), fair (0.21–0.40), moderate (0.41–0.60), substantial (0.61–0.80), and almost perfect (0.81–1.00).
The incidence of MRI lesions in axial SpA and noninflammatory back pain was compared using the propensity score adjustment method. Known or potential confounding factors for MRI lesions were included in the logistic regression analysis to generate a propensity score. Confounding factors included age, male sex, Chinese ethnicity, smoking status, regular alcohol use, age at onset of back pain, duration of back pain, and family history of SpA. Logistic and linear regressions stratified by propensity scores were used to determine the chance of occurrence of individual MRI lesions. Results were expressed as odds ratios (OR) or regression coefficients (B).
The factors associated with individual MRI lesions were identified in the axial SpA group. Univariate linear regression was tested using the number of CIL and FCL as dependent variables, and univariate logistic regression was tested using DVL without Modic type 1 lesion, and costovertebral joint lesions as dependent variables. Possible causative factors were selected as the independent variables. These included axial SpA, age, male sex, Chinese ethnicity, smoking, regular alcohol use, age at back pain onset, duration of back pain, family history of SpA, HLA-B27 positivity, and radiographic axial SpA. Independent variables with a p value less than 0.1 in univariate linear-logistic regression analyses were retested in multivariate regression models. Results were reported as OR in logistic regression models and as B and standard coefficients (β) in linear regression models. A 95% confidence interval (CI) was also reported. Unless specified otherwise, a p value of less than 0.05 was considered statistically significant. Missing values were handled using listwise deletion. All statistical analyses were performed using the Statistical Product and Service Solution package (SPSS) Version 27 (IBM Corp., Armonk, NY, USA).
Results
In total, 447 participants with axial SpA and 122 with noninflammatory back pain as controls were included in the analyses. All participants underwent MRI of the whole spine and SI joint.
The baseline characteristics of the patients are shown in Table 1. The patients in the axial SpA group were younger in age, predominantly male, had more smokers, had earlier age of onset and longer duration of back pain, and higher incidence of family history of SpA and B27 status compared with those in the noninflammatory back pain group. Participants with axial SpA were characterized by a long duration of back pain, predominantly Chinese, moderate-to-high disease activity, and moderate functional impairment. In the axial SpA group, 13.9% and 2.5% of the participants had concomitant PsO and IBD, respectively. Although mostly participants with axial SpA were HLA-B27 positive, only 55.0% had radiographic axial SpA and 60.5% had radiographic features of spinal damage (mSASSS >2). Almost half of the participants (48.7% in axial SpA versus 31.1% in noninflammatory back pain) were found to have spine inflammation on MRI.
Baseline characteristics of participants with axial SpA and noninflammatory back pain.
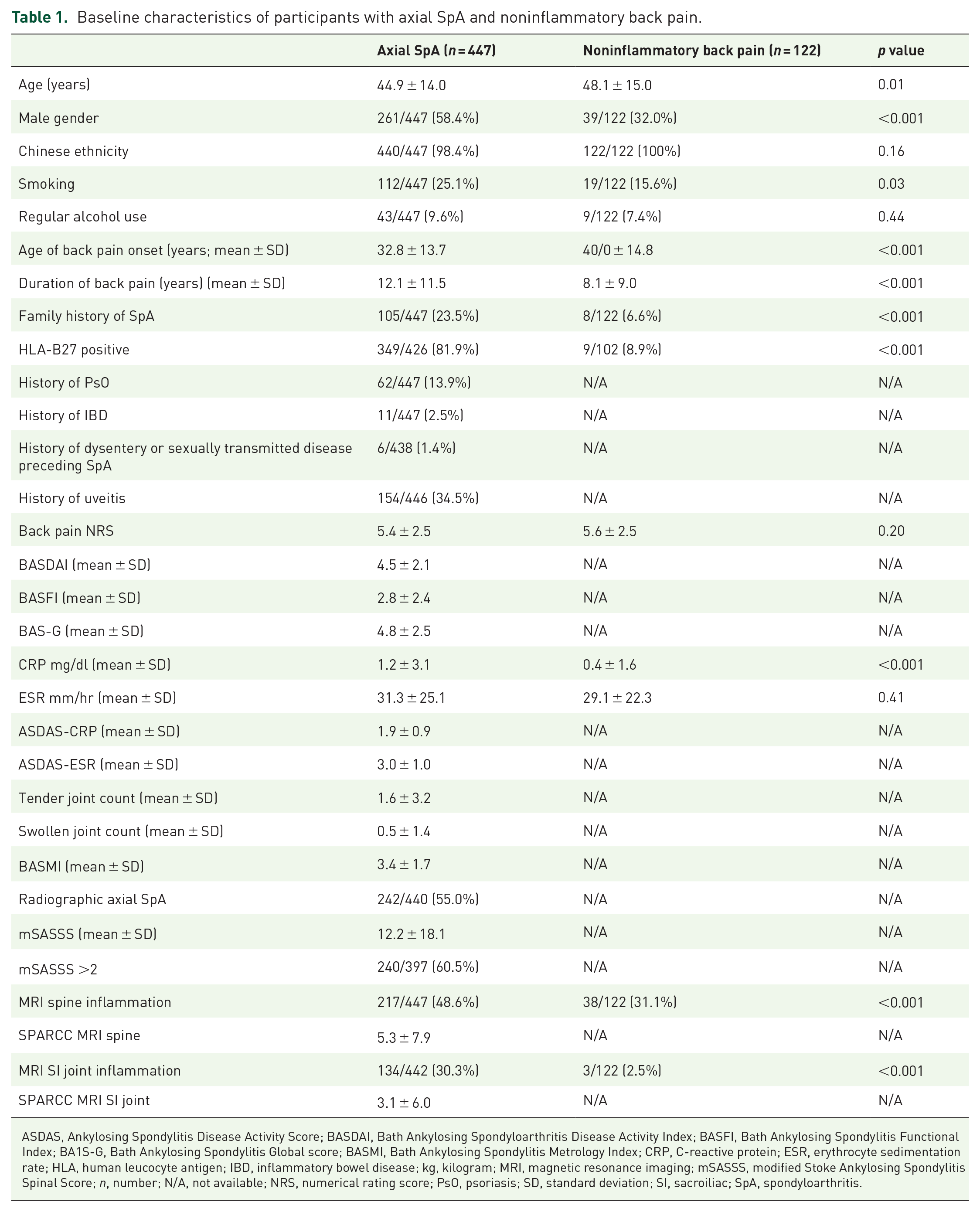
ASDAS, Ankylosing Spondylitis Disease Activity Score; BASDAI, Bath Ankylosing Spondyloarthritis Disease Activity Index; BASFI, Bath Ankylosing Spondylitis Functional Index; BA1S-G, Bath Ankylosing Spondylitis Global score; BASMI, Bath Ankylosing Spondylitis Metrology Index; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; HLA, human leucocyte antigen; IBD, inflammatory bowel disease; kg, kilogram; MRI, magnetic resonance imaging; mSASSS, modified Stoke Ankylosing Spondylitis Spinal Score; n, number; N/A, not available; NRS, numerical rating score; PsO, psoriasis; SD, standard deviation; SI, sacroiliac; SpA, spondyloarthritis.
Inter-reader reliability
The inter-reader reliability of the scores and MRI lesions were as follows: SPARCC spine score 0.85, STIR MRI sacroiliitis 0.78, DVL 0.72, cervical DVL 0.80, thoracic DVL 0.90, lumbar DVL 0.80, Modic type 1 lesion 0.68, cervical Modic type 1 lesion 0.65, thoracic Modic type 1 lesion 0.85, lumbar Modic type 1 lesion 0.61, facet joint lesion 0.64, costovertebral joint lesion 0.92, number of CIL 0.90, and number of FCL 0.95. Inter-reader reliability was substantial to almost perfect.
Description of MRI lesions
The prevalence of individual MRI lesions in both the groups is shown in Table 2. FCL and CIL were the two most prevalent lesions in axial SpA. Compared with those in the control group, participants with axial SpA had more DVL without Modic type 1 lesions, facet joint lesions, costovertebral lesions, or CIL. No significant differences were found in the DVL, Modic type 1 lesions, or FCL.
Prevalence of different MRI lesions.
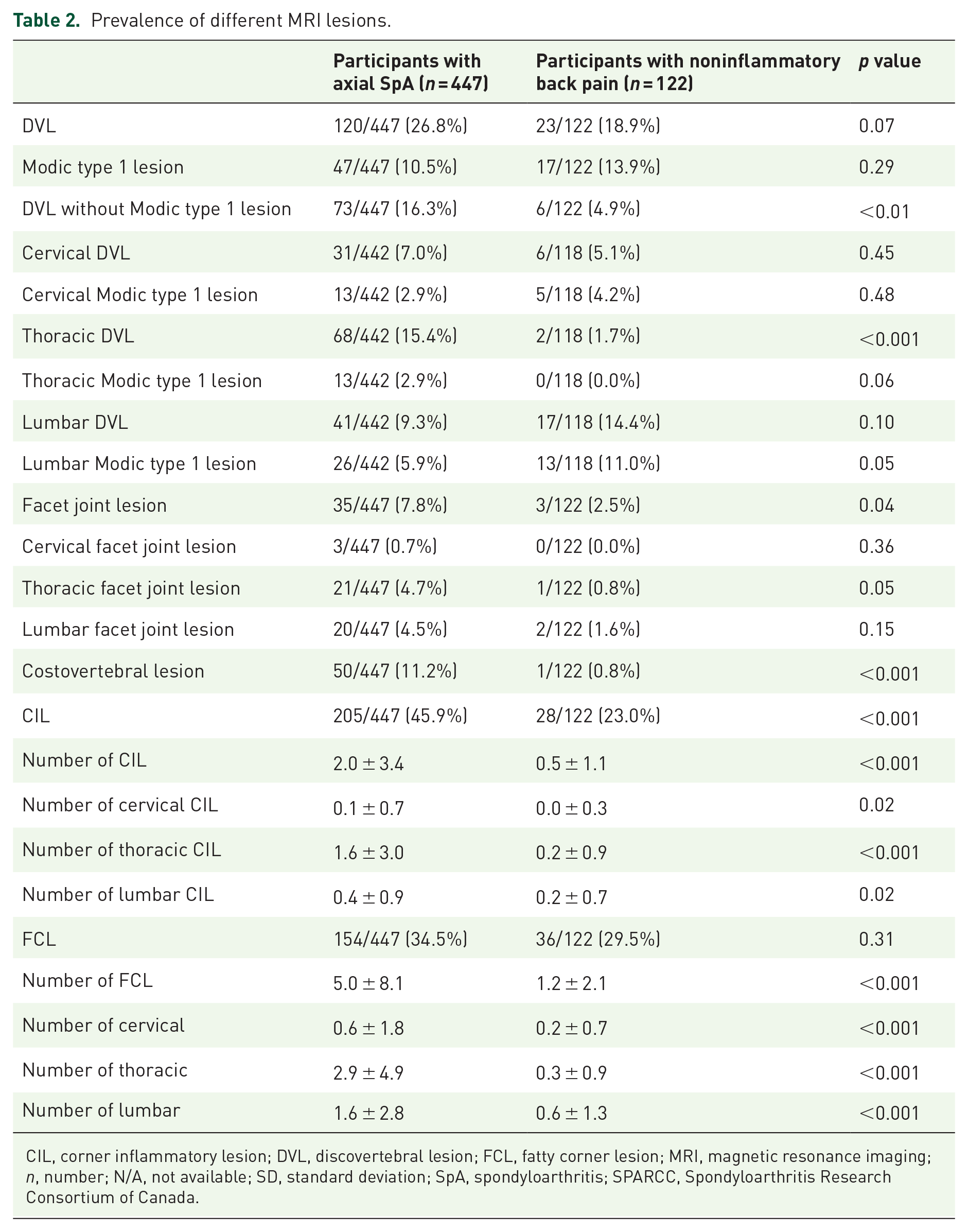
CIL, corner inflammatory lesion; DVL, discovertebral lesion; FCL, fatty corner lesion; MRI, magnetic resonance imaging; n, number; N/A, not available; SD, standard deviation; SpA, spondyloarthritis; SPARCC, Spondyloarthritis Research Consortium of Canada.
Chance of occurrence of individual MRI lesions in axial SpA
Participants with axial SpA showed an increased occurrence of DVL without Modic type 1 or costovertebral lesions (Table 3). Axial SpA also had greater numbers of CIL, thoracic CIL, FCL, and thoracic and lumbar FCL. No significant differences between the two groups were noted in DVL, Modic type 1 lesions, cervical and lumbar CIL, and cervical FCL.
Chances of occurrence of different MRI lesions in axial SpA when compared with noninflammatory back pain by propensity score adjustment.

Covariates included in the propensity score are as follows: age, male gender, Chinese ethnicity, smoker, regular alcohol use, age of onset of back pain, duration of back pain, and family history of SpA.
CI, confidence interval; CIL, corner inflammatory lesion; DVL, discovertebral lesion; FCL, fatty corner lesion; OR, odds ratio.
Regression analyses of MRI lesions in axial SpA
1. DVL without Modic type 1 lesion as dependent variable
Chinese ethnicity and radiographic axial SpA showed significant associations (p < 0.1) on univariate logistic regression analysis while multivariate logistic regression revealed that only radiographic axial SpA was independently associated with DVL without type 1 Modic lesion (Table 4).
2. Costovertebral joint lesion as dependent variable
On univariate logistic regression analyses, the male sex, smoking, and radiographic axial SpA were found to have significant associations (p < 0.1), while multivariate logistic regression showed that only radiographic axial SpA was independently associated with costovertebral joint lesion (Table 5).
3. Number of CIL and FCL as dependent variables
Using the number of CIL as the dependent variable, univariate linear regression analyses showed that male sex, smoking, age at back pain onset, HLA-B27 positivity, and radiographic axial SpA were significantly associated (p < 0.1). Multivariate linear regression analysis showed that the male sex and radiographic axial SpA were independently associated with the number of CIL (Table 6).
Logistic regression analyses of factors associated with DVL without Modic type 1 lesion.
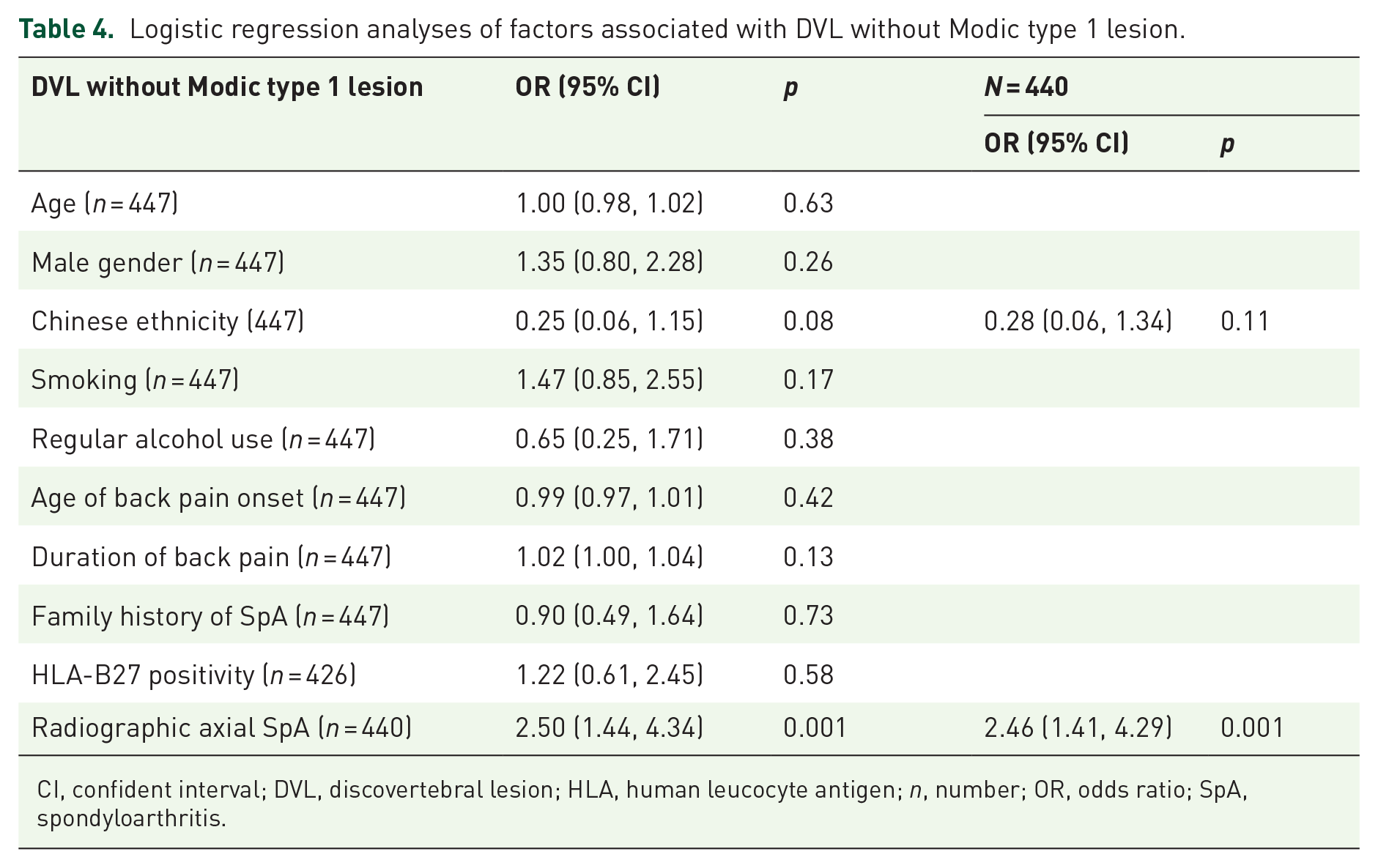
CI, confident interval; DVL, discovertebral lesion; HLA, human leucocyte antigen; n, number; OR, odds ratio; SpA, spondyloarthritis.
Logistic regression analyses of factors associated with costovertebral lesions.
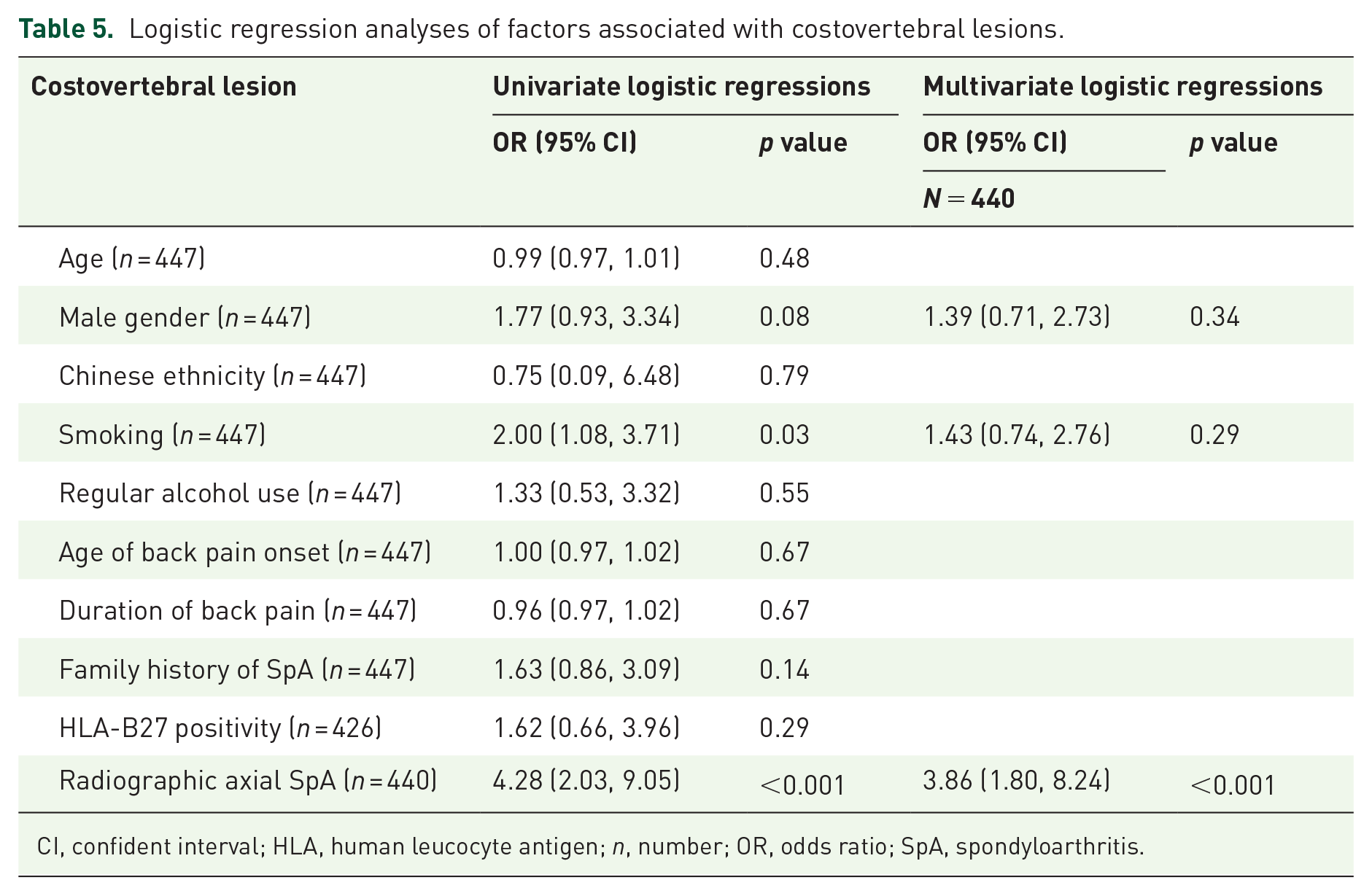
CI, confident interval; HLA, human leucocyte antigen; n, number; OR, odds ratio; SpA, spondyloarthritis.
Linear regression analyses of factors associated with CIL and FCL.

B, standard coefficient; CI, confidence interval; CIL, corner inflammatory lesion; FCL, fatty corner lesion; HLA, human leucocyte antigen; n, number; SpA, spondyloarthritis.
Using the number of FCL as the dependent variable, univariate linear regression analyses showed that age, male sex, smoking, regular alcohol use, age at back pain onset, duration of back pain, HLA-B27 positivity, and radiographic axial SpA were significantly associated (p < 0.1). Multivariate linear regression analysis revealed that male sex, HLA-B27 positivity, and radiographic axial SpA were independently associated with the number of FCL.
Missing data
HLA-B27 status was missing in 21 (4.7%) participants, and lumbosacral spinal radiographs were absent in 7 (1.6%) participants. Information regarding the level of DVLs was not recorded during image interpretation in nine participants (1.6%). All missing data were <5% and considered insignificant.
Discussion
Spinal MRI lesions were common and found in more than half of the participants with axial SpA. The three most prevalent lesions were CIL, FCL, and DVL. MRI lesions that occurred with increased incidence in axial SpA were DVL without Modic type 1 lesions, costovertebral lesions, CIL, and FCL. In contrast, the incidence of DVL, Modic type 1 lesions, and facet joint lesions were similar to those of noninflammatory back pain.
MRI is the most sensitive imaging modality for spinal inflammation in patients with SpA. With the rapid development of biological drugs and inadequately reliable serum markers, MRI has unique roles in the assessment of disease activity and monitoring the efficacy of biological disease-modifying anti-rheumatic drugs (b-DMARD). 23 This study showed that MRI lesions may be found in approximately 30% of patients with noninflammatory back pain. This ‘background noise’ is present in SI joint MRI of healthy individuals, 24 highlighting the presence of other contributing factors, such as minor trauma and mechanical stress. Our study showed that individual MRI lesions that are more specific to axial SpA might be more suggestive of disease-related changes.
Compared with those having noninflammatory back pain, participants with axial SpA were generally younger and predominantly male. Eschewing the conventional age- and sex-matched controls where much data may be lost, this study used the propensity score adjustment method to mitigate the effects of confounding factors including age and sex, among others, to give a more accurate estimate of the occurrence of lesions in axial SpA.
We observed that CIL and FCL had higher occurrences in axial SpA, which have been proposed as supplemental diagnostic aids for the ASAS classification criteria, albeit with limited added value.25–27 The pathophysiology of CIL remains unclear. The presence of FCL coupled with CIL is suggestive of resolution of inflammation.28,29 In this study, the increase in the number of CIL and FCL in the thoracic region was more likely due to disease-related primary inflammatory processes than degeneration. The thoracic spine is less affected by degeneration owing to its inherent non-weight-bearing biomechanics. 30
Persistent inflammation is associated with syndesmophyte formation; 31 however, new bone formation has been observed upon resolution of CIL. 32 Despite this apparent contradiction, prospective data have shown that both CIL and FCL predicted the development of new syndesmophytes. 33 Persistent inflammation may result in fatty changes and promotion of osteoclastogenesis via the receptor activator of nuclear factor-kappa B (RANK). 34 Both RANK and tumor necrosis factor-α (TNF-α) activate the Wnt signaling pathway, leading to new bone formation. In this study, both CIL and FCL were associated with male sex, and FCL was associated with HLA-B27 positivity. Male sex and HLA-B27 positivity are poor prognostic factors for spinal ankylosis. 35 Shared-associated factor(s) could further explain the increased risk of spinal ankylosis.
Costovertebral joint lesions were found to be specific to axial SpA. They are associated with restrictions in spinal mobility and functional impairment, 36 are not uncommon, 37 and are often overlooked. 28 In this study, costovertebral lesions were associated with radiographic axial SpA. Facet joint lesions, however, which are associated with decreased spinal mobility and increased functional impairment 36 and are present in spinal degeneration, 38 are nonspecific for SpA. Degeneration may result in varying degrees of secondary inflammation. Increased levels of inflammatory cytokines in facet joint tissue have been found in surgical cases of degenerative lumbar spinal disorders. 39 Another MRI study found that 75% of patients with Modic type 1 lesions, indicative of degeneration, also had facet joint inflammation. 40 The findings of our study are in line with those of previous international studies.
We found that the DVL without Modic type 1 lesions was the only subtype of DVL that showed a higher occurrence in axial SpA. This suggests that SpA is associated with inflammation and not with disk degeneration. The pathogenesis of DVLs may result from a combination of primary inflammation, minor trauma, and mechanical stress. 41 DVL without Modic type 1 lesions may be a result of primary inflammation irrespective of mechanical factors. 38 It has been argued that DVL with Modic type 1 lesions (disk degeneration) could be a feature of active disease; 42 however, data from the present study have shown otherwise.
Our study had a few limitations. First, the cross-sectional design allowed only the identification of associations, not the directionality of possible causation. Although propensity score adjustments were performed, other unrecognized confounding factors of the MRI lesions may have contributed to this bias. This statistical method is frequently used to compare two distinct populations to allow for the comprehensive matching of confounding factors.43,44 Missing data in less than 5% of the study population may be considered insignificant. Second, this study only investigated commonly recognized lesions. However, a sizable number of other MRI lesions (e.g. erosion, other types of Modic lesions, and enthesitis of spinal ligaments) with unknown significance in SpA are yet to be studied.
Conclusion
Spinal MRI lesions that were more specific to axial SpA included DVL without Modic type 1 lesions, costovertebral joint lesions, CIL, and FCL. In both axial SpA and noninflammatory back pain groups, however, no significant differences were noted in the occurrence of DVL, facet joint lesions, or Modic type 1 lesions. We believe that our data would help in the interpretation of individual MRI lesions in axial SpA.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X221119250 – Supplemental material for MRI lesions in SpA: a comparison with noninflammatory back pain using propensity score adjustment method
Supplemental material, sj-docx-1-tab-10.1177_1759720X221119250 for MRI lesions in SpA: a comparison with noninflammatory back pain using propensity score adjustment method by Ho Yin Chung, Jin Xian Huang, Kam Ho Lee, Helen Hoi Lun Tsang, Chak Sing Lau and Shirley Chiu Wai Chan in Therapeutic Advances in Musculoskeletal Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
