Abstract
Objectives:
The clinical benefit of conventional disease-modifying antirheumatic drugs (cDMARDs) for treating ankylosing spondylitis (AS) is generally limited to improvements in peripheral arthritis. However, cDMARDs could be conditionally considered as alternatives to established drugs for improving axial manifestations in exceptional circumstances. However, there are few studies of the impact of cDMARDs on radiographic progression outcomes. Therefore, we investigated the effectiveness of cDMARDs on radiographic progression in AS.
Methods:
Among 1280 AS patients at a single hospital from 2000 to 2018, 301 who had been treated with sulfasalazine (SSZ) or methotrexate (MTX) were enrolled. For each patient, the entire follow-up period was split into 1-year intervals. Each interval was classified as either an “on-cDMARD” interval, which was a period of treatment with SSZ alone, MTX alone, or a combination of SSZ and MTX, or an “off-cDMARD” interval, which was a period without cDMARD treatment. Radiographic progression was scored using the modified Stoke Ankylosing Spondylitis Spinal Score (mSASSS). The relationship between cDMARD use and radiographic progression within the intervals, defined as the rate of mSASSS progression, was investigated using linear models with adjustment for potential confounding covariates and for clustering among observations from the same patient.
Results:
The 732 on-cDMARD intervals and 1027 off-cDMARD intervals were obtained from enrolled patients. In multivariable regression analysis, there was no significant association between cDMARDs and the rate of mSASSS progression (β = −0.081, p = 0.418). The mean adjusted mSASSS change per year was 0.610 from on-cDMARD intervals and 0.691 from off-cDMARD intervals.
Conclusion:
Treatment with cDMARDs may not reduce radiographic progression in AS patients.
Keywords
Introduction
Conventional disease-modifying antirheumatic drugs (cDMARDs) are widely used to treat rheumatic diseases. For patients with spondyloarthritis (SpA), cDMARDs have been recommended to treat peripheral arthritis.1–3 However, in situations where non-steroidal anti-inflammatory drugs (NSAIDs) are contraindicated or not tolerated, or in resource-poor settings where tumor necrosis factor (TNF) inhibitors are not readily accessible, cDMARDs could be cautiously considered as an alternative.1–3
Although guidelines state that the conditional use of cDMARDs should be considered primarily in exceptional circumstances and with low expectations for efficacy in patients with predominantly axial manifestations, some previous studies have shown that cDMARDs offer some benefit for improving axial symptoms.4,5 Indeed, cDMARDs still constitute a portion of the treatment armamentarium for patients with ankylosing spondylitis (AS) in some regions, and the prescription frequency varies between countries, ranging from 10% to 87%.3,6–9 National reimbursement policies or financial constraints might partly contribute to inter-country variation.6,7,10 However, given the nature of slowly progressing disease, there have been few examinations of the anti-inflammatory effect of cDMARDs on the axial skeleton over a long observation period among large patient samples. Additionally, no studies have evaluated the impact of cDMARDs on radiographic progression using validated outcome measures in patients with AS.
Previously, we demonstrated TNF inhibitor effectiveness for slowing radiographic progression using time intervals during treatment. 11 However, it was difficult to evaluate the effect of cDMARDs on radiographic progression because the study design was focused on TNF inhibitors. Therefore, we investigated the effectiveness of cDMARDs on spinal radiographic progression from real-world longitudinal data based on a different design than previously used.
Materials and methods
Patients and clinical assessment
Data from a single-center cohort of patients with AS between January 2001 and December 2018 were retrospectively reviewed. Among a total of 1280 patients who satisfied the modified New York criteria, 12 301 patients treated with sulfasalazine (SSZ) or methotrexate (MTX) who also had at least two sets of spinal radiographs were included. This study was conducted in accordance with the Helsinki Declaration and was approved by the Hanyang University Seoul Hospital Institutional Review Board (HYUH 2014-04-010). The need for patient consent was waived by the institutional review board because of the retrospective nature of our study.
Demographic and clinical features including age, sex, disease duration, human leukocyte antigen (HLA)-B27 positivity, history of uveitis, peripheral arthritis, serum erythrocyte sediment rate (ESR) and C-reactive protein (CRP) levels, Bath Ankylosing Spondylitis Disease Activity Index (BASDAI), and concomitant drugs were obtained from patient medical records.
Radiographic assessment
Radiographic images of the cervical and lumbar spine were obtained from 1280 patients and were independently scored according to the modified Stoke Ankylosing Spondylitis Spinal Score (mSASSS) by two radiologists. 13 The average number (standard deviation) (SD) of mSASSS measurements per patient during the entire period was 4.6 (1.2) and the average interval (SD) between mSASSS measurements was 2.4 (0.7) years. Two readers who were blinded to patients’ clinical data scored radiographs in chronological order. The intra-class correlation coefficient (ICC) values for intra-observer reliability [ICC = 0.978 (95% confidence interval (CI)): 0.976, 0.979] and inter-observer reliability [ICC = 0.946 (95% CI: 0.941, 0.950)] were both excellent.
Time intervals according to cDMARD treatment
Time intervals were created for each of the 301 patients who were included in the analysis. The intervals were based on cDMARD treatment and the period of exposure to TNF inhibitors was not included. To capture as much data as possible, the interval included the period from the baseline to immediately before TNF inhibitor initiation if the treatment was switched to TNF inhibitors during the entire follow-up period. Then, the follow-up period was split into 1-year intervals and radiographic progression within each interval was estimated according to the rate of mSASSS increase (i.e. progression) over a year. Each interval was classified as either an “on-cDMARD” interval, which was the treatment period with SSZ alone, MTX alone, or a combination of SSZ and MTX, or an “off-cDMARD” interval, which was the period without cDMARD treatment. SSZ, MTX, NSAIDs, and glucocorticoids were considered to be sustained during the interval if they were prescribed during more than half of the interval. The mSASSS values at the beginning and end points in each interval were imputed with a linear interpolation method assuming a linear course during the time interval (Figure 1). ESR, CRP, and BASDAI were also imputed at each time point of the intervals by linear interpolation.
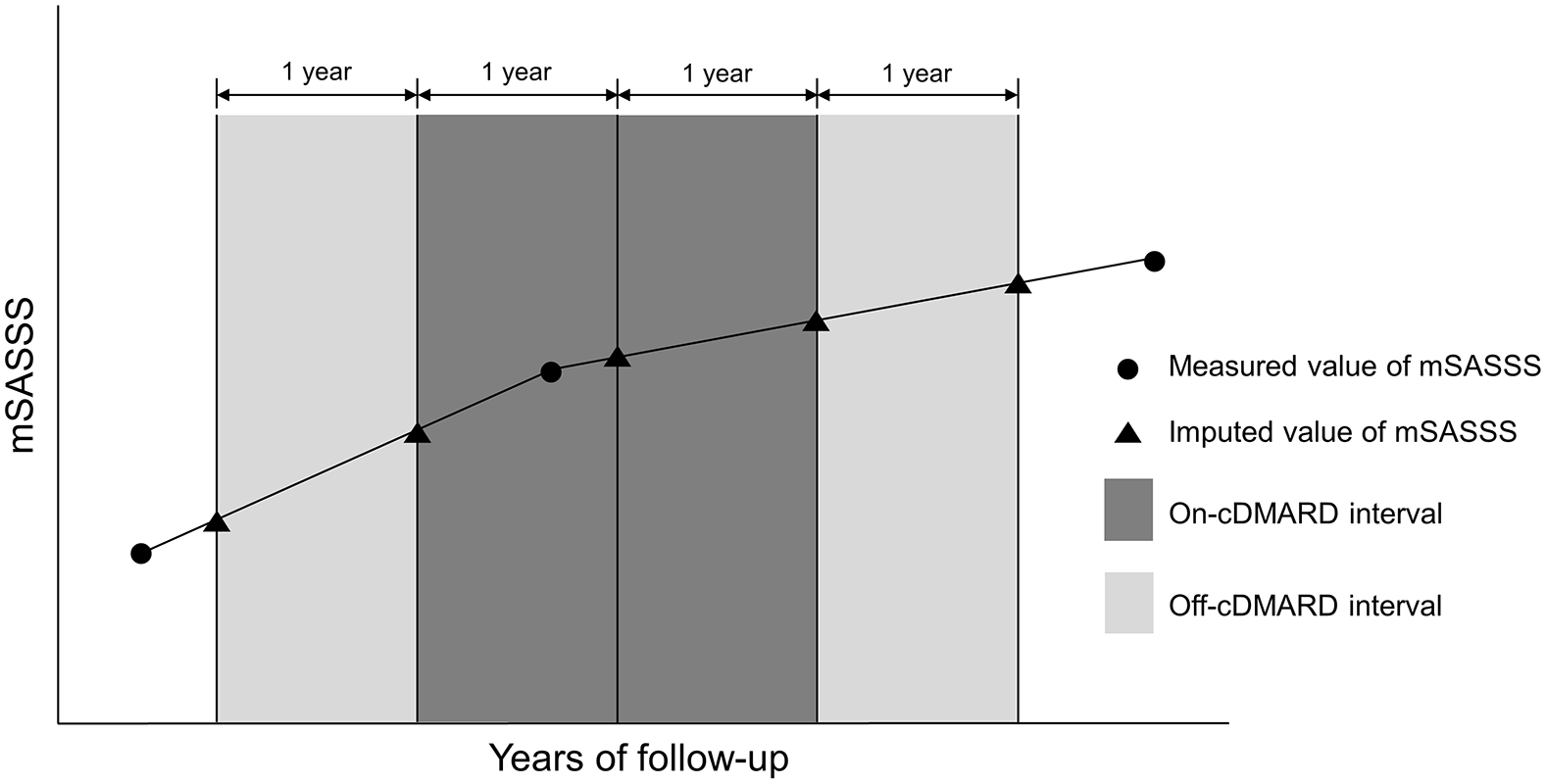
Definition of on-cDMARD intervals and off-cDMARD intervals. The mSASSS values at the beginning and end points in each interval were imputed with a linear interpolation method using the values measured before and after each time point.
Statistical analyses
The relationship between radiographic progression, which was measured as the rate of mSASSS progression, and clinical factors was investigated based on interval-level data. Highly skewed clinical factors were transformed using a log or a square-root function. To account for correlations between repeated measurements, we used linear models with an exchangeable correlation structure estimated by generalized estimating equations. Any variables with a potentially significant impact on the outcome (p-value ⩽ 0.1) in the univariate models were entered into the multivariable regression models. HLA-B27 was excluded due to its high frequency of positives and non-significant p-value. Additionally, CRP and BASDAI were excluded due to a high correlation with ESR and many missing values, respectively. The impact of cDMARDs on radiographic progression was analyzed in the multivariable model with adjustment for potential confounding covariates and imputation of missing values (model 1). In model 2, the impact of SSZ and MTX were estimated individually. The mean mSASSS change per year according to whether cDMARDs were administered was also calculated from the multivariable models with confounding factors fixed. All statistical analyses were performed using the R statistical language version 3.5.1. p-values less than 0.05 were considered statistically significant.
Results
Patient characteristics
Of the 1280 patients enrolled, 301 patients who had been treated with cDMARDs and who had sufficient clinical data and sets of radiographs to create at least one on-cDMARD interval and one off-cDMARD interval were selected. Patient clinical characteristics are described in Table 1. Most patients were male (86.0%), positive for HLA-B27 (97.0%), and the mean (SD) age at diagnosis was 31.4 (9.2) years. Eye involvement and peripheral arthritis was observed in 41.4% and 60.8% of cases, respectively.
Baseline demographic characteristics of the patients with ankylosing spondylitis.

HLA, human leukocyte antigen; SD, standard deviation.
Time interval characteristics
Among the 301 patients, 1759 intervals comprising 732 on-cDMARD intervals and 1027 off-cDMARD intervals were obtained (Figure 2). Among the on-cDMARD intervals, the number of intervals for SSZ treatment alone, MTX treatment alone, and combined SSZ and MTX was 704, 146, and 118, respectively. Clinical characteristics based on the intervals are summarized in Table 2. Gender, HLA-B27 positivity, eye involvement, and peripheral arthritis were investigated according to cDMARD interval. Additionally, mean (SD) values for age, ESR, CRP, BASDAI, and mSASSS at the start of the intervals were calculated. The mean mSASSS (SD) at the start of on-cDMARD intervals was 12.35 (13.20) and the mean at the start of off-cDMARD intervals was 14.18 (15.42).
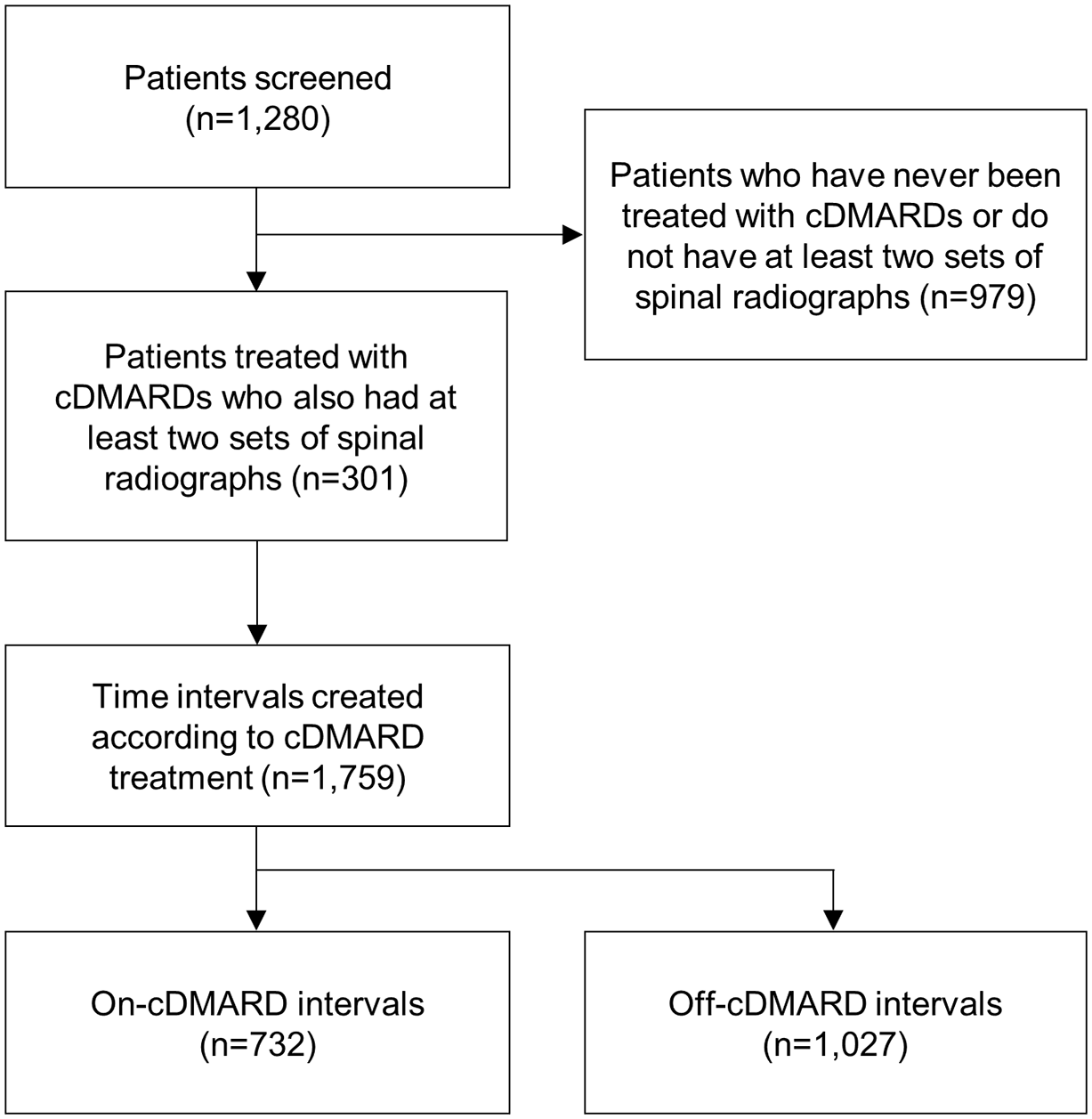
Flowchart of creating time intervals for each of the patients.
Clinical characteristics of time intervals classified according to cDMARD treatment.

Considered to be sustained during the interval if they were prescribed during more than half of the interval.
BASDAI, Bath Ankylosing Spondylitis Activity Index; cDMARD, conventional disease-modifying antirheumatic drug; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; HLA, human leukocyte antigen; mSASSS, modified Stoke Ankylosing Spondylitis Spine Score; MTX, methotrexate; NSAIDs, non-steroidal anti-inflammatory drugs; SD, standard deviation; SSZ, sulfasalazine.
Association between covariates and radiographic progression
Table 3 shows the association between covariates and the rate of mSASSS progression. The following variables were identified as potentially related to the outcome in univariate regression analyses: age [β = 0.022 (95% CI: 0.010, 0.034) p < 0.001)], ESR (log) at the interval start [β = 0.195 (95% CI: 0.107, 0.283) p < 0.001], CRP (log) at the interval start [β = 0.285 (95% CI: 0.097, 0.472) p = 0.003], and eye involvement [β = 0.623 (95% CI: 0.298, 0.948) p < 0.001] were associated with an increase in the mSASSS progression rate. Meanwhile, female patients and patients with peripheral arthritis were associated with a reduction in the mSASSS progression rate [β = −0.290 (95% CI: −0.586, 0.006) p = 0.055 and β = −0.540 (95% CI: −0.857, −0.223) p = 0.001, respectively]. Taking NSAIDs or glucocorticoids was found to have no significant effect on mSASSS progression.
Association between covariates and the rate of mSASSS progression in the regression models.

Variables that were significant in univariate analysis at p-value ⩽ 0.1 were included.
Excluded from multivariable analysis due to its well-known high correlation with ESR.
BASDAI, Bath Ankylosing Spondylitis Activity Index; cDMARDs, conventional disease-modifying antirheumatic drugs; CI, confidence interval; HLA, human leukocyte antigen; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; mSASSS, modified Stoke Ankylosing Spondylitis Spine Score; NSAIDs, non-steroidal anti-inflammatory drugs.
Impact of cDMARDs on radiographic progression
Multivariable regression analysis was performed to confirm the association between cDMARDs and radiographic progression with adjustment for variables that were potentially significant in the univariate models (Table 3). In model 1, eye involvement and log-transformed ESR at the interval start were significantly associated with an increase in progression of radiographic damage [β = 0.572 (95% CI: 0.264, 0.880) p < 0.001 and β = 0.176 (95% CI: 0.087, 0.265) p < 0.001, respectively]. Being female and peripheral arthritis remained significantly associated with a reduced rate of mSASSS progression [β = −0.449 (95% CI: −0.782, −0.117) p = 0.008 and β = −0.508 (95% CI: −0.810, −0.206) p = 0.001, respectively]. In contrast, there was no significant association between cDMARD use and the rate of mSASSS progression with adjustment for independent significant confounders [β = −0.081 (95% CI: −0.276, 0.115) p = 0.418].
Multivariable model 2 analyzed the individual associations of SSZ and MTX with radiographic progression. Significant variables included eye involvement, log-transformed ESR at the start of the interval, being female, and peripheral joint involvement, as in model 1. Neither SSZ nor MTX were significantly associated with a reduced radiographic progression rate in their respective intervals [β = −0.011 (95% CI: −0.211, 0.189) p = 0.913 and β = −0.180 (95% CI: −0.439, 0.078) p = 0.172].
Adjusted mSASSS change per year according to cDMARD treatment
Mean adjusted mSASSS changes per year are shown in Table 4. mSASSS change per year with or without cDMARDs was estimated from multivariable regression model 1, while other covariates were fixed at their mean values (0.610 versus 0.691). Results according to monotherapy or combination therapy were also estimated from model 2 (0.485 for SSZ-MTX combination therapy, 0.665 for SSZ monotherapy, and 0.496 for MTX monotherapy).
Mean mSASSS change within cDMARD intervals estimated from the multivariable models.

Adjusted for sex, eye involvement, peripheral joint involvement, and ESR at the interval start (log).
cDMARD, conventional disease-modifying antirheumatic drug; mSASSS, modified Stoke Ankylosing Spondylitis Spine Score; MTX, methotrexate; SSZ, sulfasalazine.
Discussion
In this study, we showed that treatment with cDMARDs including SSZ and MTX does not decelerate radiographic damage progression in the spine. To our knowledge, this is the first study to show that cDMARDs have no inhibitory effect on radiographic progression using mSASSS values. Our results underpin the established recommendations for a limited role of cDMARDs for treating AS.
cDMARDs have been shown to prevent radiographic progression in rheumatoid arthritis (RA), as well as decrease pain and inflammation. 14 However, cDMARDs in AS have been recognized as ineffective for suppressing inflammation in the axial skeleton,15–17 and they have only been investigated in a limited number of studies of patients with predominant axial disease.18,19 Although a placebo-controlled study showed that SSZ has some efficacy for improving axial manifestations, only a small proportion of the subjects reported an effect, and the observation period was relatively short given the slowly progressing nature of the disease. 5 On the other hand, there is growing evidence that long-term treatment with biological DMARDs (bDMARDs), such as a TNF inhibitor or an interleukin (IL)-17 inhibitor, is associated with a preventive effect on spinal radiographic progression.20,21 In contrast to bDMARDs, our results showed that cDMARDs have no efficacy of retarding radiographic damage in the spine.
Unlike RA, the major pathological process associated with SpA is confined to the entheses, which are the interfaces between tendons or ligaments and bone, and the interface between cartilage and bone predominantly in the sacroiliac joints and spine. 22 Mechanical stress or infection to these structures may trigger entheseal inflammation and adjacent osteitis, which is often followed by a subsequent tissue response process leading to syndesmophyte formation in the spine.23,24 However, cDMARDs, well known for their efficacy for synovitis, do not appear to effectively suppress entheseal inflammation.25,26 Furthermore, recent findings suggest that activation of the IL-23/IL-17 pathway is implicated in expanding entheseal inflammation and indirectly promoting new bone formation at entheseal sites. 27 Additionally, TNF has an osteogenic differentiation effect that it might exhibit at enthesitis sites where osteoclasts and tendon-derived osteoblasts may be disconnected.28,29 In the context of immunopathology, this paradoxical action of cytokines in bone dynamics can partly account for pathogenic bone formation in AS. 30 Thus, blockade of the mechanisms and involved cytokines related to enthesitis-induced bone formation would be a superior approach to cDMARDs whose anti-inflammatory effect on enthesitis is unclear. Indeed, IL-17 inhibition was observed to slow radiographic progression after 2 years of follow-up, and low radiographic changes were sustained at 4 years of follow-up.31,32
Additionally, according to studies based on magnetic resonance imaging (MRI), new syndesmophytes are more likely to develop at sites of prior inflammatory lesions in the spine and new bone formation may also proceed through a process of subsequent fat metaplasia following inflammation.33–35 Therefore, targeting early resolution of inflammatory lesions and prevention of reparative processes would support slowing radiographic progression. 36 This inflammation-suppression-mediated effect has been confirmed in recent studies.11,37,38 However, the anti-inflammatory effect of cDMARDs has been shown to be weaker than that of TNF inhibitors in a randomized controlled study, 39 and improvement of sacroiliac joint and spine inflammation according to MRI scores in patients with early axial SpA was significantly less in the SSZ-treated group than the etanercept-treated group.40,41 The inadequate efficacy of cDMARDs to suppress inflammation in the axial skeleton, despite previous reports of easing inflammatory back pain, 5 would not in turn lead to an inhibitory effect on structural damage progression. Accordingly, based on our results and established findings from previous studies, bDMARDs outperform cDMARDs with respect to the ultimate therapeutic goal—inhibition of structural progression.
In our multivariable regression analysis of clinical factors, ESR at the start of the interval was significantly associated with greater mSASSS change, as shown in previous reports,37,38 reflecting that baseline inflammation is a strong predictor of future radiographic progression. Eye involvement was also identified as a significant variable affecting radiographic progression. Given that uveitis presence in AS is associated with disease duration, 42 the long observation period of our study may have increased uveitis incidence in this study, thus contributing to the strong correlations we found. Conversely, the mSASSS progression rate in patients with peripheral arthritis was significantly lower than in patients without peripheral arthritis, which is consistent with a previous finding. 43 The inhibitory effect of NSAIDs on radiographic progression, which has been shown in some previous studies,44,45 was not confirmed in this study. There was no significant association between NSAID use, which was based on 1-year intervals with on/off periods of NSAID treatment, and a reduced mSASSS progression rate. However, because a considerable number of patients received on-demand NSAID treatment, it was difficult to collect complete information related to the dose and frequency of NSAID intake. Therefore, NSAID intake was based on the use of binary measurements and this might have influenced the outcome given that the effect of NSAIDs on structural progression may be dose-related. 45
The mean adjusted mSASSS change per year calculated from the multivariable models was lower than that of our previous study, where the measured mSASSS value was 0.914 with TNF inhibitor therapy and 0.970 without TNF inhibitor therapy. 11 The relatively lower mSASSS change value in this study might be attributable to the clinical characteristics of patients who have not been exposed to TNF inhibitors during the included observation period due to a lower risk of spinal damage progression. Meanwhile, according to two recent systematic reviews concerning the effect of therapy on radiographic progression, mean mSASSS change varied considerably between studies.20,21 Because different follow-up durations and different data-management approaches can heavily influence outcomes, especially given the slowly progressing nature and heterogeneity of AS and the added problem of confounding by indication, a direct comparison of our results with other studies would be not appropriate.
This study has several limitations. First, smoking, which has been shown to be predictive of spinal radiographic progression, 46 was not included in the covariates due to too many missing values. There was a significant lack of available data on smoking status in medical records, forcing them to be excluded from the analysis. Thus, smoking was not adjusted for and this may have influenced the outcome. Additionally, other factors that may have an impact on structural damage progression, such as obesity 47 or occupational status (white-collar versus blue-collar), 48 were not included in the covariates. Second, we performed this study under the assumption that patients took their medicine regularly and as prescribed. Therefore, there may be unmeasured confounders, such as non-adherence or a discrepancy between the date of prescription and administration. Third, because this study was based on medical records during a long-term observation period with variability in follow-up periods, continuous variables were imputed by the interpolation method at a specific time point. Missing mSASSS data at beginning and end timepoints of the intervals were also handled by linear interpolation with consideration of the slow progression of spinal structural damage. Therefore, the imputed values may be different from the actual values, which could introduce unexpected bias.
However, given that a randomized placebo-controlled comparison of a cDMARDs treatment group with an untreated group in patients with axial SpA is not feasible, our results derived from real-world data have strength in that they reflect daily clinical practice. Furthermore, as the first study to show that cDMARDs are not effective in slowing spinal radiographic progression based on validated outcome measures, this could serve as a reference study for other countries where reimbursement regulations require routine use of cDMARDs before switching to TNF inhibitor therapies or where financial constraints limit the use of TNF inhibitors.6,7,49,50
Conclusion
Our study shows that cDMARDs have no significant effect in slowing radiographic progression in AS patients. Given the recent findings of the effectiveness of biologics in slowing spinal structural damage, use of TNF inhibitors or IL-17 inhibitors, rather than cDMARDs, should be considered to inhibit spinal damage especially for patients with a high risk of radiographic progression.
Footnotes
Acknowledgements
We are grateful to the nurses who helped collect patient data in the clinics for many years.
Contributors
BSK, JSO, SYP, and THK made contributions to the study conception and design. All authors contributed to data acquisition, analysis, or interpretation. SL and KBJ scored the spinal radiographs independently. JSO and SYP were responsible for the statistical analyses. THL and BSK drafted the manuscript and all coauthors were involved in critical revisions for maintenance of intellectual content. NB and THK provided administrative, technical, or material support. THK had full access to all study data and takes responsibility for data integrity and data-analysis accuracy. All authors approved the final version to be submitted for publication.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Ethics approval
This study was approved by the Hanyang University Seoul Hospital Institutional Review Board (HYUH 2014-04-010).
