Abstract
Background:
Regenerative cell therapies, such as adipose-derived stromal vascular fraction (SVF), have been postulated as potential treatments for knee osteoarthritis (KOA).
Objectives:
To assess the efficacy and safety of SVF treatment against placebo and other standard therapies for treating KOA in adult patients.
Design:
A systematic review.
Data sources and methods:
We searched the following databases: MEDLINE via PubMed, Epistemonikos, PEDro, DynaMed, TripDatabase, Elsevier via Clinicalkey and Cochrane Controlled Trials Register. We included prospective interventional studies where treatment with SVF in adults with KOA was compared against placebo or other standard therapies, and results were objectively measured with at least one widely recognised osteoarthritis scale.
Results:
Among 266 studies published until May 2021, nine met our inclusion criteria. A total of 239 patients (274 knees) were included in our study. The follow-up ranged from 6 to 24 months. Six studies had a control group (only one being placebo). All studies showed that SVF improved pain and functionality measured, in most cases, with the visual analogue scale and the Western Ontario and McMaster Universities Osteoarthritis Index. In addition, five studies reported an improvement in anatomical structures, as detected in MR images. However, the number of cells contained in SVF varied substantially between different studies, which could induce a comparison bias.
Conclusion:
Although based on a small number of dissimilar studies, SVF was considered a safe treatment for KOA and could be promising in terms of pain, functionality and anatomical structure improvement. However, SVF products need to be standardised, the number of cells homogenised and the use of concomitant treatments reduced to establish proper comparisons.
Registration:
PROSPERO registration number: CRD42021284187.
Introduction
Osteoarthritis is a chronic degenerative joint condition characterised by the progressive destruction of the articular cartilage, leading to pain and functional loss. This disease is among the main causes of disability in adults, and the knee is the most frequent joint affected.1,2 Knee osteoarthritis (KOA) is estimated to affect 265 million people worldwide and its prevalence has risen to approximately 9% over 28 years (from 1990 to 2017).3–5 Besides, osteoarthritis prevalence is expected to continue increasing from 22% in 2003 to 25% by 2030 in the United States and from 26.6% in 2012 to 29.5% by 2032 in Sweden. 6
Several treatments have historically been explored for KOA, including pharmacological and surgical approaches, but most provide only symptomatic relief. Intra-articular treatments, such as corticoids, hyaluronic acid (HA) and platelet-rich plasma (PRP), are the most widely used drugs to treat KOA. Interestingly, recent studies have suggested that corticoids could be chondrotoxic, inducing early KOA. 7 On the other hand, HA and PRP are therapies whose effectiveness is still debated, and a unanimous consensus about their appropriate indication has not yet been reached. Hence, no widespread criteria for prescribing the abovementioned intra-articular drugs are available.8–10 At present, arthroplasty for severe cases is the only curative treatment for KOA. However, this surgery entails non-negligible complications, including infection, residual pain and stiffness. 11 Treatment with mesenchymal stem cells (MSCs), namely, multipotent stromal cells that can differentiate into osteoblast, adipocytes and chondrocytes, is among the new strategies to treat KOA. MSCs have high plasticity, self-renewal capabilities, and immune-suppressive and anti-inflammatory properties.12,13
Among the available sources of MSCs, two have received the greatest scientific attention: adipose-derived mesenchymal stem cells (ADSCs) and bone marrow mesenchymal stem cells. 14 Belonging to ADSCs, the stromal vascular fraction (SVF) isolated from adipose tissue is increasingly used to treat KOA. 15 SVF is a heterogeneous product that contains ADSCs, macrophages, blood cells, pericytes, fibroblasts, endothelial cells and their progenitors. This cellular heterogeneity entails a high therapeutic potential because of their complementary mechanism of action. Up to 60% of the cells within SVF are CD34+, a marker present on cells from the vascular microenvironment. Basic characterization of SVF has been provided by the International Society for Cellular Therapy, but the exact phenotype of ADSCs is still unknown, as biomarkers differ in vivo and in vitro. 16 SVF can be easily obtained in large amounts from autologous adipose tissue by liposuction and used without culture or differentiation. The interest in SVF stems from its extensively described immunomodulatory, anti-inflammatory, proangiogenic, antiapoptotic and antifibrotic properties.17,18 Some of these actions may be attributed to the presence of ADSC (ranging from 2% to 10%), while others are associated with the paracrine effect of cell phenotypes present in SVF. 16
Although several studies have reported the superiority of using SVF over other therapies for KOA, the evidence remains limited by the lack of randomised placebo-controlled clinical trials. 19 The most recently published systematic reviews and meta-analyses have pooled together, in the same analysis, different MSC therapy products, including SVF.20–22 The comparison of different types of cell therapies could be misleading and induce biased conclusions, as has already been warned by some authors. 23
Therefore, the aim of this systematic review was to assess the efficacy and safety of SVF against placebo and other standard therapies for treating KOA in adult patients.
Methods
Protocol and registration
We identified and examined the available literature but could not undertake a meta-analysis because of the heterogeneity of available data and study outcomes. We report our findings as suggested by the Preferred Reporting Items for Systematic Reviews and Meta-Analyses statement (PRISMA). The protocol for this systematic review was registered in the Prospective Register for Systematic Reviews (PROSPERO; registration number: CRD42021284187).
Population, intervention, comparison and outcomes
We created inclusion and exclusion criteria using the PICO (population, intervention, comparison and outcomes) model. To be eligible, a study had to meet the following PICO criteria:
P: Adults over 18 years old suffering from KOA.
I: Treatment with SVF.
C: Placebo or other therapies, including arthroscopic microfracture (AM), ADSCs, HA, or PRP.
O: Objective measure with at least one widely recognised osteoarthritis scale [e.g. WOMAC (Western Ontario and McMaster Universities Osteoarthritis Index), KOOS (Knee Injury and Osteoarthritis Outcome Score)] or a magnetic resonance imaging (MRI) scan and reported adverse effects.
Search strategy
In May 2021, we conducted a search in seven electronic databases: MEDLINE via PubMed, Epistemonikos, PEDro, DynaMed, TripDatabase, Elsevier via Clinicalkey and Cochrane Controlled Trials Register. In addition, we manually searched references from all available reviews15,20–29 on SVF to verify that none were missing from our initial search. The search included the terms ‘stromal vascular fraction’, ‘knee osteoarthritis’ or ‘osteoarthritis’, ‘cartilage’, ‘adipose-derived stem cells’ or ‘adipose-derived stromal cells’, and ‘mesenchymal stem cells’ or ‘mesenchymal stromal cells’ in the title and abstract of all trial registers and databases. Date restriction was applied to only include studies published within the last 10 years (when SVF treatments have increasingly been used). An example of a full-string search can be found in Appendix 1. We did not impose a language restriction in our search, but all articles meeting our inclusion criteria were written in English.
Study selection
We included only prospective interventional studies, either randomised or non-randomised, that met our PICO criteria. We only included published articles. We excluded articles not meeting our PICO criteria, narrative reviews, systematic reviews, meta-analysis and clinical trial registers. One reviewer (A.B.-P.) performed the eligibility assessment and two other reviewers (M.A. and E.P.-B.) revised it independently in an unblinded, standardised manner. Disagreements between reviewers were resolved by consensus. We used a designed electronic spreadsheet (Excel, Microsoft Corporation 2021) to enhance the consistency of data collected by the reviewers. First, we reviewed the titles to check for relevance and removed duplicates. Then, we screened abstracts to verify whether the article met our inclusion criteria. Finally, we retrieved and analysed full-text manuscripts.
Data extraction
Two independent reviewers (A.B.-P. and M.A.) collected the following characteristics from eligible studies: study registration, number of participants, study design, intervention, control, number of cells used, follow-up period, outcomes and adverse effects. Extracted data were summarised in an Excel spreadsheet and any disagreement between reviewers was discussed until reaching a consensus. A third reviewer (E.P.-B.) checked the extracted data to verify that the process was performed correctly. Unreported data were not considered, but reviewers asked for additional details from the authors of the article if some relevant data were missing.
Risk of bias assessment
Three independent reviewers (A.B.-P., M.A. and E.P.-B.) critically appraised each study to ensure relevance using Risk of Bias 2 (RoB2) for randomised studies and ROBINS-1 for non-randomised ones. For randomised studies, we assessed the following characteristics: randomisation process, deviations from intended interventions, missing outcome data, measurement of the outcome selection of the reported result and overall bias. For non-randomised studies, we considered the following aspects: bias due to confounding, bias in selection of participants into the study, bias in the classification of interventions, bias due to deviations from intended interventions, bias due to missing data, bias in measurement of outcomes and bias in the selection of the reported results. We classified the risk of bias as ‘low risk’, ‘some concerns’ or ‘high risk’. Any uncertainty was solved through discussion between the reviewers.
Data analysis
We performed a qualitative analysis based on extracted data by study type and population, and then by quality and specific results for each study. We also evaluated qualitatively the heterogeneity of the included studies. Furthermore, we listed the adverse effects reported to assess the safety of SVF.
Results
Study selection
The initial literature search resulted in 266 entries. Once duplicates were removed, 218 studies were screened by title and abstract. A total of 201 articles were excluded at this stage, mainly because they were not interventional trials, they studied different pathologies or joints from those of interest here, they were ongoing clinical trials or they were considered basic research articles (Figure 1). Eight of the 17 studies assessed in detail for eligibility were excluded, and the reasons can be found in Supplementary Table 1. A total of nine articles were finally analysed in this systematic review: two randomised clinical trials (RCT), two non-randomised clinical trials, three cohort studies, and two case series.

PRISMA flow diagram of article retrieval.
Characteristics of the included studies.
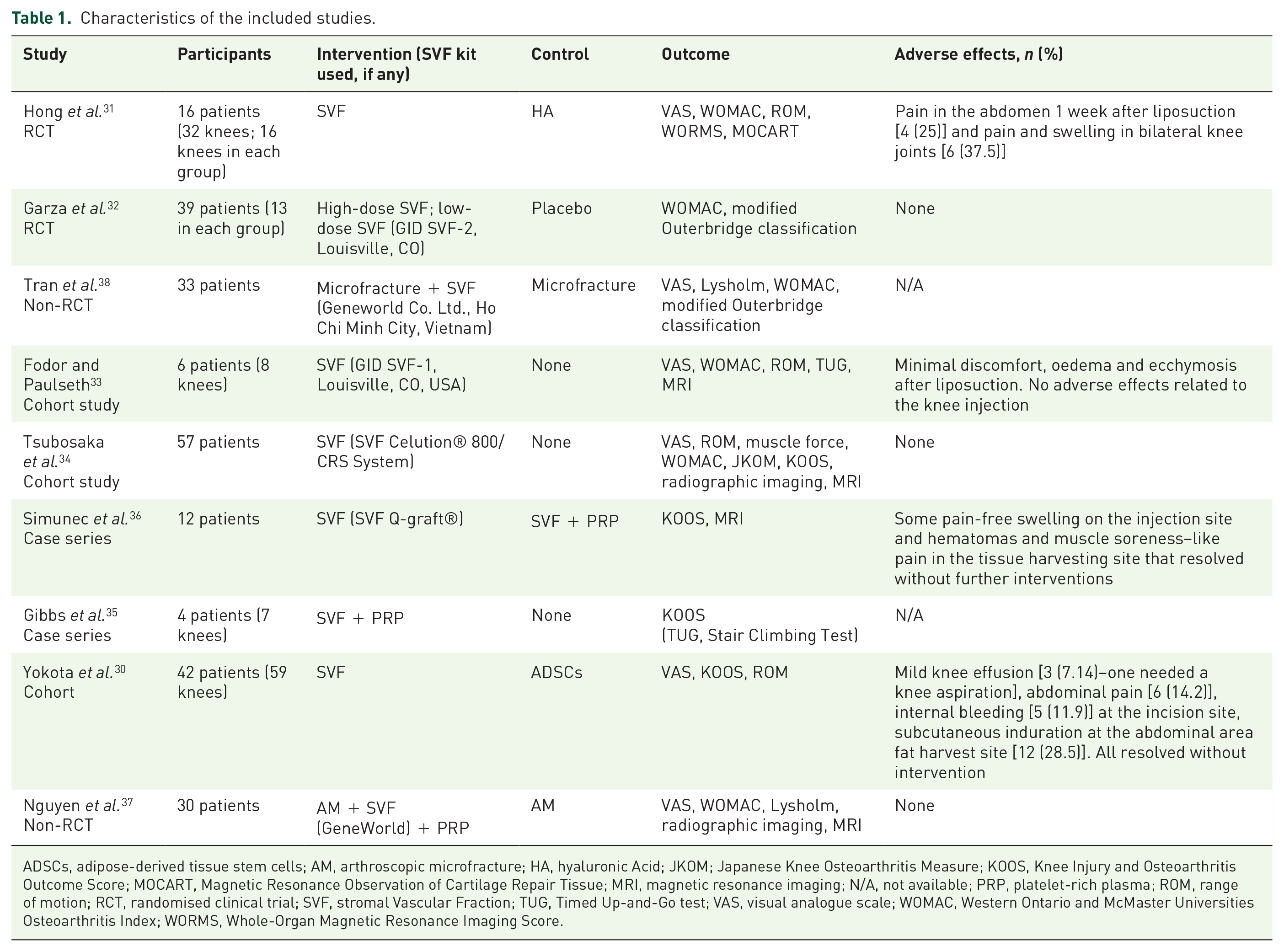
ADSCs, adipose-derived tissue stem cells; AM, arthroscopic microfracture; HA, hyaluronic Acid; JKOM; Japanese Knee Osteoarthritis Measure; KOOS, Knee Injury and Osteoarthritis Outcome Score; MOCART, Magnetic Resonance Observation of Cartilage Repair Tissue; MRI, magnetic resonance imaging; N/A, not available; PRP, platelet-rich plasma; ROM, range of motion; RCT, randomised clinical trial; SVF, stromal Vascular Fraction; TUG, Timed Up-and-Go test; VAS, visual analogue scale; WOMAC, Western Ontario and McMaster Universities Osteoarthritis Index; WORMS, Whole-Organ Magnetic Resonance Imaging Score.
Study characteristics
The characteristics of all included studies are summarised in Table 1. The duration of the follow-up ranged from 6 to 24 months (6 months in one study, 30 12 months in six studies,31–36 18 months in one study 37 and 24 months in one study). 38 The included studies involved a total of 239 participants and the equivalent of 274 knees. The main inclusion criteria were adults with unilateral or bilateral KOA, according to the Kellgren–Lawrence (KL) grading scale, and who had undergone an ineffective conservative treatment. Exclusion criteria included the following: secondary arthritis, medical conditions that precluded an anaesthetic procedure, psychiatric disorders, history of cancer, pregnancy, coagulopathy, signs of infection or syphilis- or HIV-positive serological results, knee joint surgery and intra-articular injection of any drug within the previous 3 months.
Intervention
All studies were performed in a single centre. The studies were performed in Vietnam,37,38 Japan,30,34 the United States,32,33 China, 31 Germany 36 and Australia. 35 In all studies, the intervention was a treatment with SVF. However, although the authors described the therapy as SVF, different methodological procedures and cell types were used in each study. Some of them used commercially manufactured kits,33,34,36,37 while others used a manual procedure31,32,35,38 to obtain the SVF. Three studies used concomitantly other therapies, such as AM37,38 or PRP. 36 All analysed studies employed autologous treatments.
Control
Only one study had a placebo control, 32 five studies used an active control30,31,36–38 and three were non-controlled.33–35 The active controls in the analysed studies included HA, 31 AM37,38 (both studies treated the intervention group with microfracture and SVF), PRP mixed with SVF 36 and intra-articular injection of ADSCs. 30
Outcome
In all studies, the primary outcomes were knee pain and functionality assessed with either WOMAC or KOOS scales. Radiographic imaging or MRI was also evaluated in most studies (seven out of nine).31–34,36–38
Adverse effects
Of the seven studies that assessed safety, three reported no adverse effects.32,34,37 After the liposuction, four studies described minimal discomfort, oedema and ecchymosis, which resolved without intervention, in a few cases.30,31,33,36 Two studies reported knee pain and swelling.30,31 All were described as minor adverse effects.
Results for included studies
The majority of the studies recorded two types of outcomes: clinical (concerning pain and/or functionality) and radiological outcomes. The most widely employed clinical scoring systems were the visual analogue scale (VAS)30,31,33,34,37,38 and the WOMAC.31–34,37,38 Four studies used the KOOS scale.30,34–36 Range of motion assessment,31,34 Lysholm score,37,38 Timed Up-and-Go test, 35 Stair Climbing Test 35 and the Japanese Knee Osteoarthritis Measure 30 were secondary clinical outcomes recorded in six articles. Functional outcomes improved in the intervention group in all studies.
Seven studies assessed radiological outcomes using MR images.31–34,36–38 However, only four studies used standardised radiological scoring systems. The Whole-Organ MRI Score and the Magnetic Resonance Observation of Cartilage Repair Tissue were used in one study 31 and the Outerbridge Classification System in three other studies.32,37,38 Among these seven studies, two did not observe significant differences in MR images. One performed the control MRI 3 months after intervention 33 and the other 6 and 12 months after treatment in a total of 26 patients. 32 The five remaining studies found significant improvement in MR images between baseline and a 12- or 24-month control.31,34,36–38
A pooled analysis of mean VAS and WOMAC scores 12 months after treatment, for studies with available data, can be found in Figure 2. Notably, two of these studies (Nguyen et al. 37 and Tran et al. 38 ) used AM as a coadjutant treatment in the intervention group, which may devalue the analysis. Figure 3 describes the pooled analysis of studies exclusively using SVF in the intervention group.

Forest plots showing changes in main outcomes 12 months after treatment: (a) mean VAS (Visual Analogue Scale) and (b) mean WOMAC (Western Ontario and McMaster Universities Osteoarthritis Index.

Forest plots showing changes in main outcomes 12 months after treatment in studies using exclusively SVF in the intervention group: (a) mean VAS (Visual Analogue Scale) and (b) mean WOMAC (Western Ontario and McMaster Universities Osteoarthritis Index).
Results for individual studies
The main results of included studies are summarised in Table 2. Hong et al., 31 in a RCT, showed a significant improvement in the SVF group for all clinical scores including VAS [mean: 3.19; standard deviation (SD): 0.98; p < 0.001], WOMAC pain (mean: 8; SD: 4.77; p < 0.001), WOMAC stiffness (mean: 2.25; SD: 2.11; p < 0.001) and knee range of motion (mean: 19.06; SD: 7.76; p < 0.001) 12 months after intervention. In addition, the study showed positive radiologic results: a decrease in the Whole-Organ MRI Score (mean: 15.44; SD: 21.95; p < 0.05) and a significant Magnetic Resonance Observation of Cartilage Repair Tissue score improvement (mean: 62.81; SD: 8.16; p < 0.001) compared with the control group (HA). Similarly, Tran et al. 38 found that SVF treatment improved pain and functionality since a reduction in the mean VAS (from 5.1; SD: 1.2 at 12 months to 3.4; SD: 1.8 at 24 months) and mean WOMAC (from 16.4; SD: 12.1 at 12 months to 11.1; SD: 11.9 at 24 months; p < 0.05) scores was observed. Besides, radiological results also improved 24 months after treatment as assessed by the Outerbridge score (mean: 3.0; SD: 0.8 versus mean: 2.0; SD: 0.7; p < 0.05). Characteristically, these authors observed that KL3-grade KOA patients showed a higher decrease in pain and improvement in functionality than those with a KL2 grade. On the contrary, Simunec et al. 36 suggested that, although both KL3 and KL4 grade showed an improvement in KOOS scores 12 months after treatment, the effect was milder in KL4-grade patients (7.7%) than in those with a KL3 grade (34.5%).
Main results of the included studies.
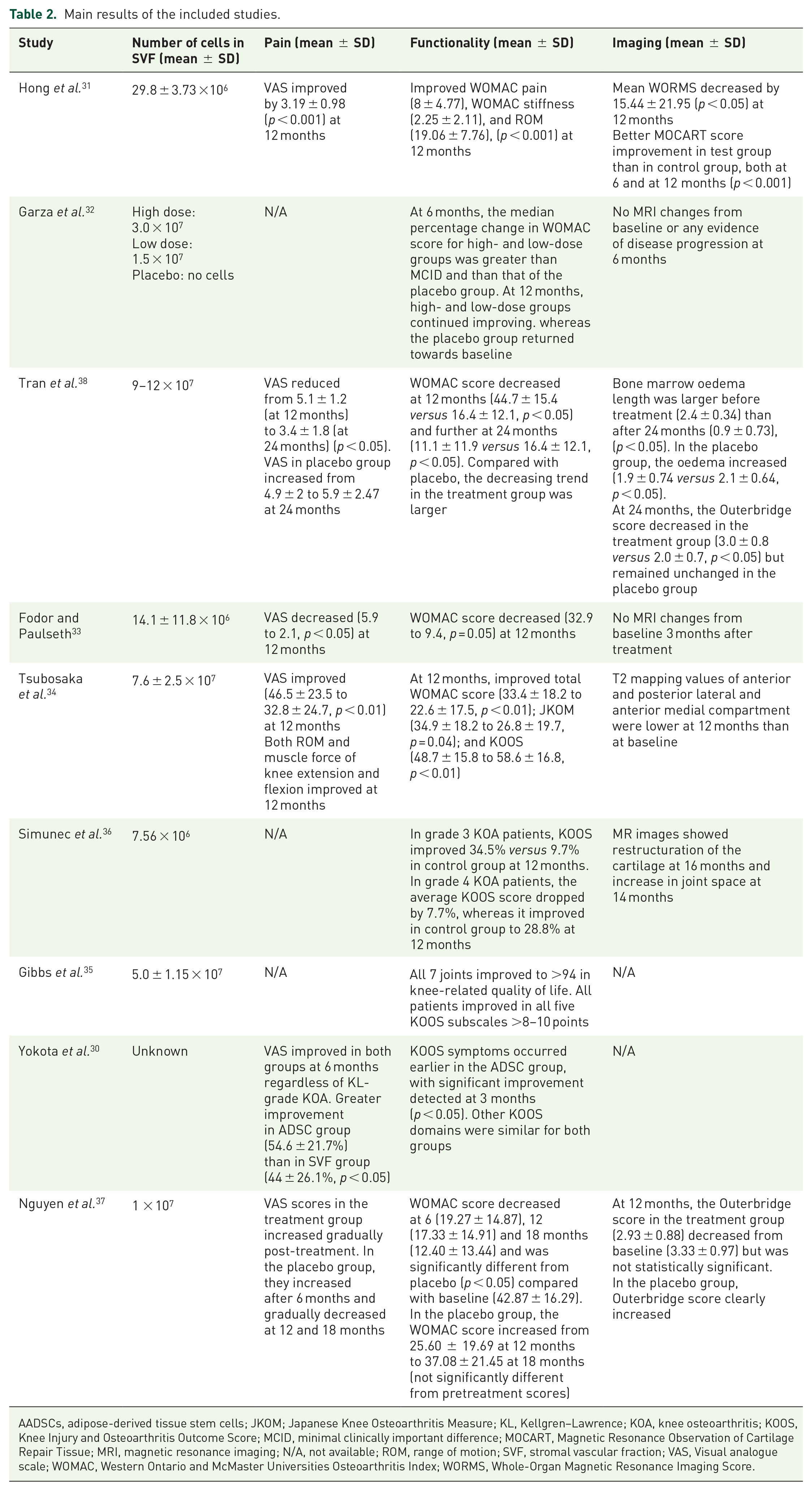
AADSCs, adipose-derived tissue stem cells; JKOM; Japanese Knee Osteoarthritis Measure; KL, Kellgren–Lawrence; KOA, knee osteoarthritis; KOOS, Knee Injury and Osteoarthritis Outcome Score; MCID, minimal clinically important difference; MOCART, Magnetic Resonance Observation of Cartilage Repair Tissue; MRI, magnetic resonance imaging; N/A, not available; ROM, range of motion; SVF, stromal vascular fraction; VAS, Visual analogue scale; WOMAC, Western Ontario and McMaster Universities Osteoarthritis Index; WORMS, Whole-Organ Magnetic Resonance Imaging Score.
On the other hand, Tsubosaka et al. 34 demonstrated a statistically significant clinical improvement with several functional scores (WOMAC, KOOS and Japanese Knee Osteoarthritis Measure) 12 months after SVF treatment. They also observed recovery in MR images 12 months after treatment, albeit no validated scoring system was used. Similarly, Nguyen et al. 37 found better Outerbridge scores in the MR images of the treatment group than in those of the control group (AM) 12 months after the intervention. In the study of Gibbs et al., 35 improvement was only reported in knee-related quality-of-life KOOS scores. In addition, both Fodor and Paulseth’s 33 and Garza et al.’s 32 studies described better WOMAC results in the treatment group. However, Garza et al. 32 could not find differences between the MR images of the SVF and the placebo group assessed at the 3-, 6-, and 12-month visits, and neither did Fodor and Paulseth 33 between MR images of their cohort taken at baseline and 3 months after treatment.
The number of cells contained in SVF varied substantially between the different studies and within participants of each study, which could induce a comparison bias. For example, Garza et al., 32 in an RCT, suggested that a high dose of SVF (3.0 × 107 SVF cells) provided an additional therapeutic relief of KOA symptoms than a low dose (1.5 × 107 SVF cells). Surprisingly, in the study of Yokota et al., 30 the authors stated that the number of cells injected was unknown.
Risk of bias within studies
The overall risk of bias in all included studies was considered high, except for the two RCTs (Figure 4). The allocation sequence was randomised and concealed until the enrolment and intervention assignment in two studies.31,32 One study was double-blinded, 31 whereas in others, the blinding process was unknown or non-existent. Data for main outcomes (pain and functionality) were available for nearly all included participants in all studies. In most studies, the measuring of the outcomes was appropriate, except for the prematurity on the MRI analysis (less than 12 months after the intervention) in two studies32,33 or the assessment of a unique outcome in another. 35 However, in several studies, outcome judgement assessors might have been influenced by the awareness of the intervention received. Finally, in most studies, results had been assessed based on multiple eligible outcome measurements within the outcome domain, but few analyses had been performed (Table 3).

Overall risk of bias for all included studies in an intention-to-treat analysis. Visualised with the ROBVIS tool.
Risk of bias for each included study.

 Low risk;
Low risk;  Some concerns;
Some concerns;  High risk.
High risk.
Discussion
To the best of our knowledge, this is the first report to review the efficacy and safety of SVF to treat KOA in adult patients against placebo and other therapies. According to the analysed studies, SVF is safe to treat KOA, entailing only a few minor adverse effects. Although low-quality studies have also been included here, all studies concluded that SVF treatment improves the symptoms and functionality of KOA patients. Inconsistencies in its effect on anatomical structures have been found, maybe due to different follow-up periods, but the majority of studies showed an improvement in MRI scans 12 months after treatment.
We have reviewed the best available evidence on SVF treatment: systematic reviews and meta-analysis. Although conclusions favour the intervention arm, a thorough analysis of the composition of cell therapies used in the studies included in these reviews unveiled very different methodologies. Like comparing apples and oranges, recent systematic reviews and meta-analyses have pooled data from studies of MSC treatments performed with different types and concentrations of cell therapy products, which could be deemed a methodological limitation of the analysis.20–23,25,28,39,40
In addition, we have detected a lack of well-categorised MSC products. Albeit MSCs are the focus of current research in multiple medical areas, many of their subtypes’ properties and therapeutic potential are still unknown. 41 Although the efficacy of these cells to modulate inflammation has been shown in different animal models, the results obtained in human clinical trials have been more modest. 42 Diverse controversial issues on their biology (including their specific phenotype, the requirement of an inflammatory environment to induce immunosuppression or the cell delivery route, among others) persist. 13 Therefore, a consensus on the definition, composition, action mechanism and production process of MSC is still missing. 43 Besides, the minimum effective dose needed for cell therapies is also unknown. Although Muthu et al. 19 already stated superior functional outcomes with non-cultured products of MSC to treat KOA over cultured ones, researchers are still considering different action mechanism hypotheses. The debate is still open on this matter with solid arguments for cultured and non-cultured products.13,44–46
Herein, to tackle the absence of a review focused solely on the efficacy of SVF, only interventional studies testing SVF treatment have been included, but a paucity of high-level evidence studies (e.g. RCTs evaluating clinical and radiological effects) has been observed. This led us to include low-level evidence studies as long as the SVF treatment was well-described and categorised. Although our inclusion criteria have been well-established, only a few articles could finally be included in our analysis. This explains why studies using cultured ADSCs,47–49 processed adipose tissue containing ADSCs,4,50 a mixture of extracellular matrix and ADSCs 51 or microfragmented lipoaspirate 52 have been withdrawn in our screening. Of the included studies, only two were RCTs31,32 and just one compared SVF treatment with placebo. 32 Despite the potential limitation of pooling and comparing such data – considered with a high risk of bias by the RoB2 and ROBINS-1 tools – and the few articles included, the trials analysed here pointed towards the superiority of SVF treatment over other therapies for KOA on all outcomes considered.
On the other hand, previous clinical trials have indicated the ability of intra-articular ADSC therapy to modify the progression of osteoarthritis.53,54 Progression rates previously reported have been variable,55,56 likely related to differences in stage of disease, definition of progression and studied population. 57 As already suggested by Iolascon et al., 58 detecting early-stage KOA (defined as early osteoarthritis) could help to better prescribe these innovative therapeutic approaches. Furthermore, the recommended follow-up period to assess cartilage changes in MRI is at least 12 months. 59 Among the studies included in our review that assessed MRI changes, Fodor and Paulseth 33 and Garza et al. 32 could not find changes in MRI scans, but their follow-up was shorter than 12 months (3 and 6 months, respectively), which could explain these observations.
Clinical and functional outcomes must also be evaluated over time. Except for the study of Yokota et al., 30 with a follow-up period of just 6 months, the follow-up of all other studies has been over 12 months, which provides some consistency to our analysis of their results. Nguyen et al. 37 presented the only negative pain result, reporting a gradually increasing mean VAS score in the treatment group. Notably, these authors have compared two groups who previously underwent an AM procedure, which can mask results due to persistent post-operative pain or worsening of the knee cartilage surface. On the contrary, the other studies using coadjutant treatments to SVF (Tran et al. 38 and Simunec et al. 36 ) have shown positive results in the intervention group.
As previously mentioned, there is yet no proper definition and reproducible manufacturing procedure for SVF treatments. Many different commercially available manufactured kits exist, but although they may look similar, they are not. Kits differ in the time allowed for the collagenase to digest, the neutralisation process, the time and intensity of centrifugation, the SVF dose cell yield, their viability and composition, cost and total processing time. 60 Given this heterogeneity, although some authors defend the use of kits instead of the reference method (manually manufactured in a laboratory by a technician), 61 the best method to obtain SVF is still debated. In our review, just three studies have used the reference manual method,30,31,35 and one of them 35 added ultrasonic cavitation to the collagenase step.
Finally, in agreement with previous systematic reviews that included SVF among analysed MSC,20–23 only minor adverse effects in the treatment group have been found, most of them caused by the liposuction procedure. They all resolved spontaneously or with mild treatment and in a short time, supporting our conclusion that SVF is a safe treatment for KOA in adult patients.
Limitations
The results of the current study should be interpreted in the context of its limitations. First, only a small number of high-quality studies could be found. Because of the lack of placebo-controlled RCTs, low-quality studies (e.g. case series) were included and a meta-analysis could not be performed. There was also significant variability in the preparation and concentration of SVF products, and, in one study, the number of injected cells was unknown. 30 Although the minimum SVF effective dose is yet to be established, these inconsistencies could have limited our analysis. In addition, although pain and functionality were assessed as the main outcomes in most studies, different validated scores were used and some data were missing. Therefore, our forest plot analysis was incomplete, missing data from some studies. Although Satué et al. 62 have described the immunomodulatory and cartilage regeneration effects of SVF in vitro, the time that SVF cells maintain their properties intra-articularly is still unknown. Consequently, a follow-up period shorter than 1 year might be insufficient to completely evaluate the effect of SVF treatment. Furthermore, not all studies have assessed anatomical changes. Albeit histological samples (which would better describe the anatomical changes) could not be analysed for obvious ethical concerns, MRI evaluation with validated scores has been rarely undertaken. All these methodological pitfalls between studies could confound our comparisons and limit our conclusions.
Performing a clinical trial following the International Standards of Good Clinical Practice 63 is a great challenge in cell therapies. Limited access to cell therapy biologists and specialised laboratories by clinicians, strict legal and governmental policies imposed by each country’s concerned authorities and the high cost of cell therapy procedures prevent the development of clinical trial protocols and their use in the clinic. From our point of view, placebo-controlled RCTs testing exclusively well-categorised SVF treatment would be mandatory to unequivocally confirm that SVF is an effective treatment for KOA. However, since the technique requires a previous liposuction procedure, a placebo-controlled trial with sham liposuction would be difficult to perform. However, comparing SVF to other approved treatments could also be a breakthrough.
Conclusion
With a low level of evidence, our systematic review suggests that SVF treatment could be a promising therapy for KOA in terms of pain, functionality and anatomical structure improvement. In addition, SVF has proved to be a safe treatment for KOA. However, SVF products need to be standardised to be compared and concomitant treatments should be avoided, as they can mask SVF’s therapeutic effect. In addition, the number of cells in the SVF should be similar to allow the comparison of different treatments. Our results have highlighted the lack of prospective RCTs exclusively comparing a well-categorised SVF treatment with placebo or standard approved treatments to better understand the effect on KOA. Therefore, we consider that the most appropriate study to evaluate SVF for KOA would be a controlled RCT testing a well-defined, standardised and categorised SVF product in a large number of patients.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X221117879 – Supplemental material for Stromal vascular fraction therapy for knee osteoarthritis: a systematic review
Supplemental material, sj-docx-1-tab-10.1177_1759720X221117879 for Stromal vascular fraction therapy for knee osteoarthritis: a systematic review by Anna Boada-Pladellorens, Mercè Avellanet, Esther Pages-Bolibar and Anna Veiga in Therapeutic Advances in Musculoskeletal Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
