Abstract
Background:
The presence of ⩾3 corner inflammatory lesions has been proposed as the definition of a positive spinal magnetic resonance imaging (MRI) for axial spondyloarthritis (axSpA), but subsequent studies showed inconclusive findings. Our objective was to evaluate whether locations of corner inflammatory lesions (CILs) would affect the diagnostic utility of MRI in axSpA.
Method:
Two groups were consecutively recruited from eight rheumatology centers in Hong Kong. The ‘axSpA’ group included 369 participants with a known diagnosis of axSpA. The ‘non-specific back pain’ (NSBP) control group consisted of 117 participants. Clinical, biochemical, and radiological parameters were collected and all patients underwent MRI of the spine and sacroiliac joints. CILs were assessed based on their locations (cervical, thoracic or lumbar) to determine the optimal cutoff for diagnosis.
Results:
The cutoff of ⩾5 whole spine CILs (W-CILs) and ⩾3 thoracic spine CILs (T-CILs) had comparable specificity to MRI sacroiliitis. Of 85/369 axSpA patients without sacroiliitis on conventional radiograph or MRI, 7 had ⩾5 W-CILs and 11 had ⩾3 T-CILs. Incorporating the proposed cutoffs into Assessment of SpondyloArthritis international Society axSpA criteria, ⩾5 W-CILs and ⩾3 T-CILs had similar performance when added to the imaging criteria for sacroiliitis (sensitivity 0.79 versus 0.80, specificity 0.92 versus 0.91).
Conclusion:
Spinal MRI provided little incremental diagnostic value in unselected axSpA patients. However, in patients without sacroiliitis on MRI or radiographs, 8–13% might be diagnosed by spinal MRI. Thoracic and whole spine MRI had similar diagnostic performance using the proposed cutoff of ⩾5 W-CILs and ⩾3 T-CILs.
Introduction
Axial spondyloarthritis (axSpA) is a spectrum of chronic inflammatory diseases affecting the axial skeleton. The presence of sacroiliitis, on either conventional radiographs or magnetic resonance imaging (MRI), forms the backbone criterion in the imaging arm of the Assessment of SpondyloArthritis international Society (ASAS) classification criteria for axSpA.1,2 However, the inflammatory process is not limited to sacroiliac (SI) joints. One study has shown that spinal inflammation was found in half of patients with active non-radiographic axSpA in the absence of sacroiliitis on MRI. 3
A corner inflammatory lesion (CIL) is defined as an increased short tau inversion recovery (STIR) sequence signal at the corner of a vertebral body, which remains normal or hypointense in T1 weighted sequence, present in one or more sagittal slices inclusive of the spinal canal. 4 Multiple CILs increase the diagnostic confidence of axSpA. 5 The presence of ⩾3 CILs has been proposed as the definition of a positive MRI of the spine for axial spondyloarthritis by the ASAS/Outcome Measures in Rheumatology (OMERACT) working group. 4 The diagnostic utility of this proposed criterion has been tested by Weber et al. in 130 patients, which has shown that when used alone, whole spine CILs have poor positive and negative likelihood ratios (LRs) (positive LR 1.74–2.36, negative LR 0.75–0.84). 6 In addition, combined spine and SI joint MRI add little incremental diagnostic value when compared with SI joint MRI alone. 7 However, no study has evaluated whether the location of CILs will affect the diagnostic utility of these lesions.
In this study, we aimed to systematically evaluate the diagnostic utility of CILs at different locations (cervical, thoracic, and lumbar) in a large tertiary cohort of 486 patients.
Methods
This was a retrospective study of prospectively acquired data from an on-going multicenter cohort. Detailed methods have been stated in our previous publication 8 and described as below. Data of participants recruited between May 2016 and August 2019 were analyzed and presented in this study.
Participant recruitment
Two groups of participants were consecutively recruited from eight rheumatology centers in Hong Kong (Queen Mary Hospital, Grantham Hospital, Tung Wah Hospital, Pamela Youde Nethersole Eastern Hospital, Caritas Medical Center, Tseung Kwan O Hospital, Kwong Wah Hospital, and Prince of Wales Hospital). The “axSpA” group included participants with a known expert diagnosis of axSpA and fulfilling the 2009 ASAS criteria for axSpA. 2 Other inclusion criteria were (i) age >18 years, (ii) current back pain of all types, (iii) ability to give written consent, and (iv) biologics-naïve. Exclusion criteria included (i) pregnancy and (ii) inability to undergo MRI examination. The “non-specific back pain” (NSBP) control group consisted of participants with chronic back pain without a diagnosis of axSpA. NSBP was defined as back pain without a specific and identified pathology such as trauma, tumor, infection, deformity, and nerve compression.
Confirmation of disease diagnosis
An independent rheumatologist (HHLT), blinded to the MRI data, confirmed correct patient allocation into the axSpA and NSBP groups according to previous clinical records.
Clinical and demographic data
Clinical and demographic data were collected at study entry from all recruited participants. These data included age, sex, smoking status, drinking status, back pain duration and severity (scored on a scale of 0–10), and family history of axSpA. Physical assessments were performed for peripheral joint counts, enthesitis, dactylitis and Bath Ankylosing Spondylitis Metrology Index. 9 All participants were asked to complete three questionnaires: Bath Ankylosing Spondylitis Disease Activity Index, 10 the Bath Ankylosing Spondylitis Functional Index, 11 and the Bath Ankylosing Spondylitis Global Index. 12 Blood parameters including HLA-B27, C-reactive protein level, and erythrocyte sedimentation rate were recorded. Ankylosing Spondylitis Disease Activity Index was calculated based on both erythrocyte sedimentation rate and C-reactive protein.
Grading of radiographs of lumbosacral (LS) spine and SI joints
Radiographs of SI joints were graded according to the Modified New York criteria. 13 Bilateral sacroiliitis grade 2 or above, or unilateral grade 3 or above were defined as radiological AS. All LS spine and SI joint radiographs were graded by a musculoskeletal radiologist (KHL) and a rheumatologist (HHLT), with disagreements resolved by consensus.
Scoring of MRI of spine and SI joints
All recruited participants underwent MRI scans of the whole spine and bilateral SI joints at study entry using a 3T Achieva scanner (Philips Healthcare, Best, The Netherlands). A single torso coil was used to image both the spine and the sacroiliac joints. STIR sequence and T1 weighted images were obtained simultaneously.
The following parameters were employed for the SI joint, both the semi-coronal and semi-axial plane: fast spin-echo STIR. The field of view (FOV) was 20 cm × 20 cm; slice thickness 5 m with no gap; the number of excitation was 2 and reptition time (TR)/ time to echo (TE) was 3500/80 ms, inversion delay being 140–160 ms; matrix size, 248 × 200. Twenty-four slices were obtained and the total scan time was 5 min 7 s.
For the spine MRI, fast spin echo STIR sagittal was performed using the following parameters: FOV 24 cm × 14 cm; 4 mm thickness without gap; matrix size 220 × 176; number of signal averages/ acquisitions (NSA). The TR/TE/TI was 4760/80/140–160 ms. Fifteen slices were obtained, making a total scan time of 3 min 38 s.
MRI of the SI joints was scored by two independent rheumatologists (HYC and SCWC, with respectively 8 years and 4 years of experience reading MRI of the spine and SI joint) according to the Spondyloarthritis Research Consortium of Canada scoring system, 14 blinded to clinical, radiological parameters and MRI of the spine. A “positive MRI” of the SI joint, indicating sacroiliitis, was defined according to the ASAS handbook 15 as one signal of subchondral bone marrow edema on at least two slices, or more than one signal on a single slice in typical anatomical position (subchondral or periarticular). Similarly, spine MRI was read by HYC and LHP (respectively with 8 years and 4 years of experience in reading MRI of the spine and SI joint) to identify CILs, defined according to the ASAS definition 5 of an increased STIR signal at a vertebral corner, which remains normal or hypointense in T1 weighted sequence, present in at least one sagittal slice (Figure 1). Vertebral levels from C2 to L5 were scored.
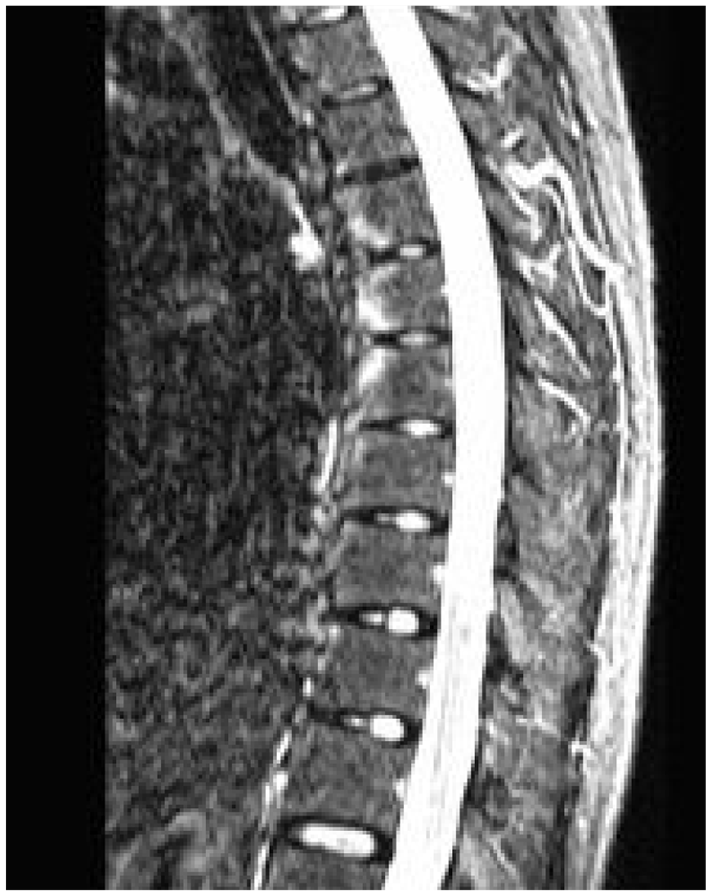
Corner inflammatory lesions.
Ethics approval
The study was approved by the Institutional Review Board of The University of Hong Kong/Hospital Authority Hong Kong West Cluster (reference no. UW 14–085). It was conducted in accordance with the Declaration of Helsinki and the guidance of Good Clinical Practice, 30 November 2006.
Statistical analyses
Baseline demographics and clinical characteristics in the axSpA and NSBP groups were compared using Student’s t-tests and chi square tests. The prevalence of at least one CIL at different vertebral levels in the two groups was expressed as percentages. The receiver operating characteristic (ROC) curve was constructed, and the area under curve (AUC) was used to determine the diagnostic utility of CILs at different locations (anterior lesions, posterior lesions, and CIL at different vertebral levels). Diagnostic utility of CILs at different cutoff values was determined by calculating the sensitivity, specificity, positive and negative predictive values, and positive and negative LRs. The locations of CILs with the highest sensitivity and specificity greater than that of “positive MRI” sacroiliitis were chosen for further analyses.
The diagnostic utility of these CIL cutoff numbers was further tested when incorporated into the imaging arm of the ASAS criteria. The sensitivity, specificity, positive and negative predictive values, and positive and negative LRs were calculated for the imaging arm of the original ASAS criteria. Similarly, we evaluated the utility of the ASAS criteria when we replaced positive MRI sacroiliitis with MRI spine using the CIL cutoff values, and when we combined both MRI SI joint and MRI spine. Clinical utility was determined according to specifications established by Jaeschke et al. 16 where a positive LR of 10 and a negative LR of 0.1, positive LR of 5–10 and a negative LR of 0.1–0.2, positive LR of 2–5 and a negative LR of 0.2–0.5, and positive LR of 1–2 and a negative LR of 0.5–1 correspond to substantially, moderately, small, and poor/rarely clinically relevant values respectively.
Cohen’s kappa was used to measure agreement in MRI scoring (sacroiliitis and CILs) by the two readers. Value ranges of 0.00–0.20, 0.21–0.40, 0.41–0.60, 0.61–0.80, 0.81–1.00 represented slight, fair, moderate, substantial, and near perfect agreement, respectively. 17
All statistics were performed using the International Business Machines Corporation Statistical Package for the Social Sciences (IBM SPSS) package 25.0. A p-value of less than 0.05 was defined as statistically significant in all analyses.
Results
Three hundred and sixty-nine patients with axSpA and 117 participants with NSBP were included. Baseline demographics, clinical parameters, and number of MRI spinal lesions of the axSpA and NSBP groups are described in Tables 1 and 2. Our cohort is characterized by long disease duration, with axSpA group having younger age (43.8 ± 13.6 years versus 49.1 ± 15.5 years; p = <0.01), male predominance (59.9% versus 29.2%; p < 0.01), a greater proportion of smokers (27.9% versus 13.7%), and higher levels of CRP (1.2 ± 2.2 mg/dL versus 0.5 ± 1.8 mg/dL; p < 0.01).
Baseline characteristics.

N/A = no sacroiliitis; absence of sacroiliitis on X-ray or magnetic resonance imaging.
AS, ankylosing spondylitis; ASDAS, Ankylosing Spondylitis Disease Activity Index; axSpA, axial spondyloarthritis; BASDAI, Bath Ankylosing Spondylitis Disease Activity Index; BASFI, Bath Ankylosing Spondylitis Functional Index; BASGI, Bath Ankylosing Spondylitis Global Index; BASMI, Bath Ankylosing Spondylitis Metrology Index; CIL, corner inflammatory lesion; CRP, C-reactive protein; C-spine, cervical spine; ESR, erythrocyte sedimentation rate; HLA, human leucocyte antigen; IBP, inflammatory back pain; L-spine, lumbar spine; mSASSS, modified stoke ankylosing spondylitis spinal score; nr-axSpA, non-radiographic axial spondyloarthritis; NSBP, non-specific back pain; T-spine, thoracic spine; W-spine, whole spine.
Frequency of CILs in different subgroups of patients.

AS, ankylosing spondylitis; axSpA, axial spondyloarthritis; CIL, corner inflammatory lesion; nr-axSpA, non-radiographic axial spondyloarthritis; MRI, magnetic resonance imaging; NSBP, non-specific back pain; SI, sacroiliac.
Inter-reader agreement of positive MRI sacroiliitis was almost perfect (Cohen’s kappa values of 0.81). The inter-reader reliability of the number of CILs was excellent (Pearson’s correlation coefficient 0.94).
ROC curves were constructed to determine the diagnostic utility of CILs at different vertebral levels (Figure 2). Whole spine CILs (AUC 0.67; 95% confidence interval (CI) 0.62–0.72; p = <0.01) and thoracic spine CILs (AUC 0.66; 95% CI 0.61–0.72; p = <0.01) had the ability to differentiate between patients without and without axSpA.

Test precision of corner inflammatory lesions at different spinal levels in axial spondyloarthritis diagnosis.
MRI sacroiliitis had a sensitivity of 33.6% and specificity of 97.4%. The different cut-off thresholds of CILs when assessed as whole spine and thoracic spine are presented in Table 3. Using a specificity of 97.4% (specificity of MRI sacroiliitis) as threshold, the optimal cutoff values were ⩾5 whole spine CILs and ⩾3 thoracic spine CILs.
Sensitivity and specificity of corner inflammatory lesion cutoff thresholds in diagnosis of axial spondyloarthritis.
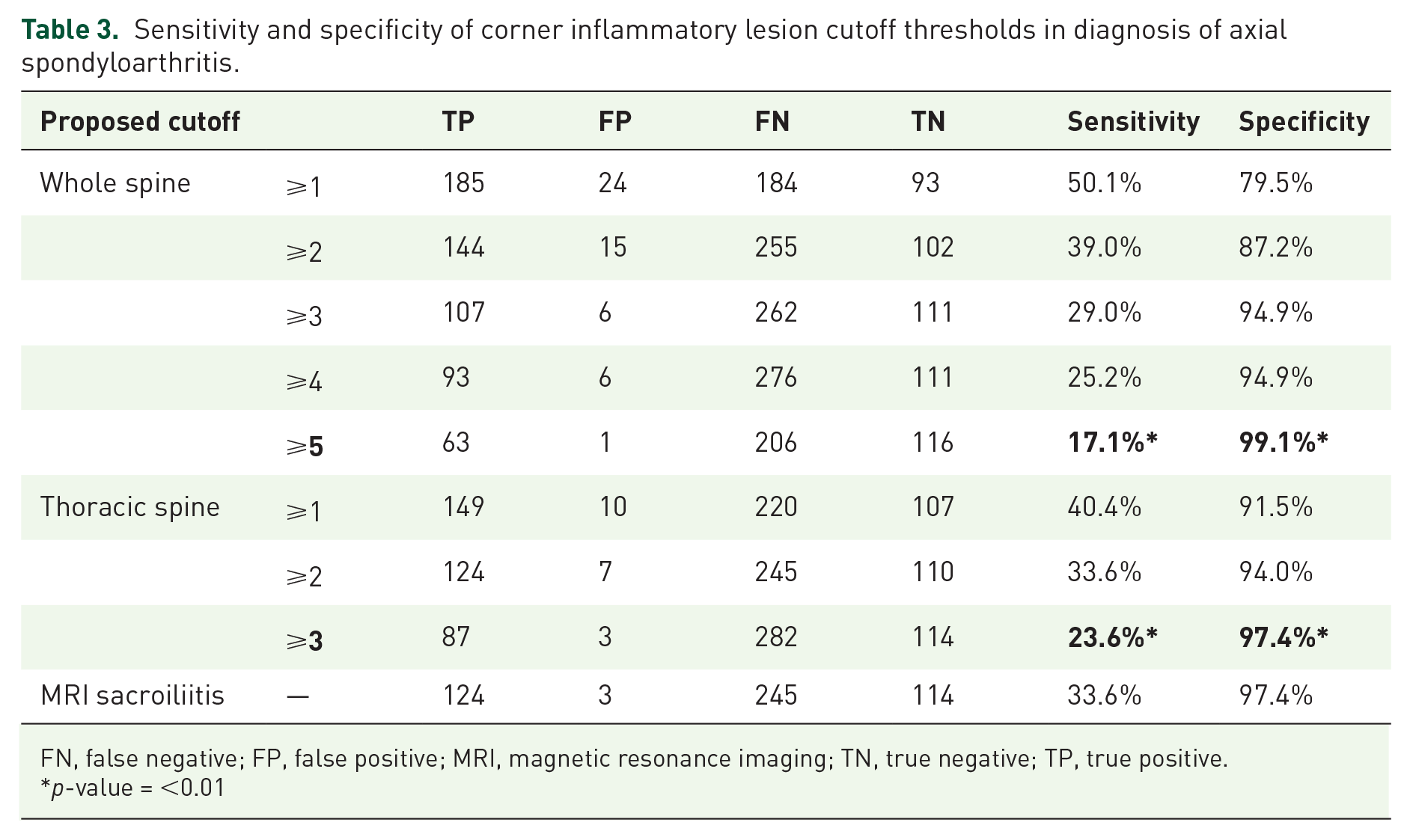
FN, false negative; FP, false positive; MRI, magnetic resonance imaging; TN, true negative; TP, true positive.
p-value = <0.01
In the axSpA group, 85 of 369 (23.0%) patients had no sacroiliitis on MRI or X-ray. Of these 85 patients, 7/85 (8.2%) had ⩾5 whole spine CILs and 11/85 (12.9%) had ⩾3 thoracic CILs. Among NSBP patients, only 1/117 (0.9%) have ⩾5 whole spine CILs and 3/117 (2.6%) have ⩾3 thoracic CILs.
The diagnostic utility of adding the respective proposed CIL cutoff values to the imaging arm of the ASAS criteria for axSpA diagnosis are presented in Tables 4 and 5. Adding the CIL cutoff values to the existing ASAS imaging criteria only marginally improved the sensitivity except in the non-radiographic subgroup. In the non-radiographic axSpA subgroup, incorporating CIL criteria into the imaging criteria of ASAS axSpA criteria improve the sensitivity (sensitivity 0.38 to 0.43, ⩾5 whole spine CILs; sensitivity 0.38 to 0.46, ⩾3 thoracic spine CILs) with similar specificity (specificity 0.93 to 0.92, ⩾5 whole spine CILs; specificity 0.93 to 0.91, ⩾3 thoracic spine CILs).
Sensitivity, specificity, positive predictive value, negative predictive value, positive and negative likelihood ratio of different classification criteria.
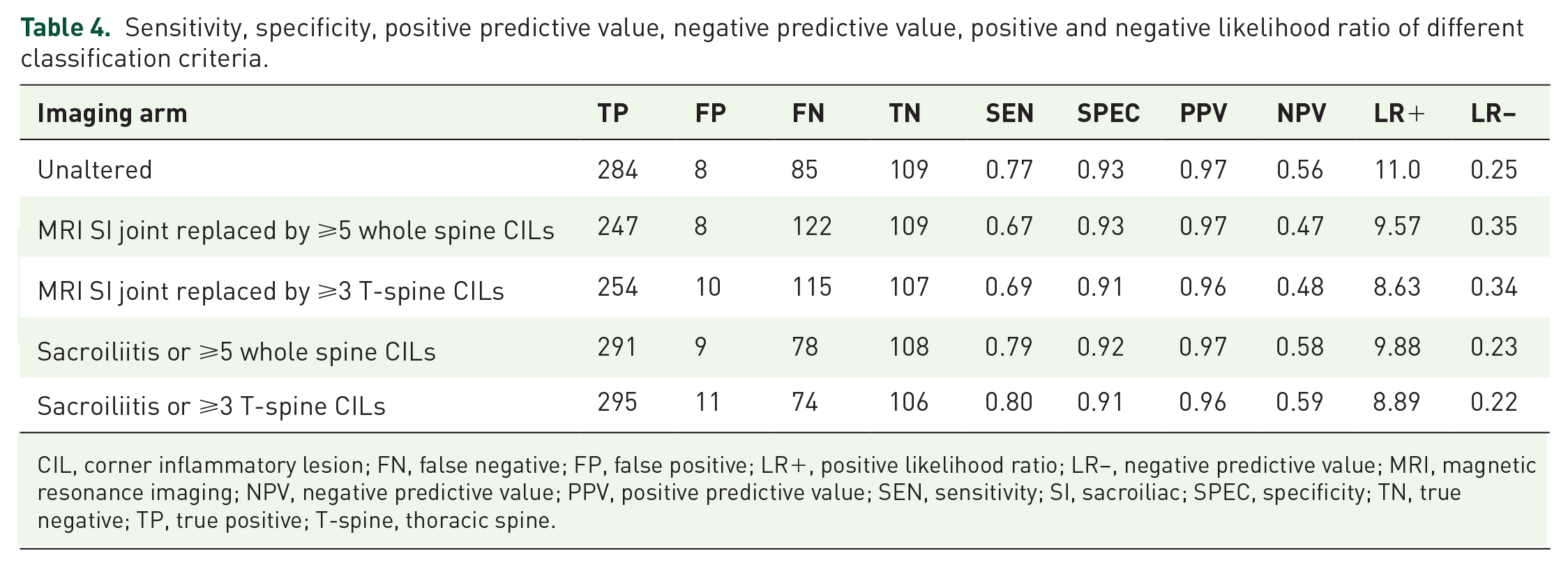
CIL, corner inflammatory lesion; FN, false negative; FP, false positive; LR+, positive likelihood ratio; LR−, negative predictive value; MRI, magnetic resonance imaging; NPV, negative predictive value; PPV, positive predictive value; SEN, sensitivity; SI, sacroiliac; SPEC, specificity; TN, true negative; TP, true positive; T-spine, thoracic spine.
Subgroup analysis in non-radiographic axial spondyloarthritis. Sensitivity, specificity, positive predictive value, negative predictive value, positive and negative likelihood ratio of different classification criteria.

CIL, corner inflammatory lesion; FN, false negative; FP, false positive; LR+, positive likelihood ratio; LR−, negative predictive value; MRI, magnetic resonance imaging; NPV, negative predictive value; PPV, positive predictive value; SEN, sensitivity; SI, sacroiliac; SPEC, specificity; TN, true negative; TP, true positive.
Discussion
This study systemically investigated the diagnostic utility of CILs at different locations (cervical, thoracic, and lumbar) in a large group of participants with back pain of which 369 participants had axSpA and 117 patients had NSBP. Previous studies have proposed different CIL cutoff values for positive MRI of the spine,4,18 but its usefulness in the diagnosis of axSpA has been questioned in a number of studies.6,7,19,20 Our study evaluated CILs further grouped by vertebral levels. Similar to previous studies, our study showed that spinal MRI provided little incremental diagnostic value in axSpA when added to standard SI joint imaging among unselected patients, but might be useful in 8–13% of those without sacroiliitis on MRI or radiographs. Thoracic and whole spine MRI had similar diagnostic performance using the proposed cutoff value of ⩾5 whole spine CILs (W-CILs) and ⩾3 thoracic CILs (T-CILs) as “positive MRI” for spinal inflammation.
Compared with a study examining the SPACE and DESIR cohort by Ez-Zaitouni et al. 19 we demonstrated a higher frequency of positive MRI of the spine in participants without sacroiliitis using the above proposed criteria. In our cohort of participants with axSpA, 85 out of 369 (23.0%) patients had negative imaging for sacroiliitis by MRI or X-ray. Of these 85 axSpA patients, seven out of 85 (8.2%) had ⩾5 whole spine CILs and 11 out of 85 (12.9%) had ⩾3 thoracic CILs. This might be due to the difference in patient characteristics between the two studies. In the abovementioned study involving the SPACE and DESIR cohort, 19 it was shown that among patients with early disease (defined as chronic back pain of maximally 3 years’ duration) only 1–2% of patients without sacroiliitis had a positive MRI spine (defined as the presence of ⩾3 CILs). In contrast, our cohort is made up of participants with longer disease duration, older age, higher percentage of HLA-B27 positivity and radiographic sacroiliitis compared with the SPACE and DESIR cohorts. Furthermore, our study included a control group of participants with NSBP. We demonstrated that the diagnostic utility of thoracic spine MRI was comparable to whole spine MRI. The use of whole spine or thoracic spine MRI might be considered in axSpA patients, especially in those without sacroiliitis on conventional radiograph or MRI.
The thoracic spine has been shown to have the lowest prevalence of degenerative lesions due to its inherent biomechanics, 21 hence, we proposed its use in the differentiation between axSpA and other NSBP. We found that CILs in the thoracic spinal segments were the most frequent, accounting for 74% of the total number of CILs. This study demonstrated the diagnostic usefulness of CIL in axSpA similar to the result from previous studies.22,23 From our data, CILs were the most frequent in the thoracic spine even after adjustment for number of vertebrae (Table 6). Also, thoracic CILs were more commonly found in axSpA (40.4%), and less frequently in NSBP patients (8.5%). Furthermore, the average number of thoracic CILs present in SpA patients was greater than that in NSBP patients (mean n = 4.3 versus 2.7). A previous study has proposed ⩾3 CIL as the cutoff value for the diagnosis of axSpA. 4 If we assess thoracic spinal segment alone, the cutoff value of ⩾3 thoracic CILs had a specificity comparable to positive MRI for sacroiliitis but ⩾5 CILs were needed to achieve comparable specificity when the whole spine was considered.
Frequency of corner inflammatory lesions by spinal segments with adjustment for numbers of vertebrae.
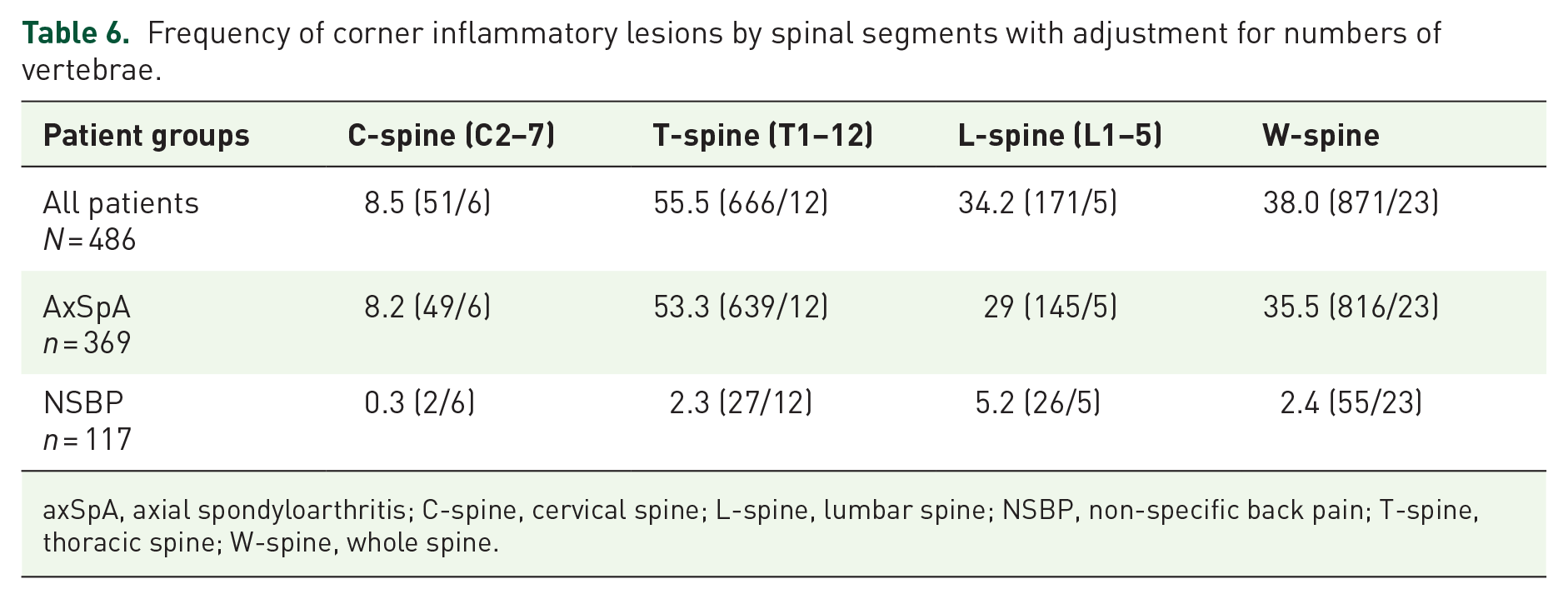
axSpA, axial spondyloarthritis; C-spine, cervical spine; L-spine, lumbar spine; NSBP, non-specific back pain; T-spine, thoracic spine; W-spine, whole spine.
Corresponding to CIL is fatty corner lesion (FCL). It is a well-demarcated fat infiltration in the corner of a vertebral body on T1 MRI sequence. 24 Fatty lesion is proposed to be the result of resolution of inflammation. 25 We have previously reported the differential diagnostic utility of FCL at different levels 26 and shown that thoracic FCLs are more frequent and have a greater contribution to axSpA diagnosis.
Our study showed that when incorporated into the ASAS criteria, the proposed MRI spine criterion was particularly useful in the group of non-radiographic-axSpA in improving the sensitivity of the imaging arm. In non-radiographic axSpA patients, only 38% have MRI sacroiliitis. By incorporating MRI spine assessment into the ASAS axSpA criteria, the sensitivity of the imaging arm improved (sensitivity 0.38 to 0.43, ⩾5 whole spine CILs; sensitivity 0.38 to 0.46, ⩾3 thoracic spine CILs) with similar specificity (specificity 0.93 to 0.92, ⩾5 whole spine CILs; specificity 0.93 to 0.91, ⩾3 thoracic spine CILs).
Limitation
Similar to many other rheumatic diseases, axSpA remained a clinical diagnosis and in this study physician diagnosis was used as the gold standard. Positive MRI sacroiliitis was defined according to ASAS definition and structural lesions were not considered. Even though the overall prevalence of positive MRI sacroiliitis was relatively low, the frequency of positive MRI sacroiliitis in the non-radiographic axSpA subgroup was similar to other cohorts. 27 As a result, the overall performance of the imaging arm was comparable to other international cohorts. 28 Our cohort was characterized by participants with long-standing disease instead of a group of new undiagnosed patients. It is also known that agreement in reading radiographs of sacroiliac joints is only moderate. 29 This could have potentially affected the interpretation of our results. Furthermore, the baseline characteristics between axSpA and NSBP groups were different. However, the frequency of CILs in our control cohort was similar to that reported in a recent study by Baraliokos et al. among healthy individuals. 30 This supports the validity of our cohort as the control group for comparison.
Conclusion
Spinal MRI provided little incremental diagnostic value in unselected axSpA patients and should not be incorporated as a routine assessment. However, among the subgroup of patients without sacroiliitis on MRI or radiographs, 8–13% might be diagnosed by spinal MRI using the proposed criteria. Thoracic and whole spine MRI had similar diagnostic performance using the proposed cutoff value of ⩾5 W-CILs and ⩾3 T-CILs as “positive MRI” for spinal inflammation.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X20973922 – Supplemental material for Diagnostic utility of whole spine and thoracic spine MRI corner inflammatory lesions in axial spondyloarthritis
Supplemental material, sj-docx-1-tab-10.1177_1759720X20973922 for Diagnostic utility of whole spine and thoracic spine MRI corner inflammatory lesions in axial spondyloarthritis by Shirley Chiu Wai Chan, Philip Hei Li, Kam Ho Lee, Helen Hoi Lun Tsang, Chak Sing Lau and Ho Yin Chung in Therapeutic Advances in Musculoskeletal Disease
Footnotes
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by the Hong Kong Society of Rheumatology, and Novartis Research.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
