Abstract
The antimalarial hydroxychloroquine (HCQ) has demonstrated several crucial properties for the treatment of systemic lupus erythematosus (SLE). Herein, we reviewed the main HCQ pharmacologic features, detailed its mechanism of action, and summarized the existing guidelines and recommendations for HCQ use in rheumatology with a systematic literature search for the randomized controlled trials focused on lupus. HCQ has been shown to decrease SLE activity, especially in mild and moderate disease, to prevent disease flare and to lower the long-term glucocorticoid need. The numerous benefits of HCQ are extended to pregnancy and breastfeeding period. Based on cohort studies, antithrombotic and metabolic HCQ’s effects were shown, including lipid-lowering properties, which might contribute to an improved cardiovascular risk. Moreover, early HCQ use in antinuclear antibodies positive individuals might delay the progression to SLE. Finally, HCQ has a significant favorable impact on long-term outcomes such as damage accrual and mortality in SLE. Based on these multiple benefits, HCQ is now the mainstay long-term treatment in SLE, recommended by current guidelines in all patients unless contraindications or side effects. The daily dose associated with the best compromise between efficacy and safety is matter of debate. The concern regarding retinal toxicity rather than proper efficacy data is the one that dictated the daily dosage of ⩽5 mg/kg/day actual body weight currently agreed upon.
Keywords
Background
Hydroxychloroquine (HCQ) is an antimalarial drug used initially for the treatment of Plasmodium parasitic infection, from where the name of the drug class came from. Beyond its initial indication as antimalarial, HCQ has been used in autoimmune and infectious diseases, as well as in metabolic or neoplastic disorders. 1 But, as recently reviewed, 2 clear benefits were reported mainly in systemic lupus erythematosus (SLE).
Thus, HCQ is now one of the most valuable therapies in SLE, showing multiple benefits over several outcomes associated with the disease itself, but also to its related comorbidities. HCQ is an inexpensive, generally available, well-tolerated immunomodulator. 3 For more than a decade, different authors emphasized that all patients with SLE should be given HCQ4–7 and the latest guidelines’ recommendations also stated the HCQ importance in SLE unless there are contraindications or side effects.8–11
The history of HCQ is supposed to start circa 1600 with the Incas in Chile, from whom the cinchona bark properties were learned by the Jesuits. The main alkaloids of quinine and cinchonine were isolated in 1820 and subsequently chloroquine (CQ) was obtained much later in 1934. 12 HCQ sulfate is the hydroxylated analogue of CQ, synthesized in 1946. Due to a better safety profile, HCQ was given since 1955 as an alternative to CQ.12,13
For SLE, the first report of the antimalarials use dates back to 1894, regarding the improvement of cutaneous lupus lesions with quinine.14,15 In the United States, HCQ was approved for SLE in 1955 for symptoms like fatigue, rashes, joint pain, and mouth sores 16 and, with specific approval and license characteristics for each country, is now among the main drugs used for SLE treatment worldwide.
Pharmacology of hydroxychloroquine
Molecular structure
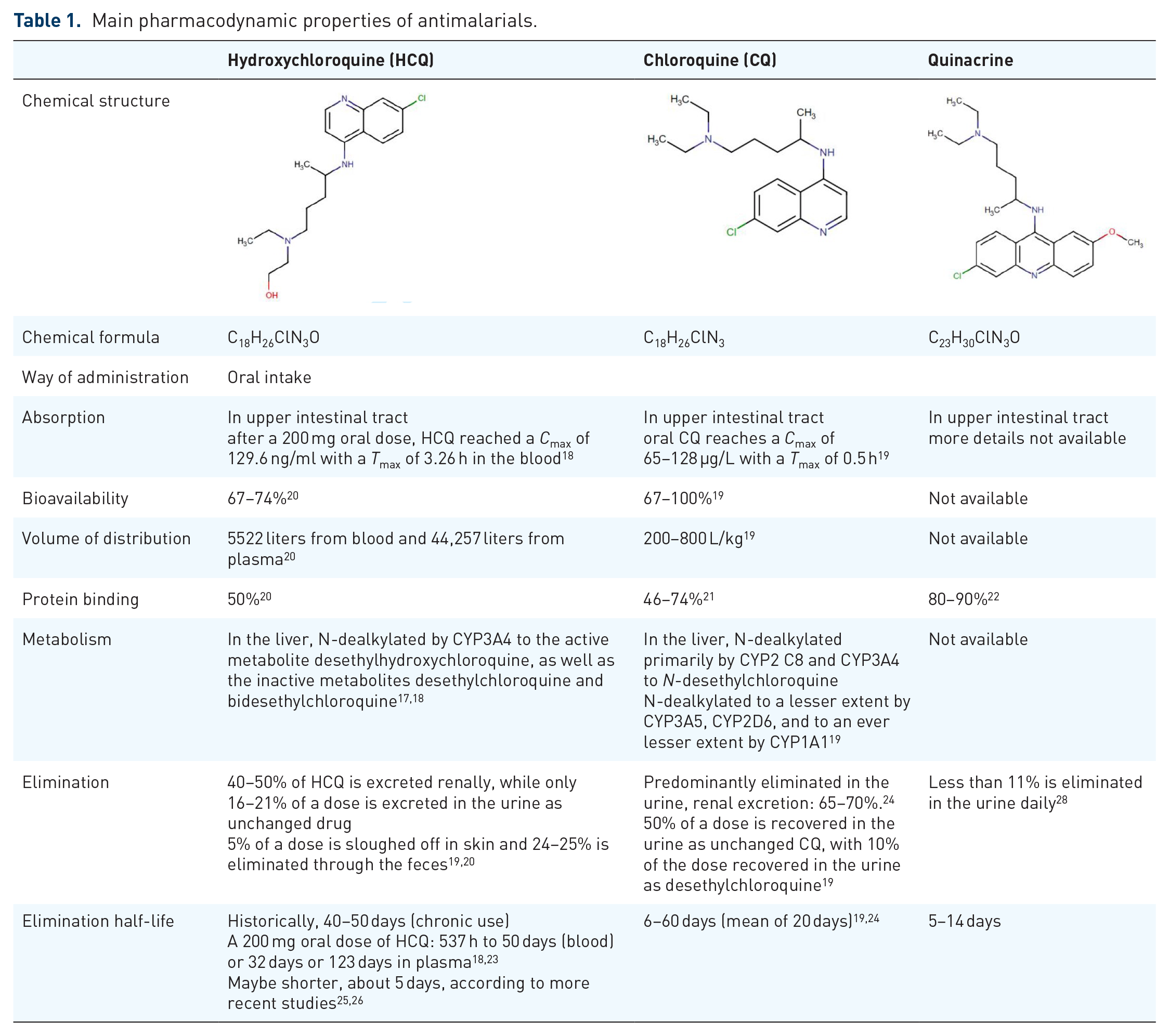
The knowledge about the pharmacokinetics of antimalarials is not completely understood and still debated. These pharmacokinetic characteristics are complex17–19 due to the large volume of distribution,19,20 significant tissue binding,20–22 and long terminal elimination half-life.18,19,23,24 Indeed, important differences have been observed between HCQ pharmacokinetic parameters as evidenced recently by its use in the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) disease (COVID-19). 25 Historically, terminal elimination half-lives were considered very long, 40–50 days for HCQ18,23 and up to 60 days for CQ.19,24 More recent studies suggest a shorter half-life of about 5 days.25,26 A long HCQ half-life can be attributed to extensive tissue uptake rather than to an intrinsic inability to clear the drug. The expected delay in the attainment of steady-state concentrations (3–4 months) may be in part responsible for the slow therapeutic response observed with HCQ. 27 Renal clearance is an important consideration for both drugs as reduced clearance increases the bioavailability 28 and subsequently the related side effects.19,20,24 Finally, dose–response relationships and toxicity thresholds have not yet been fully defined. The main pharmacodynamic properties of antimalarials are shown in Table 1.
Main pharmacodynamic properties of antimalarials.
Galenic and commercial presentations
HCQ is commercialized as 200 mg HCQ sulfate tablets corresponding to 155 mg HCQ base for each tablet. 29 The daily dosage of HCQ varies accordingly to its indication, 29 with the American Academy of Ophthalmology (2016-AAO) recommending no more than 5 mg/kg/day of real body weight in SLE to decrease retinopathy occurrence, 30 recommendation that has been recently reinforced by agreement of four medical societies. 31 The indication is based on an ophthalmological study by Melles and Marmor 32 of nearly 2500 patients in whom daily HCQ intake below 5 mg/kg/day of regular body weight was associated with a low risk of toxicity, <2% within the first 10 years of use. However, some authors highlighted that in that study, the dose of HCQ was based on pharmacy refill information and not on prescribed dose. 33
Dose adjustments with 50% reduction of posology are needed for patients with renal impairment and lower than 30 ml/min filtration rate. 34 For patients weighting more than 80 kg, a maximum daily dose of 400 mg is recommended in SLE. Doses for CQ were established only from extrapolation of HCQ and those lower than 2.3 mg/kg/day were considered safe.30,35
As the terminal elimination half time is not short, 36 dosing can be adjusted by alternate day regimens, such as 200 mg on the first day and 400 mg on the second day, yielding a mean dose equivalent to 300 mg per day. 32 Based on recent surveys, the most common daily dosage for HCQ is 400 mg daily.37,38
Mechanism of action
The mechanisms of action for HCQ are complex and still not completely understood (see Table 2 and Figure 1). Because of its high lipophilicity, lysosomotropism, and pH,39,40 HCQ can pass through cell membranes and accumulate into lysosomes 40 where it disrupts key important cellular functions via the inhibition of the Toll-like receptors (TLRs)41–43 and of the Cyclic GMP-AMP synthase–Stimulator of Interferon Genes (cGAS-STING) pathway. 44 The main effects include the inhibition of enzyme and cytokine release,45–47 receptor recycling, plasma membrane repair, cell signaling, apoptosis,48–50 autophagy,39,51 antigen presentation, 52 T-cell polarization,53–56 inhibition of the natural killer (NK) cells,57,58 energy metabolism, 40 and increases photoprotection against ultraviolet (UV)-A and B.59–65
Mechanisms of action of hydroxychloroquine.
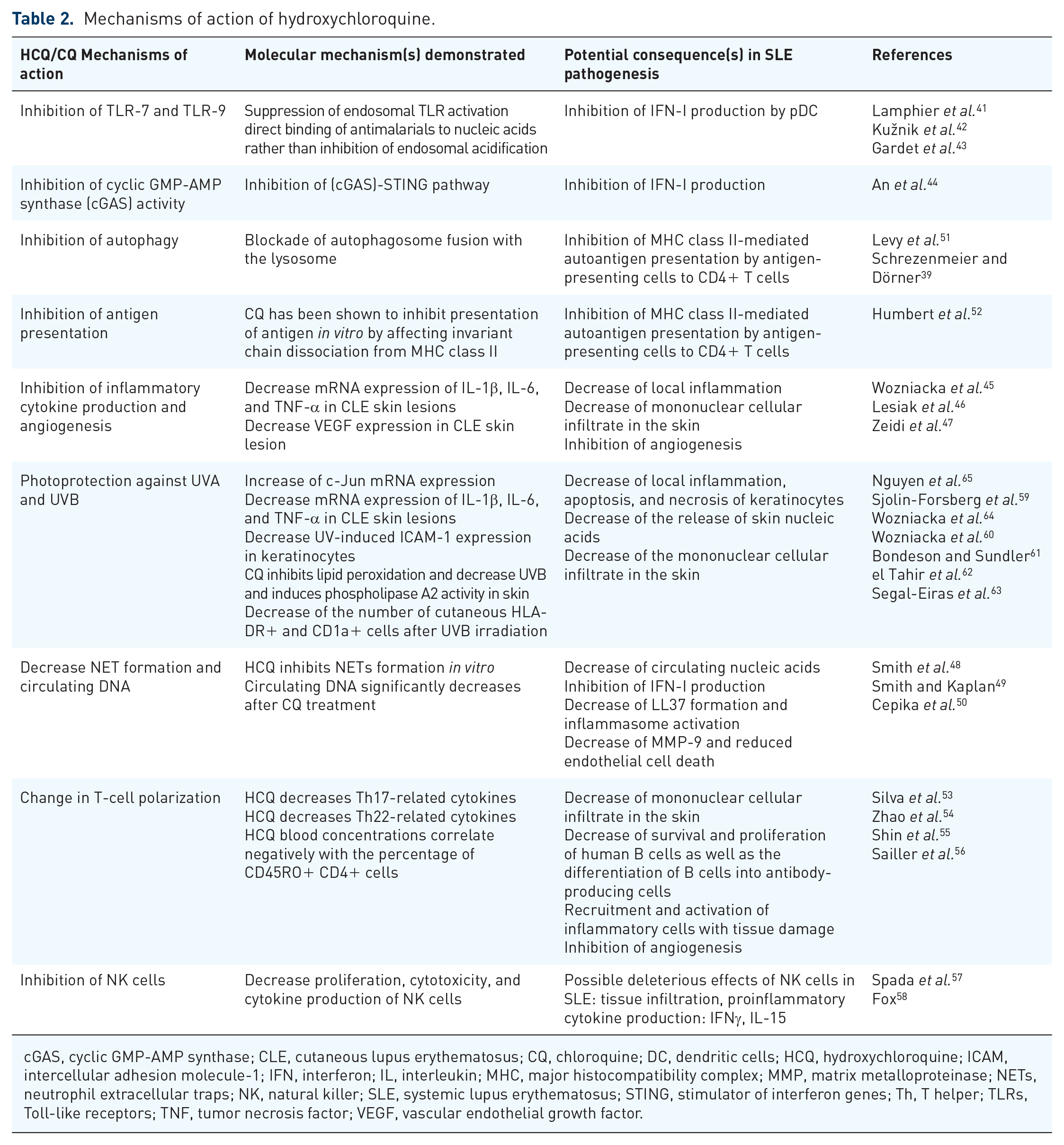
cGAS, cyclic GMP-AMP synthase; CLE, cutaneous lupus erythematosus; CQ, chloroquine; DC, dendritic cells; HCQ, hydroxychloroquine; ICAM, intercellular adhesion molecule-1; IFN, interferon; IL, interleukin; MHC, major histocompatibility complex; MMP, matrix metalloproteinase; NETs, neutrophil extracellular traps; NK, natural killer; SLE, systemic lupus erythematosus; STING, stimulator of interferon genes; Th, T helper; TLRs, Toll-like receptors; TNF, tumor necrosis factor; VEGF, vascular endothelial growth factor.

Hydroxychloroquine’s mechanisms of action.
Efficacy in systemic lupus erythematosus
Systematic search of randomized controlled trials regarding hydroxychloroquine in systemic lupus erythematosus
A systematic search for randomized controlled trials (RCTs) regarding HCQ treatment in SLE was performed using the medical subject headings (MeSH) terms ‘Hydroxychloroquine’ AND ‘Lupus Erythematosus, Systemic’ AND ‘Clinical Trials, Randomized’. The search was performed on Excerpta Medica/EMBASE, MEDLINE via PubMed, Cochrane Library, and Thomson Reuters’ Web of Science Core Collection using the same combination of relevant keywords (see Supplemental File 1). The four databases were systematically searched from inception to 1 February 2021, without any language, geographic, or type of article restrictions. The references and citations of the articles identified were also screened.
Reports not referring to HCQ or CQ use in SLE, not involving human subjects, not including adult cases, and presenting other types of studies than RCTs were excluded. A total of eight RCTs were identified in the initial search with one more identified after the references and citations screen (see Supplemental Figure 1–Flowchart Diagram, Supplemental Table 1). For each RCT included, the following information was extracted: study design, drug posology, time of follow-up, study’s endpoints, proven efficacy, and side effects noted (as presented in Supplemental Table 2a, 2b).
To the best of our knowledge, the first RCT involving antimalarial therapy in SLE was published in 1991 by Canadian Hydroxychloroquine Study Group 66 and reported a 2.5-fold increase in the risk of mild flare after HCQ withdrawal in the placebo group. 66 In 1998, Tsakonas et al. 67 presented an extension phase in 1991 and evaluated the risk of major flare after HCQ withdrawal. 67 The endpoint considered, namely flare, subtype of flare, and hospitalization, were all improved under long-term HCQ therapy; however, the results did not reach statistical significance most probably due to the small sample size. 67
Other RCTs have also demonstrated improvement of arthralgia 14 even if without a significant impact over arthritis, 68 prevention of SLE flares and reduction of the corticosteroids dose, 14 improvement of lipid metabolism 69 with decrease in total cholesterol and triglycerides, while increase in HDL-cholesterol, 70 and a safety profile of administration during pregnancy. 71 Also, the PLUS (Plaquenil LUpus Systemic) failed to demonstrate that adjusted HCQ dosing schedules targeting [HCQ] ⩾1000 ng/ml might reduce the occurrence of SLE flares. 72 Most recently, Zanetti et al. 73 tested the efficacy of lower HCQ doses (2–3 mg/kg/day) 30 and found similar 6- and 12-month flare rates between groups. 73
For cutaneous lupus erythematosus (CLE), the first RCT by Kraak et al. 74 in 1965 tested HCQ up to a maximum posology of 1200 mg daily. Furthermore, the efficacy of antimalarials has been tested in RCTs against placebo, 75 acitrecin, 76 or clofazimine 77 in RCTs showing proven efficacy in RCT with better safety profile than clofazimine or acitrecin.
Observational data for hydroxychloroquine in systemic lupus erythematosus
Currently published RCTs do not cover the whole spectrum of SLE features. Many of the data regarding HCQ benefits are from prospective SLE cohorts, such as the Hopkins Lupus Cohort,78–83 LUMINA (Lupus in Minorities: Nature versus Nurture) Cohort,84–89 Toronto Lupus Cohort,90,91 or GLADEL (multinational Latin American lupus) Cohort92–94 (see Table 3; Supplemental Table 3).
Research for antimalarials in systemic lupus erythematosus.

CC, case-control; CLE, cutaneous lupus erythematosus; CS, cross-sectional; DLE, discoid lupus erythematosus; DS, descriptive studies; GLADEL, Grupo Latino Americano de Estudio del Lupus; HCQ, hydroxychloroquine; LAC, lupus anticoagulant; LN, lupus nephritis; LUMINA, Lupus in Minorities: Nature vs Nurture; PC, prospective cohort; RA, retrospective analysis; RC, retrospective cohort; RCT, randomized controlled trial; SCLE, subacute cutaneous lupus erythematosus; SLICC, Systemic Lupus International Collaborating Clinics.
Antimalarials: chloroquine diphosphate (CDP) or hydroxychloroquine sulfate (HCQ).The most significant HCQ effect is the control of SLE disease activity itself, which implies amelioration of active clinical involvements, decrease in serum markers, decrease in activity scores, prevention of disease flares, and sustained remission on long-term use.
Therefore, decrease in disease activity,84,85,95–101 prevention of disease flares,78,100,103–105 and improvement of proinflammatory cytokine profiles85,95,97,104,180,181 have been highlighted with HCQ.
Moreover, delay of the immune clinical spectrum to overt SLE was described in antinuclear antibodies (ANA)-positive patients.94,179 A recent study showed that HCQ might suppress early mediators like the B cell activating factor (BAFF) and interferon (IFN), lowering the IFN-γ-induced protein 10 (IP-10) levels in incomplete or new-onset SLE, supporting the hypothesis that HCQ could influence disease progression. 182
In observational studies, HCQ has been shown beneficial for cutaneous lupus,95,102,106–112 musculoskeletal involvement, 99 and various other key manifestations of SLE. The management of lupus nephritis (LN) remains suboptimal 183 and HCQ is adjuvant therapy to the immunosuppressive regimens in obtaining remission.79,84,115–117
HCQ decreases disease activity and prevents SLE flare during pregnancy,80,118,119,122 and furthermore, there are reports sustaining a possible protective role for preeclampsia,120–123 fetal growth restriction, and prematurity. 98 Current data regarding HCQ efficacy during pregnancy are conclusive, however for other outcomes the results are contradictory. Thus, there are reports that did not found the impact of HCQ on pregnancy loss, preterm delivery or intrauterine growth retardation, 119 or upon miscarriage, stillbirth, pregnancy loss, or congenital abnormality rates. 80
For neonatal lupus, one retrospective study that analyzed data of a historical cohort counting more than 200 pregnancies in SLE patients with positive anti-Ro/SS-A antibodies found HCQ benefits over recurrence and outcome of the neonatal lupus. 184 In another research, HCQ was not identified as independent protective factor for neonatal lupus after adjusting for confounders like age, race, antibodies status, corticosteroids, and prior cardiac-neonatal lupus risk, even if the neonatal lupus cases were less frequent in pregnancies treated by HCQ (14% versus 37%). 185
Despite potential benefits of HCQ during pregnancy, adherence seems to be low. A population-based registry identified 376 pregnancies in which discontinuation of antimalarials occurred in 16.7% of cases in the year prior to pregnancy, 29.8% in the first trimester, 9.7% in the second, and 26.0% in the third. 186
Importantly, HCQ passes the placenta and has fetal serum concentrations equal to those measured in the maternal blood. However, HCQ use during pregnancy80,119,120,123,187–189 and breastfeeding is considered safe.5,190 During lactation, HCQ passes in the maternal milk, but with lower concentrations than in maternal blood, estimated to be 0.2 mg/kg/day. 5
There are reports of CQ overdose in children and, by parallel, cautions are related to HCQ. Antimalarials might be toxic in children in relatively small doses and patients should be counseled to keep these drugs out of children. 5
SLE disease itself is a risk factor for thrombosis. Also, about 20% of patients with SLE have antiphospholipid syndrome (APS). 191 Antimalarials might reduce the antiphospholipid antibodies titers 124 and the risk of thrombosis,116,125–135 but not all published studies reported a protective effect over thrombosis.192–194
HCQ has also some metabolic effects by lowering fasting glucose, 136 yielding protection against diabetes, 137 and improvement of the lipids profile in most81,90,138–147 but not all195,196 studies. However, the efficacy of HCQ upon atherosclerosis is more controversial.151,152,197,198
It is to remember that smoking might inhibit HCQ effects7,109,110,112 and determine a twofold lower response of cutaneous involvement under HCQ; 199 counseling for smoking cessation is therefore important. Possible anti-neoplastic properties of HCQ have been poorly assessed in SLE. 168
HCQ might inhibit the conversion of 25-(OH)-vitamin D to 1,25-(OH)2-vitamin D. 200 However, data regarding the impact of HCQ on bone metabolism in SLE remain controversial.86,162,163,201,202 Many data suggest that HCQ has a protective role against infections92,116,154–160 and severe events included92,154–156 in SLE.
Corticosteroids are widely prescribed, but also important determinants of cardiovascular, gastrointestinal, and metabolic comorbidities as well as of accrual damage and impaired quality of life in SLE. Thus, another important role for HCQ in SLE is that of corticosteroid-sparing agent.80,84,103 However, as for other outcomes, there are also studies with negative results. 102
SLE is a severe disease with survival rates at 5 years of only 50% in early studies, which now exceed 90%. 203 While mortality in early stages is usually related to severe organ involvement and SLE disease activity itself, in late, long-standing SLE, accrual damage, and cardiovascular risk are the main determinants. In spite of some contrary results, 204 many studies reported HCQ protective effects for accrual damage3,83,84,87–89,164–167 and HCQ has also been associated with shorter SLE-related hospitalization length.159,169,170 And last, but not least, HCQ is one of the few treatments that has been shown to improve survival rates in SLE.87,91,93,116,127,171–178
Therefore, based on its wide spectrum of effects, HCQ should probably be considered a possible confounder in all research involving patients with SLE.
Systematic reviews and meta-analyses on hydroxychloroquine use in systemic lupus erythematosus
The first systematic review regarding HCQ in SLE included a total of 95 studies published between 1982 and 2007. 13 All studies which considered disease activity as the main outcome (11 articles) found positive results, with more than 50% reduction in disease activity in most reports and a decrease in corticosteroid needs in three studies; 13 however, the risk of severe SLE flare was reduced only with borderline significance. 13 Also, the HCQ benefits as adjuvant therapy for LN was also confirmed. 13 The potential benefits upon accrual damage and survival were reported in a limited number of studies. 13 This systematic review was continued by another one using a similar methodology for the 2007–2012 period. 3 The authors reported further evidence thrombosis prevention, increased survival, control of disease activity, lipid profile improvement, and prevention of damage accrual 3 (see Supplemental Table 4).
Side effects of hydroxychloroquine.
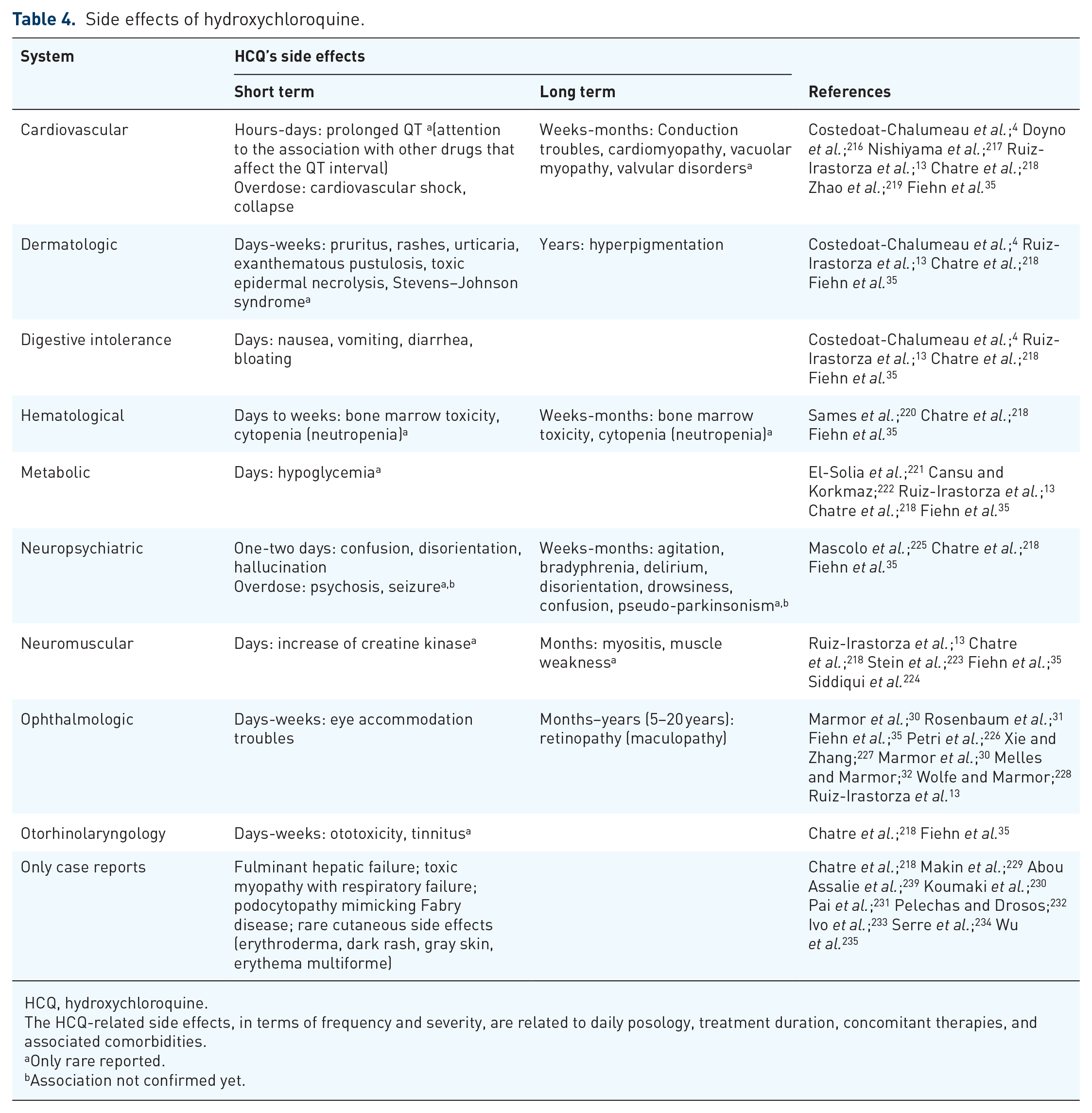
HCQ, hydroxychloroquine.
The HCQ-related side effects, in terms of frequency and severity, are related to daily posology, treatment duration, concomitant therapies, and associated comorbidities.
Only rare reported.
Association not confirmed yet.
The protective effect of HCQ against infections was further confirmed in two systematic reviews and meta-analysis.153,161 Also, two meta-analyses reported improvement of the lipid profile under HCQ in SLE.149,150 For cutaneous involvement, Fairley et al. 114 reported in one systematic review only moderate HCQ efficacy.
A 2018 meta-analysis of observational data failed to identify any significant beneficial effect of HCQ over fetal growth restriction and prematurity. However, the authors mentioned that these results should be regarded with caution due to lack of RCTs, high heterogeneity among reported data, and of numerous missing data like those on the antiphospholipid antibodies status. 205
Overview of guidelines
We reviewed here systematically the European League against Rheumatism (EULAR) recommendations referring to the use of HCQ. We identified all EULAR guidelines (www.eular.org) for the last 5 years and searched for HCQ-related paragraphs using the terms ‘Hydroxychloroquine’ and the respective abbreviation ‘HCQ’. All paragraphs found were extracted (see Supplemental Table 5) and data were further analyzed and summarized (see Supplemental Figure 2).
From the total 30 EULAR management guidelines published since 2016, 10 referring to HCQ were identified, and main indications were noted (see Supplemental Table 6). Recommendations addressing specifically to HCQ were found in seven guidelines8,34,206–210 while in others, HCQ was included as part of Disease Modifying AntiRheumatic Drugs (DMARDs).211–213 The EULAR Guidelines recommendations referring mainly to SLE and related conditions are summarized in Figure 2.
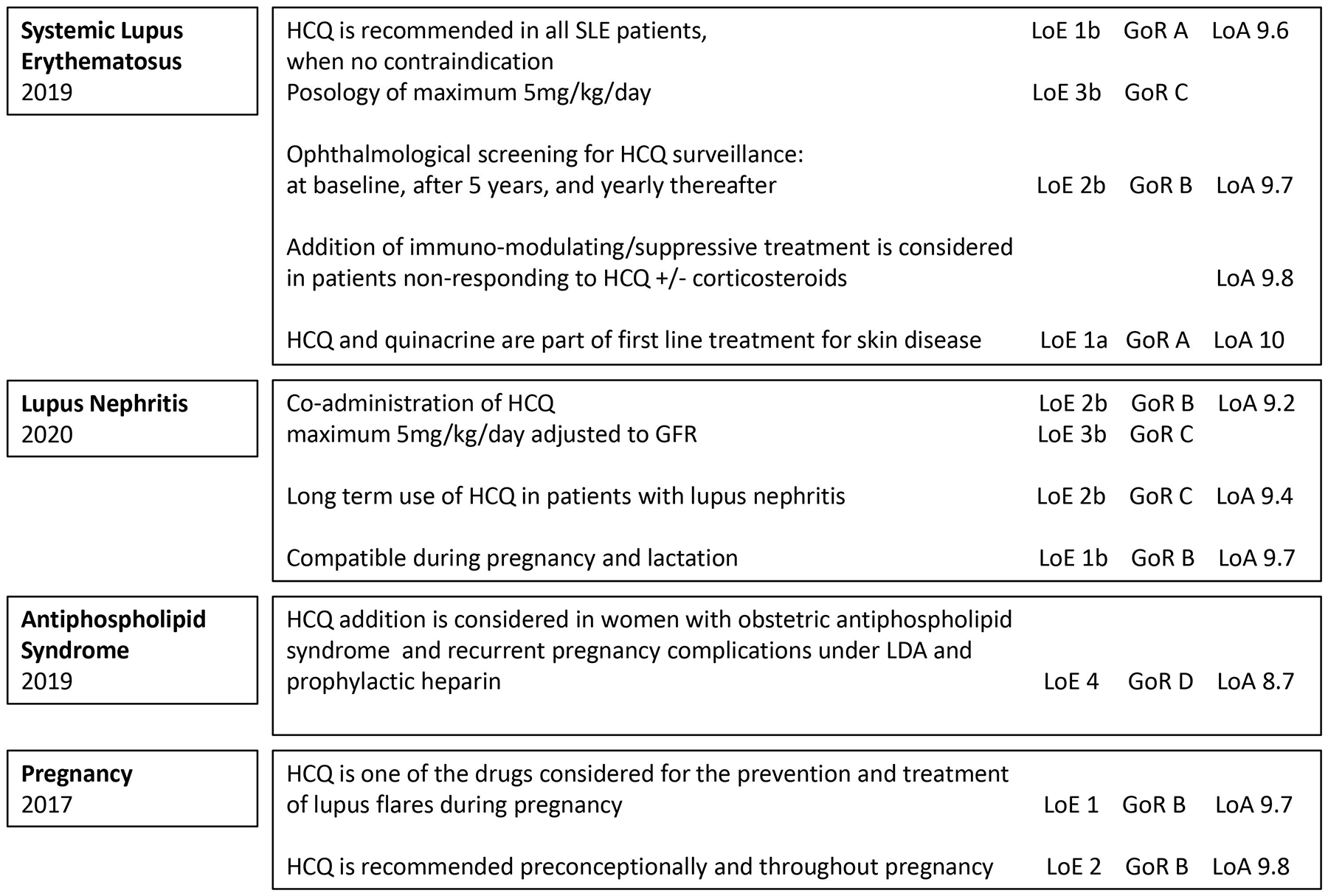
Recommendations for hydroxychloroquine (HCQ) use according to the European League against Rheumatism (EULAR) guidelines.
Tunnincliffe et al. 214 and Tamirou et al. 215 reviewed SLE recommendations published up to 2014 and between 2004 and 2017, respectively, and identified not least than 14 and 23, respectively, original clinical guidelines or original statements with focus on SLE.
The 2020 American College of Rheumatology (ACR) Guideline for the Management of Reproductive Health in Rheumatic and Musculoskeletal Diseases 190 advise for HCQ use during pregnancy and breastfeeding, in cases with positive anti-Ro/SS-A and anti-La/SS-B antibodies as well as additional or alternative therapy in SLE women with refractory obstetric APS. HCQ continuation is strongly recommended in men who are planning to father a pregnancy. 190 The 2012 ACR Guidelines for Screening, Treatment, and Management of Lupus Nephritis specifies that all SLE patients with nephritis should be treated with HCQ as background therapy. 9
The 2018 British Society for Rheumatology guideline for the management of SLE in adults 10 identified 45 studies to sustain the recommendation of antimalarial use (<6.5 mg/kg/day) for mild disease, prevention of flare in all patients, prevention of damage, and as steroid-sparing agent (overall SIGN level of evidence 1+++ and grade A of recommendation). 10
Finally, the Latin American Group for the Study of Lupus (GLADEL, Grupo Latino Americano de Estudio del Lupus)–Pan-American League of Associations of Rheumatology (PANLAR) stated also that antimalarials should be used in all SLE patients with exception of those who refuse or who have absolute contraindications, as first line for musculoskeletal or cutaneous involvement as well as associated with immunosuppressive treatments for other SLE organ involvements. 11
Hydroxychloroquine safety profile
A wide range of side effects such as cardiovascular, dermatological, digestive, hematological, metabolic, ophthalmologic, as well as other rare side effects were reported to be associated with HCQ use.4,13,30,31,32,35,216–238 The main side effects of HCQ are summarized in Table 4.
Reviewing the antimalarials’ safety profile in SLE, Ruiz-Irastorza et al. 13 noted low prevalence of antimalarials’ toxicity, mainly mild gastrointestinal and cutaneous side effects. These were significantly more frequent under CQ when compared with HCQ, results parallel by higher discontinuation rates for CQ. Overall, the HCQ global safety was rated as high. 13 Eljaaly et al. 236 published recently a meta-analysis for the HCQ safety when administrated for different pathologies (chronic urticaria, RA, SLE, osteoarthritis, IgA nephropathy, asymptomatic HIV infection, Alzheimer disease, cutaneous lupus) in daily doses of 200–400 mg and presented also encouraging results. Besides significant more frequent occurrence of skin pigmentation under HCQ, no other side effect reached a significant difference (rash, gastrointestinal complaints, headache, fatigue, visual troubles) and also no cardiac toxicity was reported. 236
Thus, for long-term HCQ use, medium uptake duration of 32 months, 35 the skin hyperpigmentation is not rarely reported and might be favored by factors like ecchymosis, bruising, platelet antiaggregant, and oral anticoagulants. Beside hyperpigmentation, all other HCQ-related side effects are only rarely encountered.
On short-term use, the digestive intolerance is the most frequently encountered side effect, with occurrence possible since first HCQ administration.237,238
A wide range of mild neuropsychiatric manifestations, but also psychosis, was reported in relation to HCQ use, especially in elderly. However, this relation remains controversial as other concomitant factors like concomitant drugs, alcohol intake, use of glucocorticoids, or background disease itself could originate the neuropsychiatric manifestations occurrence in patients with SLE under HCQ. 225
Retinopathy occurrence remains the most discussed and studied HCQ’s side effect in SLE. The main risk factors for HCQ-related retinopathy are the treatment duration, daily and cumulative dose, chronic kidney disease, as well as pre-existent retinal disease. 34 Ophthalmologic screening is mandatory, yearly from baseline if there are known risk factors or at baseline, after 5 years on HCQ, and yearly therefore in patients without retinopathy risk factors.8,30,31,34 The current 2020 Joint Statement on HCQ 31 reinforced the old recommendations8,30,34,32 of the need of sensitive testing modalities such as optical coherence tomography (OCT) and automated visual fields that could detect early toxicity. 31 When available, quinacrine (mepacrine) might be considered as an alternative in SLE patients with HCQ-related ocular or cutaneous side effects.
As the eye side effects are dose-related, not only the duration of use but also the blood levels are predictors of retinopathy development with a statistical association in patients with [HCQ] blood levels >1200 ng/ml.226,227 However, association between HCQ blood concentration and retinopathy has not been confirmed in another study. 240 For non-rheumatic diseases, doses of up to 1000 mg daily (up to 20 mg/kg daily) showed eye toxicity within 2 years in 25–40% of the patients exposed, 30 while for the doses up to 5 mg/kg of real body weight, the risk of retinopathy within 10 years was 2%. 32 For lifetime HCQ users, definite or probable toxicity was documented in only 0.65% even if 6.5% patients discontinued therapy because of eye-related side effects. 228 One longitudinal study showed ophthalmological alterations confirmed by ophthalmological examination in 5.5% of cases. 241
When compared with HCQ, the risk of retinopathy related to CQ seems to be much higher, hence CQ is not recommended as the first-line antimalarial for the SLE treatment. One systematic review including four studies for CQ versus six studies for HCQ found definite retinal toxicity in 2.5% versus 0.1% and probable retinopathy in 2.6% versus 0.3% patients. 13 A recent report from the Hopkins cohort showed a higher overall frequency of retinopathy of 4.3%, but the risk increased significantly after 15 years of HCQ use, 226 namely 1% in the first 5 years, 1.8% for 6–10 years, 3.3% for 11–15 years, and 11.5% for 16–20 years. 226
For antimalarials cardiac toxicity, the results of 86 articles were systematically reviewed and a total of 127 patients (65.4% female) were identified, of which about 60% had taken CQ, while the rest HCQ. 218 The most frequent cardiac side effects reported were conduction disorders (85%), followed by cardiac hypertrophy (22%), hypokinesia (9.4%), cardiac failure (26.8%), pulmonary arterial hypertension (3.9%), and valvular dysfunction (7.1%). Less than half of the patients (44.9%) recovered normal heart function after the antimalarial drug withdrawal. 218
Disparate cases of HCQ-related neuromyopathy, particularly manifested as insidious onset of proximal myopathy that may be later associated with peripheral neuropathy and cardiac myotoxicity, are reported. The frequency of HCQ-related myopathies is not known, but is probably extremely rare. 35 Early recognition is important as the recovery after the drug withdrawal might be incomplete. 223
Different case reports presented rare and very rare sides effects attributable to HCQ in the absence of other identifiable causes, like early fulminant hepatic failure, 229 toxic myopathy with respiratory failure, 224 and rare cutaneous lesions.230–235,239
Hydroxychloroquine blood level monitoring and withdrawal
Even if the HCQ role in SLE is acknowledged, less than half of the patients are taking HCQ as prescribed. 242 Measurement of HCQ in whole blood was proposed to monitor both response and adherence to treatment, but an appropriate cut-off for defining efficient HCQ’s blood levels remains under debate. For CLE, one prospective multicenter study found significantly higher median blood [HCQ] levels in patients with complete remission (910 ng/ml in remission versus 692 ng/ml when partial remission and 569 ng/ml in treatment failure, p = 0.007). 107 In a prospective study, improvement of cutaneous lesions was observed when [HCQ] blood levels higher than 750 ng/ml were reached. 113 Also, one study defined subtherapeutic [HCQ] levels, associated with trend of more disease flares, as less than 500 ng/ml. 243 A recent report showed that low [HCQ] blood levels are associated with thrombotic events (720 ng/ml versus 935 ng/ml; p = 0.025). 135
On one hand, a decrease in the flare rate was not observed when [HCQ] level was maintained over 1000 ng/ml. 72 On the other hand, decrease to 2–3 mg/kg/day did not modify serum [HCQ] levels significantly at 3 and 6 months, but only at 12 months. 73
One of the main reasons for using [HCQ] blood levels in daily practice is the great interindividual variability, of which determinants are not completely characterized. 5 [HCQ] levels were found to be related to its major metabolite, N-desethylhydroxychloroquine (DHCQ), to HCQ weight-adjusted oral dose and also to the time since last dose taken.243,244
Analyzing a longitudinal cohort, Mok et al. 243 found that the majority of SLE patients screened had mainly [HCQ] subtherapeutic levels: <10 ng/ml (defined as total non-adherence) in 11%, 10–500 ng/ml (subtherapeutic levels) in 77%, and >500 ng/ml (therapeutic levels) in only 12% patients. Levels correlated with the dose prescribed 243 and, importantly, higher [HCQ] levels were associated with less SLE flare occurrence over time. 243
Monitoring HCQ levels might allow identification of early nonadherence 243 and improve nonadherence. 72 HCQ levels measurement might help in counseling before the treatment change in regard to lack of adherence versus lack of treatment efficacy. 5
Finally, considering the HCQ’s side effects related to long-term use, one important question is how to identify the appropriate moment for stopping the treatment. The first RCT designed for HCQ66,67 showed efficacy of long-term HCQ use in sustaining remission. In this RCT, the average HCQ total treatment duration before withdrawal was about 3 years.66,67 A more recent retrospective study showed that HCQ discontinuation in patients older than 55 years with quiescent SLE and more than 5 years treatment, due to retinal toxicity, patient’s preference, cardiac toxicity, or other suspected adverse effects, did not result in significant increase in flare occurrence. 245 Finally, a recent survey across large international sample of physicians has shown that in case of sustained remission, 49.7% maintained the same dose indefinitely, 48.3% reduced the dose, while only 2.0% discontinued antimalarials. 37
Conclusion
In summary, HCQ is indicated in all patients with SLE in the absence of any contraindications or side effects, with high grade evidence in case of LN, cutaneous involvement, or during pregnancy and breastfeeding. However, there is a relatively small effect size for the prevention of severe flares in SLE. Monitoring HCQ blood levels might help to overcome adherence issues, which are quite common in SLE and adjust the daily dosage based on individual pharmacokinetic variability. Still, there is a need for additional research focused on defining the optimal conditions for HCQ withdrawal.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X211073001 – Supplemental material for Hydroxychloroquine in systemic lupus erythematosus: overview of current knowledge
Supplemental material, sj-docx-1-tab-10.1177_1759720X211073001 for Hydroxychloroquine in systemic lupus erythematosus: overview of current knowledge by Alina Dima, Ciprian Jurcut, François Chasset, Renaud Felten and Laurent Arnaud in Therapeutic Advances in Musculoskeletal Disease
Footnotes
Author contributions
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
