Abstract
Background:
Randomized clinical trials have demonstrated the efficacy of secukinumab (SECU) in reducing disease activity in psoriatic arthritis (PsA), while real-world studies prove a broader perspective on SECU’s usefulness in everyday clinical practice.
Objectives:
To assess the effectiveness of SECU by evaluating drug survival and identifying potential predictors of clinical response and treatment discontinuation in patients with moderate-to-severe PsA, using real-world data from the Italian Group for the Study of Early Arthritis (GISEA) registry.
Design:
This longitudinal retrospective study included PsA patients treated with SECU, spanning from May 2016 to November 2023.
Methods:
Data from 1045 PsA patients, including 783 with peripheral-only PsA (perPsA) and 262 with peripheral and axial involvement (mixed PsA) were analyzed. Drug survival was estimated by Kaplan–Meier analysis. Clinical outcomes, including Disease Activity Index for Psoriatic Arthritis (DAPSA), Psoriasis Area Severity Index (PASI), Ankylosing Spondylitis Disease Activity Score (ASDAS, C-Reactive Protein (CRP)-based), and Visual Analogue Scale (VAS) measures, were evaluated at baseline and at 6, 12, and 24 months. Adjusted hazard ratios (aHRs) for discontinuing SECU were determined using multivariate Cox regression models.
Results:
SECU survival at 24 months was 63.24%, significantly higher in mixed PsA compared to perPsA (p = 0.036). In the overall PsA population, DAPSA scores decreased significantly at 6 months, and further at 24 months (all p < 0.0001). In mixed PsA, ASDAS-CRP scores were significantly reduced at 6 months and remained stable through 24 months (all p < 0.0001). VAS pain scores also improved already at 6 months and continued to improve at 24 months (all p < 0.0001). Higher age (aHR = 0.98, 95% confidence interval (CI): 0.96–0.99, p = 0.007) and lower baseline DAPSA scores (aHR = 1.02, 95% CI: 1.01–1.03, p = 0.014) were associated with greater persistence of SECU treatment. SECU was well tolerated, with no serious adverse events.
Conclusion:
SECU showed sustained clinical improvements in both peripheral and axial involvement of PsA patients over 24 months, with higher persistence observed in mixed PsA patients. Our findings highlight the favorable clinical and safety profile of SECU in real world.
Introduction
Psoriatic arthritis (PsA) is a chronic, systemic inflammatory disease affecting the joints, skin, and nails, significantly impairing patients’ quality of life. 1 This condition may be characterized by peripheral arthritis, axial involvement, dactylitis, enthesitis, and psoriasis. 2 These different manifestations may contribute to a multifaceted disease burden, marked by pain, stiffness, and functional impairment, which interfere with daily activities and overall well-being. 3 The heterogeneous phenotype of PsA complicates its management, requiring a comprehensive treatment approach to address the wide range of disease manifestations. 4
The advent of biologic therapies has changed the treatment landscape for PsA, especially for patients inadequately responsive to conventional synthetic disease-modifying antirheumatic drugs (csDMARDs) and non-steroidal anti-inflammatory drugs (NSAIDs). 5 Recent studies have shed light on the pivotal role of interleukin-17 (IL-17), a key pro-inflammatory cytokine in PsA pathogenesis. Dysregulation of the immune system, influenced by genetic predisposition and driven by environmental factors, leads to the overproduction of IL-17 mainly produced by Th17 cells. This excess of IL-17 triggers a cascade of inflammatory events, promoting joint inflammation, bone erosion, and new bone formation.6,7 Among biologic DMARDs, secukinumab (SECU), a monoclonal antibody that specifically neutralizes IL-17A, stands out as a highly effective treatment option for PsA. However, a major challenge in managing PsA lies in diagnosing the disease during its subclinical and early phases in patients with psoriasis. Early identification and timely use of IL-17 inhibitors, such as SECU, may help prevent the progression from skin to joint disease, potentially altering the disease course and reducing long-term damage.8,9 In this regard, randomized clinical trials (RCTs) have demonstrated the efficacy and safety of SECU in improving clinical outcomes for PsA patients, with significant reductions in symptoms and disease activity.10,11 While RCTs provide valuable insights, they partially capture the complexities and variabilities of PsA patients encountered in everyday clinical practice. In contrast, real-world studies complement clinical trial data, providing a broader perspective on SECU performance in routine healthcare settings, helping clinicians make informed treatment decisions based on evidence mirroring current clinical practice.12,13 These studies also provide valuable information on patient adherence, satisfaction, and the drug’s overall tolerability.14–16 Additionally, real-world data can help identify specific patient profiles that may benefit most from SECU, enabling more personalized treatments, especially in a protean disease like PsA.12–16
In this context, the Italian Group for the Study of Early Arthritis (GISEA) Registry, a large database that tracks long-term outcomes in patients with inflammatory arthropathies treated with biologic DMARDs, provides an invaluable resource for examining real-world characteristics in terms of disease activity, comorbidities, and treatment survival among PsA patients. 17 This observational study leverages the GISEA dataset to assess SECU effectiveness by exploring drug survival and potential predictors of clinical response and treatment discontinuation in moderate-to-severe PsA patients over time.
Materials and methods
Study design and patients’ enrolment
This is a retrospective cohort study performed using data from the GISEA registry, including patients from Italian rheumatology outpatient clinics listed in the registry (for details, see Supplemental Table 1). Patients were consecutively selected, aged 18 years or older, and had a diagnosis of PsA according to the CASPAR criteria. 18 Axial involvement was assessed based on the ASAS criteria for axial spondyloarthritis. 19 Patients were either biologic-naïve or had shown an inadequate response to previous biologic DMARDs and began treatment with SECU between May 2016 and November 2023. Patients were followed until the end of the observation period (November 30, 2023) or until censored due to remission, pregnancy, or loss to follow-up. All patients received SECU subcutaneously at a dosage of 150 or 300 mg, as per national registration indications and at the discretion of the treating rheumatologist. Most patients presented with moderate-to-severe active PsA at the initiation of SECU treatment. However, SECU was also prescribed for some patients to manage uncontrolled skin symptoms, while others, although in remission, required a switch to SECU due to adverse events (AEs) with prior conventional or biologic DMARDs. Outcomes were evaluated by the rheumatologist at each center where the patients were treated, and not all the authors were directly involved in the assessment of the outcomes. This study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 20
Clinical assessment and outcome measures
Data collected included demographic, clinical, biochemical, and metrological information, such as age, gender, weight, height, HLA-B27 status for axial PsA, C-Reactive Protein (CRP), Erythrocyte Sedimentation Rate (ESR), and disease duration. Arthritis was categorized as peripheral, axial, and mixed (peripheral and axial). Extra-articular manifestations (psoriasis, ocular, inflammatory bowel diseases) and comorbidities (classified using ICD-9-CM codes) were also recorded. Body mass index (BMI) was calculated and categorized according to WHO classifications: underweight/normal weight (BMI <25.0 kg/m2), overweight (25.0 kg/m2 ⩽ BMI < 30 kg/m2), and obese (BMI ⩾30.0 kg/m2). Information on previous biologic DMARDs, concomitant csDMARDs, and corticosteroids or NSAIDs therapy was also collected.
Patient-reported outcomes (PROs) included the Visual Analogue Scale for pain (VAS pain, 0-100), Patient Global Assessment (VAS PtGA, 0-100), Physician Global Assessment (VAS PhGA, 0-100), Health Assessment Questionnaire-Disability Index (HAQ-DI, 0-3), and Bath Ankylosing Spondylitis Disease Activity Index (BASDAI, 0-10). Clinical evaluations encompassed the Psoriasis Area Severity Index (PASI, 0-72), assessment of psoriatic onychopathy (yes/no), tender joint count (TJC, 0-68), and swollen joint count (SJC, 0-66). Patients with peripheral involvement were categorized based on their Disease Activity Index for Psoriatic Arthritis (DAPSA) scores as follows: remission (DAPSA ⩽4), low disease activity (LDA; DAPSA >4 and ⩽14), moderate disease activity (DAPSA >14 and ⩽28), or high disease activity (DAPSA >28). 21 For patients with axial involvement, disease activity was categorized based on their Ankylosing Spondylitis Disease Activity Score (ASDAS, CRP based) as follows: inactive disease (ID) (<1.3), LDA (1.3 to <2.1), high disease activity (2.1–3.5), and very high disease activity (>3.5).22,23 Enthesitis was assessed using the Leeds Enthesitis Index (LEI, 0–6), and dactylitis was measured by the number of affected digits (0–20). Minimal disease activity (MDA) or very LDA (VLDA) criteria were met if patients achieved five (MDA) or seven (VLDA) of the following seven outcomes 24 : TJC68 ⩽1, SJC66 ⩽1, LEI ⩽1, PASI score ⩽1, patient VAS pain ⩽15 mm, VAS PtGA ⩽20 mm, and HAQ-DI score ⩽0.5. The proportions of patients achieving DAPSA remission/LDA, MDA, VLDA, and ASDAS-CRP ID/LDA were assessed at each time point. Patient evaluations were performed at baseline and after 6, 12, and 24 months of SECU treatment.
Statistical analysis
Data were entered into a database created with an Excel spreadsheet and analyzed using Stata MP18 software. Continuous variables are described as mean ± standard deviation (SD) and range, or median and interquartile range (IQR), while categorical variables are described as percentages. The skewness and kurtosis test were used to evaluate the normality of continuous variables. When the data were not normally distributed, a normalization model was applied, when possible. The student’s t-test for independent data or the Wilcoxon rank-sum test was used to compare continuous variables between two groups. The repeated measures ANOVA or the Friedman test were used to compare continuous variables across detection times, depending on the data distribution. Repeated measures mixed models were used to compare continuous variables between groups and detection times, followed by post hoc analysis using the test of simple effects to estimate the variation of each outcome for each group per detection time. The Chi-square test or Fisher’s exact test was employed to compare proportions between groups and detection times.
To assess the determinants of several outcomes, univariate logistic regression was used. The variables considered relevant comprised age, sex, axial involvement, disease duration, baseline scores for DAPSA, LEI, HAQ-DI, and VAS pain, csDMARD use (yes/no), steroid use (yes/no), BMI, SECU treatment line, and the presence of comorbidities and fibromyalgia. The odds ratio (OR) with 95% confidence intervals (CI) was calculated. Subsequently, for each outcome, a multivariate logistic regression was built using age, sex, and variables statistically significant related in the univariate regression as determinants. The adjusted OR (aOR) with 95% CI was calculated. The Hosmer–Lemeshow test was performed to evaluate the goodness-of-fit of the multivariate logistic regression models. SECU persistence was calculated from the first dose to treatment interruption and assessed using Kaplan–Meier curves. Group differences were analyzed with the log-rank test. Patients lost to follow-up or those who stopped therapy for reasons unrelated to SECU (such as pregnancy, relocation, clinical remission, or other reasons) were right censored. Reasons for SECU discontinuation included primary failure (lack of response within 6 months), secondary failure (loss of effectiveness after initial response), AEs, and unknown reasons. Determinants of SECU discontinuation were identified using univariate Cox semiparametric regression, with several variables serving as risk predictors. The hazard ratio (HR) with 95% CI was calculated. Subsequently, a multivariate Cox semiparametric regression was built using age, sex, and variables statistically significant related in the univariate regression as determinants. The adjusted HR (aHR) with 95% CI was calculated. The Schoenfeld and scaled Schoenfeld residuals test was employed to assess the proportionality assumption of the multivariate Cox semiparametric regression model, and the Gronnesby and Borgan test were used to evaluate the model’s goodness-of-fit. A two-sided p-value <0.05 was considered statistically significant for all tests.
Results
Demographic, clinical, and therapeutic characteristics
The study included a total of 1045 PsA patients, of whom 783 had peripheral-only PsA (perPsA) and 262 both axial and peripheral involvement (mixed PsA). For 11 of these patients, data on peripheral involvement were unspecified. As detailed in Table 1, sex, disease duration, and BMI categories did not differ significantly between the perPsA and mixed PsA groups. Similarly, the prevalence of psoriasis or a history of psoriasis, as well as comorbid fibromyalgia, were comparable between the two subgroups. However, the median number of other comorbidities was higher in the mixed PsA group (p = 0.025). Glucocorticoid use was more common in perPsA (p = 0.052). Additionally, csDMARDs were used by 40.6% of perPsA patients and 31.3% of mixed PsA patients (p = 0.007). There were no significant differences in the proportion of biologic-naïve patients or inadequate responders to previous biologic DMARDs between the perPsA and mixed PsA groups (Table 1).
Demographic and clinical characteristics of patients with psoriatic arthritis stratified by mixed and peripheral-only involvement.

BMI, body mass index; cDMARDs, conventional disease-modifying anti-rheumatic drugs; IQR, interquartile range; perPsA, peripheral psoriatic arthritis; PsA, psoriatic arthritis; SD, standard deviation; SECU, secukinumab.
Drug persistence and risk predictors for SECU discontinuation
The overall drug survival for SECU was 63.24% at 24 months (Figure 1(a)). SECU persistence varied slightly by treatment line (Figure 1(b)), with first-line therapy showing a higher, though not statistically significant persistence (p = 0.083). Drug persistence did not significantly differ among patients with none, one, or multiple comorbidities (p = 0.573; Figure 1(c)). However, patients with mixed PsA had significantly higher SECU persistence compared to those with perPsA (p = 0.036; Figure 1(d)). A Cox regression analysis was performed to evaluate the potential baseline predictors of SECU discontinuation. Greater age was found to be associated with a lower likelihood of stopping SECU (aHR = 0.98, 95% CI: 0.96–0.99, p = 0.007), while higher baseline DAPSA scores were linked to an increased risk of drug discontinuation (aHR = 1.02, 95% CI: 1.01–1.03, p = 0.014). The use of SECU as a third-line or later treatment increased the risk of discontinuation compared to first-line treatment (aHR = 1.60, 95% CI: 1.11–2.33, p = 0.013). Although axial involvement appeared to be a protective factor in the univariate analysis, it was not significant in the multivariate model (aHR = 0.95, 95% CI: 0.67–1.34, p = 0.758; Table 2).

Persistence on SECU treatment in the entire cohort (a), SECU line of treatment (b), comorbidities (c), mixed PsA (d).
Univariate and multivariate Cox semiparametric regression analyses of the risk predictors for SECU discontinuation.

Goodness-of-fit p-value = 0.805.
aHR, adjusted hazard ratio; BMI, body mass index; CI, confidence interval; DAPSA, disease activity index for psoriatic arthritis; DMARDs, disease-modifying anti-rheumatic drugs; HAQ, Health Assessment Questionnaire; LEI, Leeds Enthesitis Index; HR, hazard ratio; SECU, secukinumab; VAS pain, Visual Analog Scale for pain.
Therapeutic effectiveness of SECU in the overall PsA population
Compared to baseline, a significant decrease in disease activity was observed over time. At 6 months, the composite DAPSA score dropped from 19.1 to 13.9 (p < 0.0001), continuing to improve to 12.0 at 24 months (p < 0.0001). PASI scores improved from 2.0 to 0.7 at 6 months (p < 0.0001), remaining low at 24 months (p < 0.0001). Joint involvement showed similar improvements, with TJC68 and SJC66 decreasing significantly at 6 and 24 months (p < 0.0001; Table 3). VAS pain scores reduced from 57.6 to 46.8 at 6 months (p < 0.0001) and to 40.7 at 24 months (p < 0.0001). There was also a simultaneous reduction in VAS PtGA scores from 57.2 to 47.1 at 6 months (p < 0.0001) and to 39.3 at 24 months (p < 0.0001), and in VAS PhGA scores from 41.0 to 30.4 at 6 months (p < 0.0001) and to 22.4 at 24 months (p < 0.0001; Table 3). Overall, SECU effectively managed both skin and joint symptoms in PsA, providing sustained benefits across treatment lines, especially for articular involvement (Supplemental Tables 2 and 3).
Therapeutic effectiveness of SECU in the overall PsA population.

CRP, C-reactive protein; DAPSA, Disease Activity index for Psoriatic Arthritis; ESR, Erythrocyte Sedimentation Rate; PASI, Psoriasis Area and Severity Index; PsA, psoriatic arthritis; SD, Standard Deviation; SECU, Secukinumab; SJC66, Swollen Joint Count (66 joints); TJC68, Tender Joint Count (68 joints); VAS pain, Visual Analog Scale for pain; VAS PhGA, Visual Analog Scale for Physician’s Global Assessment; VAS PtGA, Visual Analog Scale for Patient’s Global Assessment.
Disease targets
At 6 months, 14.1% of patients achieved DAPSA remission, and 43.5% reached LDA (p < 0.0001). At 12 months, these rates increased to 19.2% for DAPSA remission and to 44% for LDA (p < 0.0001 from baseline). At 24 months, the percentages remained approximately stable with 18.3% for DAPSA remission and 47% for LDA (p < 0.0001 from baseline). Moreover, the percentage of patients achieving MDA rose significantly to 17.2% at 6 months (p < 0.0001 compared to baseline), peaking at 22.7% at 12 months (p < 0.0001 compared to baseline). Although the percentage slightly decreased to 19.5% at 24 months, it remained significantly improved from baseline (p < 0.0001; Figure 2(a)).

Percentage of patients in DAPSA remission, DAPSA LDA, and MDA (a). Percentage of patients in ASDAS-CRP ID and ASDAS-CRP LDA (b).
Predictors of DAPSA remission/LDA and MDA
Key predictors for achieving DAPSA remission, LDA (Supplemental Table 4), and MDA (Supplemental Table 5) were evaluated at 6 months. The variables considered relevant for building the multivariate logistic regression models are reported in the “Materials and methods” section under Statistical analysis. Male sex significantly increased the likelihood of achieving DAPSA remission or LDA (aOR: 1.93, 95% CI: 1.06–3.52, p = 0.031). Lower baseline DAPSA scores were also associated with a higher probability of these favorable outcomes (aOR: 0.95, 95% CI: 0.92–0.99, p = 0.015). However, no significant predictors for achieving MDA were identified in the multivariate analysis (Supplemental Table 5).
Therapeutic effectiveness of SECU in the mixed PsA group
All clinical changes reported over time were compared to baseline values. The mean ASDAS-CRP score decreased from 2.6 to 2.0 at 6 months (p < 0.0001) and remained stable at 24 months (p < 0.0001). Similarly, the BASDAI score improved from 5.0 to 4.0 at 6 months (p < 0.0001), steadying at 4.2 at 24 months (p < 0.0001). SECU also significantly reduced disease activity in peripheral joints within the mixed PsA group (Table 4). For psoriasis severity, the PASI score decreased from 1.6 to 0.4 at 6 months (p < 0.0001), with a slight increase to 0.6 at 24 months (p < 0.0001). Notably, both patient and physician-reported outcomes showed consistent improvements during SECU treatment (Table 4). Overall, SECU effectively reduced disease activity and improved clinical outcomes in mixed PsA patients, particularly when used as a first-line treatment (Supplemental Tables 6 and 7).
Therapeutic effectiveness of SECU in the mixed PsA group.
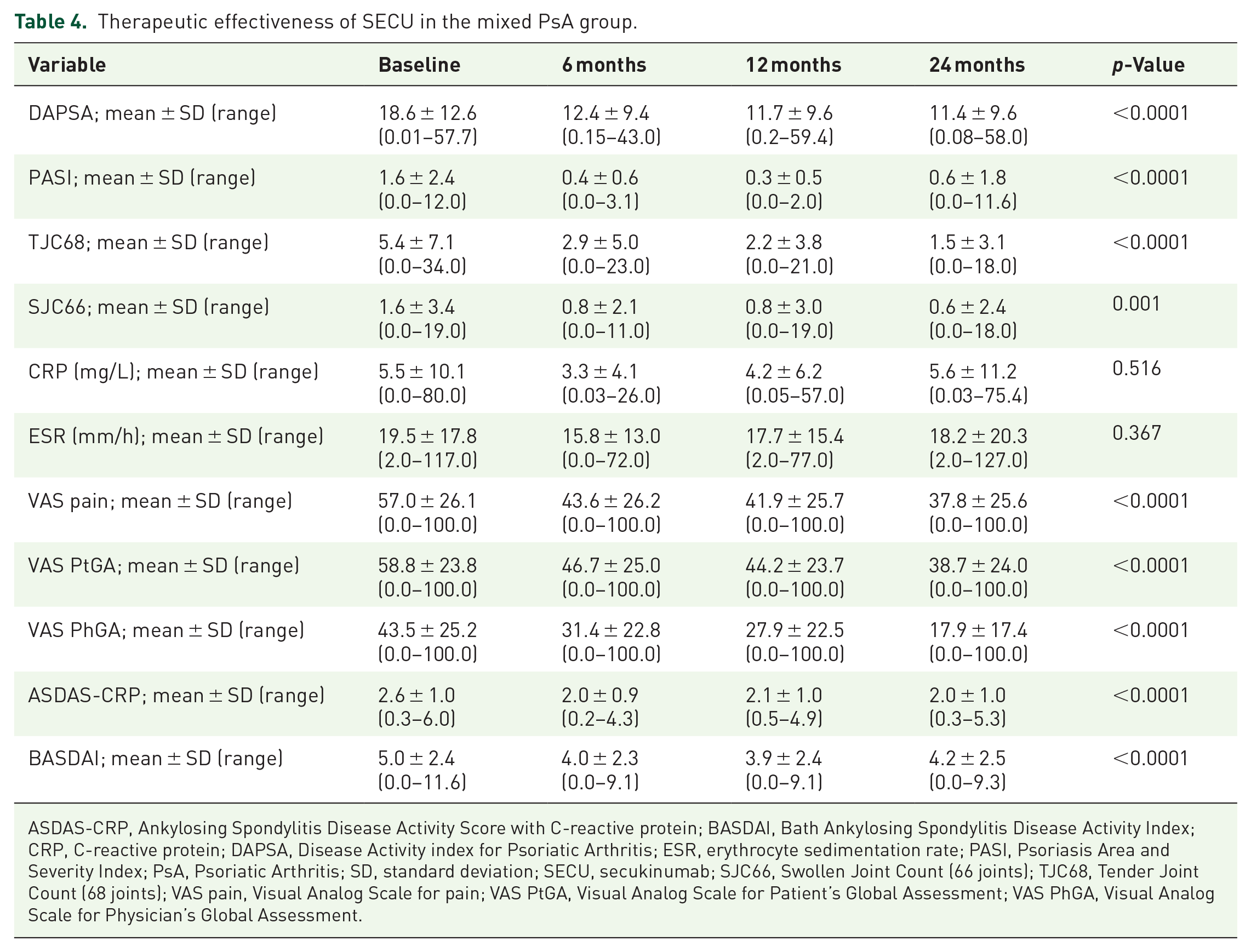
ASDAS-CRP, Ankylosing Spondylitis Disease Activity Score with C-reactive protein; BASDAI, Bath Ankylosing Spondylitis Disease Activity Index; CRP, C-reactive protein; DAPSA, Disease Activity index for Psoriatic Arthritis; ESR, erythrocyte sedimentation rate; PASI, Psoriasis Area and Severity Index; PsA, Psoriatic Arthritis; SD, standard deviation; SECU, secukinumab; SJC66, Swollen Joint Count (66 joints); TJC68, Tender Joint Count (68 joints); VAS pain, Visual Analog Scale for pain; VAS PtGA, Visual Analog Scale for Patient’s Global Assessment; VAS PhGA, Visual Analog Scale for Physician’s Global Assessment.
Achievement of ID/LDA and BASDAI < 4
Significant increases were observed in the proportions of patients achieving ASDAS-CRP ID or LDA over time. For ID, the percentage increased from baseline to 18.8% at 6 months (p = 0.001), further to 21.6% at 12 months (p < 0.0001), and 22.6% at 24 months (p = 0.023). Similarly, for LDA the percentage rose from baseline to 32.3% at 6 months (p = 0.001), then slightly decreased to 29.7% at 12 months (p < 0.0001) and 28.3% at 24 months (p = 0.023; Figure 2(b)). The percentage of patients with a BASDAI score < 4 increased significantly from baseline to 48.6% at 6 months (p < 0.0001), improved further to 49.5% at 12 months (p < 0.0001), and slightly decreased to 43.4% at 24 months, though it remained significantly higher than baseline (p = 0.002; data not shown).
Predictors of achieving ASDAS-CRP ID or LDA
A logistic regression analysis was performed to identify potential baseline predictors for achieving ASDAS-CRP ID and LDA (the relevant variables included in the models are reported in the “Materials and methods” section under Statistical analysis). VAS pain scores were identified as the only factor significantly associated with a higher likelihood of achieving ASDAS-CRP ID or LDA at the 6-month follow-up (aOR = 0.95, 95% CI: 0.92–0.99, p = 0.011; Supplemental Table 8).
Summary of SECU effectiveness in PsA management
SECU demonstrated broad effectiveness in managing PsA, with marked improvements in joint symptoms, pain, and overall disease activity that were sustained up to 24 months. Many patients reached LDA or remission, particularly in the articular domains. In patients with axial involvement, SECU also effectively reduced disease activity and improved PROs, showing particular benefit as a first-line treatment. Figure 3 depicts a flow diagram showing patient cohort selection, treatment persistence, and effectiveness outcomes for SECU.

Flow diagram showing patient cohort selection, treatment persistence, and clinical outcomes for SECU in PsA.
SECU discontinuation and safety profile
SECU treatment was generally well tolerated across the PsA cohort. Of the 1045 patients, 346 eventually discontinued therapy. Among these, 5.8% of all patients (17.6% of those who discontinued) stopped due to primary ineffectiveness, while 11.7% of the total cohort (35.6% of those who discontinued) suspended treatment due to secondary ineffectiveness. AEs led to treatment cessation in 2.2% of the entire cohort (6.9% of those who discontinued), with specific cases including three instances of paradoxical psoriasis, two cases of gastrointestinal discomfort, and one case of Crohn’s disease. Additionally, 13.2% of all patients (39.9% of those who discontinued) stopped SECU for reasons that were unknown. Notably, no serious AEs was reported, and there were no significant differences in reasons for discontinuation between patients with perPsA and those with mixed PsA (Table 1 and Figure 3).
Discussion
This study aimed to assess the real-world effectiveness of SECU in patients with PsA, with a focus on clinical outcomes for both peripheral and axial involvement. Our findings revealed a higher prevalence of comorbidities among mixed PsA patients compared to perPsA patients. Notably, the ASAS-COMOSPA study, which included SpA patients with and without psoriasis, found that SpA patients with peripheral involvement and psoriasis had higher rates of cardiovascular comorbidities compared to those with axial involvement. However, within the group with axial involvement, those with psoriasis had a significantly higher prevalence of hypertension, dyslipidemia, and diabetes compared to those without psoriasis. This suggests that psoriasis is a factor strongly associated with comorbidities, regardless of the articular pattern. 25 These differences might also be attributed to the diverse ethnicities represented in the ASAS-COMOSPA study, which included participants from 22 countries across four continents (Africa, America, Asia, and Europe). 25
Regarding BMI, our analysis found no significant differences between groups, although a substantial proportion of patients were overweight or obese. This aligns with results from the Swiss Clinical Quality Management registry, which compared 1153 patients with axial PsA to 1478 with perPsA 26 and highlighted the prevalence of obesity in PsA 27 and its role in disease development. 28 This is particularly meaningful when considering that adipokines, especially leptin, may play a noteworthy role in the inflammatory processes of both obesity and PsA. 29 Obesity may negatively impact the efficacy of biologic DMARDs, particularly TNF inhibitors 30 ; however, we did not find any influence of BMI on SECU survival, whose rate was 63.24%, with higher persistence observed in mixed PsA patients.
Our research revealed a high likelihood of SECU survival, consistent with earlier studies,31–34 though, 12-month SECU persistence was generally higher in European registries33,34 compared to our cohort. Noteworthy, the percentage of patients receiving SECU as a second or subsequent line of treatment was higher in our cohort than the EuroSpA cohort (59% vs 55%). This could still be one of several factors influencing the observed persistence rates. 33 However, variations in patient demographics and comorbidities may provide further insight into the observed differences in SECU persistence. 33
A significant finding was that mixed PsA patients had higher drug survival rates than perPsA patients, suggesting greater perceived effectiveness or tolerability in this subgroup. Axial PsA has become a focus of increased interest in recent years. The prevalence of axial involvement in PsA patients varies widely, ranging from 25% to 70%, depending on the criteria used.35–40 Axial involvement typically presents with higher disease activity than peripheral arthritis, leading patients with axial PsA to suffer from more intense symptoms such as chronic back pain and stiffness. This greater disease burden may motivate patients to adhere more closely to treatment as they experience a rapid reduction in the severity of clinical manifestations and an improvement in clinical outcomes. 41 Furthermore, recent studies have highlighted the effectiveness of SECU in managing comorbid conditions such as psoriasis,42–44 which may be more severe in axial PsA. 40 This dual benefit not only contributes to the overall improvement in patient outcomes and quality of life but also reduces the need for NSAIDs and other conventional treatments such as methotrexate, emphasizing SECU comprehensive impact on disease management.42–44
Interestingly, our findings revealed that patients with increasing age and those with lower baseline DAPSA scores were more likely to continue SECU treatment. SECU was effective in both bio-naïve patients and those previously treated with other biologic DMARDs, although those using SECU as a third or further line of treatment had a higher risk of discontinuation. Similarly, exploratory subgroup analyses from various RCTs demonstrated that SECU provides benefits regardless of prior treatment with TNF inhibitors.45–49 Significant improvements were observed in several clinical parameters, including DAPSA, PASI, ASDAS-CRP, and VAS pain assessments, which are consistent with previous studies.50–52 These results underscore the coherent and robust effectiveness of SECU in improving clinical outcomes for PsA patients in real-world. Intriguingly, we found that for mixed PsA patients, a lower baseline VAS pain score was the only significant predictor for achieving ASDAS-CRP ID or LDA. This finding is aligned with data from the Corrona Psoriatic Arthritis/Spondyloarthritis Registry, which shows that higher pain levels are associated with worse disease outcomes, including a lower likelihood of reaching MDA and ASDAS-CRP LDA in patients with axial PsA. 40 The safety profile of SECU observed in our study demonstrates that it is generally well-tolerated, with only 6.9% of patients discontinuing due to AEs. Notably, no serious AEs were reported, and the most common AEs leading to discontinuation were paradoxical psoriasis, gastrointestinal discomfort, and one case of Crohn’s disease. These findings are consistent with earlier data, where SECU has shown a favorable long-term safety profile in patients with psoriasis and PsA. 53 In summary, our study highlights SECU’s role in effectively managing PsA, with notable reductions in disease activity and pain, alongside improved PROs sustained over 24 months in both disease peripheral and axial involvement. Key findings include a marked decrease in disease activity indices and greater SECU persistence in patients with mixed PsA compared to those with perPsA. Future studies should focus on larger, prospective cohorts to validate predictors of treatment response. Additionally, exploring biomarkers associated with treatment outcomes could enable a more individualized approach, further enhancing SECU effectiveness in the comprehensive management of PsA.
Conclusion
Our study provides valuable insights into the effectiveness of SECU in managing both peripheral and axial involvement of PsA, suggesting sustained improvement in patient outcomes and quality of life. SECU was also effective in patients previously treated with biologic DMARDs. Our results underscore SECU usefulness aligning with findings from prior studies.15,31,33,34,52,54,55 Limitations of our study include its retrospective design, which may introduce biases. The potential presence of incomplete data could have affected the results, although appropriate statistical methodologies were applied to ensure reliable and generalizable conclusions. Additionally, the presence of a mixed PsA cluster (axial and peripheral) and the lack of precise information on the exact number of patients with exclusively axial involvement, even if small, may have impacted the findings specific to this group. However, the strengths of the study are the real-world setting, which provides evidence reflecting routine clinical practice. The large cohort of PsA patients and comprehensive clinical assessments enable a thorough evaluation of SECU effectiveness across different phenotypes. Moreover, the 24-month follow-up offers robust data on drug persistence and patient outcomes. Future studies, examining specific patient demographics and comorbidities could offer deeper insights into the factors influencing differences in SECU persistence.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X251315138 – Supplemental material for Efficacy and retention rate of secukinumab in psoriatic arthritis across different clinical phenotypes: insights from the Italian GISEA Registry
Supplemental material, sj-docx-1-tab-10.1177_1759720X251315138 for Efficacy and retention rate of secukinumab in psoriatic arthritis across different clinical phenotypes: insights from the Italian GISEA Registry by Giuseppe Lopalco, Maria Morrone, Fabiola Atzeni, Chiara Bazzani, Francesco Paolo Bianchi, Francesco Paolo Cantatore, Roberto Caporali, Antonio Carletto, Alberto Cauli, Maria Sole Chimenti, Sergio Colella, Fabrizio Conti, Addolorata Corrado, Ennio Giulio Favalli, Alberto Floris, Marco Fornaro, Rosario Foti, Roberta Foti, Elena Fracassi, Bruno Frediani, Stefano Gentileschi, Roberto Gorla, Elisa Gremese, Emanuela Praino, Roberta Ramonda, Cinzia Rotondo, Marco Sebastiani, Angelo Semeraro, Gianfranco Ferraccioli, Giovanni Lapadula and Florenzo Iannone in Therapeutic Advances in Musculoskeletal Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
