Abstract
Objectives:
To compare healthcare utilization and medical costs between patients with seronegative (SN) and seropositive (SP) rheumatoid arthritis (RA).
Methods:
We conducted a nationwide population study using the Korean health insurance claims database in 2016. We divided patients with RA into SN and SP groups and compared healthcare utilization including medications, medical utilization, and direct medical costs for 1 year between the groups in a cross-sectional analysis. Differences in costs between patients with SPRA and SNRA were assessed using the quantile regression model. We performed longitudinal analysis using data from 2012 and 2016 to examine changes over time.
Results:
A total of 103,815 SPRA and 75,809 SNRA patients were included in the analyses. The SPRA group used significantly more methotrexate (73.2% versus 30.3%) and biologic agents (7.9% versus 2.9%) than the SNRA group. The number of RA-related outpatient visits [6.0 ± 3.7 versus 4.4 ± 4.0 times/year, standardized difference (SD) = 0.41] and annual medical costs per patient ($1027 versus $450/year, SD = 0.25) were higher in the SPRA group than the SNRA group. Quantile regression results indicated that the incremental cost of seropositivity on total medical costs of RA patients gradually increased as medical costs approached the upper quantile. The annual direct medical costs for each patient between 2012 and 2016 increased in both groups: by 25.1% in the SPRA group and 37.6% in the SNRA group.
Conclusion:
Annual RA-related direct medical costs and RA-related healthcare utilization per patient are higher in patients with SPRA than those with SNRA.
Introduction
Patients with rheumatoid arthritis (RA) can be divided into two major subsets characterized by the presence or absence of antibodies to anti-citrullinated protein antibodies (ACPA) or rheumatoid factor (RF), whereby the presence of either or both of these types of autoantibody defines the seropositive subset of disease. 1 This subdivision of patients with seropositive RA (SPRA) has greatly enhanced pathogenic studies and has been a somewhat homogenous subgroup of RA with certain genetic and environmental risk factors. 2 Associations of serological status with clinical phenotype, treatment response, and disease outcome in patients with RA have been suggested. 3 Previous observational studies reported that patients with RF or ACPA were more likely to have joint erosion, 4 and were more likely to present with radiographic progression or lower bone mass.5–9 These serologic markers are regarded as poor prognostic markers of RA, and are used as evidence to justify intensive treatment in SPRA. 8
However, other studies have reported that seronegative RA (SNRA) is associated with more severe inflammatory activity than SPRA when assessed clinically and by ultrasound.10,11 These higher levels of inflammation of SNRA may reflect the high number of involved joints required for SNRA to fulfil the 2010 ACR/EULAR classification criteria for RA. 10 Furthermore, SNRA patients often experience delays in diagnosis, as well as delays in the initiation of disease modifying anti-rheumatic drugs (DMARDs), and are less likely to attain remission. 12 Therefore, SNRA cannot be considered simply a mild form of disease. However, differences in healthcare utilization and medical costs between SPRA and SNRA patients remain largely unexplored.
In this study, we compare healthcare utilization between patients with SNRA and SPRA using data from the Korean nationwide health insurance claims database.
Materials and methods
Data source and study population
Data source
We used the Korean National Health Insurance Service (NHIS) database, which contains individual beneficiary information, in addition to healthcare service information such as diagnosis, procedures, prescriptions, and tests. 13 We used NHIS data collected during 2016 for a cross-sectional study to analyze healthcare utilization and medical costs. To estimate changes in population characteristics over time, we performed a longitudinal study using data for both 2012 and 2016.
Study population
Patients with RA were identified by searching for the diagnostic codes for RA (M05 and M06) and prescriptions for any DMARDs based on previous validated operational definitions of RA in the NHIS claims database with positive predictive value (PPV) of 92.3%. 14 All patients with RA were identified and divided into two groups. SNRA was described as the absence of the ICD-10 diagnostic code of M05, while seropositivity was recognized by having more than one inpatient or outpatient claim with an ICD-10 diagnostic code of M05. We defined the index date as the earliest diagnosis of RA. The first application of the ICD-10 diagnostic code of M05 was defined the index date in RA patients who were charged for both M05 and M06.
Data collection
Demographic and clinical characteristics of patients with RA
Demographic and clinical characteristics of patients with RA were collected and analyzed. Payer type was defined as type of insurance on the index date. The types of institutions and departments that patients visited most often for RA management over a period of 1 year were examined. Comorbidities of RA patients were measured using Charlson comorbidity index (CCI) scores for a period of 1 year. In addition, osteoporosis, interstitial lung disease (ILD), and fibromyalgia were also determined based on ICD 10 codes.
Cross -sectional comparison of healthcare utilization and medical costs between SPRA and SNRA
Using data from 2016, medication use was characterized using information about the domestic approval status of medications including DMARDs, non-steroidal anti-inflammatory drugs (NSAIDs), and glucocorticoids. Biologic DMARDs (bDMARDs) including TNF inhibitors (etanercept, adalimumab, infliximab, golimumab) and non-TNF inhibitors (abatacept, tocilizumab, rituximab) are approved for management of RA in Korea. The pattern of medications was presented the percentage of patients treated with each medication for 1 year.
Medical utilization for a period of 1 year was examined using the number of outpatient department (OPD) visits, the number of hospitalizations, and average length of hospitalization for each individual. Medical utilization charged using diagnostic codes for RA was defined as RA-related medical utilization.
Annual direct medical costs per patient were also calculated by dividing the annual total direct medical costs by the number of patients with RA for each year. Medical costs were estimated in both inpatient and outpatient settings. Expenditures for RA based on claims with RA diagnostic codes were used to calculate RA-related costs. All values were converted from Korean won to United States (US) dollars (USD) at an exchange rate of 1 USD equalling 1000 Korean won. The impact of seropositivity on direct medical costs was assessed after adjusting for covariates.
Longitudinal changes of healthcare utilization and medical costs in SPRA and SNRA groups
Using data from 2012 and 2016, we calculated healthcare utilization and medical costs per patient for each year and analyzed changes in utilization and costs over time for patients with SNRA and SPRA.
Statistical analysis
We used standardized differences (SD) to assess the balance of baseline covariates between the two groups; SD >0.1 suggests a significant imbalance between the two groups. 15 We give medical utilization and direct medical costs as means (±standard deviations) and medians (quantiles).
We used quantile regression models to evaluate the impact of seropositivity on direct medical costs while adjusting for covariates because our data included some very high cost patients, leading to a skewed distribution. The percentiles selected for modelling were the 10th, 25th, 50th (median), 75th, and 90th. Covariates in the models included age, sex, payer type, type of institution, type of department, CCI score, and medication, all of which influence costs. All statistical analyses were performed using SAS 9.4 (SAS Institute, Cary, NC, USA).
Ethical considerations
This study was submitted to our institutional review board (IRB) and exempted from IRB review because we used existing, publicly available data and the data could not be identified directly or through identifiers linked to the subjects (IRB file No. HYUH 2020-05-005). The requirement for informed consent was waived because we utilized a de-identified database that is open to the public.
Results
Demographic and clinical characteristics of patients with RA
In 2016, we identified a total of 103,815 of patients with SPRA and 75,809 of patients with SNRA. Demographic and clinical characteristics of patients with SPRA and SNRA are presented in Table 1. Mean age was higher in the SPRA group (58.6 ± 12.8 years) compared with the SNRA group (55.2 ± 0.2 years). The percentage of females was higher in the SPRA group than the SNRA group (80.4% versus 72.4%). CCI scores were similar between groups (2.9 ± 2.0 in SPRA and 3.0 ± 1.9 in SNRA). Comorbidity with ILD was more common in the SPRA group than in the SNRA group (2.5% versus 1.3%).
Demographic and clinical characteristics of patients with RA. Values are presented as the number and percentage, or mean with standard deviation.

Frequently visited hospitals are noted.
Other institutions included traditional medicine.
AIDS, acquired immune deficiency syndrome; CCI score, Charlson comorbidity index score; RA, rheumatoid arthritis.
Differences in healthcare utilization between SPRA and SNRA patients
Patterns of RA treatment
Medications for patients with RA are listed in Table 2. Methotrexate was used more in the SPRA group than the SNRA group (73.2% versus 30.3%, SD = 0.95), while hydroxychloroquine and sulfasalazine were used more in the SNRA group than the SPRA group. Patients with SPRA tended to take more bDMARDs (7.9% versus 2.9%, SD = 0.22), along with more frequent use of corticosteroids through oral administration (81.9% versus 73.6%, SD = 0.20) or intra-articular injections (27.3% versus 19.2%, SD = 0.19) compared with patients with SNRA.
Comparison of treatment patterns between patients with seropositive and seronegative RA. Values are presented as the number and percentage, or mean with standard deviation.
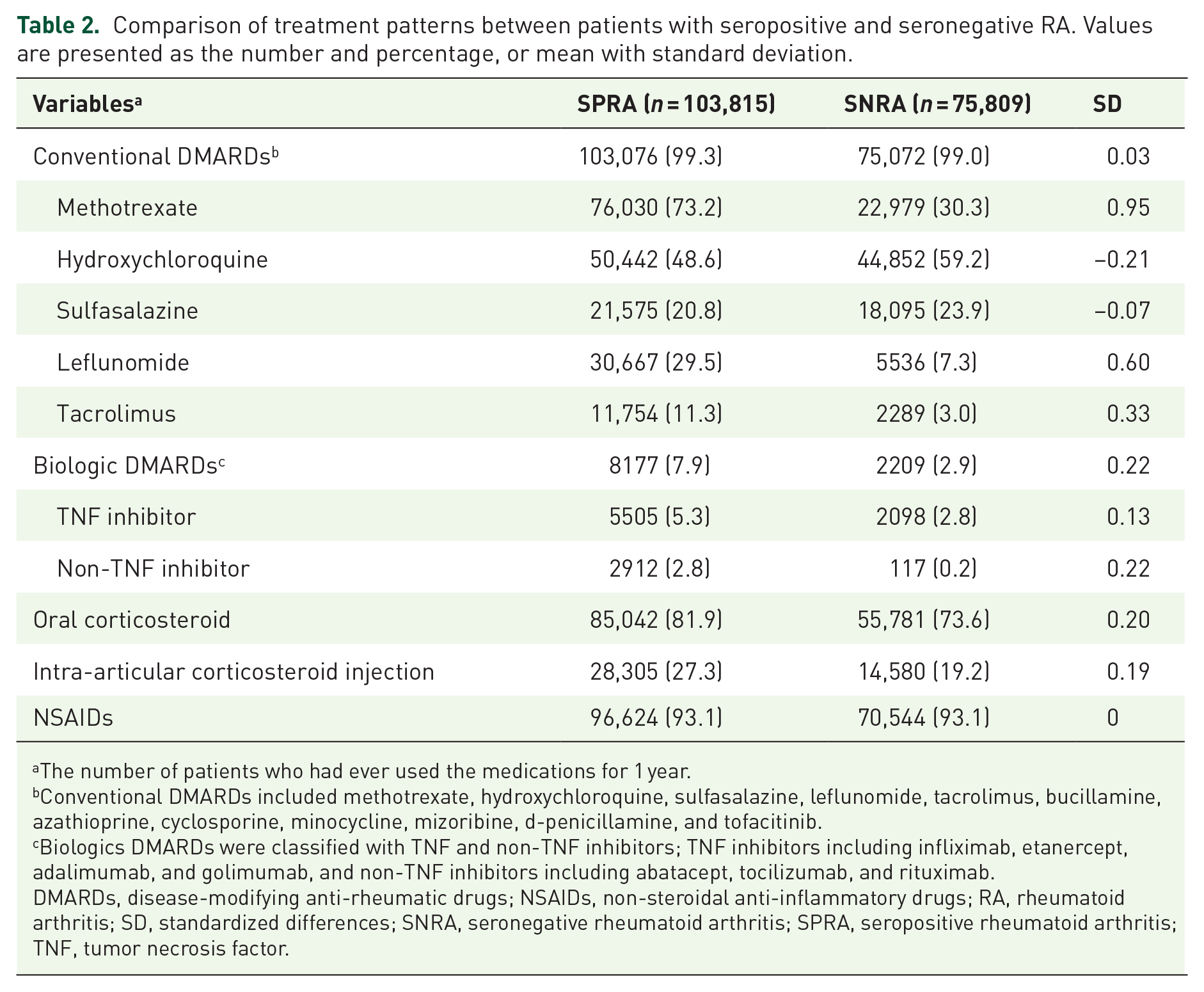
The number of patients who had ever used the medications for 1 year.
Conventional DMARDs included methotrexate, hydroxychloroquine, sulfasalazine, leflunomide, tacrolimus, bucillamine, azathioprine, cyclosporine, minocycline, mizoribine,
Biologics DMARDs were classified with TNF and non-TNF inhibitors; TNF inhibitors including infliximab, etanercept, adalimumab, and golimumab, and non-TNF inhibitors including abatacept, tocilizumab, and rituximab.
DMARDs, disease-modifying anti-rheumatic drugs; NSAIDs, non-steroidal anti-inflammatory drugs; RA, rheumatoid arthritis; SD, standardized differences; SNRA, seronegative rheumatoid arthritis; SPRA, seropositive rheumatoid arthritis; TNF, tumor necrosis factor.
Overall healthcare utilization and surgery
The mean number of RA-related OPD visits per patient with SPRA was higher than that of patients with SNRA (6.0 versus 4.4 times per year, SD = 0.41), while overall OPD visits were not different between two groups.
With regard to hospitalization, the numbers of all cause hospitalizations per year (2.4 ± 2.5 versus 2.3 ± 2.5, SD = 0.04) and RA-related hospitalizations per year (1.5 ± 1.1 versus 1.4 ± 1.2, SD = 0.06) were numerically higher in patients with SPRA than with SNRA, although the difference was not significant. The average length of hospital stay was not different between the two groups (Table 2).
In terms of orthopaedic surgery, total hip replacement (0.08% versus 0.03%, SD = 0.02) and other joint replacements (shoulders, elbows, wrists, and ankle) (0.03% versus 0.01%, SD = 0.01) were more frequent numerically in patients with SPRA than those with SNRA, although this difference was not significant. The frequencies of total knee replacement (0.21% in SNRA versus 0.18% in SPRA, SD = −0.01) and soft tissue surgery (0.22% in SNRA versus 0.19% in SPRA, SD = −0.01) were similar between groups.
Medical costs of SPRA and SNRA
Outpatient direct medical costs per patient with SPRA were higher than for patients with SNRA ($1624 and $1333, SD = 0.11 for all cause cost), while total direct medical costs were not different. However, RA-related direct medical costs were much higher for patients with SPRA than with SNRA ($1027 and $450, SD = 0.25) (Table 3).
Comparison of medical utilization and direct medical costs per patient between patients with seropositive and seronegative RA.

Presented in USD at the exchange rate of 1000 won (KRW) per 1 USD.
Direct medical cost which was charged for the treatment of RA under the diagnostic code of M05 in SPRA patients and M06 in SNRA patients.
RA, rheumatoid arthritis; SD, standardized differences; SNRA, seronegative rheumatoid arthritis; SPRA, seropositive rheumatoid arthritis; USD, United States dollars.
In the multivariable quantile regression analysis, the incremental costs in patients with SPRA increased over a period of 1 year by $179.8 at the 10th percentile, $202.8 at the 25th percentile, $252.6 at the 50th percentile, $319.2 at the 75th percentile, and $296.9 at the 90th percentile of the annual direct medical cost, compared with patients with SNRA. The effects of SPRA on attributable costs were more pronounced at higher quantiles (Table 4).
Incremental adjusted direct medical costs of SPRA patients compared with SNRA patients. a

Presented in USD at the exchange rate of 1000 won (KRW) per 1 USD. Incremental adjusted direct medical cost from multivariable quantile regression model, adjusted with covariates such as age, sex, payer type, type of institution, type of department, CCI score, and medication, which influence costs.
Other institutions included traditional medicine.
CCI, Charlson comorbidity index; CI, confidence interval; DMARDs, disease-modifying anti-rheumatic drugs; RA, rheumatoid arthritis; SNRA, seronegative rheumatoid arthritis; SPRA, seropositive rheumatoid arthritis; USD, United States dollars.
Healthcare utilization by RA patients between 2012 and 2016
From 2012 to 2016, the mean numbers of OPD visits for management of RA decreased in both groups (by 0.5 times in SPRA and 0.2 times in SNRA, respectively) (Figure 1a). On the other hand, the number of RA-related hospitalizations increased from 1.3 to 1.4 among SNRA patients, while there was no change in SPRA patients (Figure 1b).

Changes of healthcare utilization in seropositive and seronegative RA patients between 2012 and 2016. (a) Outpatient visit. (b) Hospitalization. (c) Direct medical costs. All values for costs were presented in USD at the exchange rate of 1000 won (KRW) per 1 USD.
Annual direct medical costs per patient numerically increased between 2012 and 2016 in both SPRA (from $2358 to $2919) and SNRA (from $2002 to $2598) group. RA-related medical cost per patient increased $206 in SPRA and $123 in SNRA. Theses increase were more abrupt in SNRA patients (37.6%) than SPRA patients (25.1%) (Figure 1c).
Discussion
Using the nationwide claims database for Korea, which includes all patients with RA in Korea, we compared annual healthcare utilization direct medical costs per patient between SPRA and SNRA patients. In 2016, RA-related healthcare utilization and direct medical costs per patient were higher in the SPRA group than in the SNRA group. Patients with SPRA tend to receive more intensive treatment than SNRA patients in terms of bDMARDs use and receiving joint surgery. Furthermore, the consistent increase in medical costs attributable to SPRA revealed by the quantile regression model was detected alongside increasing total medical costs. However, the annual increase of direct medical costs between 2012 and 2016 was greater in the SNRA group than in the SPRA group.
The greater healthcare utilization and medical costs of SPRA patients compared with SNRA patients could be explained in several ways. First, it may be related to differences in treatment patterns. Potent conventional DMARDs such as methotrexate, leflunomide, tacrolimus, bDMARDs, and corticosteroids were prescribed more commonly in the SPRA group, while less potent conventional DMARDs such as hydroxychloroquine and sulfasalazine were prescribed in the SNRA group. Previous US studies have also shown that the difference in medication utilization and its cost was a major factor underlying the difference in medical expenses between SPRA and SNRA.16,17 In our study, RA-related OPD visits by SPRA patients were more frequent than in SNRA. Intensive managements of RA may lead to higher drug and laboratory test costs for monitoring of safety and effectiveness of medication, including bDMARDs. Additionally, these different treatment patterns may be associated with differences in disease severity between groups, and may be reflected in more frequent healthcare utilization. Second, it may be related to the Korean individual copayment beneficiaries program (ICBP) system, which applies only to patients with SPRA. In Korea, the ICBP governs care of rare and intractable diseases. An analysis of RA patients diagnosed by physicians using the 1987 ACR criteria or 2010 ACR/EULAR classification criteria found that only SPRA are responsible for ICBP and patients with SPRA are required to copay only 10% of their medical expenses.18,19 This policy may lead to more intensive treatment and frequent healthcare utilization in SPRA patients, whereas patients with SNRA will be required to copay about 30% of total expenses and therefore may not opt for expensive medications or treatments. However, ICBP benefits are applied only to RA-related care, and the overall outpatient medical costs were also different between the two patient groups. Furthermore, there was no significant difference in the number of OPD visits between groups, although the benefits of ICBP apply only to SPRA. Therefore, other factors as well as ICBP, which is related to the high medical costs associated with SPRA, were more frequent. The quantile regression model was used to derive the incremental costs for 1 year among patients with SPRA, which increased as medical costs approached the upper quantile compared with patients with SNRA. This finding suggests that the impact of SPRA on medical costs could be higher for more complicated or advanced patients with RA. In addition, the medical costs attributable to use of nursing hospitals or public health centres, comorbidity, and the use of biologic DMARDs were also substantial, especially more pronounced at higher quantiles. The relatively low out-pocket cost in the nursing hospitals or public health centres maybe related with this effect.
From 2012 to 2016, annual direct medical costs increased to $560–590 in both SPRA and SNRA groups. RA-related medical costs per patient increased abruptly in SNRA patients (27.3%) compared with SPRA patients (20.1%). The increasing medical costs of patients with RA is noteworthy, because they could lead to increases in national burden along with increased prevalence of RA. 20 As a countermeasure, we should consider whether it is better to restrict healthcare utilization by reducing copayment benefits, or to slow the increase in access to expensive drugs, such as bDMARDs. However, such an approach may not be appropriate if it might worsen patient outcomes, and ultimately result in higher costs. Instead, there may be a group with poor prognosis among patients with SNRA who require intensive treatment. It is necessary to review and expand the healthcare supportive system for these patients.
In our study, mean age was lower and the percentage of male was higher in SNRA (27.6%) than in SPRA (19.6%). SPRA patients visited tertiary referral hospital more frequently than SNRA patients. In terms of comorbidities, the prevalence of osteoporosis was higher in the SPRA than the SNRA group. With regard to differences of demographics according to seropositivity, results have been inconsistent. In a previous US study using claims data, there was no significant difference in age and sex according to seropositivity. 21 However, analysis of a Canadian registry showed that SNRA patients were older and more often male (31%) than SPRA patients (23%). 22 In addition, the variation in the pattern of utilization of type of institution may be related to the Korean national support system, ICBP. Further, the high prevalence of osteoporosis in SPRA could in part be explained by strict monitoring in clinical practice rather than the impact of autoantibodies on bone loss.
This study has several strengths. First, this study is highly representative because we used nationwide claims data that cover the entire Korean population. Our comparisons between SNRA and SPRA provided an opportunity to review the healthcare policy covering RA patients by dividing them into two groups. Second, since our analysis was conducted using a variety of variables such as medical utilization divided by inpatient/outpatient visit, and overall medical cost, we were able to compare present healthcare utilization comprehensively in SNRA and SPRA.
There were several limitations to this study. First, it is difficult to obtain generalizability with other countries as healthcare utilization and medical costs are related to the health care environment of each country. Second, we used ICD-10 codes for dividing patients according to seropositivity. Since our claims data do not include the results of laboratory tests, we used the seropositive RA diagnostic code of ICD-10 for SPRA, which is assigned to patients with RF or anti-CCP required to register to ICBP for reducing medical expenses in Korea. In our study, the prevalence of SPRA, 57.8%, was lower than in previous observational registry data showing that 60–85% of SPRA.23–25 It is possible that other inflammatory arthritis such as undifferentiated arthritis, palindromic rheumatism or seronegative spondylopathy may be included in the group of SNRA. These diseases with low disease severity could influence low healthcare utilization and medical costs. Third, we could not estimate indirect medical costs that were incurred by sick leave or work disability, so we were limited to evaluating the direct medical costs of RA patients in society. Fourth, we did not analyse the burden of traditional oriental medicine, even though there were frequent visits to such institutions. However, previous research has suggested that musculoskeletal patients use traditional oriental medicine as a complement to Western medicine rather than an alternative, and this use may not impose a large financial burden on the patients because of the low cost of treatments, such as acupuncture and physical therapy. 26 Fifth, we could not consider the use of private insurance for health care that is not covered by national insurance.
We compared patients with RA by dividing them into homogenous groups with or without antibodies, but there were still heterogeneous elements in each group. Therefore, further study is required to evaluate high risk patients likely to incur high costs and have poor outcomes, and to identify preventable factors. In addition, the impacts of differences in healthcare utilization and medical costs on clinical outcomes of RA need to be studied.
Footnotes
Conflict of interest statement
YKS has received research grants from Bristol-Myers Squibb, Eisai, Pfizer, and JW Pharmaceutical. HK, SKC, SC, SGI, SYJ, and EJJ have nothing to declare.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by a grant of Patient-Centered Clinical Research Coordinating Center funded by the Ministry of Health and Welfare, Republic of Korea (grant number : HI19C0481, HC19C0052).
