Abstract
Background:
In previous studies, cardiovascular (CV) risk was increased in patients with gout. The effects of uric acid–lowering therapy on CV risk in gout patients have been investigated in numerous studies; however, allopurinol and benzbromarone have rarely been compared.
Objectives:
To compare CV risk based on allopurinol and benzbromarone treatment in Korean gout patients.
Design:
A nationwide population-based retrospective cohort study.
Methods:
We used South Korea database of the Health Insurance Review and Assessment (HIRA) service to identify gout patients ⩾18 years of age who newly started allopurinol or benzbromarone between 2009 and 2015. The primary outcome of the study was the occurrence of a composite CV endpoint, which included coronary revascularization, hospitalization due to myocardial infarction, ischemic stroke, and transient ischemic attack. Cox proportional hazard regression analysis and Kaplan–Meier curves were used for analysis.
Results:
The study included 257,097 allopurinol initiators and 7868 benzbromarone initiators. Compared with allopurinol initiators, the adjusted hazard ratio (aHR) of the composite CV endpoint of benzbromarone initiators was 1.01 [95% confidence interval (CI): 0.83−1.21], which was not significantly different. The results did not change even when 1:3 propensity score matching was performed for baseline characteristics. In subgroup analysis of high-risk patients with CV disease, significant difference was not observed between allopurinol and benzbromarone initiators.
Conclusion:
In this study, significant difference was not found in CV risk between allopurinol and benzbromarone initiators. In the high-CV-risk group, the incidence of CV events did not differ between allopurinol and benzbromarone initiators.
Introduction
Gout is characterized by the deposition of monosodium urate crystals in joints and soft tissues and is the most common inflammatory arthritis. Hyperuricemia plays a central role in the pathogenesis of gout, resulting from increased production or decreased secretion of uric acid. The prevalence of gout has been continuously increasing over the past few decades,1,2 as well as the burden of gout worldwide. 3 Gout is associated with comorbidities such as metabolic syndrome, chronic renal disease, cardiovascular (CV) disease, and an increase in all-cause mortality.4,5
In many studies, an association has been shown between gout and CV disease. In previous large-scale studies and recent meta-analysis, the risk of CV disease, including myocardial infarction (MI), heart failure (HF), and cerebrovascular accident was shown to be increased in gout patients compared with non-gout controls.6,7 Uric acid–lowering agents used in the treatment of gout have also been associated with CV risk. In a recent meta-analysis, the use of xanthine oxidase inhibitors (XOIs) was associated with a reduced risk of CV events compared with placebo. 8 In a CARES trial in which the CV outcome of febuxostat and allopurinol in gout patients was compared, febuxostat was not inferior to allopurinol in adverse CV events but had higher all-cause and CV mortality. 9 However, in the subsequent FAST trial, difference in all-cause mortality was not reported between the febuxostat and allopurinol groups. 10 Fewer data exist on the association between the use of uricosuric agents and CV outcome compared with XOIs. In a study comparing the CV risk of probenecid and allopurinol in the Medicare population, probenecid treatment showed a modestly decreased risk of CV events compared with allopurinol. 11 In a cohort study using a Taiwan claim database, the risk of coronary artery disease and stroke was reduced in patients with gout treated with uricosuric agents, such as benzbromarone, probenecid, and sulfinpyrazone. However, in that study, comparisons between uricosuric agents and XOIs or between different uricosuric agents were not performed. 12 In another cohort study using the Taiwan claim database to investigate the incidence of coronary artery disease in gout patients treated with allopurinol or benzbromarone, comparison between the two drugs was not performed. 13 To date, comparison of benzbromarone and XOIs has been conducted in only a few studies.
The uric acid–lowering agents approved and mainly used for the treatment of gout in Korea include allopurinol and febuxostat, which are XOIs, and benzbromarone, a uricosuric agent. In this study, the incidence of CV events was compared in patients with gout who were newly started on allopurinol or benzbromarone in a nationwide population-based cohort.
Methods
We conducted a retrospective cohort study using health administrative data of South Korea. This study reported according to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines for reporting observational studies. 14
Data resources
The Korean National Health Insurance Service (NHIS) is a government insurance provider that covers 97% of the population living in South Korea. The remaining 3% of the population in the lowest income bracket is also covered through medical aid programs operated by the NHIS. The Health Insurance Review and Assessment (HIRA) service is responsible for reviewing medical fees for reimbursement decisions and assessing the quality of healthcare services. Healthcare providers electronically submit claims to HIRA. Billing statements of claims that have been reimbursed are stored in the HIRA data warehouse which includes information on sociodemographic variables and health care utilization of treatments such as procedures, surgeries, examinations, and prescriptions for the entire population of Korea. 15 HIRA data are widely used in various epidemiologic studies to determine the burden of disease or outcome of treatment.16,17
Study population
Among adults ⩾18 years of age who were diagnosed with gout [International Classification of Disease 10th revision (ICD-10) code M10] from 1 January 2009 to 31 December 2015, patients who newly started allopurinol or benzbromarone were included in this study. Data were collected from 2008, and patients who had already started allopurinol or benzbromarone for 1 year prior to the index date were excluded from initiators. Index date was defined as the first prescription date of allopurinol or benzbromarone. Patients who started other uric acid–lowering agents, such as febuxostat, rasburicase, probenecid, or sulfinpyrazone for 1 year prior to the index date and had a history of hemodialysis or peritoneal dialysis were excluded from the study. In addition, patients who took allopurinol and benzbromarone concurrently during the study period and patients whose prescription date was <30 days were excluded from the study. Finally, 257,097 allopurinol initiators and 7868 benzbromarone initiators were included in the analysis (Figure 1).

Flowchart of study population.
Study outcomes and follow-up
The primary outcome was a composite CV outcome, hospitalization for MI, stroke, or transient ischemic attack (TIA), or coronary revascularization. The secondary outcomes were components of the primary outcome and hospitalization for HF. MI, stroke, TIA, and HF were defined using ICD-10 codes (MI: I21, I22; stroke: I63; TIA: G45, G46; HF: I50). Coronary revascularization was defined using the procedure codes in the claim record. Information on the patient baseline characteristics including demographics, baseline comorbidities, and medication during the previous year from the index date was collected. The study participants were followed up from index date until the date of outcome occurrence, drug discontinuation, or the end of the study (31 December 2018), whichever came first (Figure 2). Drug discontinuation date was defined as the last dispensing date plus days of supply with 30-day grace period or the date of change with other uric acid–lowering agents.
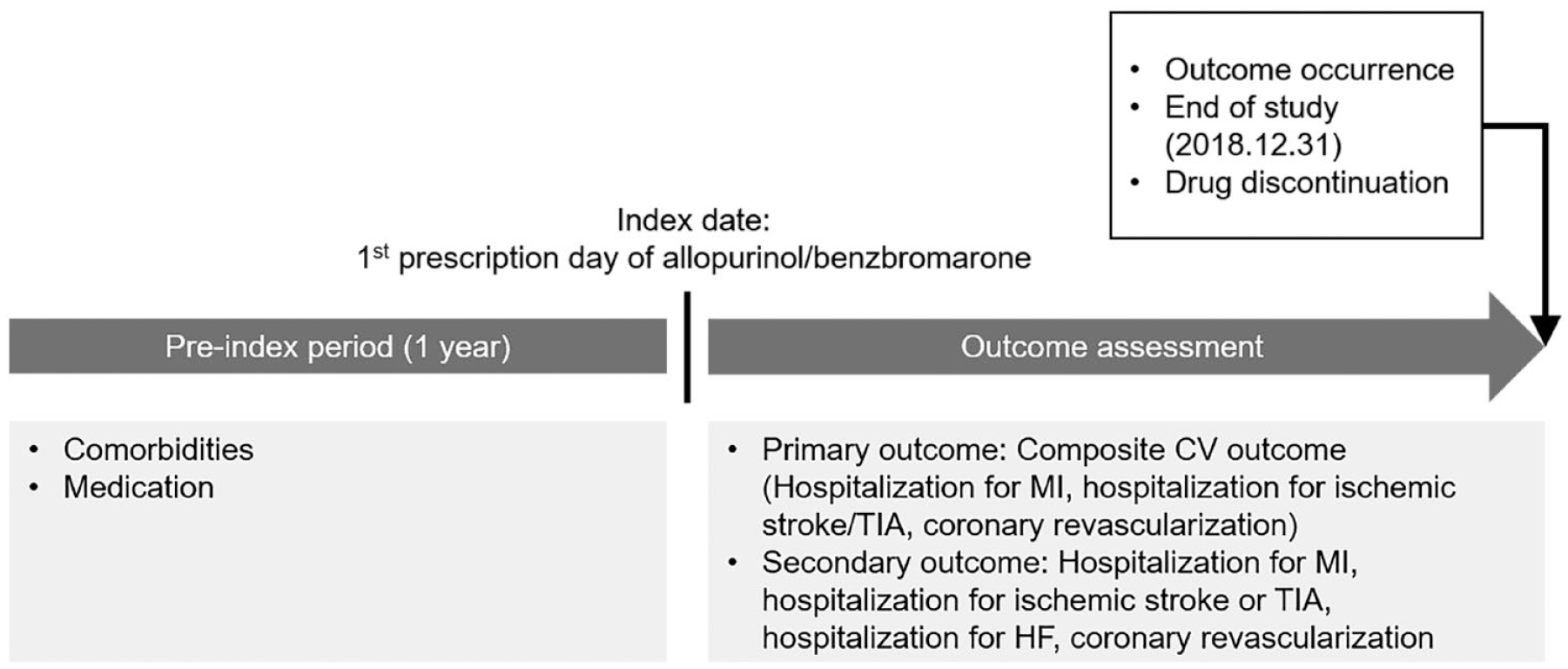
Study design.
Statistical analysis
Data distribution was evaluated using Shapiro–Wilk’s test. Mann–Whitney’s U-test and chi-square test were used for comparison between groups. The incidence rate of CV outcome was calculated by dividing incident cases by the total follow-up period. Cox proportional hazard models were used to analyze the hazard ratios (HRs) and 95% confidence intervals (CIs) of the risk of CV outcome following the use of allopurinol or benzbromarone. Kaplan–Meier curves were used to compare the CV outcome of allopurinol and benzbromarone initiators.
Propensity score (PS) matching was used to minimize the effect of differences in baseline characteristics between allopurinol and benzbromarone initiators on outcome development. PS was estimated using a multivariable logistic regression model including variables of baseline characteristics in Table 1, and allopurinol and benzbromarone initiators were matched 3:1 using nearest PS on the logit scale (caliper of 0.25). After PS matching, the standardized mean differences were within 0.1. Cox proportional hazard analysis was performed on the risk of CV outcome in allopurinol and benzbromarone initiators after PS matching.
Baseline characteristics of allopurinol and benzbromarone initiators.

ACE, angiotensin-converting enzyme; ARB, angiotensin II receptor blocker; CCI, Charlson Comorbidity Index; CV, cardiovascular; HF, heart failure; MI, myocardial infarction; NSAID, non-steroidal anti-inflammatory drug; SD, standard deviation; SMD, standard mean difference; TIA, transient ischemic attack.
Post hoc power analysis was performed on the data before and after matching. The minimum value of the adjusted HR that can secure statistical significance with 80% power was 1.401 in the data before matching and 1.389 in the data after matching.
Subgroup analysis was performed for the high CV disease risk group. The high-risk group was defined as males >50 years of age and females >55 years of age diagnosed with angina, MI, stroke, TIA, peripheral vascular disease, or diabetes in the 1 year prior to the index date. In addition, a subgroup analysis was performed in equivalent dose users using allopurinol ⩾200 mg or benzbromarone ⩾50 mg.
Sensitivity analyses were performed based on grace period, adherence, follow-up duration, and chronic kidney disease. The CV outcomes when the grace period was adjusted to 60 and 90 days were analyzed. Adherence was calculated by dividing the total number of prescription days for a drug by the follow-up time in days and multiplying by 100. Previous study has described that the median medication possession ratio for any uric acid−lowering drugs in gout patient was 0.68. 18 Therefore, we set 60% as a cut-off that reflects the mean adherence, and CV outcomes were assessed in patients with adherence ⩾60%. To compare the CV outcome in highly adherent patients, we compared the CV outcome in patients with adherence ⩾90%. Stratified analysis according to follow-up duration was performed by dividing it into less than 6 months, 6 months to 1 year, 1–2 years, and more than 2 years. In addition, stratified analysis was performed according to the presence or absence of baseline chronic kidney disease.
Statistical analysis was performed using SAS version 9.4 (SAS Institute Inc., Cary, NC, USA), and p value < 0.05 was considered statistically significant.
Results
Baseline characteristics
The baseline characteristics of allopurinol and benzbromarone initiators are presented in Table 1. The mean age of subjects included in the study was 54 years and 86% were male. Benzbromarone initiators had more comorbidities, such as MI, angina, stroke, hypertension, diabetes mellitus, and hyperlipidemia, and used more related drugs than allopurinol initiators. Compared with allopurinol initiators, benzbromarone initiators had more chronic kidney disease, used less non-steroid anti-inflammatory drugs (NSAIDs) and used more colchicine and oral steroids.
The mean daily dose of the drug was 186 mg [standard deviation (SD) 82 mg] in allopurinol initiators and 62 mg (SD 27 mg) in benzbromarone initiators. The mean drug adherence was 68.2% (SD 17.9%) in allopurinol initiators and 74.4% (SD 17.4%) in benzbromarone initiators. The number of patients in each group according to the follow-up period is presented in Supplementary Table S1, 72.7% of allopurinol initiators and 57.8% of benzbromarone initiators followed-up within 6 months. The number of patients followed up for more than 2 years was 19,550 (7.6%) in the allopurinol group and 965 (12.3%) in the benzbromarone group, respectively.
CV risk in allopurinol and benzbromarone initiators
The incidence rate of composite CV outcome was 1.89 per 100 person-years (PYs) during follow-up of 152,332 PYs in allopurinol initiators and 1.70 per 100 PYs during 6705 PYs in benzbromarone initiators (Table 2). When the allopurinol group was used as a reference, the adjusted HR (aHR) for the primary outcome in the benzbromarone group was 1.01 (95% CI: 0.83−1.21), indicating no statistical difference in the incidence of composite CV outcome between allopurinol and benzbromarone initiators. Significant differences between the two groups were not observed in secondary outcomes such as hospitalization for MI, stroke, TIA, or HF, and coronary revascularization (Figure 3).
Comparison of CV risk between allopurinol and benzbromarone initiators.

aHR, adjusted hazard ratio; CI, confidence interval; CV, cardiovascular; HF, heart failure; HR, hazard ratio; IR, incidence rate; MI, myocardial infarction; PYs, person-years; Ref, reference; TIA, transient ischemic attack.
Adjusted for age, sex, hypertension, diabetes mellitus, hyperlipidemia, chronic kidney disease, MI, angina, stroke, TIA, peripheral vascular disease, HF, angiotensin converting enzyme inhibitor or angiotensin II receptor blocker, beta blocker, calcium channel blocker, diuretics, anticoagulant, antiplatelet, statin, other antilipidemic agent, insulin, and non-insulin antidiabetic agent.
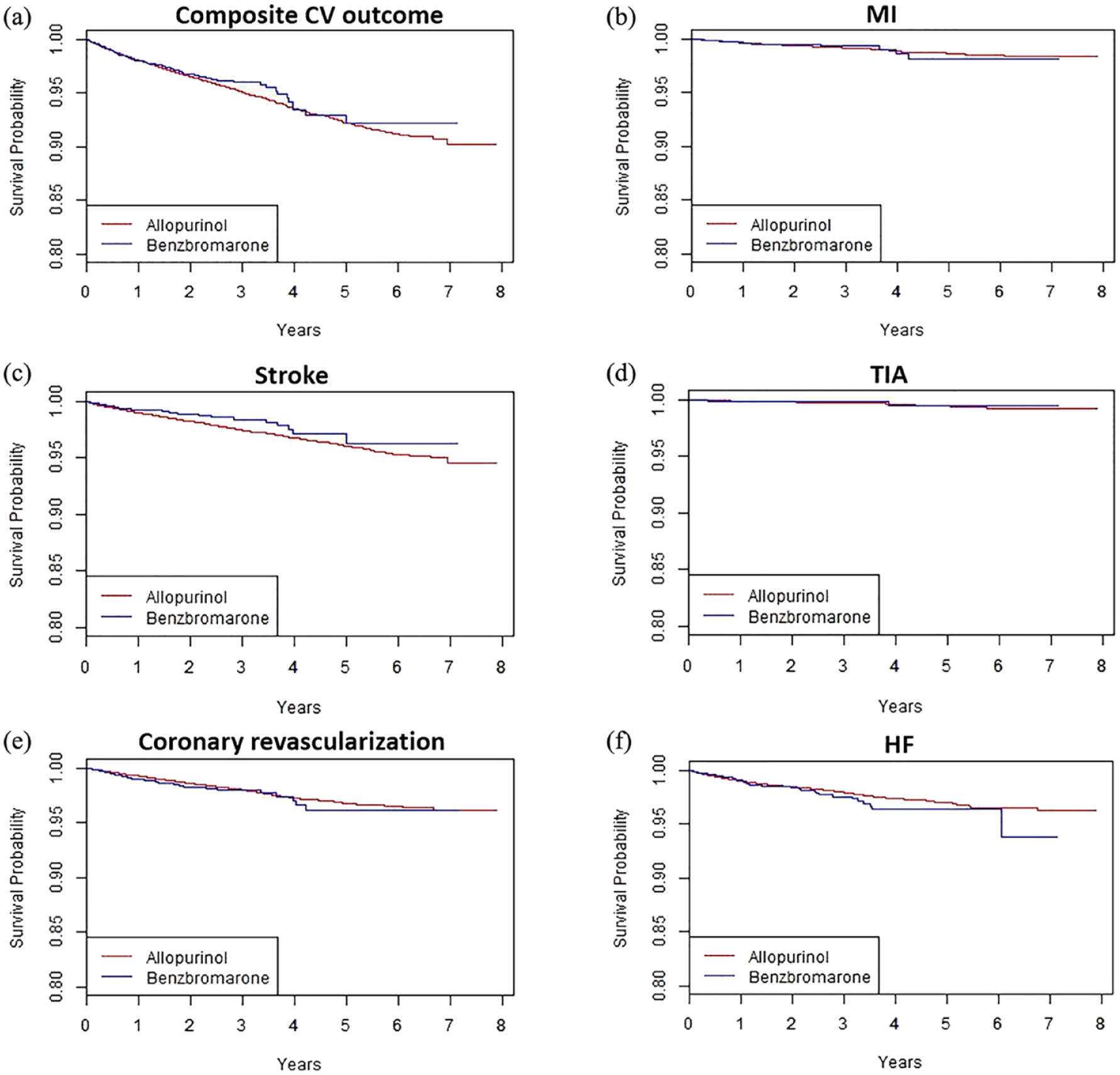
Kaplan–Meier curves for cardiovascular (CV) outcome in allopurinol and benzbromarone initiators: (a) composite CV outcome; (b) hospitalization for myocardial infarction (MI); (c) hospitalization for stroke; (d) hospitalization for transient ischemic attack (TIA); (e) coronary revascularization; and (f) hospitalization for heart failure (HF).
After 3:1 PS matching of allopurinol and benzbromarone initiators using the baseline characteristic variables, the standardized mean difference was<0.1 (Supplementary Table S2). In the cohort based on PS matching, the composite CV outcome did not show any statistical difference between allopurinol and benzbromarone initiators (aHR 0.96, 95% CI 0.77−1.20; Table 3), and significant difference was not observed in the secondary outcome based on drug (Supplementary Figure S1).
Comparison of CV risk between allopurinol and benzbromarone initiators after 3:1 PS matching.

aHR, adjusted hazard ratio; CI, confidence interval; CV, cardiovascular; HF, heart failure; HR, hazard ratio; IR, incidence rate; MI, myocardial infarction; PS, propensity score; PYs, person-years; Ref, reference; TIA, transient ischemic attack.
Adjusted for age, sex, hypertension, diabetes mellitus, hyperlipidemia, chronic kidney disease, MI, angina, stroke, TIA, peripheral vascular disease, HF, angiotensin converting enzyme inhibitor or angiotensin II receptor blocker, beta blocker, calcium channel blocker, diuretics, anticoagulant, antiplatelet, statin, other antilipidemic agent, insulin, and non-insulin antidiabetic agent.
Subgroup analysis
When analyzing the CV outcome in the high CV disease risk group, the incidence rate of the composite CV outcome was 3.18 per 100 PYs for allopurinol initiators and 3.26 per 100 PYs for benzbromarone initiators; significant difference was not observed between groups (aHR 1.03, 95% CI 0.83−1.27). Among secondary outcomes, the risk of coronary revascularization was slightly higher in benzbromarone initiators, but non-significant after adjustment (Table 4).
Comparison of CV risk between allopurinol and benzbromarone initiators at high risk of CV disease.

aHR, adjusted hazard ratio; CI, confidence interval; CV, cardiovascular; CV, cardiovascular; HF, heart failure; HR, hazard ratio; IR, incidence rate; MI, myocardial infarction; PYs, person-years; Ref, reference; TIA, transient ischemic attack.
Adjusted for age, sex, hypertension, diabetes mellitus, hyperlipidemia, chronic kidney disease, MI, angina, stroke, TIA, peripheral vascular disease, HF, angiotensin converting enzyme inhibitor or angiotensin II receptor blocker, beta blocker, calcium channel blocker, diuretics, anticoagulant, antiplatelet, statin, other antilipidemic agent, insulin, and non-insulin antidiabetic agent.
In patients taking allopurinol ⩾200 mg or benzbromarone ⩾50 mg, other outcomes were not significantly different; however, coronary revascularization was higher in benzbromarone initiators (aHR 1.58, 95% CI 1.16−2.15; Supplementary Table S3).
Sensitivity analysis
Sensitivity analysis with grace period adjusted to 60 and 90 days showed no difference in primary outcome from main analysis; however, the risk of coronary revascularization was higher in benzbromarone initiators than in allopurinol initiators (Supplementary Table S4).
The CV risk was compared between allopurinol and benzbromarone initiators in subjects with drug adherence ⩾60% and in those with drug adherence ⩾90% (Supplementary Tables S5 and S6). Similar to the main analysis, significant difference was not observed in CV risk between the allopurinol and benzbromarone initiators in the adjusted model.
In sensitivity analysis according to follow-up duration and baseline chronic kidney disease, composite CV endpoints did not differ between allopurinol and benzbromarone initiators (Supplementary Tables S7–S10, S11–12).
Discussion
In a nationwide population-based cohort of gout patients who started taking allopurinol or benzbromarone, the incidence of CV outcomes did not differ based on which drug was used. Difference was not found in CV outcome based on allopurinol or benzbromarone treatment in the high CV disease risk group. The difference in CV risk based on treatment with allopurinol or benzbromarone was not observed even in the group with high drug adherence. However, in the group using allopurinol ⩾200 mg or benzbromarone ⩾50 mg, the risk of coronary revascularization was slightly higher in benzbromarone initiators.
Several factors have been suggested as the cause of the increased CV risk in gout patients compared with non-gout controls. Previous studies have shown that traditional CV risk factors, such as hypertension, diabetes mellitus, and metabolic syndrome, were shown more common in gout patients, which may be associated with increased CV risk in gout patients.19–22 Hyperuricemia, which plays a central role in the pathophysiology of gout, has been suggested to increase CV risk by inducing endothelial dysfunction.23,24 Xanthine oxidase uses molecular oxygen as an electron acceptor in the purine oxidation process, resulting in the generation of reactive oxygen species (ROS), such as superoxide anion and hydrogen peroxide.25,26 ROS reacts directly with nitric oxide (NO) to inhibit the bioavailability of NO and reduces tetrahydrobiopterin, a cofactor of NO synthase (NOS), causing endothelial NOS (eNOS) uncoupling. 27 Uncoupled eNOS contributes to endothelial dysfunction by shifting the nitroso-redox balance to favor superoxide anion formation over NO. A recent finding that a neutrophil activation signature was associated with an increased CV risk in gout patients indicates that inflammation also contributes to an increased CV risk in gout patients. 28
Although controversy remains, in previous studies, uric acid–lowering therapy used as a treatment for gout ameliorated the increased CV risk in patients with gout.8,12,29 Allopurinol, a purine analog XOI, has been suggested to have beneficial effects on CV disease by reducing uric acid as well as by inhibiting xanthine oxidase to inhibit ROS generation, thereby reducing oxidative stress and improving endothelial dysfunction.30–32 Conversely, benzbromarone is a representative uricosuric agent and inhibits the dominant apical (luminal) urate exchanger of urate anion transporter 1 (URAT1), which is a urate transporter in the proximal tubule of the kidney, thereby reducing urate re-absorption and, consequently, promoting urate excretion into urine. 33 Therefore, benzbromarone may be involved in CV risk through a mechanism different from that of allopurinol. In experimental studies, uric acid transporters were shown expressed not only in renal tubular cells, but also in endothelial cells and vascular smooth muscle cells (VSMCs).34–36 Organic anion transporter (OAT) inhibitors, such as probenecid and benzbromarone, hindered uric acid uptake into the VSMC and decreased cell proliferation and expression of monocyte chemoattractant protein-1 (MCP-1). 37 In addition, benzbromarone had direct scavenging activity against superoxide anion radical in an in vitro study 38 and an antioxidant effect by reducing the levels of advanced oxidation protein products in an in vivo study using a rat model of hypertension. 39
In this study, significant difference was not observed in CV outcome between allopurinol and benzbromarone initiators, and although statistically non-significant, benzbromarone showed a more beneficial effect than allopurinol in stroke. Results of a recent in vitro study indicated that benzbromarone might have a protective effect against vascular diseases of the brain. In human brain microvascular endothelial cells, benzbromarone inhibited propofol-induced production of mitochondrial ROS, inflammatory factors, and adhesion molecules, indicating that benzbromarone may have protective properties against endothelial injury of the brain. 40
In subgroup analysis of patients taking allopurinol ⩾200 mg and benzbromarone ⩾50 mg, a higher aHR was found for coronary revascularization in benzbromarone initiators than in allopurinol initiators. This can be considered a result reflecting the difference in baseline characteristics between the two groups. In this study, benzbromarone initiators had more comorbidities and took more related medications than allopurinol initiators. In previous studies, high morbidity burden resulted in higher use of specialist physicians 41 and was associated with increased healthcare utilization. 42 Therefore, coronary revascularization was possibly observed more in benzbromarone initiators with higher comorbidity burden.
It can be explained in the same context that the benzbromarone group had an adherence of 75%, which was higher than that of the allopurinol group, which was 67%, and that of uric acid–lowering agents reported in a previous study, which was 68%. 18 Previous studies have shown that gout patients with many comorbidities have higher adherence than those without,43,44 and such comorbidities may affect the occurrence of CV events. The incidence rate of CV events was higher in the group with higher drug adherence, which was also considered as a finding that reflects the patient’s characteristics related to high adherence rather than the true drug effect.
The incidence rates of the composite CV endpoint in patients taking the equipotent dose of allopurinol and benzbromarone were 1.51/100 PYs and 1.83/100 PYs, respectively, lower than in the total group. In stratified analyses according to follow-up duration, the incidence rate was lower as the follow-up period was longer. These results suggest the possibility that uric acid–lowering therapy may have a protective role in CV events.
This study had several limitations. First, because information on the serum uric acid level was lacking, analysis of whether the degree of uric acid–lowering differed between the two drugs and whether a difference existed in the CV risk based on the uric acid differences could not be performed. Second, HIRA data did not include death information, rendering analysis CV mortality or all-cause mortality impossible. Third, as real-world data reflecting actual clinical practice, differences existed in the number and baseline characteristics between allopurinol and benzbromarone initiators. However, to overcome this, outcomes in the two groups after PS matching were also compared and the same results obtained as in the main analysis. Fourth, the analysis could not be performed with respect to the dose of corticosteroids or the use of colchicine that could affect the CV risk. However, the standardized mean difference of colchicine use of allopurinol and benzbromarone initiators at baseline was less than 0.1, indicating that there was no meaningful difference in colchicine use between the two groups. Finally, since our study was conducted on Koreans, there is a limitation in generalizability to other races.
The association between gout and hyperuricemia and CV disease has been emphasized and the effect of uric acid–lowering therapy on CV risk investigated in numerous studies; however, a conclusion has not yet been reached. This study results showed benzbromarone had a similar effect on CV risk compared with allopurinol, and indicating benzbromarone may have a protective effect against stroke. This is important for gout patients who need to decide on uric acid–lowering treatment in consideration of future CV risk.
Conclusion
Difference was not found in the incidence of CV events among gout patients treated with allopurinol or benzbromarone in a nationwide population-based cohort. In the high-CV-risk group, the incidence of CV events did not differ between allopurinol and benzbromarone initiators. A prospective study to confirm the effects of allopurinol and benzbromarone treatment on CV risk is warranted.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X221116409 – Supplemental material for Cardiovascular risk associated with allopurinol or benzbromarone treatment in patients with gout
Supplemental material, sj-docx-1-tab-10.1177_1759720X221116409 for Cardiovascular risk associated with allopurinol or benzbromarone treatment in patients with gout by Yeonghee Eun, Heewon Han, Kyunga Kim, Seonyoung Kang, Seulkee Lee, Hyungjin Kim, Jaejoon Lee, Eun-Mi Koh and Hoon-Suk Cha in Therapeutic Advances in Musculoskeletal Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
