Abstract
Spondyloarthritides (SpA) like psoriatic arthritis, axial spondyloarthritis/ankylosing spondylitis, reactive arthritis and inflammatory bowel disease (IBD)-associated SpA can present with characteristic skin manifestations. These SpA-associated skin disorders may precede joint involvement, reflect a loss of efficacy of a current systemic treatment or can even be treatment associated. Cutaneous manifestations in SpA not only add additional morbidity with physical impact but also impose a psychosocial burden on affected patients. Psoriasis (PsO) – the main skin disease in SpA – has a variety of clinical presentations, including plaque-type PsO, inverse PsO, guttate PsO, erythrodermic PsO, nail PsO and pustular types. SpA associated with IBD presents with neutrophilic and granulomatous skin disorders, including pyoderma gangrenosum, hidradenitis suppurativa and cutaneous Crohn’s disease. Reactive arthritides has a favourable prognosis and may feature keratoderma blenorrhagicum or balanitis circinatum as typical skin manifestations. Immunologically, SpA-associated skin diseases share interleukin (IL)-17 and IL-23 dysregulation but show distinctive genetic and immunological profiles. Therefore, they vary in their treatment responses to targeted therapies with biologicals or small molecules. In this review, we highlight the clinical presentation of skin manifestations in SpA and discuss therapeutic approaches in this interdisciplinary field.
Keywords
Introduction
Spondyloarthritis (SpA) is one of the most common rheumatic diseases, affecting up to 1.9% of the population. 1 It describes a variety of chronic inflammatory conditions that affect primarily the axial skeleton with the sacroiliac joints and the spine, but may also present with peripheral joint involvement. SpA can be further distinguished into non-radiographic and radiographic axial SpA [the latter also known as ankylosing spondylitis (AS)], psoriatic arthritis (PsA), SpA associated with inflammatory bowel diseases (IBD) and reactive arthritis (ReA). 2 Progressive spinal stiffness along with chronic back pain are the most common symptoms of axial involvement, whereas asymmetrical joint stiffness and pain along with dactylitis and enthesitis, mono- or oligo-arthritis, mainly in lower extremities, define the peripheral inflammatory joint diseases. All of them are associated with distinct extra articular manifestations affecting the gastrointestinal system with IBD, anterior uveitis (AU) of the eye and various diseases of the skin. 3
Genetic links between the aforementioned diseases include positivity for the major histocompatibility complex human leucocyte antigen B27 (HLA-B27) allele and genetic polymorphisms within genes of the Th17/interleukin (IL)-23 pathway. 4 These factors play a major role in the inflammation causing SpA and associated diseases.5–7
In this review, we focus on the skin manifestations associated with SpA, and state-of-the-art treatments based on currently approved drugs for inflammatory disorders. Finally, we provide some insights regarding progress achieved in the understanding of these disorders and related future therapeutics. 8
Psoriasis
Psoriasis (PsO) is one of the most common inflammatory skin diseases, with an estimated prevalence of 0.6–4.8% of the global population.9,10 Patients typically present with characteristic erythematous plaques with a coarse overlying scale on specific sites such as elbows, knees, scalp, umbilicus, rima ani and palms. The transition from a cutaneous to a synovio-entheseal disease affects about one-third of PsO patients, who develop a PsA over time. 11 PsO precedes joint involvement on average by 7 years (range 0–20 years), creating a unique opportunity for early intervention in populations at risk.12,13 The importance of early diagnosis and early intervention in articular disease is highlighted by the fact that a delay of 6 months in initiating PsA treatment already can cause peripheral joint erosions, with subsequent impairment of joint function.13,14 In peripheral SpA patients, an early anti-tumour necrosis factor (TNF) treatment leads to a remarkable sustained clinical remission. This shows that there is a window of opportunity in the course of inflammatory diseases. 14 Therefore, a thorough clinical examination of joints and the whole skin by dermatologists and rheumatologists is key to allow early intervention. 15 Ideally, selected patients should be seen in interdisciplinary medical settings.
PsO is a frequent and early manifestation of SpA. 16 About one-third of SpA patients are affected by PsO. On the other hand, a significant number of PsA patients are asymptomatic concerning back pain, but show radiographic signs of axial involvement. 17 In SpA, PsO most frequently presents as a plaque-type disease (66.7%), which is the commonest form of PsO in general. Also, scalp involvement is very common in SpA patients (65.5%). Psoriasis pustulosa palmoplantaris (20.2%) and nail PsO (19.1%) are described in four out of five patients. 16 Of note, nail involvement, psoriasis capitis and inverse psoriasis have been reported to show an increased risk for PsA.18–20
The diagnosis of PsO is generally a clinical diagnosis based on its characteristic morphological appearance. There are no specific blood tests recommended and a skin biopsy is rarely needed. Psoriasis vulgaris refers to the classic plaque type PsO (PV), initially with erythematous macules and papules coalescing to well demarcated erythematous silver-white scaly round plaques on the body with a symmetrical predilection on the elbows and knees, the dorsum of the hands and feet, scalp, umbilicus and rima ani (Figure 1).

Clinical presentation of PsO. (A) PV; (B) erythrodermic PsO; (C) guttate PsO; and (D) inverse PsO.
Mild psoriasis is treated by topical agents like vitamin D analoga (calcipotriol, tacalcitol) and corticosteroids. In flexural sites, or on the face, either calcipotriol or non-steroidal topicals like pimecrolimus or tacrolimus (off-label) are recommend to avoid corticosteroid-induced skin atrophy. Phototherapy with narrow-band ultraviolet (UV)B and topical dithranol are effective, but usually used in inpatient settings for treatment initiation because application is inconvenient in a domestic setting. Further systemic treatments with methotrexate, fumarates and acitretin are generally considered as first line and cost effective.21,22 Although cyclosporine is generally recommended in guidelines, it should not be used due to its toxicities and pro-tumorigenic aspects. As the aforementioned agents are generally rarely effective in SpA, they will not be discussed in this review.
Inverse PsO
In contrast to psoriasis vulgaris, inverse PsO affects the flexural sites and intertriginous areas. Most common affected skin sites are axillae, inguinal folds, inframammary and perineal regions. Clinically, an inverse PsO presents with sharply edged erythematous and mildly elevated plaques with little or no scales and occasionally rhagades, and therefore is easily confused with candida or dermatophytic intertrigo (Figure 1D). Inverse psoriasis affecting the genitals significantly affects the patient’s quality of life.
Guttate PsO
Guttate PsO often presents with an acute onset of multiple small round erythematous plaques on the trunk and extremities. The term guttata refers to droplets (‘gutta’) representing the small plaques, which are less than 1 cm in size and usually present monomorphically due to a timely similar onset of the lesions (Figure 1C). PsO guttata is associated with streptococcal infections of the throat, especially in children and young adults. Although PsO guttata has a favorable prognosis, it may also appear as the first manifestation of an underlying chronic plaque PsO.23–25 About one-third of patients will develop classical PsO over time. 25 Although streptococcal infections are often associated, there is no reproducible data on the efficacy of antibiotic treatment to give general recommendations. Yet, some patients may benefit from antistreptococcal antibiotics or tonsillectomy. A rapid involuting course is associated with younger age, higher anti-streptolysin titres and a negative family history for PSO.25,26
Erythrodermic PsO
If the majority of the body surface (>80–90%) is affected with erythematous maculae or plaques, an erythrodermic PsO is a concerning differential diagnosis. It is associated with hypothermia, fevers with dehydration and tachycardia and often displays with peripheral oedema. Abrupt discontinuation of systemic treatments, especially corticosteroids or PsO-triggering medication such as lithium or beta blockers, can lead to this more exanthematous clinical picture. In this case, skin biopsies are helpful to rule out differential diagnosis like pityriasis rubra pilaris, an erythroderma drug rash, a cutaneous T cell lymphoma or an atopic dermatitis. In addition, in Fitzpatrick skin types V and VI visualizing the erythema can be challenging (Figure 1B).
Nail PsO
The spatial proximity of the inflammation in the nail bed and matrix along with the enthesitis suggests a close relationship of nail involvement and enthesitides.27,28 Up to 80% of PsA patients show nail involvement, and nails are affected in more than half of all PsO patients. 29 In fact, almost all patients with proof of ultrasonic enthesitis show nail abnormalities. 28 PsO of the nail may present without classic skin manifestations. While nail PsO of the finger shows a very typical picture, toenails might present only with subungual hyperkeratosis, which is seen regularly in onychomycosis and therefore an important differential. Psoriasiform nail changes include changes of the nail matrix, such as pitting, nail plate crumbling, leukonychia and red spots in the lunula. Pitting, irregular deep small depressions in the nail plate, is highly characteristic of nail PsO and reflects a focal parakeratinization in the proximal nail matrix, leaving nucleated cells in the upper cornified layers. 30 If the nail bed is affected subungual hyperkeratosis, splinter haemorrhages, oil drop dyschromia and onycholysis is shown (Figure 2C, D).

Clinical presentation of PsO affecting the hands and feet. (A) Plantar PsO with hyperkeratotic plaques (left and middle) and pustular presentation (right). (B) Palmar PsO with hyperkeratotic plaques (left and middle) and pustular presentation (right). (C) Acrodermatitis continua of Hallopeau with severe onychodystrophy and oil drop dyschromia (*); (D) Nail PsO with onychodystrophy, pitting (*) and onycholysis (°).
Treatment of nail PsO still is a challenging and tedious process since nail renewal takes several months. Topical therapies usually underperform. Calcipotriol ointment, intralesional and topical corticosteroid show no, or limited, effects. 31 Nail PsO should be treated with systemic drugs when it has a high impact on quality of life and presents with functional impairment.
Pustular PsO, palmoplantar pustular PsO and acrodermatitis continua of hallopeau
Neutrophilic accumulation is histologically found as microabscesses in the skin of psoriatic plaques. Macroabscesses form clinically visible pustules. Pustular PsO (PP) presents mostly with an acute onset of multiple small-sized sterile pustules on an erythematous base. Its generalised form [generalised pustular psoriasis (GPP)], with rapidly evolving monomorphic pustules coalescing to confluent pustules, is a rare but serious condition with fevers, myalgia and elevated inflammatory serum markers. Causally, abrupt corticosteroid withdrawal, infections, certain drugs and pregnancy have to be evaluated. Further, impaired liver function, hypocalcemia and dehydration may appear and complicate the condition. In GPP, mutations within the IL36RN or CARD14 genes have been found that promote inflammatory cell recruitment by keratinocytes and uncontrolled release of inflammatory cytokines.32,33 However, only a minority of PP are actually caused by IL36RN mutation and PV-associated PP correlates with this mutation in only 17%.34,35 Despite the genetic association with the IL-36 pathway, all forms of pustular PsO are characterised by an increased transcriptional expression of IL-1β, IL-36α, and IL-36γ compared with PV. 36 The IL-17 axis plays also an important role in PP, independent of IL36RN mutation status. 37
A severe pustulosis affecting the tips of fingers and toes is called acropustulosis or acrodermatitis continua of Hallopeau (ACH). It generally affects nail growth, from severe onychodystrophy up to anonychia, and may lead to osteolysis of the distal phalanx (Figure 2C).
A manifestation of palmoplantar pustular PsO (PPP) features erythematous plaques with multiple sterile pustules on the palms and soles, demarcated mostly by a thin red ring and scales (Figure 2A, B). PPP is the most common of the pustular PsO types. 38 It is more common in females and smokers and may present as a paradoxical skin manifestation in patients without a history of PsO that are treated with anti-TNF-α therapeutics, for example, inflammatory joint diseases or IBD.
The treatment of all pustular PsO types is challenging and there is a need for treatments with proven efficacy.38–40
IBD-associated skin manifestations in spondyloarthritis
Crohn’s disease (CD) and ulcerative colitis (UC) are chronic IBDs, which can present with several extra intestinal manifestations (EIM) that affect mortality and morbidity. Up to 50% of IBD patients experience at least one EIM.41–43 EIM of the skin typically appear years after the first diagnosis of an IBD, whereas axSpA and uveitis can manifest before the diagnosis of IBD. 43 More than one-third of patients with CD and up to 15% of patient with UC present with EIM, and more than 10% of all Crohn’s patients present with cutaneous manifestations.44,45 Within the latter group, erythema nodosum (EN), pyoderma gangrenosum (PG) and psoriasis are the most common EIM. 46
Pyoderma gangrenosum
PG are rare chronic or recurrent cutaneous ulcers that typically present with a necrolytic undermined violaceous border rising rapidly from a papule or pustule of the skin (Figure 3). The incidence of PG is described as 3–10 per million per year, with a female predilection, occurring mainly in mid-adulthood. 47 It may occur idiopathically, in association with inflammatory diseases such as IBD or as inherited inflammatory syndrome, such as PAPA (pyogenic arthritis, PG, and acne), PASH (PG, acne, and hidradenitis suppurativa), or PAPASH (pyogenic arthritis, acne, PG, and hidradenitis suppurativa). 48 Histologically, it shows a massive sterile neutrophilic infiltrate but often lacks a fibrinoid necrosis of blood vessels and therefore represents a neutrophilic dermatosis along with Behçet’s disease, hidradenitis suppurativa and Sweet syndrome. Nonsyndromic PG shows a decrease in regulatory T cells and an increase in IL-17 in affected skin facilitating the recruitment of neutrophils.49–51 Syndromic PG has been recently classified as auto-inflammatory diseases with inflammasome activation leading to excessive IL-1β, TNF-α and IL-8 production. 52 Interestingly, this signature has also been found in nonsyndromic PG. 53

Clinical presentation of pyoderma gangrenosum of the axilla (left) and the buttocks (right).
A retrospective cohort study aiming to investigate comorbidities in PG showed that 20.5% of all PG patients were affected by inflammatory arthritides defined as RA, PsA, and AS. 54 Most commonly, PG is associated with IBD, especially UC, but also hematologic malignancies have been described. In IBD associated PG, IBD should be treated first. 55 Pathergy is a well-known trigger mechanism in PG and postsurgical reoccurrence is published regularly. 56
There are various therapeutic approaches but, due to disease rarity, controlled clinical trials are lacking. Systemic corticosteroids are most frequently used for treatment initiation. For long-term immunosuppression in PG cyclosporine, infliximab, mycophenolat mofetil, azathioprine, intravenous immunoglobulins and methotrexate as well as cyclophosphamide have been reported.47,57 Although there is an excessive infiltration with neutrophils, anti-neutrophilic therapies with dapsone or colchicine showed limited efficacy and are used as adjunct agents.
Erythema nodosum
Erythema nodosum (EN) is the most commonly reported cutaneous manifestation in rheumatic diseases and IBD. It presents clinically as painful, often symmetric erythematous plaques and nodules on the extensor surfaces, mainly of the lower limbs (Figure 4). The clinical presentation is often classical but can be clinically challenging when it lacks the symmetrical appearance or its nodular aspect. In this case, differential diagnoses such as a cutaneous CD, a cellulitis due to a bacterial infection and cutaneous lymphoma should be considered and a biopsy should be taken. Histologically, EN presents as a septal panniculitis, characterized by septal fibrosis with infiltration of lymphocytes, neutrophils (especially in the early phase), histiocytes and granulomas with giant cells. In most cases, EN is self-limiting when the underlying disease is being treated. Since EN often presents painful, anti-inflammatory analgesics and compression are a helpful supportive treatment. In more severe cases, systemic corticosteroids up to 1 mg/kg/day are recommended and generally lead to a rapid improvement. 58 EN may also present as a manifestation of an infection with Mycobacterium tuberculosis or as a symptom of a manifest tuberculosis. 59 Especially in the scope of reactivations of latent tuberculosis in patients treated with TNF-α inhibitors, this rare differential diagnosis has to be considered.
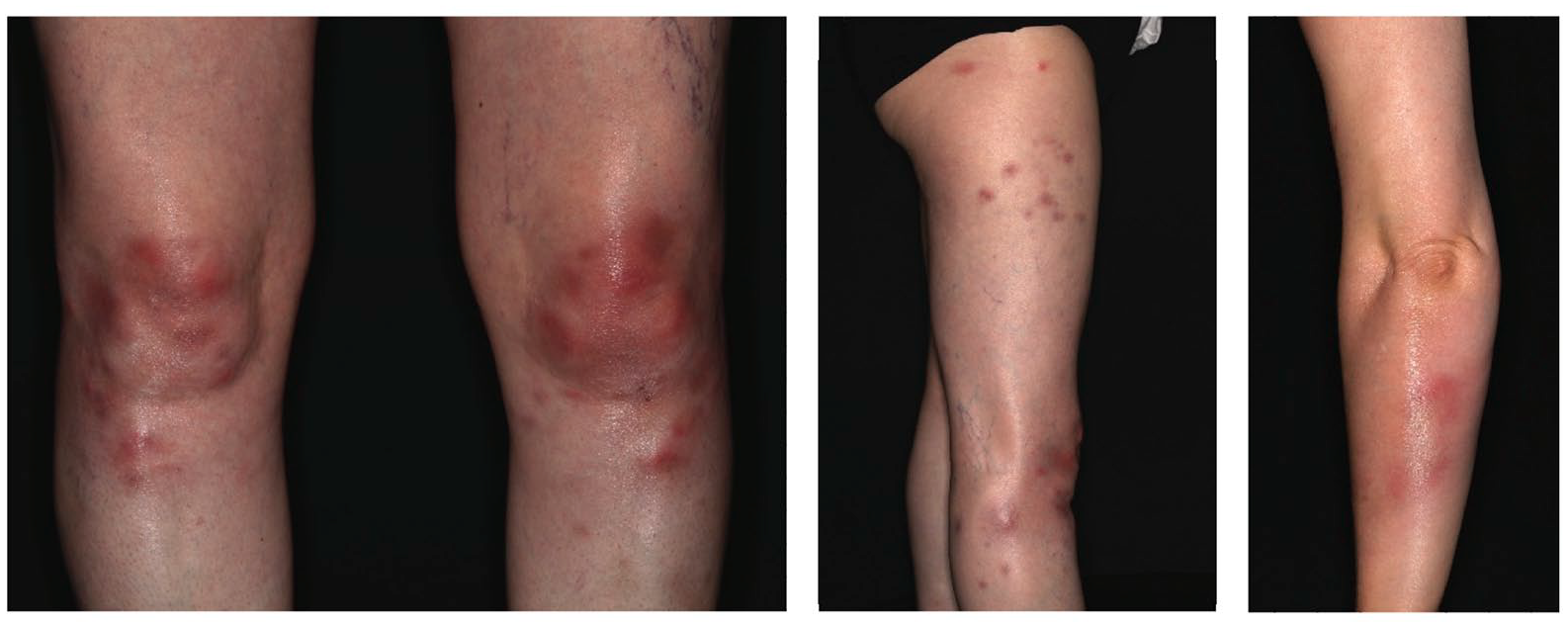
Clinical presentation of EN of the lower limbs.
Cutaneous CD
Cutaneous CD (cCD) – often referred to metastatic CD – is a very rare disease with less than 100 reported cases since its first description in 1956.60,61 It presents in tissues noncontiguous with the intestine and histologically presents, as CD, with noncaseating epitheloid cell granulomas with various giant cells reaching into the deep dermis. It often shows a perivascular lymphohistiocytic infiltrate with plasma cells. Clinically, it presents as erythematous swollen plaques or nodules that may ulcerate and discharge pus (Figure 5). Due to its rarity, there are no clinical trials regarding treatment of cCD. Topical and intralesional corticosteroids may be used for small lesions, while, for larger lesions oral antibiotics, especially metronidazole, traditional immunosuppressive agents and surgery, have shown mixed results. 62 For systemic treatment, biologics should be used as known from classic CD treatment.

Clinical presentation of cutaneous Crohn’s disease of the lower left extremity.
Hidradenitis suppurativa
Hidradenitis suppurativa (HS) is a strongly impairing chronic inflammatory disease affecting primarily apocrine gland-bearing areas such as the groin and axillaries. HS leads, via recurrent painful deep tissue inflammation, to purulent discharge and debilitating scars and is associated with a massive impact on quality of life (Figure 6). 63 Affecting 1–4% of the general population, it is a rather common disease with an association with smoking and overweight. 64 It is three times more common in females and affects generally young adults. 65

Clinical presentation of HS of the axilla (left) and the groin (right).
The clinical presentation can be challenging. On average, HS patients see more than three physicians and it takes about 10 years from first symptoms to the final diagnosis. 66 HS shows an association with a variety of skin diseases, such as acne conglobate and neutrophilic dermatoses. 67 Recent transcriptomic analysis supported the hypothesis that the underlying pathogenic mechanism of these diseases link to an infiltrate of neutrophils and macrophages in affected tissues and increased levels of inflammatory cytokines IL-1α, IL-1β, IL-17A/F, IL-23 and TNF-α, an involvement of the JAK–STAT pathway and genetic variants of B-cell co-stimulation, such as CXCR5.68–70 In lesional skin of HS patients, frequencies of CD4+ T cells expressing IL-17 and TNF were elevated significantly and anti-TNF treatment resulted in a significant reduction of IL-17 expressing CD4+ T cells in HS skin. 71
Importantly, 2–28% of HS patients show an axial involvement, and almost 10% of patients with axSpA develop a HS. Their temporally correlating appearance reflects a possible immunogenic link.72–74 In SpA patients, HS precedes the diagnosis of SpA in most cases and axial involvement is frequent within this group, which also shares a strong HLA-B-27 positivity. 72 Since SpA patients with axial involvement and HS tend to have a higher disease activity, clinical screening for HS in SpA patients is crucial. 75 On the other hand, HS patients with lower back pain and dactylitis or other oesteoarticular symptoms should be screened for SpA. 74 Besides IBD and inflammatory arthropathies, common comorbidities of HS are cardiovascular disease, diabetes mellitus, metabolic syndrome, polycystic ovary syndrome, depression and anxiety and these conditions should be regularly screened in HS patients. 76
The treatment of HS is mostly an interdisciplinary approach, with surgical, topical and systemic treatments and supportive therapies such as analgesics, especially nonsteroidal anti-inflammatory drugs (NSAIDs) and weight reduction and smoking cessation.76,77 Topical therapies focusing on, and antibiotic strategies to reduce, bacterial triggered inflammation as well as keratolytic ointments reducing follicular clogging are considered as initial treatments, intralesional corticosteroid injections are considered as second-line therapies and should be used only for acutely inflamed HS lesions. 76
Guidelines recommend oral tetracyclines as first-line therapy and should be administered over a minimum period of 12 weeks.78,79 Coadministration of clindamycin and rifampicin over at least 4 weeks is a common long-term treatment, although there is low evidence that the combination reduces the presumed antibiotic resistances. 80 Retinoids, especially acitretin and dapsone, are recommended as third-line therapies in most guidelines. A systemic treatment does benefit inflammation, with a decrease of nodules and discharge, but does not benefit fistulas. A surgical evaluation therefore is key to efficacious treatment of moderate-to-severe HS.
Reactive arthritis-associated skin manifestations
Reactive arthritis (ReA) is defined as a form of peripheral SpA occurring after an infection affecting not primarily the joint, most commonly of the gastrointestinal or genitourinary tract. ReA frequently affects young adults and is five times more common in individuals with a positive HLA-B27 genotype. 81 HLA-B27-related ReA presents in a more chronic manner, with frequent extra-articular manifestations and an unfavorable prognosis compared with non HLA-B27 ReA. 82 In up to 50% of patients, it presents with distinct dermatological manifestations such as keratoderma blenorrhagicum (KB) and circinate balanitis (CB), may show an ulcerative vulvitis, nail changes, oral lesions and conjunctivitis. Since symptoms occur 1–4 weeks after the causing infection, microbial swaps and cultures often remain negative, but microbial DNA and RNA have been found in affected joints.83,84 Pathomechanistically, it is rather a stimulation of inflammatory cytokines by bacterial antigens and its immune-mediated reaction. Depending on the region, sex and age group, Chlamydia trachomatis, Enterobacteriae and Campylobacter, Yersinia and Shigella are well known causes.85,86
CB is characterised by vesicles and pustules of the glans penis developing into small annular painless erosions and is very common (Figure 7A). In circumcised males, it may present more hyperkeratotic. Ulcerative vulvitis shows a similar pattern on small labia and the vulva, and is often associated with vaginal discharge. 87 KB typically manifests at the plantae and palmae, beginning with erythematous macules and papules rising into vesicular, often hyperkeratotic plaques and sterile pustules (Figure 7B). It presents in about 10% of HLA-B27-positive ReA patients. Nail and mucosal changes are common. 88

Skin manifestations in ReA. (A) BC of the glans and penis. (B) KB of the soles of the feet.
ReA is frequently self-limiting, and first line treatment is generally with NSAIDs. 89 Treatment of cutaneous symptoms are similar to other inflammatory cutaneous diseases and often respond to topical corticosteroids and keratolytics. The use of long-term antibiotics has been controversially been discussed. 90 Biologics rapidly improve skin and joint manifestations but relapses are common after discontinuation. 91 There has been a broad spectrum of the use of anti-inflammatory biologics with an emerging role of the IL-17/IL-23 pathway. 92
Paradoxical skin reactions to biologicals
Paradoxical skin reactions (PSR) in patients being treated with targeted biologicals appear with a prevalence of 0.6–5.3% in the general population, and are most common within patients being treated with TNF-α-antagonists.93,94 They have to be considered when new cutaneous lesions occur during targeted- or immune-modulating therapies in immune-mediated inflammatory diseases. PSR present most commonly within the first year of biological treatment (60%), and often have a psoriasiform or pustular appearance. 95 Especially in patients being treated with TNF-antagonists, paradoxical PsO may present palmoplantar with an acute onset of pustules. PPP is diagnosed in 36.3% of PSR in TNF-inhibitors, but plaques PsO (44.8%) and guttate PsO (11%) are also reported.95,96 Hair loss is also a common PSR in biological therapy, and presents with clinical and histopathological finding of alopecia areata and PSO capitis.97,98 There are also almost 50 cases of HS as PSR in the literature, with adalimumab and infliximab as the commonest causing agents. Notably, one-third of these patients had an additional PSR, with PsO and CD being the commonest. 99 Autoimmune-bullous disorders such as pemphigus foliaceus, bullous pemphigoid or linear IgA dermatosis, as well as lupus-like syndromes, vasculitis and lichenoid skin eruptions have also been described. 100 Commonly, PSR improve after discontinuation of the culprit agent, even within the same drug group. About one-third of patients with PSR need a discontinuation of the drug and these patients do not response to topical treatments. 101 The underlying mechanism is most likely is a cytokine imbalance between TNF-α and type 1-interferons (IFN-α) favouring development of inflammatory plasmacytoid dendritic cells (pDCs) and increased type-1 IFN production.102,103 As a consequence of anti-TNF, type I IFNs are increased. Therefore, in PSR anti-TNF medication should be discontinued and a possible class switch should be considered. 104
Treatment
Treatment goals
With the introduction of a variety of new systemic therapeutic options over the last decade, treatment goals in inflammatory skin diseases and rheumatic disorders had to be reconsidered.
In PsO, the Psoriasis Area and Severity Index (PASI) and Body Surface Area (BSA) scores are the gold standards in measuring skin involvement. The PASI has a maximum score of 72 and is calculated on the basis of the percentage of affected skin in combination with its erythema, induration and scaling. The BSA represents the percentage of affected skin. The patient’s palm presents 1% of the body surface. Through therapies targeting TNF-α, IL-17/IL-23, PDE4 and JAK-STAT the total clearance of the skin, a PASI 100 and BSA < 1, may be achieved while the placebo effect on PsO is minimal.105–108
Unfortunately, the treatment targets in joint involvement in current clinical trials are far from a total 100% clearance of inflammatory activity. This being said, primary endpoints with validated measurements of the American College of Rheumatology (ACR20, ACR50 or ACR70) responses in PsA and ASAS20 or ASAS40 in axSpA are the gold standard of musculoskeletal manifestation response measurement. In addition, the effects of placebo treatment are much higher for articular inflammation (up to 30%) than for skin inflammation. 12 There is an ongoing discussion regarding whether articular diseases should be treated to target and what these targets are. 109 While an elevated C-reactive protein is a SpA feature in its current classification and correlates with disease activity, in inflammatory skin conditions it represents disease severity only on severe skin conditions and might not reflect the need for treatment of moderate and mild disease. 110
In HS and PG, treatment goals have to consider that existing scarring will not be reversible. Pain, odour and daily wound care massively impact daily life. To measure the impact of patient’s quality of life, the Dermatological Life Quality Index (DLQI) is recommended, and should be used in combination with validated scoring systems for each disease. To evaluate efficacy in HS, the Hidradenitis Suppurativa Clinical Response (HiSCR) scoring system is validated. It defines a >50% reduction in total abscess and inflammatory nodule count without any increase in abscess and draining fistula count in comparison with baseline. 76 In HS, response rates of more than 50% are considered high.
In rare disorders like PG or CCD, clinical trials are rare and validated scoring systems lacking. A regular clinical picture along with the size of the lesion, together with a DLQI and a pain assessment on a visual analogue scale (VAS) should be monitored to evaluate efficacy.
TNF-α
TNF-α belongs to the first generation of biologicals. Current approved anti-TNF agents include adalimumab, certolizumab pegol, etanercept, golimumab and infliximab. Because of relatively long data accumulation, safety data for pregnancy for the use of certolizumab, etanercept and adalimumab are available.111–113
While the response rates in PsA did not differ largely between adalimumab, certolizumab pegol, infliximab and etanercept, in PsO PASI, 90 responses ranged from 54% in infliximab, 30% in adalimumab, 48% in certolizumab and 24% in etanercept.114,115 Concerning skin involvement, response rates of etanercept are similar to those of methotrexate. 116 A retrospective analysis of 52 patients suffering from PG showed complete remission with infliximab in 63.6% (n = 33), for adalimumab in 57.1% (n = 28), for etanercept in 71.4% (n = 7) and for ustekinumab in 66.6% (n = 9) of patients. Notably, all of these were superior to corticosteroids. 117
In HS, adalimumab is the only approved biologic and is recommended for moderate-to-severe HS that is unresponsive to antibiotics, with relatively good response rates. 118 No significant difference comparing infliximab and placebo was observed for >50% improvement HiSCR, but the improvement rate of 25–50% was significantly higher for infliximab versus placebo. 119 Etanercept does not benefit HS patients compared with placebo. 120 There is limited data concerning the therapy of HS with golimumab or certolizumab, which was found to be ineffective in case series. To our knowledge, infliximab is the only biologic agent being assessed in a randomized controlled trial in PG showing superiority over placebo, 121 whereas systemic prednisone and cyclosporine were assessed as classic immunosuppressants with no superiority of either drug. 122 Recently, adalimumab was shown to be effective in an open-label phase III trial in PG (Figure 8). 123 In therapy-resistant EN, anti-TNF-α therapy also shows a response rate of up to 80%.42,58 There is very limited data for the use of anti-TNF in ReA. In a retrospective study, skin and joint inflammation improved rapidly with biologicals but disease exacerbation followed cessation of the drug within the first 6 months. 91

Efficacies targeting TNF-α in PsO, HS, PG, rSpA, nrSpA and PsA. The size of the circle presents the level of evidence, the colour represents the level of efficacy in the indicated disease.124–138 As standards of therapeutic efficacies in PsO PASI90, in HS HiSCR, in SpA ASAS40 and in PsA ACR50 are used. The scheme is adapted from Eyerich et al. 139
IL-17
Currently, three monoclonal antibodies targeting IL-17 are approved: brodalumab, ixekizumab and secukinumab. The latter two target IL-17A directly, while brodalumab targets the IL-17 receptor A leading to the inhibition of both IL-17A, F and other family members.
In PsO, secukinumab showed PASI 90 scores of 76.6% and was superior in a head-to-head study against ustekinumab.140,141 In contrast to anti-TNF, the onset of efficacy is earlier in secukinumab. Ixekizumab also presents a fast onset with response rate superior to placebo at week 1 and a PASI 90 of 50% at week 8. Brodalumab has a similar response with PASI 90 at 70% at week 12 and showed, along with the others, no discernible loss of efficacy.142,143 Ixekizumab and secukinumab are also efficacious in nail and scalp psoriasis and approved for PsA.144–146 Interestingly, IL-17 blockade with secukinumab failed in patients with palmoplantar pustular PsO. 147 Anti-IL-17 targets have been suggested to be associated with clinically relevant mucocutaneous candida infections as IL-17 pays an important role in defending candida infections. 148 However, the risk seems to be limited. In addition, data are limited on the association of new-onset IBD and secukinumab, and recent insights loosen that association.142,149,150
Secukinumab has been evaluated in clinical trials for moderate-to-severe HS and preliminary reports state a HiSCR of 67–70% at week 24 in cohort of TNF-naïve and TNF-refractory patients.120,151 Ixekizumab has been reported to be effective in HS in case studies. Other IL-17 antibodies such as secukinumab are currently in clinical trials for HS (Figure 9). 152

Efficacies targeting IL-17 in PsO, HS, PG, rSpA, nrSpA and PsA. The size of the circle presents the level of evidence, the colour represents the level of efficacy in the indicated disease.126,153–158 As standards of therapeutic efficacies in PsO PASI90, in HS HiSCR, in SpA ASAS40 and in PsA ACR50 are used. The scheme is adapted from Eyerich et al. 139
IL-12/IL-23 and IL-23
Ustekinumab is the only approved biologic targeting the p40 unit shared by IL-12 and IL-23. For a more specific IL-23 blockade, the following antibodies bind to the specific p19 unit of IL-23: guselkumab, risankizumab and tildrakizumab. Ustekinumab’s response rates in PsO are 46% PASI 90.114,115 Risankizumab presented with 75.3% PASI 90 at week 16 and 82% at week 52. Impressively, the PASI 100 Score at week 52 was 60%, and patients showed a relatively long efficacy despite the cessation of the agent.159,160 However, it did not show efficacy in AS. 161 Similarly, ustekinumab does not play a therapeutic role in axial SpA following negative phase III program results in axSpA, in contrast to joint inflammation in PsA with moderate clinical responses and beneficial effects of ustekinumab and guselkumab in axial involvement in PsA to some extent.161–163 To date, it is unclear why IL-23 targets do not improve axSpA in contrast to anti-IL-17 therapeutics. 164 Tildrakizumab showed a PASI 90 response of 35% (week 12) and 59% (week 28) and guselkumab 72% PASI 90 (week 16) and 76% (week 48).165–167
Ustekinumab has been evaluated in a randomised controlled trial (RCT) showing clinical improvement of HS (HiSCR 47%, week 40) compared with placebo. 105 In addition, risankizumab shows promising efficacy in case reports and is currently in phase II trials for CD and HS. In HS, guselkumab has been studied in case series. Systemic literature reviews suggest significant reduction in pain with improvements in lesions. RCT are currently recruiting with no published results yet. 106
An increased expression of IL-23 and IL-17 in lesional PG skin led to numerous case reports of the efficacious use of ustekinumab, ixekizumab, risankizumab and brodalumab although clinical trials are missing (Figure 10).168,169

Efficacies targeting IL-12/23 and IL-23 in PsO, HS, PG, rSpA, nrSpA and PsA. The size of the circle presents the level of evidence, the colour represents the level of efficacy in the indicated disease.161–163,170–172 As standards of therapeutic efficacies in PsO PASI90, in HS HiSCR, in SpA ASAS40 and in PsA ACR50 are used. The scheme is adapted from Eyerich et al. 139
IL-1
Targeting IL-1 led to a revolutionary change in the treatment of auto-inflammatory skin diseases. Currently, canakinumab, rilonaccept [not available in the European Union (EU)] and anakinra are approved anti-IL-1 drugs. Anakinra has been evaluated for PsO. As in PsA, anakinra showed only a modest benefits in PsO, with partially worsening of skin involvement. 173 In contrast to plaques PsO, IL-1 seems to play an important role in pustular PsO, and the results of a RTC are awaited. 174
In syndromic PG, TNF-α-inhibitors and anti-IL-1 agents are most frequently used. Anakinra and canakinumab targeting IL-1 show limited efficacy in PG.175,176 In HS, anakinra is considered as third line in some countries. In a relatively small cohort, anakinra did initially show good efficacy, with HiSCR of 78% versus 30% in the placebo group after 3 months, but after 24 weeks no difference was found between the two groups. Evidence to support reproducible efficacy is limited.76,177 Canakinumab is used in Brazil because of unavailability of anakinra, but data show mixed results and RCTs are missing (Figure 11).

Efficacies targeting IL-1 in PsO, HS, PG, rSpA, nrSpA and PsA. The size of the circle presents the level of evidence, the colour represents the level of efficacy in the indicated disease.173,177–181 As standards of therapeutic efficacies in PsO PASI90, in HS HiSCR, in SpA ASAS40 and in PsA ACR50 are used. The scheme is adapted from Eyerich et al. 139
IL-6
Since IL-6 plays a role in IL-23 induced differentiation of Th17 cells, targeting IL-6 led to new therapies in rheumatoid arthritis (RA), giant cell arteritis and is currently under evaluation for PsA. Targeting IL-6 may play an important role in palmoplantar pustulosis. Tocilizumab has been reported to be efficacious in TNF-triggered pustular PsO. 182 On the other hand, several case reports of tocilizumab-induced psoriasis exist.94,183 (Figure 12)

Efficacies targeting IL-6 in PsO, HS, PG, rSpA, nrSpA and PsA. The size of the circle presents the level of evidence, the colour represents the level of efficacy in the indicated disease.184–188 As standards of therapeutic efficacies in PsO PASI90, in HS HiSCR, in SpA ASAS40 and in PsA ACR50 are used. The scheme is adapted from Eyerich et al. 139
IL-36
IL-36 is a member of the IL-1 cytokine family that plays a central role in immunity and inflammation. It is overexpressed in PsO and RA patients is various stimuli, such as TNF-α, IL-17 lipopolysaccharides and double-stranded RNA.189–191 Comparing PsA and RA, IL-36 levels do not differ in the synovia but PsA shows a significant higher activation of IL-36 due to a lack of antagonist IL-36-RA and IL-38 in comparison with RA. 192 Therefore, targeting IL-36 in PsA and pustular PsO seems to be a promising therapeutic approach and is currently being tested in clinical trials.36,193 Antagonizing IL-36 may also be beneficial in other neutrophil-dominated diseases.
Janus tyrosine kinase inhibitors and small molecules
Janus tyrosine kinase inhibitors (JAKi) are small molecules and taken orally. To date, there are four members in the JAK family (JAK 1-3 and TYK2) with five approved agents: Baricitinib, filgotinib, tofacitinib, upadacitinib and ruxolitinib.33,194 These molecules are gaining increasing attention and are currently being tested in several dermatologic disorders.194–197 Tofacitinib shows efficacy in PsO, with PASI 75 responses of 55.2% (10 mg/day) and efficacy rates comparable with those of TNF- and IL-17 inhibitors in axSpA.198–200 Topical tofacitinib also improves PsO but is not approved. 201 Baricitinib reached PASI 90 responses between 20% and 30% depending on the dose. 202 JAKi may also be beneficial in palmoplantar PsO. 203 So far, tofacitinib is the only JAKi approved for PsA but not for PsO. However, a large JAKi program studying the safety and efficacy of multiple JAKi for different PsO entities is ongoing.195,196
There is very limited data for JAKi in HS. Tofacitinib shows good clinical results in two recalcitrant severe HS cases, not responding to targeted biologic therapies. 204 Upadacitinib, topical ruxolitinib and INCB-054707 are currently under clinical investigation in HS (Figure 13).

Efficacies targeting JAK in PsO, HS, PG, rSpA, nrSpA and PsA. The size of the circle presents the level of evidence, the colour represents the level of efficacy in the indicated disease.202,205–212 As standards of therapeutic efficacies in PsO PASI90, in HS HiSCR, in SpA ASAS40 and in PsA ACR50 are used. The scheme is adapted from Eyerich et al. 139
Apremilast is a phosphodiesterase-4 inhibitor (PDE4) and inhibits the hydrolyzation of cAMP, leading to a decreased expression of the proinflammatory cytokines TNF-α, IFNγ and IL-12/IL-23p40. 213 It is approved for the treatment of moderate-to-severe plaque PsO, PsA and oral ulcers in Behçet’s disease, and did show beneficial effects in PsO in the difficult-to-treat sites nail, scalp and palmoplantar.106,214,215 In contrast, in AS, apremilast did not meet the primary endpoint in a phase III clinical trial with a higher ASAS40 in the placebo group [ClinicalTrials.gov identifier: NCT00944658]. 216 Apremilast has been described as an effective treatment for PG, although RCT are lacking.217,218 PDE4 has recently been approved for Behçet’s disease, which belongs to the group of ulcerative neutrophilic disorders. 219 Apremilast has also been evaluated in mild-to-moderate HS with improvement in disease activity (HiSCR 53.3%, week 16), pain, and quality of life in patients with less severe HS (Figure 14).220,221
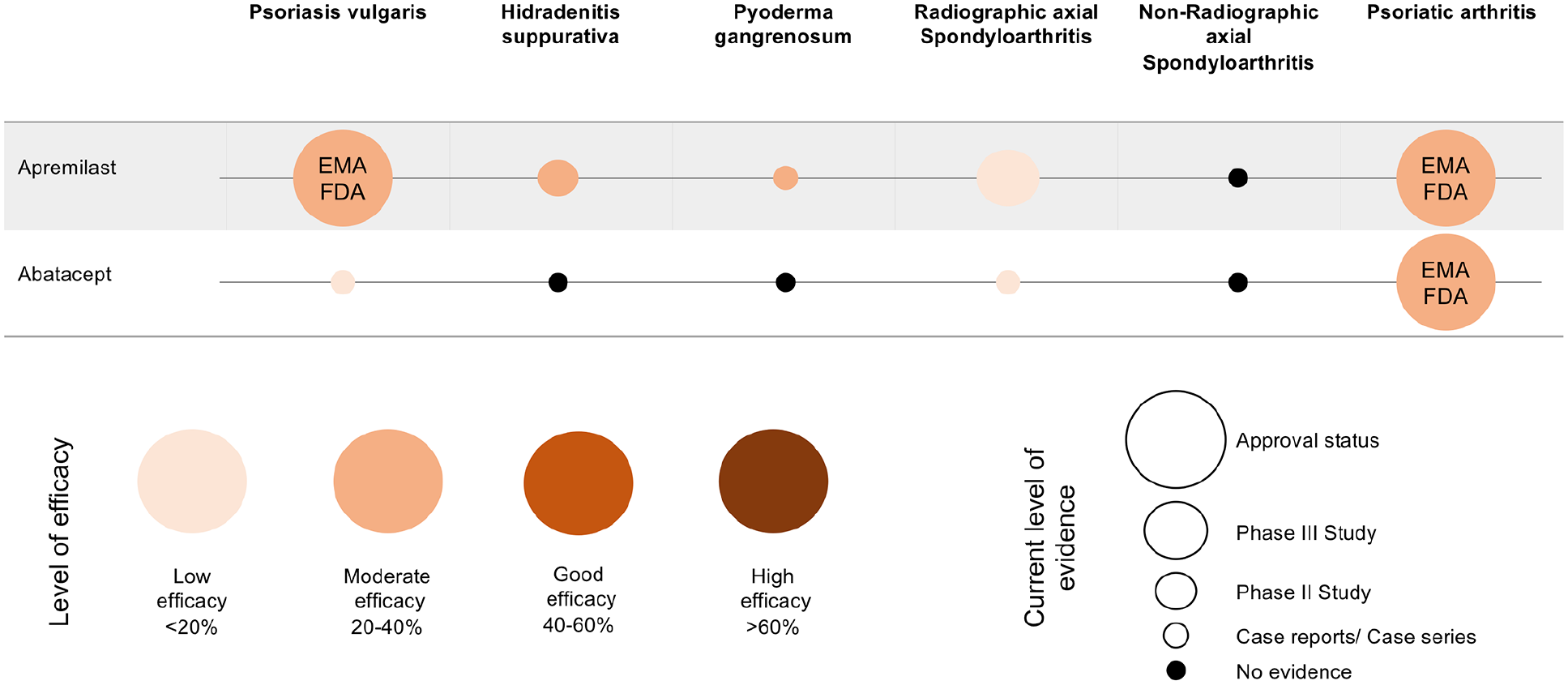
Efficacies of abatacept and apremilast in PsO, HS, PG, rSpA, nrSpA and PsA. The size of the circle presents the level of evidence, the colour represents the level of efficacy in the indicated disease.217,222–228 As standards of therapeutic efficacies in PsO PASI90, in HS HiSCR, in SpA ASAS40 and in PsA ACR50 are used. The scheme is adapted from Eyerich et al. 139
Conclusion
Skin manifestations in SpA are various. A knowledge of associated disorders and their therapeutic approaches are key to an interdisciplinary approach and optimal patient care. SpAs present in various clinical pictures and generally add an increased impact on quality of life since excessive scaling, discharge, itch and pain are common symptoms of skin inflammation. This review focussed on the most frequent comorbidities and their approved therapies. Most commonly, PsO manifests with numerous clinical presentations. In SpA associated with IBD, another distinct group of skin manifestation such as PG, HS and EN are often seen. Thus, there have been reports of a variety of inflammatory skin responses such as erythema elevatum diutinum, discoid lupus and other vasculitides in patients with AS.229–231 Pathomechanistically, they all seem to share a common pathway, with dysregulation of classic inflammatory cytokines such as IL-1, IL-12/23, IL-17 and IL-23. Common genetic factors like HLA-B27 are also typical in these cohorts. Strikingly, IL-23 inhibitors show impressive response rates in PsO, with a high number of patients achieving complete remission and good efficacy in associated peripheral arthritis but are less effective in axial involvement. Hence, the efficacy of well-established targeted therapies differs greatly in associated morbidities. As of today, we need to find out why certain diseases respond differently, whether we can combine targeted therapies in order to achieve adequate responses in different entities without increasing the risk of side effects in our patients. Current clinical trials for several agents such as bimekizumab, filgotinib or bermekimab are likely to widen the number of effective therapeutic options for patients with inflammatory diseases.232,233
In order to achieve optimal patient care, individuals with complex inflammatory diseases need to be screened for these comorbidities in daily practice. In fact, knowledge of associated disorders, their pathomechanisms and therapeutic approaches may lead to different therapeutic strategies and may open doors for in-label therapies of new anti-inflammatory agents.
Footnotes
Conflict of interest statement
KM has received honoraria or travel expenses for lecture and research activities from Abbvie, Biogen, Celgene, Janssen-Cilag and UCB Pharma.
AS has received travel expenses from Janssen-Cilag and Celgene
DP has received grant/research support from AbbVie, Lilly, MSD, Novartis, and Pfizer; has been a consultant for AbbVie, Biocad, Gilead, GSK, Lilly, MSD, Novartis, Pfizer, Samsung and UCB; and served on the speakers bureau for AbbVie, BMS, Lilly, MSD, Novartis, Pfizer and UCB.
KG has received honoraria or travel expenses for lecture and research activities from Abbvie, Almirall, Biogen, Boehringer Ingelheim, Bristol-Myers Squibb, Celgene, Delenex, Eli Lilly, Galderma, Janssen-Cilag, Medac, MSD, Novartis, Pfizer and UCB Pharma.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
