Abstract
Background:
Early stage of osteoarthritis (OA) is characterized by joint stiffness and pain as well as by subclinical structural changes that may affect cartilage, synovium, and bone. At the moment, the lack of a validated definition of early osteoarthritis (EOA) does not allow to make an early diagnosis and adopt a therapeutic strategy to slow disease progression. Also, no questionnaires are available to evaluate the early stage, and therefore this remains an unmet need.
Objective:
Therefore, the purpose of the technical experts panel (TEP) of ‘International Symposium of intra-articular treatment’ (ISIAT) was to create a specific questionnaire to evaluate and monitor the follow-up and clinical progress of patients affected by early knee OA.
Design:
The items for the Early Osteoarthritis Questionnaire (EOAQ) were identified according to the following steps: items generation, items reduction, and pre-test submission.
Methods:
During the first step, literature has been reviewed and a comprehensive list of items about pain and function in knee EOA was drafted. Then, during the ISIAT (5th edition 2019), the draft has been discussed by the board, which reformulated, deleted, or subdivided some of the items. After the ISIAT symposium, the draft was submitted to 24 subjects affected by knee OA. A score based on the importance and the frequency was created and the items with a score ⩾0.75 were selected. After intermediate evaluation made by a sample of patients, the second and final version of the questionnaire EOAQ was submitted to the whole board for final analysis and acceptance in a second meeting (29 January 2021).
Results:
After an exhaustive elaboration, the final version of the questionnaire contains two domains (Clinical Features and Patients Reported Outcome) with respectively 2 and 9 questions, for a total of 11 questions. Questions mainly explored the fields of early symptoms and patients reported outcomes. Marginally, the need of the symptoms treatment and the use of painkillers were investigated.
Conclusions:
Adoption of diagnostic criteria of early OA is strongly encouraged and a specific questionnaire for the whole management of the clinical features and patients’ outcome might really improve the evolution of OA in the early stages of the disease, when the treatment is expected to be more effective.
Introduction
Osteoarthritis (OA) is a health burden that affects more than 300 million people across the world, 1 and is the leading cause of disability. The disease is characterized by joint pain, loss of function, and a decreased quality of life (QoL).2,3
Usually, OA management in not focused on the oligosymptomatic early stages that is characterized by milder intensity and less frequent symptoms. In this phase, the interventions are more likely associated with success both from a symptomatic and structural point of view.
In particular, early-stage OA is characterized by joint stiffness and pain as well as by subclinical structural changes that may affect cartilage, synovium, and bone, 4 which are detectable by imaging (X-ray and magnetic resonance – MRI). 5
Moreover, numerous risk factors are involved in the alteration and progressive joint damage,6–8 such as older age, obesity, and metabolic diseases or sex, since the disease prevalence is higher in women after 50, because of the cartilage hormone sensitivity and anatomical differences between genders. 9
At the moment, the lack of a validated definition of early OA (EOA) does not allow to make an early diagnosis and adopt therapeutic strategies to slow the disease progression. However, in the literature, some definitions are available and three constructs of EOA can be found: early occurrence of symptom, early onset in young adult patients, and initial radiological changes [Kellgren and Lawrence (KL) grade 0-1-2].10–13 In addition, OA is characterized by different phenotypes but their definitions are still discordant.6,14–16
In established stage of OA, some questionnaires may assess the disease severity and the response to treatment. However, no questionnaires are available to evaluate the early stage, and therefore this remains an unmet need. It has been known that, since the symptoms of EOA are quite similar but still different from the ones involved in the established OA (especially in frequency and intensity), nowadays there are no questionnaires available to deeply investigate the status and the progression of EOA.
Therefore, the purpose of the technical experts panel (TEP) of International Symposium Intra Articular Treatment (ISIAT) was to create a specific questionnaire to evaluate and monitor the follow-up and clinical progress of patients affected by early knee OA (KOA).
Materials and methods
Survey design
The items for the Early Osteoarthritis Questionnaire (EOAQ) were identified, according to the procedure used for the realization of other questionnaires, through the following steps: items generation, items reduction, and pre-test submission. 17 The reporting of this study conforms to the TRIPOD statement. 18
First step: literature review and first draft creation
During the first step, an exhaustive review of the literature was made and a comprehensive list of items about pain and function in EOA of knee was drafted. This list of items was obtained from generic OA questionnaires for patients suffering from KOA or knee pain.
Five subjects with EOA (KL 0-1) were interviewed in a face-to-face discussion about quality, degree, and recurrence of symptoms. All interviews, using open discussion and open-ended questions, were performed by the same clinical researcher. During these interviews, all subjects described symptoms related to KOA, and after that, the draft was modified accordingly. Finally, this list was completed by the feedback of the steering committee who received a semi-structured questionnaire.
Second step: draft correction and evaluation
During the ISIAT (5th edition 2019), the draft has been discussed by the board, which reformulated, deleted, or subdivided some of the items. During the meeting, the board selected the most pertinent items to be included in the final questionnaire, amending the items into clear, brief, unambiguous, and relevant questions. Redundancy of items was also taken into consideration throughout the item reduction process. During this step, the layout of the questionnaire and the response format were defined.
After the ISIAT symposium, the draft was submitted to 24 subjects affected by KOA (12 EOA KL grade 0-1, 12 established OA KL grade 3), different from those included in creation step, and to the experts. The grade of relevance of each item was voted on a 4-point Likert-type scale ranging from ‘1, not relevant’ to ‘4, extremely relevant’. Then, for each item, it was evaluated for its ‘frequency’ (the proportion of subjects or experts who identified the item as ‘extremely relevant’) and its importance (the mean importance score was based on the 1–4 Likert-type scale for this item). A score based on the importance and the frequency was created and the items with a score ⩾0.75 were selected.
Third step: draft first submission and final questionnaire creation
During the third step, the questionnaire was submitted by the steering committee to a sample of 20 KOA patients, to ensure a satisfactory understanding of each question and the acceptability of the questionnaire’s format. Subjects were invited to express their misunderstanding and to formulate recommendations over the questions. Following this pre-test, the second and final version of the questionnaire EOAQ was submitted to the whole board for final analysis and acceptance in a second meeting (29 January 2021).
Literature review
Databases
The literature review strategy consisted in an active search in different scientific databases, such as PubMed, Medline, PreMedline, where publications about patients affected by EOA were selected.
Selection criteria
Literature search results were subsequently examined and further selected according to specific criteria (human) and depending on the subject. Due to the few specific articles in literature, no selection criteria were applied to the publication type (case report and minor publications were included).
The literature research was performed by a librarian and the subsequent review of the literature results was performed by AM and GG. In Appendix 1 is reported the extraction form.
Results
Board members
The board included 23 members from different specializations. Most of them (52.17%, n = 12) were rheumatologists, followed by orthopedics (26%) and physiatrists (13.04%). Also one epidemiologist and one patients’ association representative participate in the board.
Literature review
Search results
A total 643 publications were examined. After a first screening, 43 publications were selected according to the selection criteria, as reported in Appendix Figure 1. All the publications have been reported in Supplemental Table 1.
Evaluation of early KOA
Eleven publications were focused on the evaluation of the early KOA-associated symptoms. Eijkenboom et al. 19 conducted a cross-sectional case–control study, where they defined the early KOA based on the presence of one of the following symptoms: crepitus or pain while climbing, squatting, running, cycling, or sitting for a prolonged period with the knee flexed.
Emery et al. 20 wrote a review defining early KOA according to the presence of new-onset symptoms and MRI evaluation of damage (cartilage, meniscal, and synovitis).
Some other authors, such as Luyten et al. 21 and Sancheti et al. 22 focused the definition on knee pain, in presence or not of other symptoms.
In 2017, Migliore et al. 23 defined the criteria to diagnose the early KOA in presence of: (a) two mandatory symptoms (knee pain in the absence of any recent trauma or injury and very short joint stiffness, lasting for less than 10 min, when starting movement even in the absence of risk factors) or (b) knee pain, and one or two risk factors, or (c) three or more risk factors in the presence of at least one mandatory symptom, with symptoms lasting less than 6 months starting movement even in the absence of risk factors. In the less recent publications, other authors defined the presence of early KOA based on different associated symptoms, especially during activities.24–41
Although most of the authors focused their attention on pain and on other associated symptoms, some of them included the radiological evaluation in the definition of the early KOA.42,43
OA misdiagnoses or underdiagnosis
According to the literature search, often early KOA diagnosis is difficult because patients tend to avoid the medical consultation. Moreover, some of them never consulted a general practitioner.33,38,30,42–45
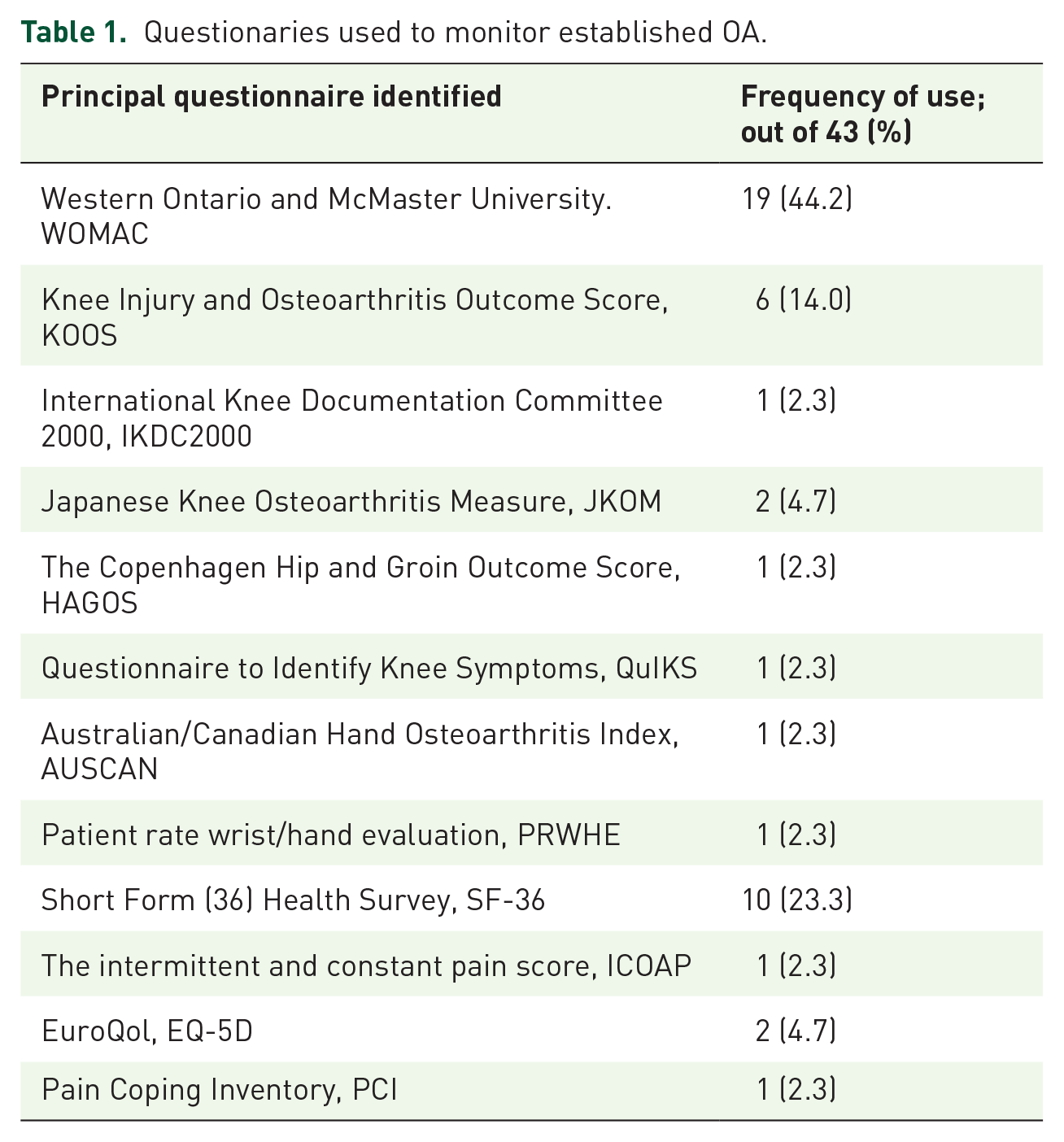
According to the literature analysis, different questionnaires have been adopted by the authors to evaluate the disease. The most used one (44.2% of the authors) was the Western Ontario and McMaster University (WOMAC), followed by the Knee Injury and Osteoarthritis Outcome Score (KOOS), used by 14% of the authors. The complete list of the questionnaires and their frequency is reported in Table 1.
Questionaries used to monitor established OA.
Questionnaire
First draft of the questionnaire
The first version of the questionnaire was discussed in October 2019 by patients and by the member of the board. A first draft containing 17 questions was created. Questions were grouped into two sections (clinical feature and patient-reported outcome), and a score was assigned to each item. Each question has been voted and only the ones that reached the full agreements were used to write the final version of the draft. The first draft is reported in Table 2.
First draft of the questionnaire.

Final version
During the discussion, 6 questions were rejected and a final version of the questionnaire containing 11 questions was created. As in the first draft, questions were divided into two different groups: clinical feature (two questions) and patient-reported outcome (nine questions). For each question, three answers were available, based on the number of episodes: never, rarely (one to three episodes) and frequently (more than three episodes). The final version of the questionnaire is reported in Table 3.
Final version of the questionnaire.

Discussion
OA is a health burden, which affects more than 300 million people across the world, 1 and is the leading cause of disability. Moreover, some authors reported an association between OA and a reduced time-to-mortality, independent of age, sex, and race. This could be due to some functional consequence of pain, which increases walking disability or reduces physical activity. 46 In EOA, patients at high risk for OA progression might be identified to halt or modulate the structural progression of the damage.47,48 Nowadays, different approaching criteria have been proposed, to detect and treat EOA but diagnostic criteria are not yet available. It is worth to note that often general practitioners are not well-trained to perform the diagnosis, and this lack is often associated with an increase in burden and disability. To address this problem, Rannou et al. recently developed visual decision trees to facilitate the diagnosis and management of OA of the hip, knee, and hand or finger in primary care. 49
After the publication of a composite algorithm combining exclusion criteria, symptoms, and risks factors 22 to help physicians and patients to refer properly to specialists to confirm the diagnosis of early KOA, the development of an EOA Questionnaire grade and evolution of EOA is mandatory.
This was the first international effort with the aim to try to provide a specific questionnaire for the early KOA.
In EOA, symptoms are similar to those of established phase, differing mainly for severity, intensity, and frequency. The TEP detected what were the domains and items to be investigated and what items should be considered in monitoring the response to pharmacological and non-pharmacological therapy.
EOA clinical features and patients’ outcomes were the main topics selected for the questionnaire. Clinical features should be evaluated in patients with a new onset of pain, stiffness, and other symptoms, since they might be associated with the development of OA in the future. 19 Also, it is fundamental to receive a report of the outcome directly from the patients, without the interpretation of the clinicians. Only in this way an effective questionnaire could be developed. 19 For this reason, the first version of the questionnaire was discussed in October 2019 by patients and by the members of the board. During the discussion, score was assigned to each item. Each question has been voted and only the ones that reached the full agreements were used to write the final version of the draft. The aim of this work was to create a complete but still short and easy-to-understand questionnaire, to let respondents to complete it shortly and easily.
After an exhaustive elaboration, a final version of the questionnaire containing 11 questions was created. The first two questions about clinical features explore early symptoms, in particular, the appearance of pain when a normal activity (i.e. walking) is performed much more than usual and the initial perception of knee locked or giving way. The proposal of questions about initial knee warming or noise have not been accepted, since these symptoms were considered affecting established OA.
Nine questions have been accepted to investigate patient-reported outcome. Many questions were asked about the presence of pain or discomfort during normal daily activities, which are then limited or painful during the established phase of the disease. In particular, knee pain climbing the stairs, or a sensation of discomfort in the knee when getting up after sitting for a long period, when walking on an uneven surface, using the toilet, squatting, kneeling, or following a short run such as to catch a bus. Question number 8 outlined the appearance of initial knee qualitative and quantitative limitations when performing routine or preferred sport activities (e.g. jogging), since these are first signs of joint alteration.
There was one question about the use of painkillers. In the established OA painkillers, analgesics and or NSAIDs are commonly and frequently used, often daily or many days for a month. On the contrary, at the beginning of the disease, their use is none or sporadic. The last question explores the behavior or mood of the subject when he starts to understand or to perceive that something is changing in his or her knee and consequently his worrying about his future.
Four questions about patient outcomes have been not accepted for the questionnaire. The appearance of discomfort or pain in the knee during weather changes was considered relevant only by some representatives of southern European countries and did not reach the final agreement. The other three unaccepted questions were considered a repetition or already incorporated into the previously accepted questions.
For each question, three answers were available, based on the number of episodes: never, rarely (one to three episodes), and frequently (more than three episodes) in a 6-month period. This was a decision only based on the clinical experience of the board members, since there are actually no published data available.
The real strength of this work is that for the first time, an attempt was made to fill the gap existing in the evaluation of early stages of OA through the formulation of a specific questionnaire different from the current questionnaires available in established OA, which are not able to catch symptoms and signs characteristic of early stages of the disease.
This was the first attempt to understand the main items that are meaningful in the early phases of KOA, to create a dedicated questionnaire. The most informative items included the evaluation of some normal daily activities (such as walking and climbing the stairs), since these are the actions that have been first influenced by EOA. During the next steps, authors would like to assess the clinimetric properties of the questionnaire through a punctual evaluation of some characteristics, such as its reliability, measurement error, construct validity, and responsiveness.
In the light of a ‘treat to target strategy’ proposed for the management of KOA, 50 some good clinical practice statements suggest that pharmacological and non-pharmacological treatment should begin as early as possible with an early diagnosis of symptomatic KOA. Similarly, early management of KOA is recommended by several guidelines, since this approach might modify the course of the disease and clinical features, especially considering disease-modifying agents that may delay disease progression. Understanding of disease pathogenesis and progression is increasing as well as the investigation on biomarkers capable to diagnose EOA and to monitor clinical evolution, but they are not applicable in clinical practice. In this scenario, an EOA questionnaire is an evaluable tool to cover this clinical gap.
Nowadays, we have both clear diagnostic criteria and well-validated, worldwide used questionnaire exploring symptoms, function, and outcome to manage established OA. In the case of EOA, these are unmet needs; while some efforts have been made to propose diagnostic criteria of EOA, this is the first proposal of a specific questionnaire for EOA.
The adoption of diagnostic criteria of the EOA is strongly encouraged and a specific questionnaire for the evaluation of the clinical features and patients’ outcome might really improve the evolution of OA in the early stages of the disease, when the treatment is likely to be more effective. This approach would allow to decrease disability and improve patients’ QoL.
Conclusion
We present a novel EOA questionnaire designed for diagnostic and follow-up assessment. The creation of this tool is particularly relevant, since there are no other questionnaires able to evaluate the EOA at the moment. Also, it is important to have a guide to decide when a pharmacological or non-pharmacological treatment would be recommended. Finally, this tool would help clinicians to monitor symptoms to prevent the progression of the disease, through non-pharmacological treatments or lifestyle changes.
Even with its limited validation, the adoption of this questionnaire is strongly encouraged to allow earlier diagnosis and intervention.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X221131604 – Supplemental material for Early Osteoarthritis Questionnaire (EOAQ): a tool to assess knee osteoarthritis at initial stage
Supplemental material, sj-docx-1-tab-10.1177_1759720X221131604 for Early Osteoarthritis Questionnaire (EOAQ): a tool to assess knee osteoarthritis at initial stage by Alberto Migliore, Liudmila Alekseeva, Sachin R. Avasthi, Raveendhara R. Bannuru, Xavier Chevalier, Thierry Conrozier, Sergio Crimaldi, Gustavo C. de Campos, Demirhan Diracoglu, Gianfranco Gigliucci, Gabriel Herrero-Beaumont, Giovanni Iolascon, Ruxandra Ionescu, Jörg Jerosch, Jorge Lains, Emmanuel Maheu, Souz Makri, Natalia Martusevich, Marco Matucci-Cerinic, Karen Pavelka, Robert J. Petrella, Raghu Raman and Umberto Tarantino in Therapeutic Advances in Musculoskeletal Disease
Footnotes
Appendix 1
Keywords (for Systematic Literature Review)
The search strategy included the following keywords: ‘osteoarthritis’ or ‘osteoarthrosis’, ‘osteoarthritis’ and ‘early’ or ‘early osteoarthritis’ and ‘questionnaire’, or ‘osteoarthritis’ and ‘early diagnosis’. Also, keywords such as ‘surveys and questionnaire’, ‘score’(or ‘scores’ or ‘scale’ or ‘scales’), ‘PROM’ or ‘PROMS’, ‘physical function’, ‘physical examination’, ‘NRS pain’, ‘level walking’, ‘walking test’, ‘sit to stand movement’, ‘stair negotiation’, ‘stair climbing’ (or ‘stair climb test’), ‘EQ-5D’ (or ‘30-s CST’, ‘FPWT’), or ‘stiffness’, ‘pain’, ‘pain measurement’, ‘WOMAC’, ‘AUSCAN’, ‘LEQUESNE index’, ‘KHOA-SQ’, ‘QoL’ (or ‘quality of life’), ‘analgesic use’ (or ‘analgesics use’, ‘analgesics/therapeutic use’, ‘pain killer use’), ‘measures’, ‘evaluation’ (or ‘evaluating’, ‘assessing’, ‘assessment’), ‘detect*’, ‘estimat*’. The search was conducted in English.
Acknowledgements
Medical editorial assistance was provided by A.I.C., Asti Incentives & Congressi Srl.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
