Abstract
Objectives:
To analyse the accuracy of plasma calprotectin in patients with rheumatoid arthritis (RA) receiving monoclonal antibodies against IL-6 receptors (anti-rIL-6) or JAK inhibitors (JAKis) in detecting ultrasound (US) synovitis and compare it with acute phase reactants [high-sensitivity C-reactive protein (hs-CRP) and ESR].
Methods:
An observational cross-sectional study of RA patients receiving anti-rIL-6 (tocilizumab or sarilumab) or JAKi, (baricitinib or tofacitinib) was made. Plasma calprotectin for the diagnosis of US synovitis [synovial hypertrophy grade (SH) ⩾ 2 plus power Doppler signal (PD) ⩾ 1] was analysed using receiver operating characteristic curves (ROCs). The performance of ESR and hs-CRP was also studied. The three ROC curves were compared to determine which had the highest discriminatory power. Associations between plasma calprotectin and US scores were made using correlation analysis.
Results:
Sixty-three RA patients were included. Mean plasma calprotectin levels were significantly higher in patients with US synovitis than in those without (0.89 ± 0.85 vs 0.30 ± 0.12 μg/ml; p = 0.0003). A moderate correlation between calprotectin and all US scores (HS score Rho = 0.479; PD score Rho = 0.492; and global score Rho = 0.495) was found. The discriminatory capacity of plasma calprotectin showed an AUC of 0.795 (95% CI: 0.687–0.904). The AUC of hs-CRP and ESR was 0.721 and 0.564, respectively. hs-CRP serum levels showed a low positive correlation with the three US scores (Rho < 0.40). After analysis according to the drugs administered, the correlation disappeared in patients receiving anti-rIL-6.
Conclusion:
Plasma calprotectin may be a sensitive biomarker of synovial inflammation in RA patients treated with anti-rIL-6 or JAKi.
Keywords
Introduction
The prognosis of rheumatoid arthritis (RA) has significantly improved in recent decades. In patients who do not achieve the therapeutic goal (remission or low disease activity) with a first strategy using conventional synthetic disease-modifying antirheumatic drugs (DMARDs), e.g. methotrexate, a second strategy using targeted therapies, biologics, or selective Janus kinase inhibitors (JAKi) is recommended. 1 Monoclonal antibodies against interleukin (IL)-6 receptors (anti-rIL-6) (tocilizumab and sarilumab) and JAKi (tofacitinib, baricitinib and, more recently, upadacitinib) are included in this strategy, with significant improvements in clinical and radiographic outcomes.2,3 It has been shown that both families of targeted therapies have a profound impact on the acute phase response, including C-reactive protein (CRP) and erythrocyte sedimentation rate (ESR), due to inhibition of hepatocyte stimuli via IL-6.4,5 Acute phase reactants (APRs) form part of the composite activity indices [e.g. Disease Activity Score (DAS) and Simplified Disease Activity Index (SDAI)] used to measure inflammatory activity in RA and may lead to an overestimate of the clinical response when using these targeted drugs. 6
Calprotectin is a heterodimeric complex of two S100 calcium-binding myeloid-related proteins (MRP8 [or S100A8] and MRP14 [or S100A9]) which is released from cells of the innate immunity, such as neutrophils or monocytes, which have proinflammatory activities and act as endogenous-associated molecular patterns via Toll-like receptor activation. 7 Calprotectin is strongly expressed in rheumatoid synovial membrane. 8 Recently, serum and plasma calprotectin have been found to be sensitive markers of inflammatory activity in RA patients 9 and have been associated with radiographic damage, 10 as a biomarker of clinical response to antirheumatic drugs 11 and as a predictive factor for disease relapse. 12
Joint ultrasound (US) is a validated imaging technique for synovitis evaluation in RA, with a higher sensitivity than the clinical examination in detecting active synovitis. 13 A significant proportion of patients in clinical remission may exhibit active synovitis on US. 14 Recent studies have shown that calprotectin is more associated closely than APR with US synovitis in RA, even in patients in remission or with low disease activity.15,16
We have shown that in RA patients treated with tocilizumab, serum calprotectin but not serum CRP correlates with disease activity, 17 a finding also observed by other authors. 18 To our knowledge, there are no studies on calprotectin as a biomarker of US synovitis in RA patients treated with anti-rIL-6 or JAKi. The objective of the study was to analyse the accuracy of plasma calprotectin in patients with RA receiving anti-rIL-6 or JAKi in detecting US synovitis and compare it with traditional APR (CRP and ESR). We hypothesized that calprotectin has a higher discriminatory capacity than APR in detecting US synovitis in these patients.
Methods
Design and study population
An observational cross-sectional study of RA patients (ACR/EULAR 2010 criteria) 19 from our arthritis unit receiving anti-IL-6 receptor monoclonal antibodies (tocilizumab or sarilumab) or JAKi (baricitinib or tofacitinib) for ⩾3 months who were consecutively included. Patients were included on the day of the scheduled routine follow-up visit from September 2020 to September 2021. Patients were included regardless of disease activity status, previous DMARDs (including biological therapies or JAKi), and concomitant treatment (methotrexate or others). Patients who, at the study visit presented signs of active infection or other clinical conditions that, in the opinion of the investigator, could modify the results of CRP, ESR, or calprotectin determinations were excluded. Demographic data, disease duration, autoantibody status (ACPA/RF), radiological data (erosive disease), previous biologic therapy, and concomitant therapy were collected.
Measurement of clinical disease activity
All patients underwent a clinical assessment, including 28 swollen and tender joint counts (28-SJC and 28-TJC) and physician and patient global assessment (PhGA and PGA) with visual analogue scales (0–100 mm). Disease activity indices were subsequently calculated (DAS28, SDAI, and CDAI). In addition, participants also were asked to complete two questionnaires: the Health Assessment Questionnaire (HAQ) and the Routine Assessment of Patient Index Data 3 (Rapid3).
Assessment of blood biomarkers
Blood samples were obtained in the clinical evaluation. ESR was measured using the Westergren method (NV < 20 mm/h), and high-sensitivity C-reactive protein (hs-CRP) using an immunoturbidimetric method measured by Siemens Atellica® Solution (lowest detection limit of 0.02 mg/dl; NV < 0.4 mg/dl). Calprotectin plasma levels were determined using an ELISA Test Kit [CALPROLAB ALP (CALPRO), Menarini Diagnósticos S.A.] according to the manufacturer’s protocol. Briefly, 100 µl of each standard, control, and diluted 1:20 sample in duplicate wells were incubated at room temperature for 40 min; three washings were made, 100 µl of the conjugated enzyme was added, and plates were incubated at room temperature for 40 min. After three washes and the addition of the enzyme-substrate, the optical density values at 405 nm were determined using an ELISA reader. To reduce variations in calprotectin determination, the whole procedure was performed in a Triturus autoanalyzer; the coefficients of variation were 5% within and 13% between assays.
Imaging biomarkers: US score
Sonographic assessments were carried out using high-sensitivity US equipment (MyLab9®; Esaote, Genoa, Italy), with a longitudinal probe, frequency range from 10 to 14 MHz and a pulse repetition frequency between 800 and 900 Hz. Joint musculoskeletal US findings were defined according to published Outcome Measures in Rheumatoid Arthritis Clinical Trial (OMERACT) definitions. 20
A single experienced sonographer (AP), who was blinded to the results of the clinical joint examination, evaluated 11 joints and tendons of each hand (including the proximal interphalangeal joints, metacarpophalangeal joints, and wrists) for synovial hypertrophy (SH) and intra-articular power Doppler (PD) signalling according to EULAR guidelines. 21 SH and PD signals were graded using a four-grade semi-quantitative scoring system (0 = no, 1 = mild, 2 = moderate, and 3 = severe) according to the methodology of Szkudlarek et al. 22 The highest SH and PD grade detected during the scans was adopted as representative of each joint, respectively. We also evaluated synovitis in symptomatic joints outside the hands and graded it using the methodology of Szkudlarek et al. 22
By summing the scores for elementary lesions in each joint, we calculated the PD score (sum of PD scores in all joints, range 0–66), the SH score (sum of SH scores in all joints, range 0–66) and the global score (sum of the PD and SH scores, range 0–132). The score does not include joints outside the hands. To ensure a stringent definition of US synovitis, only patients with SH grade ⩾ 2 plus PD signal (⩾1) were classified as having active synovitis. 23
Statistical analysis
Continuous data were presented as the mean (SD) and categorical variables as absolute frequencies with percentages. Groups were compared using parametric or nonparametric tests according to the distribution of the variables.
The performance of calprotectin in the diagnosis of US synovitis was analysed using receiver operating characteristic curves (ROC) with US synovitis yes/no (yes: SH grade ⩾ 2 plus PD signal ⩾ 1) as the gold standard. The ROC curves made it possible to identify the best cut-off point in terms of sensitivity, specificity, and likelihood ratios, and to calculate the area under the curve (AUC) as a measure of the overall discriminative power. The performance of ESR and hs-CRP was also studied, and the three ROC curves were compared to determine which of the three parameters had the highest discriminatory power for the diagnosis of synovitis.
Correlation analysis (Spearman’s correlation coefficient) was used to assess the association between plasma calprotectin and US scores (PD score, SH score, and global score). The analysis was made using STATA version 12 (STATA Corp, College Station, TX, USA).
The study was conducted in accordance with the Declaration of Helsinki and was approved by the Clinical Research Ethics Committee of the Hospital Clinic of Barcelona (Reg. HCB20210783). Written informed consent was obtained from all patients before study enrolment and patients were de-identified. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 24
No specific funding was received from any bodies in the public, commercial, or not-for-profit sectors to carry out the work described in this article.
Results
Demographic, clinical, and therapeutic characteristics
Although the initial sample consisted of 78 patients, US studies were performed in 63 (42 receiving anti-rIL-6 and 21 JAKi), which was the final sample included in the study. Ninety-two percent of patients were female with a mean age of 56 (±12) years. Mean disease duration was 15 (±8.9) years, 90.5% were seropositive (RF and/or ACPA) and 75.8% had erosive disease. In general, disease activity was low, with a median CDAI value of 11.3 (±8.7). DAS28 remission was observed in 29 patients (46%). The mean duration of drug therapy (anti-rIL-6 or JAKi) was 45 (±42.4) months.
Patient with US synovitis had a significantly higher prevalence of radiographic erosive disease and a trend to a higher disease duration. As expected, higher median values of the disease activity composite index were observed in patients with US synovitis than in those without, although the difference was not significant measured by DAS28. Furthermore, remission rates were higher in patients without US synovitis. No differences were observed according to the drugs used (anti-rIL-6 or JAKi; Table 1).
Demographic, clinical and laboratory characteristics of patients with RA (N = 63). Demographic, clinical and treatment variables, and biomarkers of patients classifying according to ultrasound activity [ultrasound synovitis in ⩾1 joint (SH ⩾ 2 + PD ⩾ 1)].

CDAI, Clinical Disease Activity Index; DAS28, Disease Activity Score; HAQ, Health Assessment Questionnaire; hs-CRP, high-sensitivity C-reactive protein; PGA, patient global assessment, PhGA, global assessment; Rapid3, Routine Assessment of Patient Index Data 3; SD, standard deviation; SDAI, simplified Disease Activity Index; 28 SJC, 28 swollen joint counts; 28 TJC tender joint count; US synovitis, ultrasound synovitis; VAS pain, Visual Analogue Scale.
Remission: DAS28 ⩽ 2.6.
Low disease activity: 2.6–3.2.
Moderate disease activity: DAS28 3.2–5.1.
High disease activity: DAS28 ⩾ 5.1.
Calprotectin and APR as biomarkers of US synovitis
Mean plasma calprotectin levels were significantly higher in patients with US synovitis than in those without (0.89 ± 0.85 vs 0.30 ± 0.12 μg/ml; p = 0.0003), as occurred with hs-CRP serum levels (0.05 ± 0.08 vs 0.27 ± 0.41 mg/dL; p = 0.007) but not with ESR (16.9 ± 25.6 vs 7.8 ± 4.2 p = 0.474; Table 1 and Figure 1).
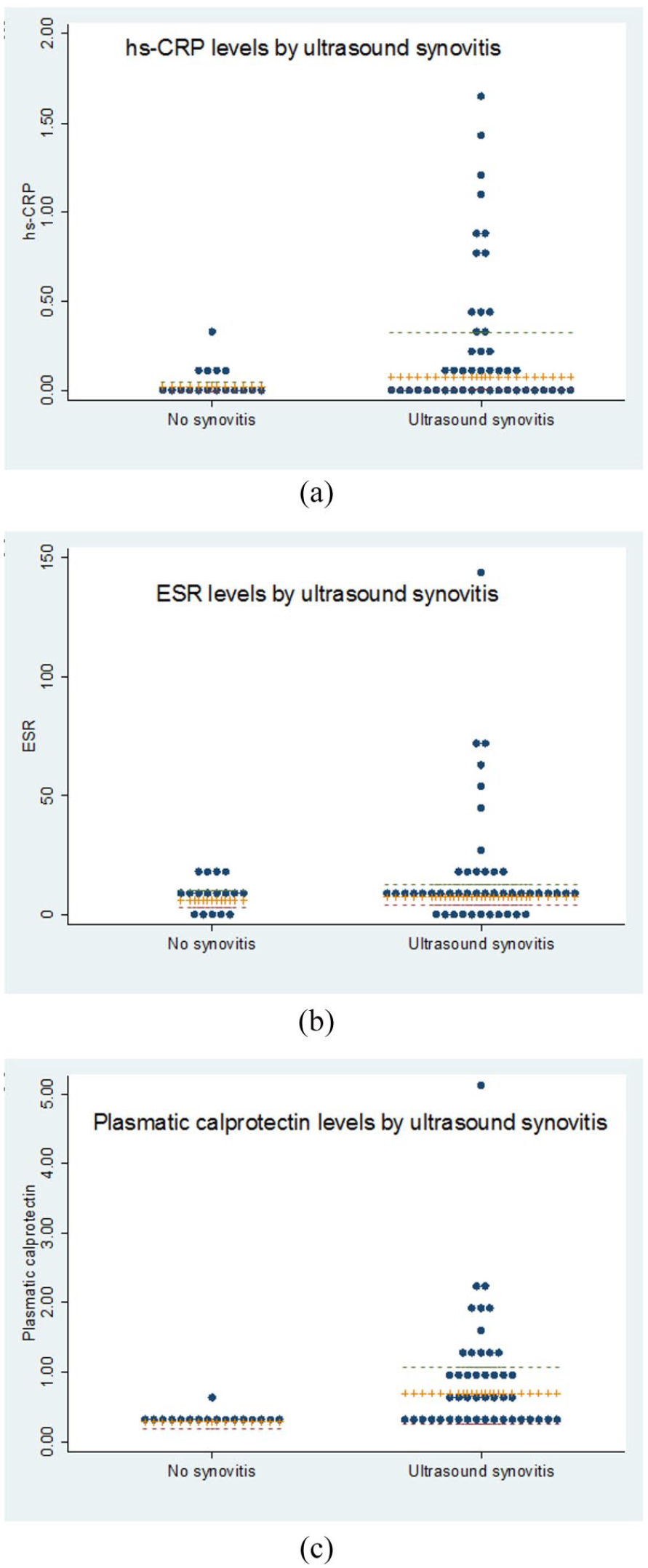
Blood biomarkers classifying patients according to ultrasound synovitis [ultrasound synovitis in ⩾1 joint (SH ⩾ 2 + PD ⩾ 1)]: (a) hs-CRP levels according to ultrasound synovitis, (b) ESR levels according to ultrasound synovitis, and (c) plasma calprotectin levels according to ultrasound synovitis. The central line (orange) represents the median. The lateral lines the 25th (red) and 75th (green) percentiles.
Analysis of the correlation between blood biomarkers and US scores showed a moderate correlation between calprotectin and the three US scores (HS score Rho = 0.479; PD score Rho = 0.492; and global score Rho = 0.495). The association of calprotectin with these three US parameters was found in patients treated with anti-rIL-6 and JAKi, although the magnitude of the association was greater in patients treated with JAKi (Table 2).
Correlation between ultrasound synovitis scores and blood biomarkers.
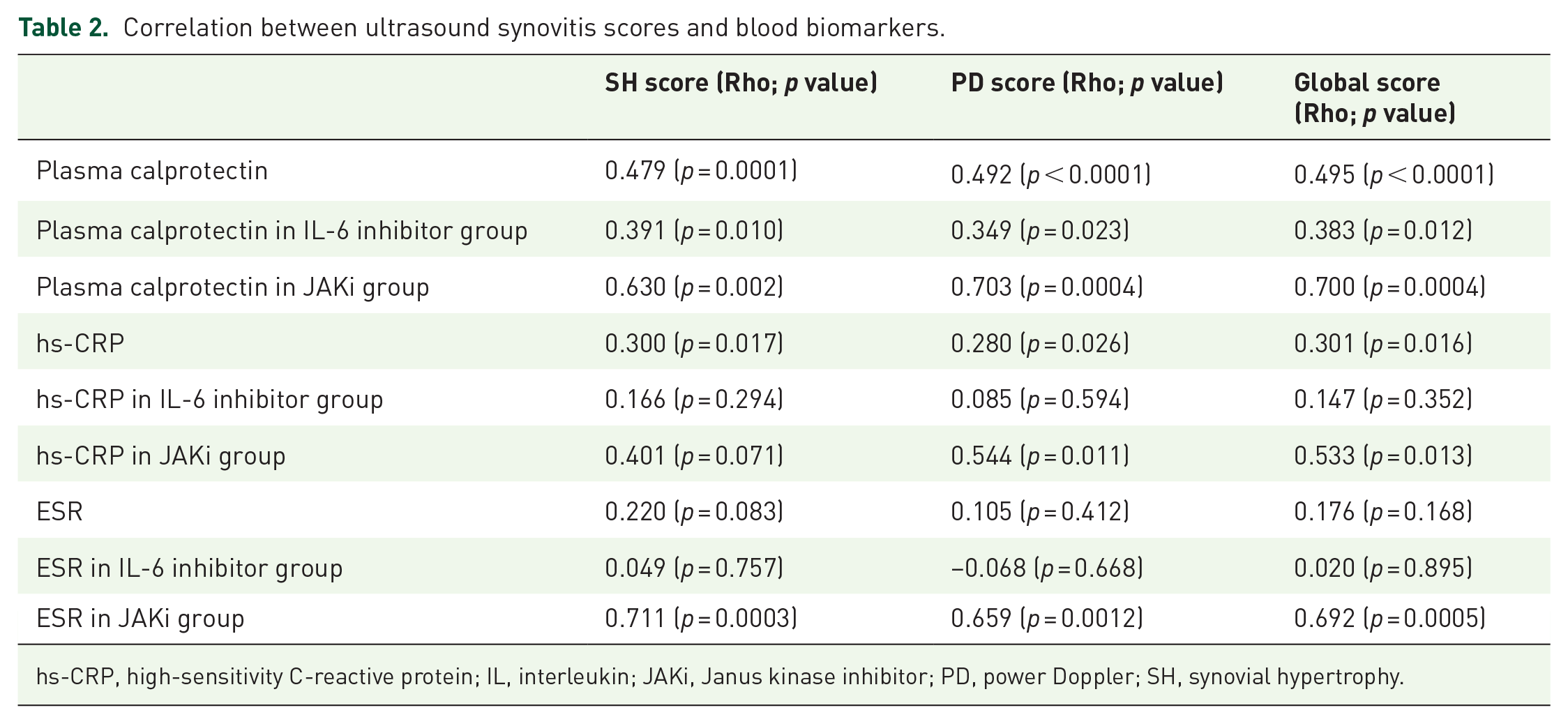
hs-CRP, high-sensitivity C-reactive protein; IL, interleukin; JAKi, Janus kinase inhibitor; PD, power Doppler; SH, synovial hypertrophy.
hs-CRP serum levels also showed a correlation with the three US scores, although with a low positive correlation (Rho < 0.40). However, analysis of the correlation according to treatment showed that the correlation disappeared in patients receiving anti-rIL-6 (Table 2). ESR showed a good correlation with US scores in patients receiving JAKi but not in those treated with anti-rIL-6 (Table 2).
Plasma calprotectin had a very good discriminatory capacity, with an AUC of 0.795 [95% confidence interval (CI): 0.687–0.904]. The best cut-off was ⩾0.38, with a sensitivity of 67.4% and a specificity of 88.2%, which correctly classified 73% of patients with US synovitis. The positive likelihood ratio was 5.7. The discriminatory capacity of hs-CRP and ESR was lower, with an AUC of 0.721 and 0.564, respectively (Figure 2). The ability of plasma calprotectin to discriminate US synovitis was higher in patients treated with JAKi (AUC = 0.931, 95% CI: 0.820–1.000) than in those receiving anti-rIL-6 (AUC = 0.712; 95% CI: 0.558–0.867; Figure 2). hs-CRP and ESR had a good discriminatory capacity to detect US synovitis in patients treated with JAKi but not in those receiving anti-rIL-6 (Figure 3).

ROC curves of blood biomarkers for ultrasound synovitis.

ROC curves of calprotectin, hs-CRP And ESR vs ultrasound synovitis according to type of treatment.
When analysing the correlation between CDAI index and US score, we have found a lower correlation than that observed with calprotectin (correlation between CDAI and US scores: CDAI and HS score Rho = 0.298; CDAI and PD score Rho = 0.354; CDAI and global score Rho = 0.334, p value < 0.05).
Discussion
We evaluated the accuracy of plasma calprotectin in detecting US synovitis in patients with RA treated with anti-rIL-6 or JAKi. Our results show that plasma calprotectin levels are associated with US synovitis in these patients. Calprotectin demonstrated a higher capacity than traditional biomarkers, such as CRP or ESR, in detecting US synovitis in RA patients treated with anti-rIL-6 and a better correlation with US scores in patients receiving JAKi. Therefore, plasma calprotectin may be considered a sensitive biomarker of synovial inflammation in RA patients treated with anti-rIL-6 or JAKi.
The role of APR in the evaluation of disease activity in RA has been questioned in patients receiving anti-rIL-6, such as tocilizumab and sarilumab, both of which have an important effect on APR due to the reduction in hepatic synthesis of acute phase proteins due to the inhibition of IL6 stimuli.6,25 As demonstrated with tocilizumab, CRP serum levels decrease dramatically independently of the improvement in inflammatory activity, while there are detectable drug levels in the blood. 26 Therefore, CRP serum levels should not be used as a biomarker of inflammation in patients treated with anti-rIL-6 and the composite indices including APR, such as DAS28, should be avoided. Furthermore, significant residual synovial inflammation, even in patients in clinical remission using lower cut-off values than previously validated, has been demonstrated in patients receiving tocilizumab. 27
Calprotectin may be a sensitive biomarker of synovitis in RA patients treated with anti-rIL-6, due to the high levels observed in the blood, which reflect local synovial inflammation rather than a systemic effect as occurs with APR. 28 Recent studies have shown that calprotectin is increased in the serum or plasma of patients with RA and other immunodetected diseases, reflecting a relevant role of neutrophilic activation in these diseases. 29 Calprotectin correlates better with active inflammatory active disease in RA than traditional APR such as CRP and ESR. 30
To our knowledge, this is the first study to evaluate the performance of blood calprotectin in RA patients treated with JAKi, a family of targeted therapies that also have also an important impact on APR. 3 Our results show that, in patients treated with JAKi, plasma calprotectin has a higher correlation with US synovitis than hs-CRP. Also, this correlation is higher than those observed with a clinical activity index such as CDAI. The discriminatory capacity of calprotectin in detecting US synovitis was also very high, but similar to that observed with hs-CRP. By contrast, hs-CRP and ESR did not show the same relevance in patients treated with anti-rIL-6, where only calprotectin was a biomarker of US synovitis.
US is a sensitive imaging technique for the detection of active synovitis in patients with RA and other immune-mediated rheumatic conditions. Our results confirm the association of calprotectin with US synovitis in RA as documented in other studies.15,16,31,32 We have previously demonstrated that, in patients receiving tumour necrosis factor inhibitor (TNFi), calprotectin is a good biomarker of US synovitis, even in patients in remission or with low disease activity. 16 We have replicated these findings for the first time in RA patients treated with anti-rIL-6 and JAKi, most of whom were in remission or with low disease activity.
Our study has some limitations. We included a relatively small sample size, especially of patients treated with JAKi. On the contrary, patients were predominantly in remission or with low disease activity. However, even in this context, calprotectin showed a good discriminatory capacity for US synovitis. No attempt was made to further distinguish the different antirheumatic drugs, to avoid very small samples that would make data interpretation difficult. However, we cannot say whether differences might be observed within the same therapeutic group.
In conclusion, plasma calprotectin is a good biomarker of US synovitis in patients with RA treated with anti-rIl-6, whereas traditional APR (CRP or ESR) do not reflect local synovial inflammation detected by US. Some differences emerged in RA patients treated with JAKi, where calprotectin is also a good biomarker of US synovitis in RA but in whom APR also reflect synovial inflammation. We suggest that plasma calprotectin is a valuable biomarker of synovitis in these patients.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X221114105 – Supplemental material for Plasma calprotectin as a biomarker of ultrasound synovitis in rheumatoid arthritis patients receiving IL-6 antagonists or JAK inhibitors
Supplemental material, sj-docx-1-tab-10.1177_1759720X221114105 for Plasma calprotectin as a biomarker of ultrasound synovitis in rheumatoid arthritis patients receiving IL-6 antagonists or JAK inhibitors by Beatriz Frade-Sosa, Andrés Ponce, José Inciarte-Mundo, Rosa Morlà, Viginia Ruiz-Esquide, Laura Macías, Ana Belen Azuaga, Julio Ramirez, Juan D. Cañete, Jordi Yague, Josep M. Auge, José A. Gomez-Puerta and Raimon Sanmarti in Therapeutic Advances in Musculoskeletal Disease
Footnotes
Acknowledgements
The authors acknowledge the assistance provided by rheumatology unit nurses (Nuria Sapena and Marta Bassas) statistical help from M Jesus Garcia Yebenes (Inmusc) and technical advice from David Buss.
Declarations
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
