Abstract
Background:
To investigate factors associated with drug survival of an alternative tumour necrosis factor inhibitor (TNFi) and secukinumab (SEC) after switching from the first TNFi in patients with ankylosing spondylitis (AS).
Methods:
We included a total of 78 patients with AS who switched to an alternative TNFi (n = 56) or SEC (n = 22) from the first TNFi. Patient characteristics at the time of switching and drug discontinuation rate were compared between the two groups. Cox regression analyses were performed to evaluate factors associated with the risk of discontinuing the alternative TNFi and SEC.
Results:
The proportion of patients with syndesmophytes was numerically lower (28.6% versus 45.5%, p = 0.155) and the C-reactive protein (CRP) level was numerically higher [3.8 (1.0–15.4) mg/L versus 1.1 (0.5–3.5) mg/L, p = 0.060] in patients who received an alternative TNFi. The drug discontinuation rate (alternative TNFi: 35.7% versus SEC: 36.4%, p = 0.957) and reasons for discontinuation were similar (primary failure, p = 0.342; secondary failure, p > 0.999; and adverse events, p = 0.670) between the two groups. A higher CRP level at switching was associated with a lower risk (adjusted HR = 0.93, 95% CI = 0.87–0.99, p = 0.022) of discontinuing the alternative TNFi, and primary failure of the first TNFi was associated with a higher risk [adjusted HR (HR) = 5.20, 95% confidence interval (CI) = 1.91–14.11, p = 0.001]. Current smokers (adjusted HR = 5.77, 95% CI = 1.20–27.74, p = 0.029) and the presence of syndesmophytes (adjusted HR = 7.49, 95% CI = 1.39–40.23, p = 0.019) were associated with a higher risk of discontinuing SEC.
Conclusion:
When switching the drug from the first TNFi in patients with AS, an alternative TNFi could be preferable in patients with higher CRP levels or syndesmophytes, or current smokers, whereas SEC could be a better choice in patients who presented primary failure of the first TNFi in terms of drug survival.
Keywords
Introduction
Ankylosing spondylitis (AS) is a chronic inflammatory disorder that mainly affects the axial skeleton, leading to bony fusion of the vertebral joints. 1 Non-steroidal anti-inflammatory drugs (NSAIDs) are effective in reducing symptoms in patients with AS and are used as a first-line treatment in these patients. 1 For patients with AS who present an insufficient response to NSAIDs, the current practice is to employ tumour necrosis factor inhibitors (TNFis).2–4 If therapy with the first TNFi fails, switching to an alternative TNFi or an interleukin 17A inhibitor (IL-17Ai) is recommended.2–4 An alternative TNFi is still efficacious after failure of the first TNFi, although the level of efficacy is lower than that observed with the first TNFi. 5 Similarly, therapy with an IL-17Ai is efficacious in patients with AS who failed to show adequate response with a TNFi, but less efficacious than in TNFi-naïve patients. 6 However, it is unclear which patients could benefit more from switching to an alternative TNFi or an IL-17Ai after failure of the first TNFi. A recent observational study has revealed the comparable effectiveness of secukinumab (SEC), an IL-17Ai, and an alternative TNFi after prior exposure to TNFis. 7 However, limited data are available on whether specific patient subsets favour IL-17Ai over an alternative TNFi, or vice versa, after failure of the first TNFi.
It could be reasonable to implement different approaches between patients who present primary failure and those who demonstrate a secondary failure of the first TNFi. Patients who presented primary failure of the first TNFi might benefit from switching to an IL-17Ai rather than an alternative TNFi, assuming that TNFα is not the main mediator of inflammation in these patients. For patients who presented secondary failure of the first TNFi, switching to an alternative TNFi could be preferable over immediately switching to an IL-17Ai, given that there are only two biological disease-modifying antirheumatic drugs (bDMARDs) with different modes of action (i.e. TNFi and IL-17Ai) available for AS. Indeed, recommendations suggest switching to an IL-17Ai in patients who presented primary failure of the first TNFi and switching to an alternative TNFi in patients who presented secondary failure of the first TNFi.2–4 However, there is a lack of data supporting this suggestion. Identifying factors that favour one approach over the other, possibly including the reasons for discontinuation of the first TNFi, is crucial for a more strategic switching of bDMARDs. As a relatively limited number of bDMARDs are available for the treatment of AS when compared with that for other rheumatic diseases, such as rheumatoid arthritis and psoriatic arthritis, drug survival is of particular importance and needs to be considered when switching bDMARDs in patients with AS. In this study, we aimed to identify patient subsets who may benefit from switching to an alternative TNFi or an IL-17Ai, respectively, in terms of drug survival after exposure to the first TNFi.
Methods
Study population
Patients with AS who were started on a new TNFi or SEC between January 2018 and June 2020 at a tertiary referral hospital in Seoul, South Korea were retrospectively reviewed for inclusion. Inclusion criteria were as follows: (i) fulfilment of the radiological criterion of the 1984 modified New York criteria (sacroiliitis grade ⩾2 bilaterally or grade ⩾3 unilaterally) 8 and (ii) prior exposure to a TNFi. The following patients were excluded: (i) prior exposure to two or more TNFis, (ii) dose reduction or prolongation of the dosing interval for the drug of interest (TNFi or SEC) during the observation period, (iii) follow-up for less than 6 months, and (iv) presence of uveitis, psoriasis, or inflammatory bowel disease, as the presence of these diseases could affect the choice of second-line bDMARDs.
In all patients, the first TNFi was initiated after insufficient response to NSAIDs, with or without conventional synthetic DMARDs (csDMARDs). Patients were switched to a second-line bDMARD (i.e. an alternative TNFi or SEC) on experiencing a primary non-response to the first TNFi (primary failure), relapse after an initial response (secondary failure) or adverse events. The choice of second-line bDMARDs was at the discretion of the treating physicians.
The patients included were retrospectively observed from the time point of initiating the second-line bDMARD (referred to as the baseline) to the last follow-up date. The following baseline data were reviewed: age, sex, symptom duration, presence of peripheral symptoms (peripheral arthritis, enthesitis, and dactylitis), smoking status (current smoker or not), body mass index, human leukocyte antigen B27 (HLA-B27) positivity, presence of syndesmophytes (based on the plain radiographs of cervical spine and lumbar spine interpreted by experienced radiologists), erythrocyte sedimentation rate, C-reactive protein (CRP), Bath Ankylosing Spondylitis Disease Activity Index (BASDAI), concurrent use of csDMARDs and NSAIDs, the type of first TNFi, and the reason for discontinuation of the first TNFi (primary failure, secondary failure, and adverse events). Primary failure was defined as cases where BASDAI improvement ⩾2 and BASDAI <4 were never achieved after the initiation of the first TNFi. Secondary failure was defined as cases where BASDAI improvement ⩾2 and BASDAI <4 were achieved, but afterwards became BASDAI ⩾4 with BASDAI worsening ⩾2.
This study was approved by the Institutional Review Board (IRB) of Gangnam Severance Hospital (IRB No: 3-2020-0463). Owing to the retrospective nature of this study, the requirement for informed consent was waived.
Outcome
The outcome was drug survival of second-line bDMARDs. Electronic medical records of each patient were reviewed from the baseline to the last follow-up date to determine whether second-line bDMARDs had been discontinued during the observation period. Patients who discontinued their second-line bDMARDs owing to primary failure, secondary failure, or adverse events were all included. Patients with ongoing treatments were censored at the last follow-up date.
Statistical analysis
Continuous variables were expressed as median (interquartile range) and categorical variables were expressed as number (%). The Mann–Whitney U test and Fisher’s exact test (or χ2 test when less than 20% of cells had an expected count less than 5) were used to compare continuous and categorical variables, respectively, between two groups. The Kaplan–Meier analysis was used to visualize drug survival of the alternative TNFi and SEC, and log-rank test was used for comparison. The risk of discontinuing the alternative TNFi and SEC was evaluated using Cox proportional hazard regression analysis. The proportional hazards assumption for each independent variable was tested using Schoenfeld partial residuals. No relevant violations were determined. Factors with a p value of <0.1 in univariable analysis were incorporated into the multivariable analysis using stepwise backward elimination method. A p value of <0.05 was considered statistically significant. All analyses were performed using the SPSS software version 25.0 (IBM Corp., Armonk, NY, USA).
Sensitivity analysis
To test the robustness of results obtained in the main analysis, we performed a sensitivity analysis using a stricter definition of drug discontinuation. Instead of including all patients who discontinued their second-line bDMARDs owing to primary failure, secondary failure, or adverse events, we excluded patients who discontinued their second-line bDMARDs due to adverse events and performed Cox proportional hazard regression analysis. We performed this sensitivity analysis because, in contrast to primary and secondary failures, discontinuation owing to adverse events does not necessarily imply that the drug was ineffective.
Results
Patient characteristics
In total, 143 patients with AS who fulfilled the radiological criterion of the 1984 modified New York criteria 8 switched to an alternative TNFi or SEC between January 2018 and June 2020. Overall, 21 patients who were previously exposed to two or more TNFis, 17 patients who did not receive a standard dose of the drug of interest throughout the observation period, three patients who were followed up for less than 6 months, and 24 patients who had uveitis, psoriasis, or inflammatory bowel disease were excluded. The remaining 78 patients with AS who received an alternative TNFi (n = 56) or SEC (n = 22) as a second-line bDMARD were included in the analysis. The baseline characteristics of patients are shown in Table 1. The proportion of patients with syndesmophytes was numerically lower (28.6% versus 45.5%, p = 0.155) and the CRP level was numerically higher [3.8 (1.0–15.4) mg/L versus 1.1 (0.5–3.5) mg/L, p = 0.060] in patients who received an alternative TNFi. The type of the first TNFi differed between the two groups: the proportion of patients with prior exposure to adalimumab (37.5% versus 63.6%, p = 0.037) and golimumab (3.6% versus 22.7%, p = 0.017) was lower, while the proportion of patients with prior exposure to infliximab (30.4% versus 0.0, p = 0.002) was higher in the alternative TNFi group.
Baseline characteristics of patients with AS initiating an alternative TNFi or SEC after previous exposure to a TNFi.

AS, ankylosing spondylitis; BASDAI, Bath Ankylosing Spondylitis Disease Activity Index; BMI, body mass index; CRP, C-reactive protein; csDMARD, conventional synthetic disease-modifying antirheumatic drug; ESR, erythrocyte sedimentation rate; HLA-B27, human leukocyte antigen B27; IQR, interquartile range; NSAID, non-steroidal anti-inflammatory drug; SEC, secukinumab; TNFi, tumour necrosis factor inhibitor.
Discontinuation of second-line bDMARDs
Overall, drug discontinuation occurred in 28 of 78 patients (35.9%) during a median observation period of 27.8 (14.6–32.6) months. The observation period [29.2 (14.8–32.9) months versus 23.1 (13.7–31.6) months, p = 0.196] and drug discontinuation rate (35.7% versus 36.4%, p = 0.957) did not differ between patients who received an alternative TNFi and those who received SEC. Following Kaplan–Meier analysis, the two groups showed no significant difference in drug survival (p = 0.492) [Figure 1(a)]. Next, as there was a numerical difference in observation period between the two groups, patients with ongoing treatments were censored at 23 months, which was the median value of observation period in the SEC group, and an additional Kaplan–Meier analysis was performed [Figure 1(b)]. The two groups still showed no significant difference in drug survival (p = 0.315). The reasons for drug discontinuation were similar between the two groups (primary failure, 5.4% versus 13.6%, p = 0.342; secondary failure, 21.4% versus 18.2%, p > 0.999; and adverse events, 8.9% versus 4.5%, p = 0.670) (Table 2).
Discontinuation of the second-line bDMARDs.
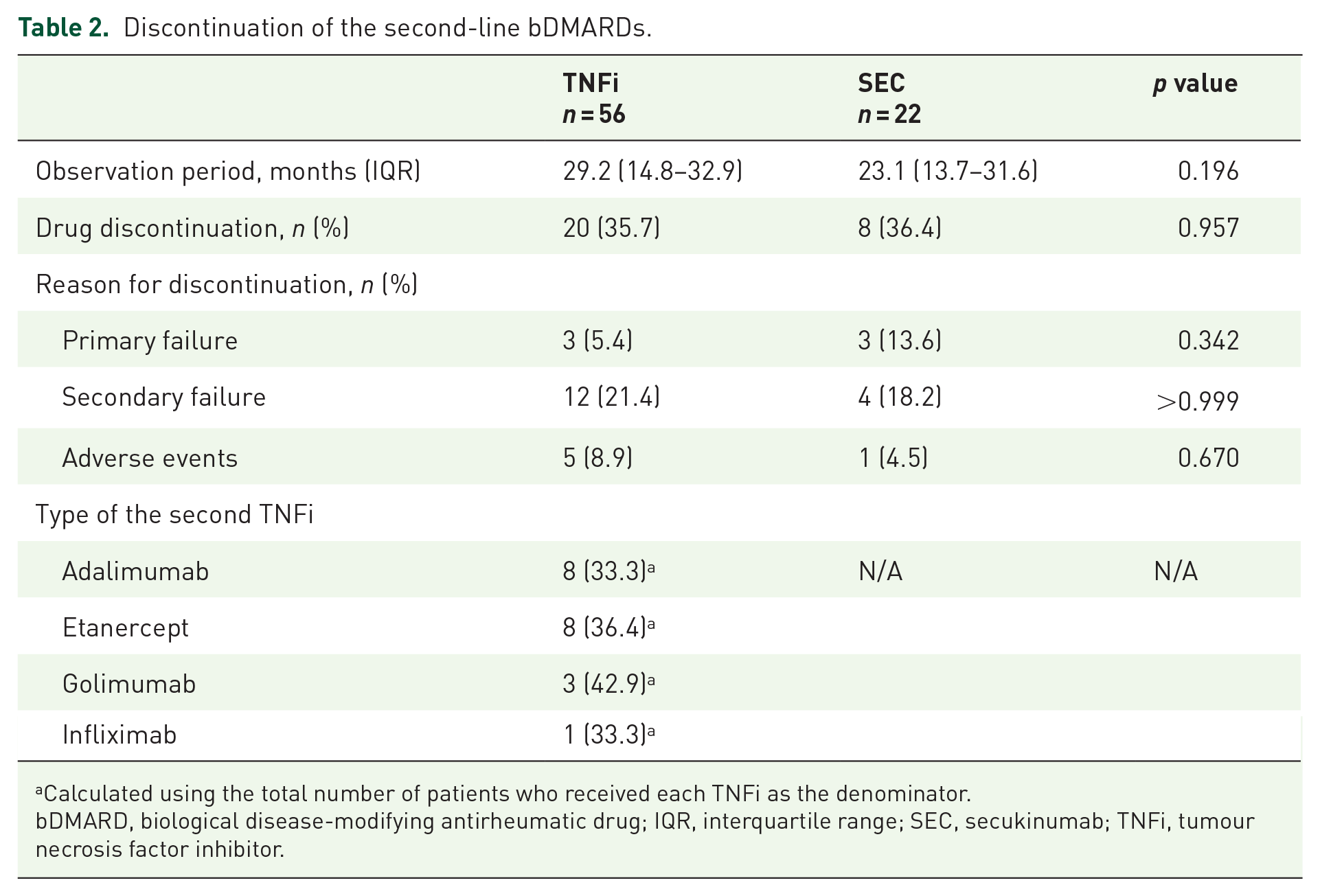
Calculated using the total number of patients who received each TNFi as the denominator.
bDMARD, biological disease-modifying antirheumatic drug; IQR, interquartile range; SEC, secukinumab; TNFi, tumour necrosis factor inhibitor.

Comparison of drug survival curves between the alternative TNFi and SEC. (a) ongoing treatments censored at last follow-up date, and (b) ongoing treatments censored at 23 months.
Drug survival analysis
For patients who received an alternative TNFi, HLA-B27 positivity [unadjusted hazard ratio (HR) = 0.33, 95% confidence interval (CI) = 0.13–0.89, p = 0.028], CRP level at baseline (unadjusted HR = 0.94, 95% CI = 0.88–0.99, p = 0.038), and primary failure of the first TNFi (unadjusted HR = 4.13, 95% CI = 1.57–10.90, p = 0.004) had a p value of <0.1 in the univariable analysis. These covariates were included in the multivariable analysis. In the final model after stepwise backward elimination, higher CRP level at baseline was associated with a lower risk (adjusted HR = 0.93, 95% CI = 0.87–0.99, p = 0.022) of discontinuing the alternative TNFi, and primary failure of the first TNFi was associated with a higher risk (adjusted HR = 5.20, 95% CI = 1.91–14.11, p = 0.001) of discontinuing the alternative TNFi.
In regard to patients who received SEC, male sex (unadjusted HR = 6.49, 95% CI = 0.80–52.94, p = 0.081), current smoker (unadjusted HR = 4.08, 95% CI = 0.95–17.51, p = 0.059), and the presence of syndesmophytes (unadjusted HR = 5.93, 95% CI = 1.18–29.86, p = 0.031) had a p value of <0.1 in the univariable analysis and hence were included in the multivariable analysis. In the final model after stepwise backward elimination, current smokers (adjusted HR = 5.77, 95% CI = 1.20–27.74, p = 0.029) and the presence of syndesmophytes (adjusted HR = 7.49, 95% CI = 1.39–40.23, p = 0.019) were significantly associated with a higher risk of discontinuing SEC (Table 3).
Factors associated with the discontinuation of TNFi and SEC.

Covariates with a p value of <0.1 in the univariable analysis included for multivariable analysis.
Not applicable because none of the patients received infliximab as the first TNFi in the SEC group.
BASDAI, Bath Ankylosing Spondylitis Disease Activity Index; BMI, body mass index; CI, confidence interval; CRP, C-reactive protein; csDMARD, conventional synthetic disease-modifying antirheumatic drug; ESR, erythrocyte sedimentation rate; HLA-B27, human leukocyte antigen B27; HR, hazard ratio; NSAID, non-steroidal anti-inflammatory drug; SEC, secukinumab; TNFi, tumour necrosis factor inhibitor.
Sensitivity analysis
Similar results were revealed in the sensitivity analysis, where patients who discontinued second-line bDMARDs owing to adverse events were excluded (Table 4). For patients who received an alternative TNFi, a higher CRP level at baseline was associated with a lower risk (adjusted HR = 0.90, 95% CI = 0.82–0.99, p = 0.022) of discontinuing the alternative TNFi, and primary failure of the first TNFi was associated with a higher risk (adjusted HR = 10.07, 95% CI = 2.99–33.94, p < 0.001) of discontinuing the alternative TNFi. For those who received SEC, current smokers (adjusted HR = 7.62, 95% CI = 1.40–41.61, p = 0.019) and the presence of syndesmophytes (adjusted HR = 7.08, 95% CI = 1.22–41.12, p = 0.029) were associated with a higher risk of SEC discontinuation.
Sensitivity analysis: factors associated with discontinuation of TNFi and SEC.

Covariates with a p value < 0.1 in the univariable analysis included for multivariable analysis.
ESR excluded in the multivariable analysis due to multicollinearity with CRP.
Not applicable because none of the patients received infliximab as the first TNFi in the SEC group.
BASDAI, Bath Ankylosing Spondylitis Disease Activity Index; BMI, body mass index; CI, confidence interval; CRP, C-reactive protein; csDMARD, conventional synthetic disease-modifying antirheumatic drug; ESR, erythrocyte sedimentation rate; HLA-B27, human leukocyte antigen B27; HR, hazard ratio; NSAID, non-steroidal anti-inflammatory drug; SEC, secukinumab; TNFi, tumour necrosis factor inhibitor.
Discussion
When treating patients with AS who were exposed to the first TNFi, whether to switch to an alternative TNFi or an IL-17Ai as a second-line bDMARD is an important clinical decision that the treating physicians need to make. Our results showed that some subsets of patients may favour one approach over the other in terms of drug survival. In particular, a higher CRP level at baseline (i.e. at the time of switching bDMARDs) was associated with a lower risk of discontinuing the alternative TNFi, primary failure of the first TNFi was associated with a higher risk of discontinuing the alternative TNFi, and current smokers and the presence of syndesmophytes were associated with a higher risk of discontinuing SEC. The results of our main analysis were confirmed in a sensitivity analysis, where patients who discontinued the second-line bDMARDs owing to adverse events were excluded, adding robustness to our findings.
Our data suggest that the reason for discontinuation of the first TNFi should be considered when choosing the second-line bDMARD in patients with AS. Primary failure of the first TNFi was associated with a higher risk of discontinuing the alternative TNFi, but not SEC. Therefore, SEC could be beneficial over an alternative TNFi, after primary failure of the first TNFi. Conversely, secondary failure of the first TNFi, as well as an adverse event with the first TNFi, were not associated with the risk of discontinuing both the alternative TNFi and SEC. Given the limited number of bDMARDs with distinct modes of action, switching to an alternative TNFi rather than to SEC would be preferable in cases of secondary failure or adverse events with the first TNFi. Our finding is meaningful as it is the first to provide evidence supporting the suggestions from the recommendations.2–4
A higher CRP level at the time of bDMARD switching was associated with a lower risk of discontinuing the alternative TNFi, but not SEC. Thus, for patients with higher CRP levels, an alternative TNFi could be better than SEC. Previous reports have consistently shown that higher CRP levels at baseline are associated with good clinical response in patients treated with TNFis.9–12 In contrast to the studies on TNFis that clearly indicate better responses in patients with higher CRP levels, one study on SEC revealed that SEC is effective in patients with both normal and elevated CRP levels. 13 Taken together with our results, patients with higher CRP levels could benefit from an alternative TNFi, whereas those with lower CRP levels could benefit from SEC.
Although the presence of radiographic damage at baseline is a well-known factor associated with radiographic progression in patients with AS,14–16 its association with drug survival is poorly studied. We observed that the presence of syndesmophytes at baseline was associated with a higher risk of discontinuing SEC but not the alternative TNFi. This suggests that for patients with syndesmophytes, which is associated with an impaired functional status of the patient, 17 an alternative TNFi would be a better choice than SEC when switching from a first TNFi.
We also found that smoking was associated with a higher risk of discontinuing SEC but not the alternative TNFi. Previous studies have reported that smoking status does not affect the response or discontinuation of TNFi.18,19 In contrast, one study has suggested a low response to SEC in patients with AS who were smokers. 20 Collectively, in patients who are current smokers, an alternative TNFi could be better than SEC.
The present study has several limitations. First, although we used strict inclusion and exclusion criteria to minimize confounding factors, this is a retrospective study; hence, there is a risk of confounding by undetermined variables such as patients’ functional status (i.e. Bath Ankylosing Spondylitis Functional Index). Second, we excluded patients with extra-articular manifestations to homogenize the study population. Although this approach has an advantage in reducing confounding by indication and selection bias, it may also have a disadvantage in that our findings cannot be generalized to those with extra-articular manifestations. Third, the number of events was small in the SEC group, and statistically powerful conclusion cannot be drawn. Fourth, the observation period was relatively short, owing to the relatively recent introduction of SEC in the treatment of AS. To balance the observation period between the alternative TNFi and SEC groups, the observation period was initiated in January 2018, the date when SEC was first used at our centre. However, although discontinuation of the first TNFi occurs mostly within 5 years, 21 the second TNFi has a shorter duration of drug retention than the first TNFi, with a pronounced discontinuation rate during the first year after switching. 22 Therefore, as the drugs of interest were second-line bDMARDs in our study, a median observation period of 27.8 (14.6–32.6) months remains clinically meaningful. Further large head-to-head prospective randomized trials with long-term follow-up would be helpful to confirm our present data.
In conclusion, we demonstrated that when switching bDMARDs in patients with AS who were previously exposed to a TNFi, an alternative TNFi could be preferable in patients with higher CRP levels or syndesmophytes, or current smokers, whereas SEC could be a better choice in patients who presented primary failure of the first TNFi in terms of drug survival. These data could be considered when choosing second-line bDMARDs in patients with AS.
Footnotes
Conflict of interest statement
The authors declare that there is no conflict of interest.
Data availability statement
All data are incorporated into the article.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
