Abstract
Background and Aims:
Glucosamine sulphate (GS) can be used as background therapy in people affected by knee osteoarthritis (OA). Knowledge regarding the efficacy and safety of GS is of importance since its use worldwide is increasing. Therefore, the present study aimed to map and grade the diverse health outcomes associated with GS using an umbrella review approach.
Methods:
Medline, Cinahl and Embase databases were searched until 1 April 2020. An umbrella review of systematic reviews and meta-analyses of randomized controlled trials (RCTs) was carried out. The evidence from the RCTs was graded using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) tool.
Results:
From 140 articles returned, 11 systematic reviews, for a total of 21 outcomes (37 RCTs; 3949 participants; almost all using 1500 mg/day), were included. No systematic reviews/meta-analyses of observational studies were included. Regarding the findings of the meta-analyses, 9/17 outcomes were statistically significant, indicating that GS is more effective than placebo. A high certainty of evidence, as assessed by GRADE, supported the use of GS (
Conclusion:
GS, when used as a prescription drug (i.e. crystalline glucosamine sulphate) at 1500 mg daily dosage, can positively affect the cartilage structure, reduce pain, improve function and glucose metabolism in people with knee OA, without having a greater incidence of adverse effects than placebo.
Introduction
Osteoarthritis (OA) is among the most common diseases in older people, defined here as those greater than or equal to 65 years. This condition is traditionally characterized by joint pain and stiffness, with relevant consequences on functional decline/disability and finally loss in quality of life.1,2 In this regard, knee OA is the most common localisation within the symptomatic forms of OA, affecting more than approximately 250 million people worldwide, with symptomatic forms occurring in 10% of men and 13% of women aged 60 years or older.3,4
Currently, there is no definitive treatment for knee OA. The current therapeutic approach combines nonpharmacological and pharmacological strategies that aim to improve function, decrease pain and, if possible, improve structural aspects, with limited adverse events. 5 In this regard, symptomatic slow-acting drugs for OA [such as chondroitin sulphate, glucosamine sulphate (GS), hyaluronic acid and diacerein] 6 are an important background therapy for people affected by knee OA.7–9 In this class of medications, however, different drugs exist exhibiting different pharmacological profiles.
Glucosamine is a natural compound, present in different preparations. Briefly, glucosamine hydrochloride is used as nutraceutical or over-the-counter (OTC) products. 10 In contrast, GS is obtained only by a proprietary semi-synthetic route and stabilisation process. 10 GS is used only as a prescription drug product, prescription crystalline glucosamine sulphate (pCGS). 10 However, multiple formulations of GS are available, 11 both as prescription-grade and OTC products, with the latter having small/varying amounts of glucosamine. 12 Moreover, GS is not available as a prescription-grade product in all countries. Importantly, there is extensive and increasing literature supporting the idea that only pCGS is able to deliver consistently high glucosamine bioavailability and plasma concentration in humans.13,14 In these experimental studies, the measurement of glucosamine concentration in patients affected by knee OA was also made as a plasma peak (7.17 µM) and as a site of action concentration (synovial fluid) equal to 4.34 µM. 14 Plasma and synovial pCGS concentrations are highly correlated and both are in 10 µM range, a cut-off that seems important for some actions of pCGS, such as an anti-inflammatory effect 15 that finally results in clinical efficacy.5,16–22
In addition to the use of GS for people affected with knee OA, GS may be an appropriate treatment for other conditions. For example, GS is used in hip OA23,24 or in other forms of OA.25–27 Moreover, the difference in efficacy and adverse effects incidence by prescription and OTC doses is still unclear.
The aim of the present work was to evaluate, through an umbrella review, the strength and credibility of the evidence derived from systematic reviews and meta-analyses of observational and/or intervention studies, that is, randomised controlled trials (RCTs), and obtain a general summary of their importance relative to health outcomes and adverse effects, in order to inform policies on the use of GS in humans.
Methods
This work followed a preplanned protocol (PROSPERO link: CRD42020179570). The Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) 28 recommendations and specific guidelines regarding how to conduct an umbrella review 29 were followed for the reporting of this study.
Data sources and searches
An umbrella review was carried out, 30 systematically searching the Medline, Cinahl and Embase databases from inception until 1 April 2020, using the terms ‘systematic reviews/meta-analyses’ and ‘glucosamine’, as free vocabulary words and/or controlled terms specific to each database, on a central platform hosted at Anglia Ruskin University. Reference lists of eligible articles and reviews in this field were also searched, including systematic reviews and meta-analyses under process.
Eligibility and selection criteria
We included systematic reviews, with or without formal meta-analysis of RCTs, in which at least one group used GS and one placebo reporting on health outcomes, both in terms of efficacy and safety. Only subjects taking GS only (not in combination with other medications)
Two reviewers (NV, JD) independently screened title/abstracts and full texts for eligibility, and when a consensus was not reached a third reviewer (SM) was consulted.
Data extraction
The following information was extracted: PMID/DOI, first author’s name, year of publication, study design (cohort, case control, RCT), number of included studies in each systematic review, the specific population under investigation (i.e. general population, subjects with OA and its location, etc.), the dosage of GS, the health outcome(s), the median follow-up period (in months), and for RCTs the risk of bias in included studies, according to the Cochrane review indications (high, unclear, low). 31 If an article presented separate meta-analyses on more than one reported outcome, each one was assessed separately. Duplicated data from same papers were eliminated including only the largest sample size of the RCTs.
Next, the RCT-specific estimated estimates for any adverse effects or negative outcome for both systematic reviews and meta-analyses outcomes [risk ratio, odds ratio (OR), hazard ratio, incident risk ratio, standardized mean differences (SMDs), mean differences (MDs)], along with their 95% confidence intervals (CIs), were extracted.
Outcomes
Any efficacy/effectiveness outcome, adverse events or adverse effects potentially associated with GS use was included.
Risk of bias assessment
The methodological quality of each included systematic review was assessed using the Assessment of Multiple Systematic Reviews (AMSTAR) 2 tool (available at https://amstar.ca/Amstar-2.php), which is a recent update of AMSTAR, 32 by two independent investigators (JD, NV). The AMSTAR 2 ranks the quality of a systematic review from critically low to high according to 16 predefined items.
Data synthesis and analysis
For each meta-analysis, we re-estimated the summary effect size and its 95% CIs under the assumption of a random-effects model. If the re-calculated effect size differed from the published effect size (e.g. in case of the use of a fixed-effects model instead of a random-effects model), we keep the re-calculated estimations. After the data extraction, we re-calculated the overall summary effect size, double checking with the original published ones. We also estimated the prediction intervals (PIs), which further accounts for between-study effects and estimates the certainty of the association if a new study addresses that same association.33–35 Between-study inconsistency was estimated with the
All statistical analyses were conducted in Stata Statistical Software, version 14.0 (StataCorp., College Station, TX, USA).
Grading the evidence
Evidence from meta-analyses of RCTs was assessed in terms of the significance of the summary effect, using a
Main descriptive findings of the systematic reviews included.

GS, glucose sulphate; JS, joint space; JSW, joint space width; K–L, Kellgren and Lawrence; KOA, knee osteoarthritis; OA, osteoarthritis; RCT, randomised controlled trial; TMJ OA, temporomandibular joint osteoarthritis; VAS, visual analogue scale; WOMAC, Western Ontario and McMaster Universities Arthritis Index.
Sensitivity analyses
For outcomes of observational studies having a class I/II evidence, it was planned to conduct sensitivity analyses including only cohort studies. Moreover, for the outcomes of RCTs, it was planned to stratify analyses for risk of bias of the RCTs included using the original data if possible or evaluating the risk of bias using the Cochrane tool for risk of bias if not available in the original meta-analysis. Finally, it was planned to stratify the analyses of RCTs by prescription and not prescription doses. However, no observational studies were included and only prescription doses (i.e. >750 mg/day) 40 were used. Only for one outcome (pain in OA) was it possible to run a sensitivity analysis, removing the RCTs at high risk of bias.
Results
Literature review
The initial search yielded 180 articles. After removing the duplicates, study selection commenced and 140 papers were evaluated, with 47 assessed as full text. As shown in the PRISMA flowchart (Figure 1), 11 articles were finally included,21,41–50 that is, 3 systematic reviews without meta-analysis, 1 network meta-analysis reporting narrative data on GS and 7 meta-analyses, for a total of 21 independent outcomes, as fully reported in Table 1. No systematic review or meta-analysis regarding observational studies was included, that is, no observational studies met the inclusion/exclusion criteria.
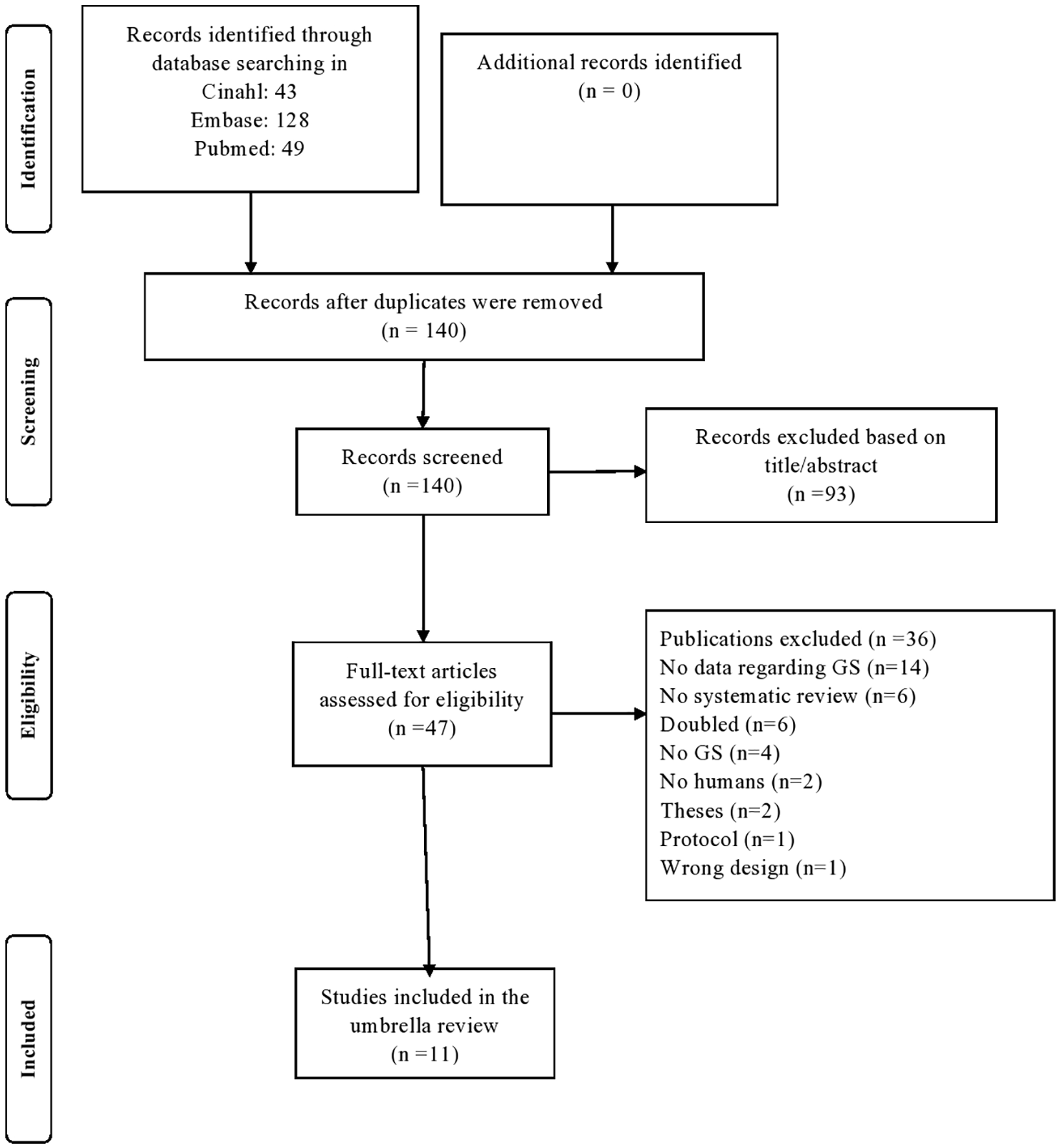
PRISMA flowchart for study selection.
Descriptive findings of the articles included
Table 1 summarises key descriptive findings regarding the 11 articles included. Overall, 37 independent RCTs for a total of 3949 participants (1987 randomised to GS and 1962 to placebo), mainly affected by knee OA with a Kellgren and Lawrence grade of 2 (definite osteophytes and possible narrowing of joint space) or 3 (moderate multiple osteophytes, definite narrowing of joint space, some sclerosis and possible deformity of bony ends) were included. For each article included, the mean number of RCTs included was 4 (range: 2–21) for a median of 414 participants (range: 18–2228). The median follow-up period was 3 months (range: 1.5–36). Almost all RCTs (30/37), used the dosage of 1500 mg daily.
Main findings of the meta-analyses
Among the 17 outcomes included in the meta-analyses, 9 were statistically significant (
Summary of the findings according to the GRADE tool for randomized controlled trials.
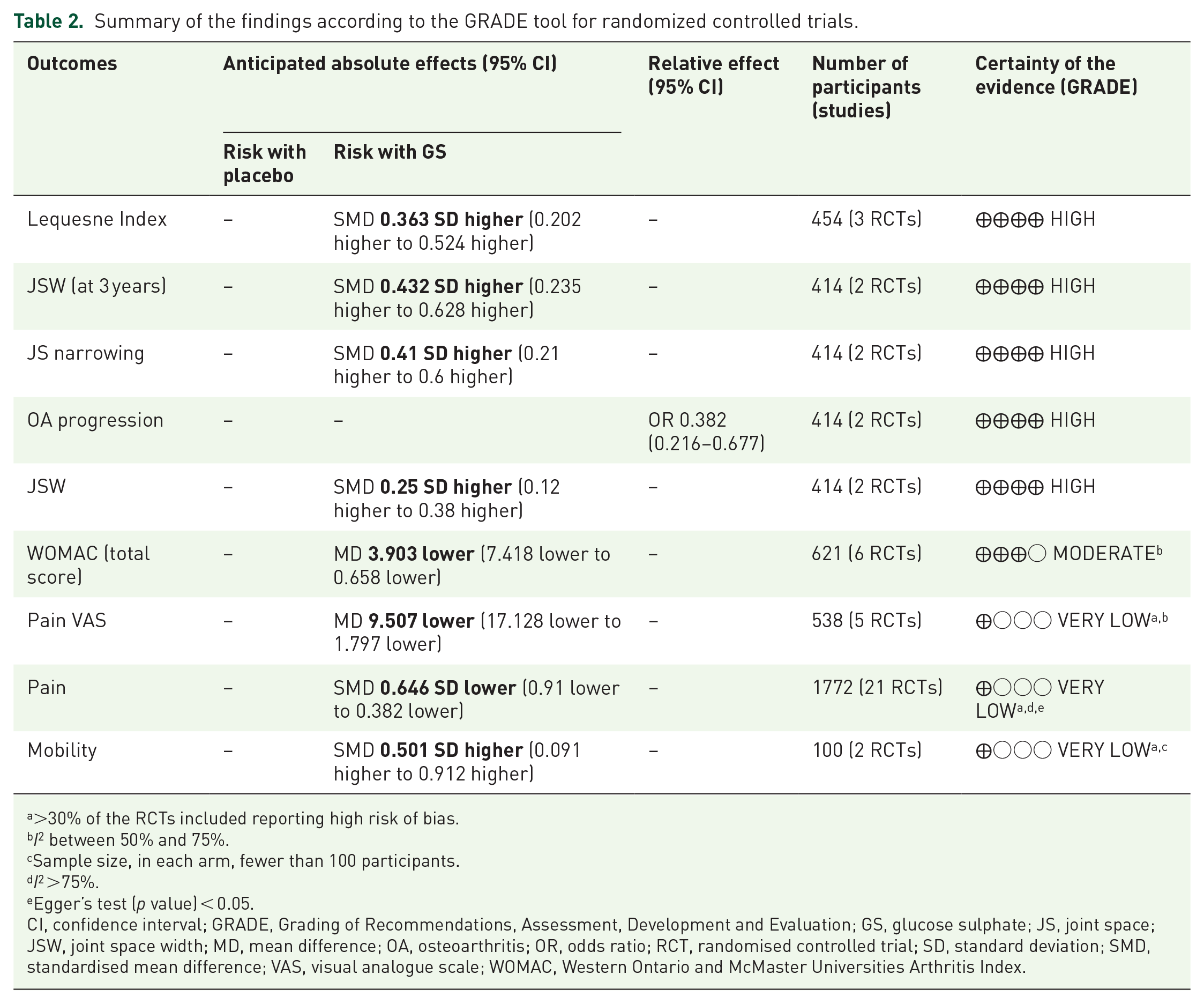
>30% of the RCTs included reporting high risk of bias.
Sample size, in each arm, fewer than 100 participants.
Egger’s test (
CI, confidence interval; GRADE, Grading of Recommendations, Assessment, Development and Evaluation; GS, glucose sulphate; JS, joint space; JSW, joint space width; MD, mean difference; OA, osteoarthritis; OR, odds ratio; RCT, randomised controlled trial; SD, standard deviation; SMD, standardised mean difference; VAS, visual analogue scale; WOMAC, Western Ontario and McMaster Universities Arthritis Index.
Table 2 shows the findings of the statistically significant outcomes using the GRADE approach, ranked by the level of evidence. A high certainty of evidence, as assessed by GRADE, supported the use of GS (
A sensitivity analysis was carried out for the outcome pain. A total of 10 RCTs at high risk of bias were removed, the re-calculated SMD was –0.298 (11 RCTs; n = 1493; 95% CI: –0.546; –0.05); however, no differences in terms of heterogeneity (
Of importance, as shown in Supplemental Table 2, no significant differences in terms of adverse events between GS and placebo were observed (5 RCTs; 632 participants; OR = 1.236; 95% CI: 0.623–2.454;
Findings from the narrative systematic reviews
As shown in Table 3, of the four outcomes included in the systematic reviews without meta-analysis, GS was associated with a better glucose profile 43 and a better physical function performance compared with placebo. 46 On the contrary, when people suffering from spine or temporomandibular joint OA were included no significant effect of GS on physical function or pain was observed.44,48
Main findings of the systematic reviews, without meta-analysis.

GS, glucosamine sulphate; KOA, knee osteoarthritis; OA, osteoarthritis; RCT, randomised controlled trial; TMJ OA, temporomandibular joint osteoarthritis.
Quality assessment
The assessment of the risk of bias in the meta-analyses included is fully reported in Supplemental Table 3. Four systematic reviews/meta-analyses were adjudicated as having high (i.e. one noncritical weakness) confidence of the results found, whilst, for the others, one was low (i.e. one critical flaw with or without noncritical weaknesses) and six were critically low (i.e. having more than one critical flaw with or without noncritical weaknesses).
Discussion
In this umbrella review, including 11 systematic reviews comprising 37 RCTs, the current research regarding GS and health outcomes in humans is reported. Overall, the findings suggest that GS is a safe product and, when used as prescription drug at 1500 mg/daily dosage, is able to positively modify the cartilage structure, reduce pain and improve function in people with knee OA, without having a greater incidence of adverse effects than placebo. The efficacy of GS was supported by different degrees of certainty of evidence, according to the GRADE evaluation, similar to that made in the 2019 European Society for Clinical and Economic Aspects of Osteoporosis, Osteoarthritis and Musculoskeletal Diseases (ESCEO) updated algorithm, which supports the use of prescription GS as background therapy for knee OA. 53 Altogether, our findings suggest that GS might provide clinical benefits at 1500 mg/daily; we should differentiate the formulation of GS that is essential for maximising the clinical benefit, patient adherence and satisfaction with treatment. 18
GS is widely used, particularly in older people, for the treatment of knee OA and its global use is increasing overall.54,55 The present umbrella review supports the assumption that, when compared with placebo, GS is able to delay the joint space narrowing, finally resulting in a minor OA progression. This analysis showed that in two RCTs,16,17 those randomised to GS had a 62% reduction in OA progression compared with those randomised to placebo. This evidence is supported by a high certainty of evidence, meaning that the role of potential biases is limited. These effects may be explained through several mechanisms. First, GS is able to reduce inflammatory parameters, in particular IL-1. 56 In this regard, GS, if it reaches appropriate doses in blood and cartilage cells, can positively interfere with the IL-1 intracellular signalling pathway and gene expression. 56 However, the dose-dependent effect of GS on IL-1β-induced gene expression of stromelysin-1 (MMP-3) and aggrecanase-2 (ADAMTS5) in human chondrocytes is optimised at clinically relevant concentrations (~10 μM) that can be obtained only at pharmacological doses of GS. 57 Through this mechanism, GS is able to reduce the degradation of cartilage, therefore improving the cartilage structure of the knee.
However, GS is able, according to this umbrella review, to improve clinical outcomes commonly affected in knee OA. In particular, it was found that GS is able to improve the Lequesne Index,
58
a tool that evaluates several aspects compromised in OA, including activities of daily living, pain and physical function. In this sense, this umbrella review indicates that GS is able to reduce pain and disability, however, this evidence is supported by a lower certainty of evidence according to GRADE. Traditionally, Cohen defined an effect size of 0.20 as small, 0.50 as moderate and 0.80 or greater as large.
59
Given this, the effect of GS, compared with placebo, is ranked between small and moderate. However, as already discussed in other relevant papers,7,11 these effects are almost doubled those of paracetamol,
60
a common medication used for knee OA pain-relief treatment. Moreover, other medications commonly used for the treatment of knee OA have a similar effect as observed for GS, as indicated in the recent network meta-analysis of Gregori
Another important aspect of this umbrella review is the potential association between GS and favourable glucose profile. Glucosamine, in fact, is an amino sugar, therefore, one might claim that this medication can lead to hyperglycaemia, insulin resistance and finally to diabetes by overactivating the hexosamine pathway. 61 In this sense, however, a large RCT comprising 407 overweight and obese women, followed up for 2.5 years, reported that there was no significant effect of a 2.5-year GS intervention on mean glycosylated haemoglobin level. 62 Present data, although limited by the narrative nature of the review, confirmed that GS is safe from a metabolic point of view, being in agreement with a large longitudinal study using the UK Biobank showing that the use of GS in OA is associated with a lower incidence of diabetes, over 8 years of follow up. 63 In the same database, it is reported that GS can lead to a reduction in cardiovascular disease 64 and all-cause and specific-cause mortality. 65 Further studies are encouraged in order to confirm these promising findings, since in the UK Biobank the data are reported for different glucosamine preparations and not specifically for GS. Regarding the mechanisms of action that can justify these epidemiological findings, we can argue that GS may interfere with some pro-inflammatory pathways such as nuclear factor kappa-B, mitogen-activated protein kinase and phosphatidyl-inositol-3-kinase-dependent pathways66,67 that are usually involved in the pathogenesis of diabetes. 68
Finally, our umbrella review is, in our opinion, important since GS use was associated with a similar incidence in adverse effects, compared with placebo, suggesting that its use is safe. One pivotal meta-analysis regarding this topic and included in this umbrella review, in fact, reports that the use of pCGS is not associated with a higher incidence of total and specific gastrointestinal, skin and subcutaneous tissue, renal and urinary adverse events when compared with placebo. 50 The topic of safety for medications is clinically relevant, particularly in older people, in which knee OA is widely diffused. It is known that the median age of knee OA detection is 55 years and typically people with this condition live about 30 years with the disease. 69 Therefore, to have medications with a good safety profile is of importance, but still debated in geriatric medicine since older people often use a high number of medications (polypharmacy) that may have unwanted interactions.
Findings from the present review should be interpreted in light of its limitations. First, the use of already established tools for quality assessment of evidence, which indirectly rely on the data reported in the selected articles, can cumulatively bring some biases. In order to overcome these potential biases we used low heterogeneity in the GRADE assessment as one of the criteria for high certainty of level. However,
In conclusion, the present umbrella review suggests that prescription GS, when used at 1500 mg/daily dosage, can positively affect the cartilage structure and improve the pain and function in people with knee OA, without having a greater incidence of adverse effects than placebo, indicating a possible role in older people. Moreover, some promising results indicated that GS is associated with a better glucose profile than placebo. Overall, these findings encourage further research regarding GS and other forms of OA not affecting the knee.
Supplemental Material
sj-pdf-1-tab-10.1177_1759720X20975927 – Supplemental material for Glucosamine sulphate: an umbrella review of health outcomes
Supplemental material, sj-pdf-1-tab-10.1177_1759720X20975927 for Glucosamine sulphate: an umbrella review of health outcomes by Nicola Veronese, Jacopo Demurtas, Lee Smith, Jean-Yves Reginster, Olivier Bruyère, Charlotte Beaudart, Germain Honvo and Stefania Maggi in Therapeutic Advances in Musculoskeletal Disease
Footnotes
Author contributions
NV: conceptualization; data curation; formal analysis; funding acquisition; methodology; writing original draft. JD: data curation; writing original draft. LS: data curation. J-YR: writing – review and editing. OB: writing – review and editing. CB: formal analysis; methodology. GH: formal analysis; methodology. SM: writing – review and editing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by an unrestricted grant from Mylan Company.
Conflict of interest statement
J-YR reports grants and personal fees from IBSA-Genevrier, Mylan, Radius Health and CNIEL, personal fees from Laboratoires Pierre Fabre, Faes Pharma, Rejuvenate Biomed, Samumed, Teva, Theramex, Pfizer, Mithra Pharmaceuticals, Dairy Research Council, Nutricia, Danone and AgNovos, and grants from TRB. GH reports lecture fees and travel support from IBSA. SM reports grants from Sanofi Pasteur, MSD, GlaxoSmithKline, Pfizer and Takeda as organiser of meetings/congresses and as principal investigator of epidemiological studies, for taking part on advisory boards and in expert meetings. OB reports grants from Biophytis, IBSA, Meda, Servier Laboratories and SMB, and personal fees from Amgen, Aptissen, Biophytis, IBSA, Meda, Novartis, Sanofi, Servier Laboratories, SMB and UCB. NV reports personal fees from Mylan, IBSA and Fidia.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
