Abstract
Psoriatic arthritis (PsA) and ulcerative colitis (UC) are immune-mediated diseases that cause significant burden worldwide. Recent advances in their management have improved patient outcomes. However, significant unmet needs still remain as not all patients respond to current treatments, and patients may lose responsiveness over time. An improved understanding of the pathophysiology of these diseases has brought about the development of novel disease-modifying agents, including interleukin inhibitors and, more recently, Janus kinase (JAK) inhibitors. With the approval of tofacitinib for the treatment of adults with active PsA and in adult patients with moderately-to-severely active UC, JAK inhibitors have recently entered the treatment armamentarium for PsA and UC. A number of other JAK inhibitors are also undergoing clinical development and are currently in phase III trials. This review provides an overview of the current therapeutic options for PsA and UC, with a focus on the JAK inhibitors.
Introduction
Psoriatic arthritis
Psoriatic arthritis (PsA) is a chronic, immune-mediated, inflammatory arthropathy that can cause permanent joint damage and disability, and is typically associated with psoriatic skin disease. 1 The clinical presentation of PsA is heterogeneous and can manifest as peripheral arthritis, axial disease, enthesitis, dactylitis, psoriasis, and psoriatic nail disease; 2 peripheral arthritis is the most common disease manifestation. 3 The presence of psoriasis is the single greatest risk factor for the development of PsA; the presence of PsA among first-degree relatives and psoriatic nail dystrophy also increases the risk of PsA. 4
There is considerable variation in the prevalence and incidence of PsA amongst different populations.3,5 Worldwide, the reported prevalence ranges from one in 100,000 persons in Japan to 500 in 100,000 persons in Australia and the annual incidence of 0.1 to 23 cases per 100,000 persons in the general population. 6 Although the prevalence of PsA is low in the general population, it is common among patients with psoriasis. Approximately one in four patients with psoriasis have PsA, with a pooled prevalence of 23.8% identified in 45 studies which applied the Classification Criteria for Psoriatic Arthritis, 7 although a prevalence as high as 41% has also been reported.3,7 In patients with psoriasis, the incidence of PsA was reported to range from 0.27 to 2.7 per 100 person-years. 7 Immune-mediated inflammation plays a major role in the initiation and maintenance of PsA. A key feature of PsA is inflammation of the synovial membrane, characterized by an increase in vascularization and infiltration of immune cells, which release pro-inflammatory mediators that activate fibroblast-like synoviocytes and invade adjacent cartilage and bone. 8 Inflammation of the enthesis is also a common feature of PsA.
Although PsA can resemble other forms of arthritis at disease onset, and especially rheumatoid arthritis, PsA has a distinctive pathology. Genetics, environmental factors, and immune-mediated inflammation are involved in the pathogenesis of PsA. However, their precise mechanisms are complex and are incompletely understood. 8 In PsA, activated immune cells release pro-inflammatory cytokines including tumor necrosis factor (TNF) α and interleukins (ILs), which drive inflammation within the joints, causing damage to the synovial membrane, articular cartilage, and bone. 9 Results from synovial biomarkers in PsA have shown the participation of several cytokines and chemokines, including IL-1, IL-8, and the IL-6/IL-17 cytokine axis and their correlation with systemic disease activity. 10 Comprehensive analysis of cytokine and chemokine activation and genes representative of the inflammatory processes in PsA skin and synovial tissue showed that gene expression in synovium was more closely related to gene expression in skin than to gene expression in synovium in other forms of arthritis. 11 PsA gene expression patterns in skin and synovium arose to be clearly distinct, showing a stronger IL-17 gene signature in skin than in synovium and more equivalent TNF and IFN gamma gene signatures in both tissues. 12 Moreover, cytokines and chemokines expression profile analysis, at synovial tissue level, of PsA patients found that PsA patients with synovial tissue aggregates exhibited an increase of IL-23 expression compared with PsA patients without synovial aggregates showing a clear predominance of plasma IL-17. 13
Comorbidities of PsA
Comorbidities are commonly associated with PsA, including cardiovascular disease, hypertension, dyslipidemia, metabolic syndrome, diabetes, obesity, inflammatory bowel disease (IBD), osteoporosis, ophthalmic disease, non-alcoholic fatty liver disease, hypothyroidism, and depression and anxiety.14–18 Indeed, their prevalence was shown to be high in patients with PsA, with 42% of patients reported to have three or more comorbid conditions. 19
Of particular relevance to this review, PsA was found to be associated with ulcerative colitis (UC) [odds ratio 1.9, 95% confidence interval (CI): 1.21–3.00; p = 0.05] after controlling for known risk factors in a retrospective analysis of data from Israel’s largest healthcare provider (from 2002 to 2013), which matched, for age and sex, 3161 PsA patients with 31,610 randomly selected patients. 17 In a separate study, the adjusted incidence rate ratio of UC in patients with PsA was 2.34 (95% CI: 1.77–3.10) after the exclusion of patients with reactive arthritis, with a 10-year UC incidence of 11 per 1000 patients. 20 However, the relative risk of developing UC was not significantly higher in a UK population-based cohort study of 6783 patients with PsA when compared with a matched cohort of 27,132 psoriasis patients without PsA and a general population cohort of 27,132 subjects. 21
The GRAPPA 2015 treatment recommendations for PsA 22 and the 2018 American College of Rheumatology (ACR)/National Psoriasis Foundation guideline for the treatment of PsA, 23 together with the 2019 European League Against Rheumatism (EULAR) recommendations for the management of PsA with pharmacological therapies, 24 focus on treatment of PsA in the presence of comorbidities.
UC
UC, which causes mucosal inflammation and is mainly confined to the large bowel and rectum, and Crohn’s disease, a transmural inflammatory disease which can affect any part of the gastrointestinal tract, are the two major disorders that constitute IBD, a chronic condition characterized by inflammation of all or part of the digestive tract.
Symptoms of UC, which are dependent on disease extent and severity, include bloody diarrhea, rectal bleeding, tenesmus, urgency, fecal incontinence, increasing bowel frequency, nocturnal defecation, abdominal pain, anorexia, fatigue, and fever. 25 UC mostly presents in adults aged 30–40 years, 26 with a second peak in incidence occurring after 50 years of age in some populations. 25
Epidemiologic studies show that the use of oral contraceptives, former smoking, and high linoleic and animal protein intake are the main risk factors for the development of UC, whereas ever being breastfed and current smoking are protective factors; antibiotic use in childhood or in adults was not associated with an increased risk of UC. 27 Other risk factors include family history, with first-degree relatives having a four-fold increased risk of developing UC. 26
The incidence and prevalence of UC in Europe is estimated to be 0.9–24.3 cases per 100,000 person-years and from 2.4 to 294 cases per 100,000 persons, respectively, which suggests there may be around 2.1 million persons with UC in the total European population and 1.5 million persons with UC in the European Union. 28
Although the pathogenesis of UC is poorly understood, genetic and environmental factors, as well as the gut microbiota and the immune response, appear to be involved.29–31 A number of pro-inflammatory cytokines are known to be involved in the progression of UC, including TNFα and ILs. Both innate and adaptive cellular immunity appear to be key to the pathogenesis of the disease, and numerous review articles of the immune mechanisms involved in the pathogenesis of UC have been published.26,29–32
Clinimetric indexes used for the assessment of PsA and UC
In the treatment of PsA and UC, the availability of new drugs that target the inflammation process is expected to provide symptomatic relief to the patient. It is therefore imperative to demonstrate that symptom control is directly related to the modification of the disease course, as persistent inflammation, despite symptomatic relief, is correlated with poorer long-term outcomes.32,33 Validated measures that quantify disease activity in PsA have been comprehensively summarized.34,35
With the arrival of new therapeutic options for PsA, it is important to understand the optimal way to measure disease activity and its impact. 2 The ACR 20% response criteria (ACR20) is a composite measure and is the primary outcome required by the US Food and Drug Administration (FDA) for the approval of a new therapy for PsA.36,37 ACR20 is defined as at least a 20% improvement in tender and swollen joint counts, as well as a 20% improvement in three of the following: Health Assessment Questionnaire, patient pain assessment, patient global assessment, Physician Global Assessment or acute phase response: C-reactive protein. However, the ACR20 fails to assess a number of areas that are relevant to PsA, including dactylitis, enthesitis, and axial involvement; it also does not provide a clinically meaningful threshold as the proportion of patients who achieve ACR20 does not provide information on the magnitude of the response. 37
GRAPPA, in conjunction with the Outcome Measures in Rheumatology Clinical Trials (OMERACT) working group, updated the PsA Core Domain Set, which defines core domains measured in randomized controlled trials and longitudinal observational studies, to include musculoskeletal disease activity, skin disease activity, fatigue, pain, patient global, physical function, health-related quality of life, and systemic inflammation. 38 In the 2019 meeting, the GRAPPA–OMERACT working group focused on four prioritized domains: musculoskeletal disease activity (enthesitis and dactylitis), fatigue, physical function, and structural damage. 39
For UC, the treatment aim is to induce and maintain symptomatic and endoscopic remission, and both the proportion of patients with symptomatic remission (based on patient-reported outcomes) and the proportion of patients with endoscopic remission should be measured as co-primary endpoints. 33 The Mayo score, originally devised in 1987 40 and used to evaluate clinical outcomes, is a composite index consisting of stool frequency, rectal bleeding, mucosal appearance at endoscopy, and Physician’s Global Assessment, each scored from 0 to 3, with a higher score (maximum 12 points) indicating a higher severity of UC. The Mayo endoscopic subscore is used to assess endoscopic remission, with endoscopic remission defined as a score of 0 or 1 and where mucosal healing may be defined as 0. 41 For an assessment of clinical symptoms using the clinical section of the Mayo score, the European Medicines Agency (EMA) defines symptomatic remission as a score of 0 or 1 and that includes cessation of rectal bleeding. 33
A focus on treatment in PsA and UC
A better understanding of the pathophysiology of PsA, in which inflammation is now believed to be driven by elevated levels of TNF and ILs in synovial tissue, synovial fluid and/or enthesis, has advanced the development of effective biologics and small-molecule drugs.8,42–46 By targeting specific pro-inflammatory cytokines and signaling pathways, disease progression can be prevented and quality of life improved.
The last decade has seen major advances in the therapeutic management of PsA, as illustrated by the recent update of EULAR recommendations. 24 Treatment recommendations depend on disease severity and should be aimed towards reaching disease remission or minimal/low disease activity via regular monitoring and appropriate therapy adjustment. 24 Symptomatic drugs, including non-steroidal anti-inflammatory drugs (NSAIDs) and corticosteroids, relieve musculoskeletal signs and symptoms; however, neither inhibits the development of structural joint damage. In patients with unequivocal enthesitis and insufficient response to NSAIDs or to local corticosteroid injections, a biologic disease-modifying anti-rheumatic drug should be considered. 24 To date, commercially available biologic treatments for PsA have revolutionized the treatment of PsA. These include TNFα inhibitors (adalimumab, certolizumab pegol, etanercept, golimumab, and infliximab) and inhibitors directed against IL-12/23 (ustekinumab) and IL-17 (secukinumab and ixekizumab).
Advances in the understanding of the pathophysiology of UC have also led to the successful use of biologic therapies in its treatment. The introduction of TNF inhibitors as biologic therapy has improved clinical outcomes for UC in the treatment of moderate-to-severe disease over the last two decades. TNF inhibitors, which bind the cytokine TNFα and inhibit its inflammatory effects in the gastrointestinal tract, are used for the treatment of moderate-to-severe UC that is refractory to anti-inflammatory and immune-modulating therapies. 47
Medical management of active UC is based mainly on disease site and activity, and on the course of disease behavior, which includes relapse frequency, disease course, prior treatment response, adverse effects, and extra-intestinal manifestations. 48 Treatment guidelines according to the Third European Crohn’s and Colitis Organisation evidence-based consensus on the current management of UC have been updated recently, 48 and treatment options for UC include 5-aminosalicylic acid drugs, steroids, and immunosuppressants. The ideal treatment goal in UC is to induce and then maintain steroid-free remission, defined as symptom resolution and endoscopic healing. Currently approved and available biologic therapies for UC include three anti-TNFα inhibitors – infliximab,49–51 adalimumab,52–54 and golimumab55–58 – and the integrin receptor antagonist vedolizumab.59,60 Table 1 summarizes current available biologics in the treatment of adult PsA and UC patients.
Currently approved TNF inhibitors, IL inhibitors and integrin inhibitors in the treatment of adult patients with psoriatic arthritis and ulcerative colitis.
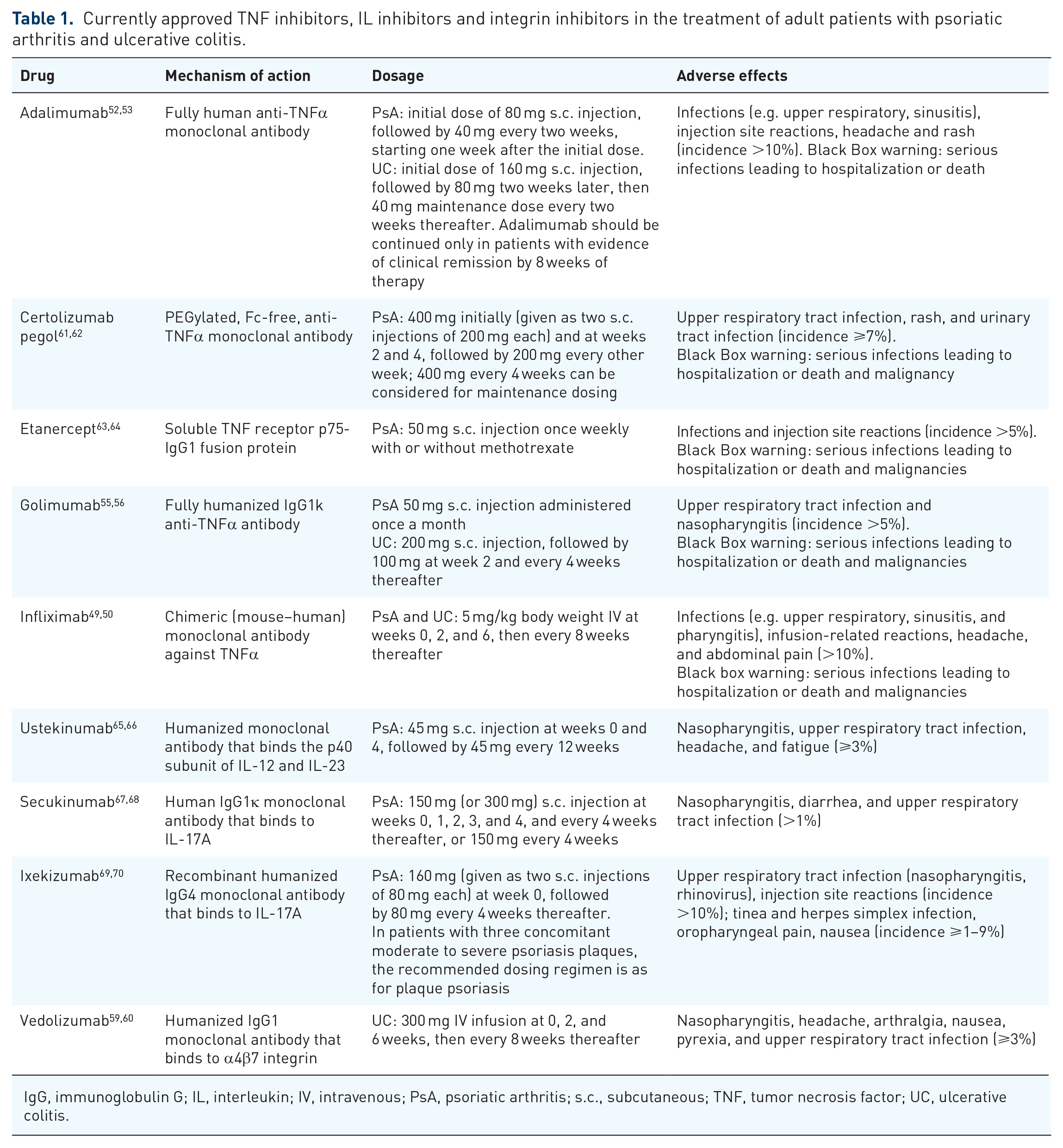
IgG, immunoglobulin G; IL, interleukin; IV, intravenous; PsA, psoriatic arthritis; s.c., subcutaneous; TNF, tumor necrosis factor; UC, ulcerative colitis.
Although biologic therapies have profoundly modified the management of both PsA and UC, there remain significant unmet needs. The induction and maintenance of remission is still problematic despite the variety of conventional and biologic therapies available, as not all patients respond to current treatments or may lose responsiveness over time. 71 Therefore, despite improved clinical outcomes, resistance or the loss of response over time to currently available biologic therapies may limit therapeutic options for a substantial number of patients. 1 Consequently, novel disease-modifying agents that are effective and safe are needed to overcome the unmet clinical needs of patients with PsA and UC.
Cytokines involved in Janus kinase inhibition
Janus kinases (JAKs) are intracellular enzymes that transfer phosphate groups from adenosine triphosphate or guanosine triphosphate to the hydroxyl groups of amino acids of their substrates. JAK signaling pathways are regulated by growth factors and ILs; in turn, the activation of pro-inflammatory genes of many cytokines involved in PsA and UC pathogenesis, such as TNF, IL1β, IL-6, IL-23, and IL-17 axis molecules, are mediated by JAK/signal transducers [JAK1–3 and tyrosine kinase 2 (TYK2)] and STAT (STAT1–5a/b, 6) intracellular pathways. 44 Cytokine binding to receptors at the cell surface activates JAKs bound to the intracellular domains of these cytokine receptors via autophosphorylation events. Subsequently, activated JAKs phosphorylate sites on intracellular domains of cytokine receptors that become docking sites for STAT molecules from the cell cytoplasm. 72 The rationale for using JAK inhibitors (JAKis) to treat PsA and UC relies on the central role of cytokines in their pathogenesis. Most of the cytokines involved, directly or indirectly, are regulated through the JAK–STAT pathway, although JAKs cannot transmit signals provided by IL-1, IL-8 TGFβ, MCSF, and IL-17. 73 Moreover, different cytokines signal using different pairings of individual JAKs. In PsA, the six g-common cytokines (IL-2, IL-4, IL-7, IL-9, IL-15, and IL-21) signal through the JAK1/JAK3 combination, modulating adaptive immune functions. PsA pathogenesis strongly depends upon IL-7 for signaling: innate lymphoid cells (ILCs), present in psoriatic skin lesions and in the pathophysiology of spondyloarthritis (SpA), are related to the presence of IL-17. 73 IFNγ and IL-12 signaling via JAK1/JAK2 and JAK2/TYK2 combinations, respectively, are critical for Th1 cell response and ultimately for production of TNFα by macrophages. 74 JAKs have an influence also in the IL-23/IL-17 axis involved in the pathogenesis of SpA. IL-23, which is produced by activated dendritic cells, uses the JAK2/TYK2 combination for intracellular signalling. 75 Furthermore, IL-6 is also involved in ILC type 3 and Th17 cell activation and functions using the JAK1/JAK2 combination. 76
For UC a number of pro-inflammatory cytokines are known to be involved, including TNF-α, IFN-γ, IL-1β, IL-6, IL-12, IL-13, IL-17, IL-2, IL-15. 29 Indeed, increased expression of both IL-17 and signal transducer and activator of transcription 3 (STAT3) have been identified in inflamed mucosa of patients with UC, which is notable since IL-17 activates STAT3 and stimulates an immune inflammatory response. 28 Th17 cells have been identified as key effectors in the immunopathogenesis of IBD, due to the balance between the proinflammatory Th17 response and anti-inflammatory regulatory response. 77
JAKis
JAKis have recently entered the treatment armamentarium for PsA and UC. The JAK protein tyrosine kinase (PTK) family is constituted by cytoplasmic PTKs that play a pivotal role in cytokine signal transduction pathways through association with various cytokine receptors by activation of the latent forms of STATs involved in the pathogenesis of PsA and UC. Targeting tyrosine kinases, which phosphorylate tyrosine residues and block their enzymatic activity, has proved to be a successful treatment strategy. The JAK–STAT pathway plays an important role in innate and adaptive immunity and is used by numerous cytokines implicated in the pathogenesis of inflammatory and autoimmune diseases.72,78,79 By binding to the kinase domain of JAK at the ATP-binding site, JAKis block cytokine signaling and have been proven to be successful in the treatment of immune-mediated disorders, including PsA and UC. 41
Tofacitinib, an orally administered small-molecule, is a potent selective inhibitor of the JAK family that in human cells preferentially inhibits signaling by heterodimeric cytokine receptors that associate with JAK3 and/or JAK1, with functional selectivity over cytokine receptors that signal via pairs of JAK2. This prevents phosphorylation and activation of STATs, thereby modulating the JAK–STAT signaling pathway at the point of JAKs. Tofacitinib was the first JAKi to be approved, in 2017 in the US and in 2018 in the EU, for the treatment of adults with active PsA and an inadequate response or intolerance to methotrexate or other disease modifying antirheumatic drugs (DMARDs).80,81 In 2018, tofacitinib was also approved for the treatment of adult patients with moderately-to-severely active UC in both the US and the EU.80,81 Clinical trials in PsA that demonstrated its efficacy are Oral Psoriatic Arthritis Trial (OPAL) Broaden and OPAL Beyond phase III trials showing superior efficacy of tofacitinib compared with placebo after 3 months’ treatment in PsA patients with an inadequate response to either conventional synthetic DMARDs 78 or to one or more TNF inhibitor (Table 2). 79 OPAL Broaden was a 12-month, double-blind, active- and placebo-controlled trial in which 422 patients were randomly assigned at a 2:2:2:1:1 ratio to receive one of the following regimens: 5 mg oral tofacitinib twice daily, 10 mg oral tofacitinib twice daily, 40 mg subcutaneous adalimumab once every 2 weeks, placebo with a blinded switch to 5 mg tofacitinib at 3 months, or placebo with a blinded switch to 10 mg tofacitinib at 3 months. 78 OPAL Beyond was a 6-month, double-blind, placebo-controlled trial in which 395 patients were randomly assigned to one of four regimens: 5 mg oral tofacitinib twice daily; 10 mg oral tofacitinib twice daily; placebo with a switch to 5 mg tofacitinib at 3 months, or placebo with a switch to 10 mg tofacitinib at 3 months. 79 For both OPAL trials, the primary endpoints at 3 months were the ACR20 response and the change from baseline in the Health Assessment Questionnaire Disability Index.
Primary endpoint outcomes of the novel Janus kinase inhibitor tofacitinib in patients with psoriatic arthritis.

Change from baseline – a decrease in the HAQ-DI score indicates clinical improvement.
p ⩽ 0.05 versus PL.
p ⩽ 0.01 versus PL.
p ⩽ 0.001 versus PL.
Tofacitinib, approved for the treatment of adult patients with moderately-to-severely active UC in both the US and EU, is recommended at a dose of 10 mg twice daily for induction for at least 8 weeks, followed by 5 mg twice daily for maintenance. For patients who do not achieve adequate therapeutic benefit by week 8, the induction dose of 10 mg twice daily can be extended for an additional 8 weeks (16 weeks total). Tofacitinib should be discontinued if an adequate therapeutic benefit is not achieved after 16 weeks of 10 mg twice daily dose.60,71 Tofacitinib was shown to be more effective than placebo as induction and maintenance therapy in patients with moderately-to-severely active UC in three phase III trials (OCTAVE Induction 1, OCTAVE Induction 2, and OCTAVE Sustain) (Table 3). 80 In OCTAVE Induction 1 and 2, patients were randomized to oral tofacitinib 10 mg twice daily or placebo for 8 weeks. Patients who were eligible to participate in the OCTAVE Sustain trial (i.e. those who had completed OCTAVE Induction 1 or 2 and had at least a clinical response) were randomly assigned again to receive maintenance therapy with tofacitinib 5 mg or 10 mg twice daily, or placebo for 52 weeks. Of the patients who entered OCTAVE Sustain, 88% had received tofacitinib during the induction trial, and 30% were in remission at entry. The primary efficacy endpoint in OCTAVE Induction 1 and 2 was remission (a total Mayo score of ⩽2, with no subscore >1 and a rectal bleeding subscore of 0) at 8 weeks; in OCTAVE Sustain the primary endpoint was remission at 52 weeks. Mucosal healing (a Mayo endoscopic subscore of ⩽1) was a key secondary endpoint at 8 and 52 weeks, respectively, in OCTAVE Induction 1 and 2 and in OCTAVE Sustain. Induction of remission and rates of mucosal healing at 8 weeks were significantly higher in patients treated with tofacitinib than with placebo, and maintenance therapy through 52 weeks sustained remission and mucosal healing. In a recent meta-analysis of oral JAKis in the treatment of UC, JAKis as induction therapy were associated with higher rates of both clinical remission and response compared with placebo, along with a greater likelihood of achieving endoscopic response, endoscopic remission, and mucosal healing. 40
Primary and key secondary endpoint outcomes of the novel Janus kinase inhibitor tofacitinib in patients with ulcerative colitis.

Primary endpoint: a total Mayo score of ⩽2, with no subscore >1 and a rectal bleeding subscore of 0.
Key secondary endpoint: a Mayo endoscopic subscore of ⩽1.
p = 0.007 versus PL.
p < 0.001 versus PL.
b.i.d., twice daily; PL, placebo; pts, patients; UC, ulcerative colitis; wks, weeks.
Adverse Events (AEs) in clinical trials for both PsA and UC reported respiratory tract infection, nasopharyngitis, arthralgia, and headache more frequently than in control groups. More infections occurred in patients treated with tofacitinib than with placebo, although most were mild or moderate in severity. Although treatment with a JAKi did not significantly increase the overall risk for AEs or serious AEs compared with placebo, an increased risk of infection and of herpes zoster was observed. On 26 July 2019 the FDA released a boxed warning to alert physicians and patients regarding the treatment with tofacitinib at the dose of 10 mg twice daily as associated with an increased risk for pulmonary embolism and death in treated patients. The higher dose of 10 mg twice daily of tofacitinib is not approved for PsA or rheumatoid arthritis but is for UC. 81 A study by Mease et al. reported post hoc data of patients with cardiovascular risk (50 years or older with at least one cardiovascular risk factor) from the tofacitinib development in the context of data submitted to the Pharmacovigilance Risk Assessment Committee (PRAC) of the EMA and other real-world data. PRAC reviewed an ongoing postauthorization study in patients with RA, PsA and psoriasis with cardiovascular risk factors treated with 5 and 10 mg tofacitinib or a TNF inhibitor (TNFi), and reported increased all-cause mortality, six-fold increased pulmonary embolism (PE) risk with 10 mg and threefold with 5 mg (not significant) versus TNFi. 82 As a result, the use of tofacitinib has been limited by regulators. Specifically, the updated US prescribing information includes thrombosis as a boxed warning and recommends that tofacitinib be avoided in patients at risk of thrombosis (including pulmonary embolism, deep vein thrombosis, and arterial thrombosis), whereas the EMA Committee for Medicinal Products for Human Use (CHMP) states that tofacitinib should be used with caution in patients with known risk factors for venous thromboembolism, regardless of indication and dosage. Questions emerged whether increased venous thromboembolic events seen in certain trials with JAKi can be attributed to the treatment, the disease, including disease activity, and/or individual patient risk factors.
A number of JAKis are currently undergoing clinical development in the treatment of PsA and/or UC including upadacitinib, filgotinib, and TD-1473 (Table 4). In August 2019, upadacitinib was approved in the US for the treatment of adult patients with moderate-to-severe rheumatoid arthritis who have had an inadequate response to or are intolerant of methotrexate. 82 In December 2019, upadacitinib, alone or in combination with methotrexate, was approved in the EU for the treatment of adult patients with moderate-to-severe rheumatoid arthritis who have had an inadequate response to or are intolerant of one or more DMARDs. 83 Phase III clinical trials that will investigate novel JAKis in PsA and/or UC are planned or underway, and results are eagerly awaited (Table 2). 84 The development of peficitinib as a treatment for UC has been discontinued due to disappointing efficacy results. 85
Ongoing phase III clinical trials with JAK inhibitors in psoriatic arthritis and ulcerative colitis. 84

ADA, adalimumab; DMARD, disease-modifying antirheumatic drug; FLG, filgotinib; JAK, Janus kinase; MTX, methotrexate; NCT, Number of clinical trial; PsA, psoriatic arthritis; Tofa, tofacitinb; UC, ulcerative colitis; UPA, upadacitinib.
Conclusions and future perspectives
The management of PsA and UC should ideally both provide symptomatic relief and modify the course of the disease. Since the two diseases may also overlap patients may be evaluated in the context of a multidisciplinary management. In our experience, a multidisciplinary approach incorporating a rheumatologist and a gastroenterologist, in patients with both PsA and UC, improves the diagnostic and therapeutic management and outcome of these patients, and allows for more comprehensive care. 86 The development of novel therapies targeting the immune system has arisen from an improved understanding of the pathogenesis of these diseases. Tofacitinib, approved either for the treatment of PsA or UC, provides an alternative treatment option that should improve the management and quality of life of adult patients living with these debilitating diseases. Its efficacy and safety profile in patients with an inadequate response to DMARDs or TNF inhibitors alongside its oral bioavailability should position tofacitinib as a potentially viable option in future treatment algorithms for patients with PsA or UC. Data on real-world life will allow the ideal positioning of the treatment. Novel therapies that target IL-23 and JAK are under investigation to provide additional therapies for physicians and ultimately offer clinically meaningful benefits to patients with PsA or UC. Maximizing treatment efficacy whilst improving the safety and tolerability of these new anti-IL-23 agents and JAKis is key to their future use.
Footnotes
Acknowledgements
We would like to thank Melanie Gatt (Ph.D.) and Gayle Robins, independent medical writers, for medical writing support on behalf of Springer Healthcare Communications.
Conflict of interest statement
Maria Sole Chimenti, Paola Coniglilaro, Livia Biancone, and Roberto Perricone received honoraria from Springer Healthcare in connection with the development of this manuscript.
Ethical statement
Our study did not require an ethical board approval because it is a review of the literature.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Pfizer provided funding for medical writing.
